Abstract
The material, δ-MnO2, has exhibited superior performance on the removal of methylene blue (MB), but the process is significantly impacted by pH, and the impacting mechanism is still unclear. In this study, the effects of pH on the removal mechanism of MB using synthesized δ-MnO2 were investigated by distinguishing the adsorption and oxidation of MB by δ-MnO2 during the removal process in the dark. The results show that the total removal efficiency of MB by δ-MnO2 decreased significantly with an increase in the pH. MB could be removed by δ-MnO2 via an adsorption mechanism and oxidation mechanism, and the proportion of adsorptive removal and oxidative removal was different under different pH conditions. With an increase in the initial pH from 2.00 to 8.05, the redox potential of δ-MnO2 decreased, and its oxidation ability for the removal of MB also gradually decreased. In contrast, the surface negative charges of δ-MnO2 increased with an increase in the pH, and the adsorption ability towards positively charged MB also gradually increased. This indicates that the effects of pH on the removal of MB by δ-MnO2 are primarily dominated by its influence on the oxidation ability of δ-MnO2. In addition, it is further proved that the pH value has a significant effect on the oxidation and adsorption of MB on δ-MnO2. Moreover, the significant effects of pH on the oxidation of MB by δ-MnO2 are further demonstrated by observing the changes in Mn2+ and the UV-Vis spectra of intermediate products during the reaction, as well as the changes in the FTIR and XPS characterizations of δ-MnO2 after the reaction.
1. Introduction
Manganese oxides widely exist in the earth’s surface and water environments, and they are important components of soil and sediment [1]. In the microstructures of manganese oxides, manganese atoms coordinate with oxygen atoms in the form of [MnO6] octahedra, and the [MnO6] octahedra form single chains through coedge connections. These single chains are connected by sharing edges or corner oxygen atoms forming manganese oxides with different crystal structures [2]. The surface of manganese oxides belongs to hydrated oxide types with variable surface charges [3]. Because the zero point of charge (ZPC) of manganese oxides is very low, they are usually negatively charged within the normal pH range, which can adsorb charged heavy metal ions or organic pollutants [4–6]. Manganese oxides are primarily composed of high valent Mn(III) and Mn(IV) [7], which have high redox potentials. Hence, the removal towards organic pollutants can be achieved via oxidation reactions [8–10]. As a semiconductor, manganese oxides also exhibit photocatalytic activity, which can photocatalytically remove organic pollutants by irradiation [11]. Manganese oxides are often used to remove organic pollutants due to their multiple environmental properties, such as adsorption, oxidation, and photocatalysis [12, 13].
More than
Therefore, in this study, the common manganese oxide, δ-MnO2, was selected as a representative for manganese oxides. The effects of pH on the removal mechanism of MB by δ-MnO2 were studied by analyzing the adsorption amount and oxidation amount of MB by δ-MnO2, as well as the changes in pH, Mn2+, and products during the reaction process. The reaction was conducted in the dark in order to avoid the photocatalytic effect of δ-MnO2.
2. Materials and Methods
2.1. Synthesis and Characterization
The used δ-MnO2 were prepared with KMnO4 and HCl, typically in a triangular flask with 300-400 mL of deionized water boiled in a constant temperature oil bath (110°C). Then, 120 mL of 6 mol/L HCl was added dropwise at a rate of 0.7 mL/min under strong stirring. After the addition was completed, the reaction was continued for 30 min, and the obtained product was aged at 60°C for 12 h [23]. After washing with deionized water to conductivity of <15 μs/cm, the final product was dried in a 40°C oven, ground and passed through a 60-mesh sieve, and then placed in a desiccator for later use. Methylene blue (
2.2. Characterization
The crystal structure of the sample was determined by the Bruker Advance D8 X-ray powder diffraction (XRD) with CuKα radiation at 40 kV tube voltage, 40 mA tube current, 1°/min scanning speed, scanning interval of 5°-85°, and step size of 0.02°. The morphology of δ-MnO2 was observed under the scanning electron microscope (SEM) of FEI Nova NanoSEM450. Transmission electron microscope (TEM) analysis was used to analyze the morphology and particle size characteristics by JEOL JEM-2100. The specific surface area (SSA) was determined using Quantachrome Autosorb-1. The zero point of charge (ZPC) was measured by the rapid potentiometric titration method. The average oxidation state (AOS) of Mn was measured by the oxalic acid-permanganate back-titration method [24].
