Abstract
The MnO
1. Introduction
The combustion of massive fossil fuels brings about the harmful emission of nitrogen oxides (NO
NH3-SCR has also exposed many problems in a wide range of industrial applications, such as high investment and operating costs, NH3 escape, N2O generation, and catalyst deactivation [11, 12]. The greenhouse effect of N2O is up to 300 times that of carbon dioxide. The almost inevitable escape of NH3 is particularly worrying. It not only increases the operation cost but also easily results in serious air preheater blocking. Therefore, how to remove NO
Although many noble metal catalysts have shown good performance in NO catalytic oxidation, the high cost limits their wide application in coal-fired power plants [18–20]. Transition metal oxides have been proven with excellent performance compared to noble metal catalysts, with a wide range of sources, low prices, simple preparation processes, and good thermal stability. Therefore, they have received extensive and in-depth research in recent years [21–23]. Wu et al. prepared a series of MnO
Active Al2O3 has the characteristics of large adsorption capacity, large specific surface area, good thermal stability, nontoxicity, and noncorrosiveness. Therefore, it is regarded as an excellent catalyst support material and has received extensive attention in the field of catalysis. Wang et al. [26] used a sol-gel method to prepare a series of Ce-based catalysts, selecting Co, Mn, Fe, Cr, and Ni as the doping metal elements. At a reaction temperature of 230°C, the order of NO catalytic activity is Co > Mn > Cr > Ni > Fe.
Although much work has been carried out on NO catalytic oxidation over transition metal oxides, there are some deficiencies along with these studies [21–25]. Firstly, only oxidation efficiency but not removal efficiency was focused on during the experiments. Secondly, the temperature window of the researched catalysts was relatively narrow, not suitable for large-scale practical application. In this investigation, we prepared a series of MnO
2. Experimental
2.1. Catalyst Preparation
All tested samples in the study were prepared via an ultrasonic impregnation method. Chemicals used here were of analytical grade. Firstly, 0.04 mol (10.04 g) Mn(NO3)2·4H2O was dissolved in 20 mL deionized water. Afterwards, with continuous magnetic stirring, 0.1 mol (10.20 g) Al2O3 was added into the solution (
For samples with different Ce addition, a specific amount (0.005 mol, 0.01 mol, and 0.02 mol, respectively) of Ce(NO3)3·6H2O was dissolved in deionized water with Mn(NO3)2·4H2O together in the first preparation step. Other preparation procedures were the same as mentioned above. The finally prepared samples were denoted as Mn0.4Ce0.05/Al, Mn0.4Ce0.1/Al, and Mn0.4Ce0.2/Al, respectively.
2.2. Catalytic Activity Test
The catalyst activity test system is shown in Figure 1. The catalytic activity test was carried out in a fixed-bed quartz tube furnace. 0.5 g sample was fixed on the bottom of the quartz glass tube by quartz wool. The total gas flow rate was fixed to 1 L/min (STP), with 600 ppm NO, 8 vol% O2, and balanced N2. The exhaust gas after the reaction at a certain temperature was introduced into a 0.5 mol/L sodium hydroxide aqueous solution for absorption, after which the outlet gas composition was examined online by a Fourier transform infrared spectroscopy gas analyzer (Gasmet DX4000, Finland).

Schematic diagram of the experimental setup.
The NO removal efficiency was calculated according to the following equation:
2.3. Catalyst Characterization
N2 adsorption-desorption measurement was performed on a full-automatic physical-chemical adsorption instrument (Micromeritics ASAP2020, USA) to determine the textural properties of samples. The specific surface area was acquired by the Brunauer-Emmett-Teller (BET) method, while the total pore volume and mean pore diameter were obtained according to the Barrett-Joyner-Halenda (BJH) method. The micromorphologies of samples were monitored by a field emission scanning electron microscope (FESEM, GeminiSEM 500, Germany). An X-ray diffraction (XRD) meter (Xpert pro, Netherlands) was adopted to identify the crystal phases of samples. The scanning angular velocity was 7°/min, and the scanning angle range was 20°-80°. An X-ray photoelectron spectroscopy (XPS, AXIS ULtrabld, UK) was employed to analyze the catalyst surface atomic concentrations, using C1s at 284.8 eV as the calibration.
3. Results and Discussion
3.1. Catalytic Activity
600 ppm NO, 8 vol% O2, and balanced N2 were introduced into the reactor to explore the performance of catalysts with different contents of active component Ce (Figure 2). As the reaction temperature increased from 20°C to 500°C, the NO removal efficiency was peaked at 400°C for all samples. The oxidation of NO to NO2 is exothermic, and the reaction is limited by the thermodynamic equilibrium: the temperature rises, and the equilibrium shifts to the left. Therefore, the temperature increased, the NO oxidation rate decreased, and the denitration efficiency decreased. It was apparent that the removal efficiency of the Ce-doped Mn0.4/Al catalyst was higher than that of the Ce-free Mn0.4/Al catalyst, indicating the promotion effect of Ce addition on NO oxidation removal. In addition, the temperature window of Mn0.4Ce