2.3. Dye Removal
2.3.1. Kinetic
100 mL 160 μmol/L dye solution (pH 3.99) was added into 100 mL 400 mg/L δ-MnO2 suspension (pH 4.00) in a 250 mL triangle bottle under magnetic stirring, and the tin paper was wrapped in a triangular bottle to avoid light. The pH of dye and δ-MnO2 was adjusted with HCl or NaOH solution until the pH value is stable in ±0.05 in 12 h. 10 mL of the supernatant was sampled using a syringe and immediately filtered through a 0.45 μm membrane filter at 0 min, 1 min, 3 min, 5 min, 9 min, 15 min, 20 min, 30 min, 60 min, 90 min, 180 min, and 240 min, respectively. In addition, two parallel experiments were set. The liquid under test was ally determined by the UV spectrophotometer (UV). The removal rate
2.3.2. Removal Mechanism
For the investigation of the mechanism, 50 mL of an 80 μmol/L MB solution was added into 50 mL of 200 mg/L δ-MnO2 suspension in a 150 mL triangle bottle using tin foil to avoid light under magnetic stirring. The pH of the dye and the δ-MnO2 was adjusted to the desired value (pH of 2.00, 4.01, 5.92, and 8.05) with the HCl or NaOH solution. The sampling times were 0.5 h, 1 h, and 3 h. At the specified time, 10 mL of the solution was filtered through a 0.45 μm membrane filter, and the removal efficiency was measured using the dye (Equation (1)) of the filter membrane as the total removal efficiency (
The δ-MnO2 that passed through the filter in the experiment was collected and repeatedly rinsed with ultrapure water and drying. The reacted δ-MnO2 was subjected to FTIR and XPS measurement and compared with the initial δ-MnO2 [26, 27]. UV-visible spectroscopy was utilized to determine dye removal efficiency and adsorption capacity. pH and the contents of Mn2+ of the solution after filtering the solution were also measured using atomic absorption spectrometry (AAS, PerkinElmer AA900). All the experiments were run in duplicate, and the data presented are averages of this duplicate analysis.
3. Results
3.1. Characterization of δ-MnO2
Table 1 shows the physicochemical properties of the as-synthesized δ-MnO2. The powder XRD pattern of the as-synthesized sample is shown in Figure 1(a). The
The physicochemical properties of δ-MnO2.
aBET surface area; bmicropore surface area; cexternal surface area; dmicropore volume obtained from
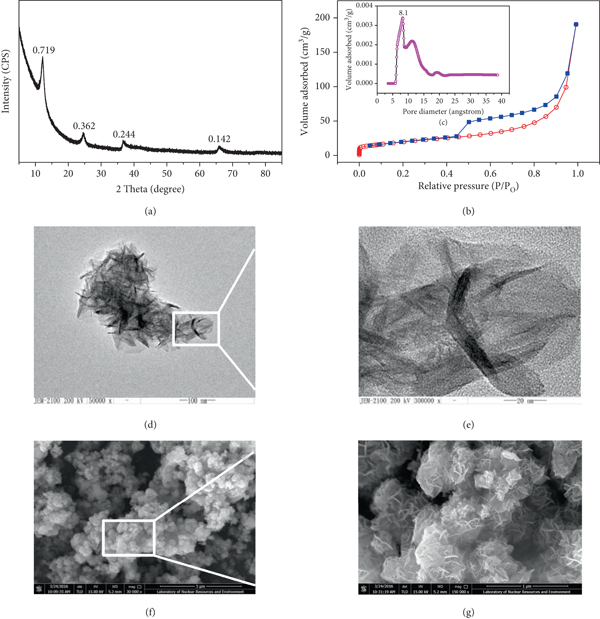
The XRD pattern (a) of δ-MnO2; the N2 adsorption/desorption isothermal plot of δ-MnO2 (b) and the microspore size distribution plot (c) by the SF method; HRTEM (d, e) and HRSEM (f, g) images of δ-MnO2.
3.2. Kinetics of the Removal of MB by δ-MnO2
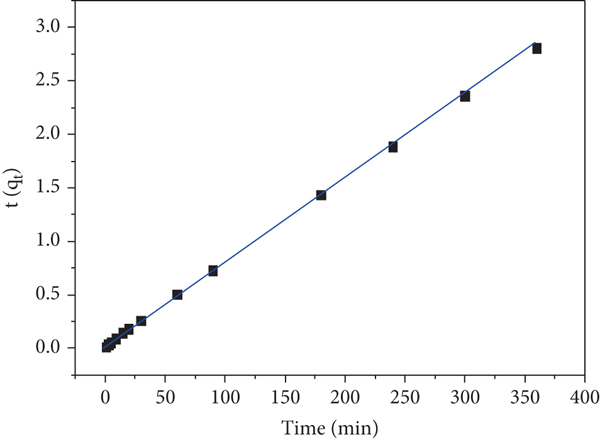
The kinetics of the removal of MB by δ-MnO2 are shown in Figure 2. The removal rate of MB was high within the initial 60 min, and the removal efficiency reached 50.93% at 1 min and 79.66% at 60 min. With an increase in the reaction time, the removal rate gradually decreased reaching equilibrium, and the removal efficiency was 85.74% (Figure 2(a)) after 6 h of reaction. The obtained kinetic data were linearly fitted using the pseudo-first-order (PFO) and the pseudo-second-order (PSO) kinetic models (Figures 2(b) and 2(c)). The fitting results are shown in Table 2. The correlation coefficients,
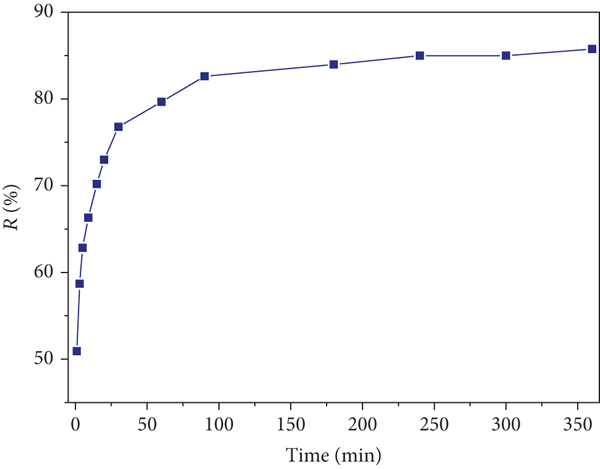
(a) Adsorption capacity as a function of contact time using the (b) pseudo-first-order, (c) pseudo-second-order, and (d) UV-Vis wavelength scan of MB dye.


Parameters of the PFO and PSO kinetic models for the adsorption of MB on δ-MnO2.