Removal efficiency of catalysts with different Ce contents.
For the Mn0.4/Al catalyst without Ce addition, the denitration efficiency increased gradually with the reaction temperature in 20-400°C, but it started to decrease slightly as the reaction temperature increased from 400°C to 500°C. The highest efficiency reached 79.5% at 400°C. When the molar ratio of Mn, Ce, and Al was 0.4 : 0.05 : 1, the efficiency reached the highest peak of 89.5%. When the molar ratio of Mn, Ce, and Al was 0.4 : 0.1 : 1, the efficiency peak reached 89.1%, similar to Mn0.4Ce0.05/Al. For Mn0.4Ce0.2/Al, the highest efficiency was 85.2%, a little lower than Mn0.4Ce0.05/Al and Mn0.4Ce0.1/Al. Doping with Ce improves the activity of the Mn/Al catalyst significantly. The temperature window moves to the left, indicating that the low-temperature activity of the catalyst is enhanced. Free Ce has excellent oxygen storage capacity, and a small amount of Ce doping increases the active sites on the catalyst surface, thereby increasing the NO removal rate. But excessive doping may aggravate the accumulation of surface crystals, cover some active centers, or block the pores, resulting in a decrease in the catalytic activity of the catalyst [27, 28]. In the subsequent surface analysis, it was found that after Ce doping, the specific surface area was significantly reduced. The active ingredient is not as much as possible and should be lower than the surface dispersion threshold. Otherwise, Ce agglomerates and stacks on the surface, so Mn0.4Ce0.05/Al with better activity is selected as the main research object in the follow-up.
3.2. XRD
The XRD patterns of Mn0.4/Al and Mn0.4Ce0.05/Al expressing the crystal phases on the catalyst surface are depicted in Figure 3. MnO

XRD patterns of Mn0.4/Al and Mn0.4Ce0.05/Al.
In the XRD patterns of Mn0.4Ce0.05/Al, the diffraction peaks of MnO2 and Mn3O4 were very weak, and no diffraction peaks of Mn2O3 existed, which revealed that the addition of Ce had a great influence on the crystal structure of Mn0.4/Al catalyst. It is worth noting that we also did not detect the crystalline phase of Ce in the XRD pattern, which indicated that Ce was evenly dispersed on the surface of the catalyst, or Ce enters the lattice of manganese. We speculate that Ce atoms may enter the lattice of Mn2O3, resulting in the disappearance of a large amount of Mn2O3 crystal structure on the catalyst surface and resulting in the increase of the crystal structure of MnO2 and Mn3O4 on the catalyst surface.
After adding active component Ce to Mn0.4/Al catalyst, manganese and cerium interacted in a solid solution manner, and manganese ions entered the cerium oxide lattice to increase the oxygen storage capacity of the cerium oxide and the oxygen migration activity of the surface oxide [29]. This interaction was related to the electron transfer between manganese and cerium and the gain and loss of oxygen, and it also influenced the crystal structure of the catalyst and the valence state of manganese and cerium compounds. Therefore, related characterization analysis was carried out.
3.3. XPS
The XPS spectra for Mn 2p of Mn0.4/Al and Mn0.4Ce0.05/Al are separately drawn in Figure 4(a) and Figure 4(b). Mn2p peaks for Mn oxides have many multiplet-split components, and the binding energy of Mn4+ is greater than Mn3+ [30]. In Figure 4(a), the binding energy peaks of 641.0 eV and 652.5 eV represent Mn3+; the binding energy peaks at 642.8 eV and 653.4 eV represent Mn4+. The relative size of the energy spectrum peak area represents the relative content of different manganese oxides on the catalyst surface. The

XPS spectra for Mn 2p of (a) Mn0.4/Al and (b) Mn0.4Ce0.05/Al.
On Mn0.4Ce0.05/Al, the binding energy peaks of 641.3 eV and 652.8 eV represent Mn3+; the binding energy peaks at 642.8 eV and 654.2 eV represent Mn4+ in Figure 4(b). Compared with Mn0.4Ce0.05/Al, the energy level was shifted upward. The
Figure 5 shows the XPS spectra for O 1s of Mn0.4/Al (Figure 5(a)) and Mn0.4Ce0.05/Al (Figure 5(b)). There were two kinds of oxygen in catalysts, i.e., surface absorbed oxygen (denoted as O
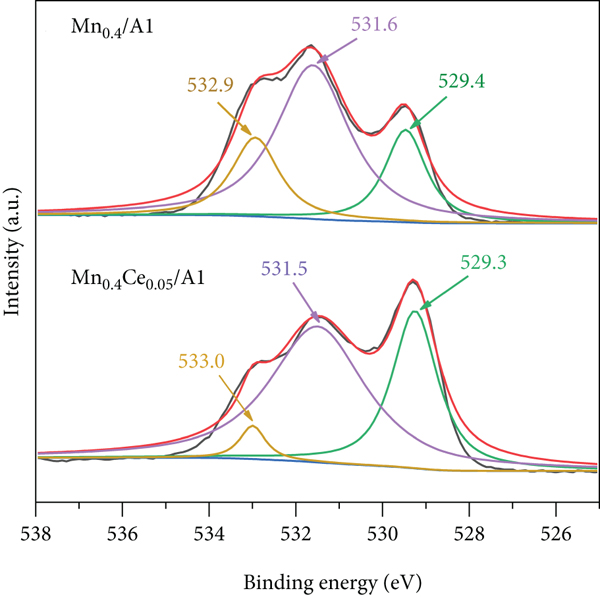
XPS spectra for O 1s of (a) the Mn0.4/Al catalyst and (b) the Mn0.4Ce0.05/Al catalyst.
Lattice oxygen played an important role in NO oxidation. After adsorption on the catalyst surface, NO was first oxidized by the active lattice oxygen to form nitrite or nitrate on the surface of the catalyst [32]. The higher proportion of O
Figure 6 shows the XPS spectra for Ce 3d of Mn0.4Ce0.05/Al. The Ce 3d spectrum consists of two series of spin-orbit lines Ce3d3/2 and Ce3d5/2 [34]. There were eight distinct characteristic peaks, of which the peaks at 881.8 eV, 889.2 eV, 897.7 eV, 902.3 eV, 907.0 eV, and 916.0 eV correspond to Ce4+ [26, 35], and the peaks at 902.2 eV and 884.0 eV correspond to Ce3+ [36, 37]. It indicated that Ce in Mn0.4Ce0.05/Al had two forms of Ce4+ and Ce3+ after calcination at high temperature. The