3.3. The Effect of pH on Removal of MB
The changes in

Effect of pH on

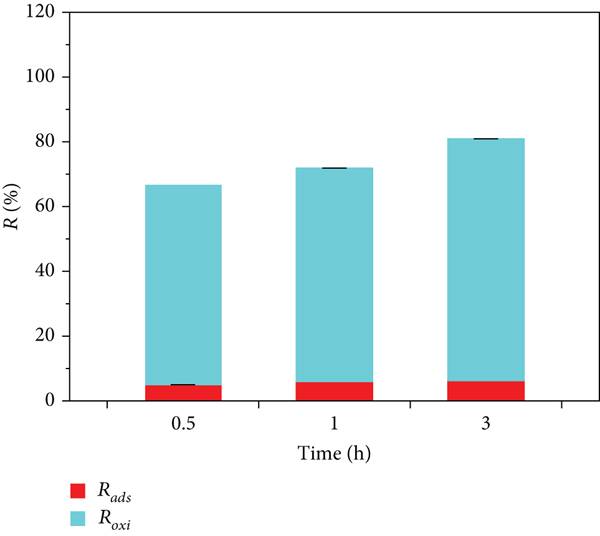

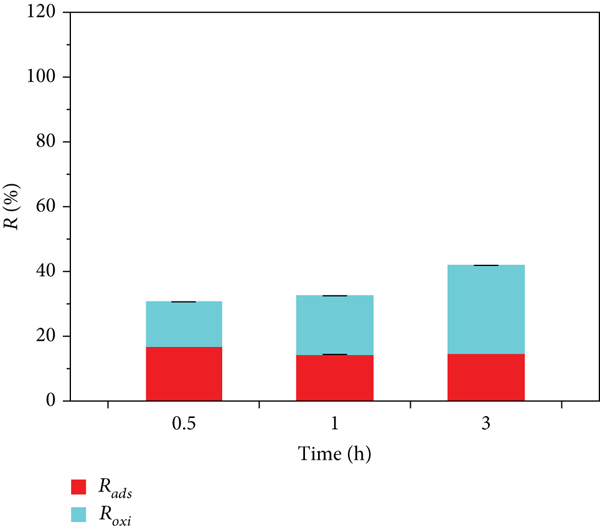
The above results indicate that there was almost no adsorption of MB on the surface of δ-MnO2 when the pH was 2.00 (Figure 3(b)), at which time, the removal of MB on the surface of δ-MnO2 was primarily dominated by oxidation reactions. In addition, there was almost no MB in the reaction system. At a
4. Discussion
The above results show that the removal of MB by δ-MnO2 includes oxidation removal and adsorption removal. In order to further explore the oxidation removal mechanism, the UV-visible spectra of the MB solution after reaction with δ-MnO2 under different pH conditions are compared (Figure 4). The change of Mn2+ in the solution (Figure 5), FTIR spectrum (Figure 6), and XPS (Figure 7).

UV-Vis of MB after 3 hours of reaction at pH 2.00, 4.01 (dilute five times), 5.92 (dilute ten times), and 8.05 (dilute ten times).

Trend of the release of Mn2+ during the reaction in different pH.
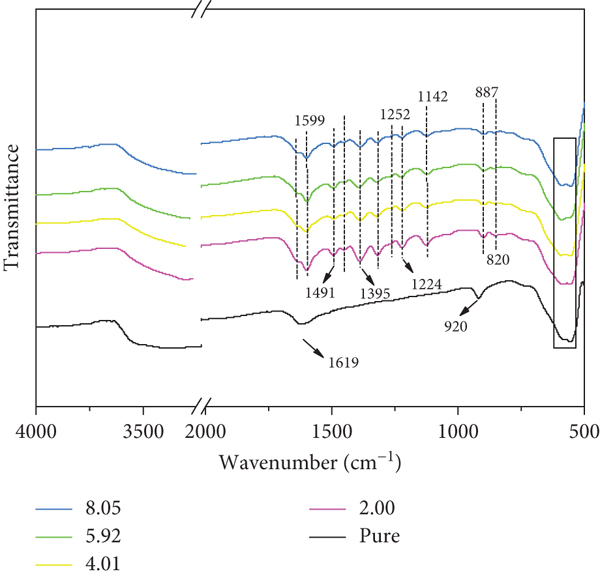
FTIR spectra of acid δ-MnO2 (pure) and acid δ-MnO2 after reaction with MB at different pH (2.00, 4.01, 5.92, and 8.05).
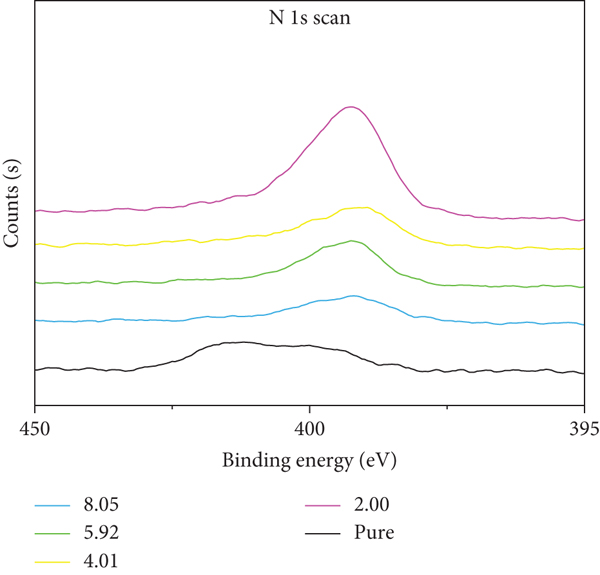
The best fit of the N 1s XPS spectra of acid δ-MnO2 (pure) and δ-MnO2 after reaction with MB at different pH (2.00, 4.01, 5.92, and 8.05).



4.1. Oxidating MB by δ-MnO2
In order to prove that the oxidation-reduction reaction occurred when δ-MnO2 removes MB, the intermediate and final products of the oxidation reaction can be analyzed. First of all, it can be seen from Figure 4 that under the reaction conditions of different pH, the absorption peaks of the UV-Vis of the solution after the reaction are significantly different. With the decrease of pH, the absorption peak of the solution after the reaction changes significantly and accompanied by the appearance of new absorption peaks, such as absorption peaks at 451, 557, 608, 632, and 657 nm. And similar to the trend reported by Zaied et al., the MB removal process will produce intermediate products such as Azure B, Azure A, Azure C, and Thionin [28], and the absorption peak gradually shifts to the left. Therefore, it can be inferred that the oxidation-reduction reaction of MB occurred during the removal of δ-MnO2, and as the pH of the reaction system gradually decreased, the oxidation capacity of δ-MnO2 gradually increased, and the degradation degree of MB also increased, and intermediate products were formed. The maximum absorption peak also gradually shifted to the left.
By detecting the change of Mn2+ in the solution after the reaction, it can also be proved that the process of removing MB by δ-MnO2 has undergone an oxidation-reduction reaction. As shown in Figure 5, at pH 2.00 and 4.01, δ-MnO2 is accompanied by the release of Mn2+ in the process of removing MB. At pH 2.00, the release of Mn2+ reaches 13.3 mmol/L. When the pH was 4.01, the release of Mn2+ was only 0.76 mmol/L, and the lower the pH, the greater the release of Mn2+. When the pH was 5.92 and 8.05, Mn2+ was not detected in the solution after the reaction. However, it can be seen from Figure 3 that the redox reaction still occurs at pH 5.92 and 8.05. It can be inferred that the negative charge on the surface of δ-MnO2 will increase with the pH under weakly acidic, neutral, and alkaline conditions. However, the corresponding adsorption sites that can adsorb positively charged Mn2+ increase accordingly; that is, the Mn2+ released during the reaction is readsorbed to the surface of δ-MnO2, so Mn2+ is not detected in the solution.
4.2. The Mechanism of Adsorption
By comparing the changes of the FTIR spectra of δ-MnO2 before and after the reaction (Figure 6), it was explored whether δ-MnO2 had an adsorption and removal effect on organic matter in the solution. As shown in Figure 6, for the FTIR spectrum of the reacted δ-MnO2, compared with the δ-MnO2 that has not reacted with MB, a new chemical bond vibration is obviously detected. The absorption bands of δ-MnO2 before the reaction are located at 1619, 920, 449, and 512 cm-1, of which 449 and 512 cm-1 are the characteristic absorption of δ-MnO2, respectively. The lattice vibration of the layered Mn-O bond has a slight blue shift [29]; 920 cm-1 is the bending vibration of manganese-oxygen octahedron-OH [30], and 1619 cm-1 is the bending vibration of H2O. After reacting with MB, δ-MnO2 still retains the characteristic peak of δ-MnO2. The layered structure of Mn-O bond lattice vibrates, but the bending vibration of the manganese oxide octahedral cavity -OH at 920 cm-1 disappears. At the same time, many new chemical bond vibrations have appeared. For example, 1395 cm-1 was assigned to the stretching vibrations of CN bonds in the heterocycle of MB; the band at about 1491 cm-1 was ascribed to the CH2 deformation vibration. In addition, the wave bands at 1252 and 1224 cm-1 are due to the Ar-N deformation vibration. For the tensile vibration of C=S and CS, the wave band is about 1142 and 887 cm-1. In the band of 820 cm-1, new chemical bonds appear, which are attributed to the rocking vibration of C-H in the benzene ring of MB [31]. In addition, no bending vibration of -OH at 920 cm-1 was detected on the surface of δ-MnO2 after reacting with MB. It can be presumed that the adsorbed material occupied the -OH site of δ-MnO2 and disappeared. It can be seen from Figure 4 that δ-MnO2 is accompanied by the production of intermediate products in the process of removing MB, and the structure of the intermediate product is similar to that of MB. This can prove that the surface of δ-MnO2 adsorbs MB or organic matter similar to MB.