XPS spectra for Ce 3d of the Mn0.4Ce0.05/Al catalyst.
Ce had strong oxygen storage capacity and stores and releases oxygen through the transformation of Ce3+ and Ce4+, which was consistent with the higher lattice oxygen content on the surface of Mn0.4Ce0.05/Al. It can be seen from Figure 1 that Ce doping improved the low-temperature activity of the catalyst, which was consistent with the study of other scholars [38, 39]. In addition, the doping of Ce element led to a reduction in the amount of O atoms combined with Mn, which in turn converted Mn4+ to Mn3+ with better activity.
The results of the XPS characteristics of Mn0.4/Al and Mn0.4Ce0.05/Al are listed in Table 1. According to the analysis above, the increase of Mn3+ and lattice oxygen was important for the effective improvement of catalyst activity.
Results of the XPS results of Mn0.4/Al and Mn0.4Ce0.05/Al.
The physical properties of Mn0.4/Al and Mn0.4Ce0.05/Al are listed in Table 2, mainly including the BET surface area, the BJH pore volume, and the BJH average pore diameter. From Table 2, it could be found that the surface area and the pore volume of the Mn0.4Ce0.05/Al catalyst were lower than those of the Mn0.4/Al catalyst. On the contrary, the pore diameter of the Mn0.4Ce0.05/Al catalyst was higher.
Physical properties of catalysts.
According to the XRD analysis results, cerium ions entered the manganese oxide lattice, resulting in an increase in the weight per unit volume of the pore structure and resulting in the decrease in the specific surface area and pore volume of the catalyst [40]. And it could be found in Table 1 that the O
3.4. FESEM
The FESEM images with magnification times (×10000) of Mn0.4/Al and Mn0.4Ce0.05/Al are shown in Figure 7. As shown in Figure 7(a), the Mn0.4/Al surface was evenly distributed with fine particles. According to the above XRD analysis results, they were likely to be Mn2O3 particles. As shown in Figure 7(b), there were many needle-like substances on the Mn0.4Ce0.05/Al surface. The surface of Mn0.4Ce0.05/Al was rougher, which was conducive to generating more active sites and also conducive to the adsorption of reactants, which strengthens the catalytic oxidation of NO on the surface.

FESEM images of (a) Mn0.4/Al and (b) Mn0.4Ce0.05/Al.

4. Conclusions
The NO oxidation removal activity of Mn0.4/Al catalysts with different Ce contents (Mn0.4/Al, Mn0.4Ce0.05/Al, Mn0.4Ce0.1/Al, and Mn0.4Ce0.2/Al) was studied experimentally. The results showed that the activity of Mn0.4/Al catalysts was effectively promoted with Ce addition, and the Mn0.4Ce0.05/Al performed the best.
Simultaneously, the physical-chemical properties and microstructures of Mn0.4/Al and Mn0.4Ce0.05/Al were compared and analyzed by various characterization methods, which was helpful to reveal the mechanism of catalytic oxidation of NO by Mn-based catalysts and the effect of Ce addition. The characterization results showed that (1) the entry of cerium ions into the manganese oxide lattice led to the change of crystal structure of the catalyst surface and the decrease of specific surface area and pore volume; (2) the decrease of Mn4+ and the increase of Mn3+ on the catalyst surface were beneficial to the NO oxidation; (3) Ce doping increased the lattice oxygen content on the surface of the Mn0.4Ce0.05/Al, which was favorable for NO oxidation.
Footnotes
Data Availability
All data, models, and code generated or used during the study appear in the submitted article.
Conflicts of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Acknowledgments
The present work was supported by the National Natural Science Foundation of China (51906193).