It can also be found from Figure 6 that the adsorbed substances on δ-MnO2 increase with the increase of pH, which is contrary to the result that the adsorption removal rate of δ-MnO2 on MB gradually increases with the increase of pH of the reaction system in Figure 3. Therefore, XPS was used to further quantitatively analyze the content of adsorbed substances on δ-MnO2.
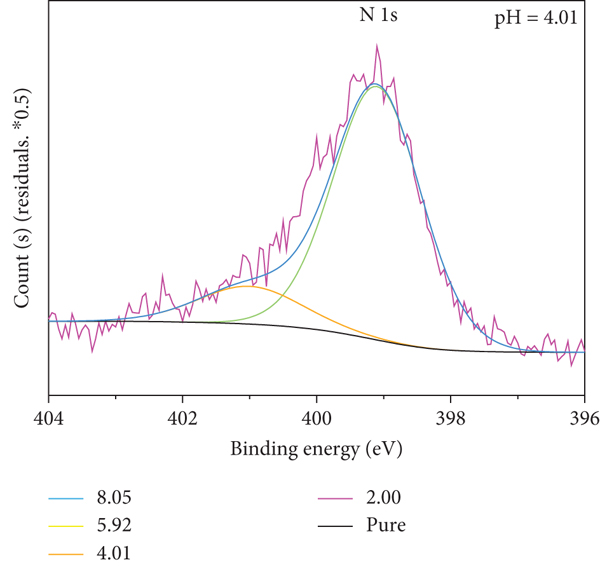
The N element is a characteristic element in the molecular structure of MB and an element that does not exist in the δ-MnO2 structure; for the N element, the δ-MnO2 before and after the reaction is analyzed by XPS (X-ray photoelectron spectroscopy); as shown in Figure 7, XPS analysis found that N element was detected on the reacted δ-MnO2, indicating that the δ-MnO2 had adsorbed the material with N element, and the peak fitting of the N 1s peak of the mineral powder after the reaction was performed (N atoms are not directly bonded to the surface of δ-MnO2, and the binding energy of N 1s is 399.2 eV; N atoms are directly bonded to the surface of δ-MnO2, and the binding energy of N 1s is 401.0 eV) [32].
The results are shown in Figure 7(a) and Table 3. As the pH increases, the N content on δ-MnO2 gradually decreases. Under the conditions of pH 2.00, 4.01, 5.92, and 8.05, the ratio of N directly adsorbed on δ-MnO2 to N not directly adsorbed on δ-MnO2 is roughly 1 : 2. It is known that both the structure of MB and its intermediate product contain 3 nitrogen atoms, and the hole -OH on the manganese oxygen octahedron of δ-MnO2 has disappeared. It can be speculated that one N atom in the molecule of the adsorbed substance is adsorbed on the -OH site on the δ-MnO2 hole, while the remaining two N atoms in the molecule do not directly contact the δ-MnO2. Therefore, the δ-MnO2 decolorization MB has an adsorption decolorization process, and the content of adsorbed substances gradually decreases with the increase of pH.
The best fit of N 1s XPS spectra for MB adsorption.
4.3. Removal Mechanism
Based on the above results, it is reasonable to speculate that the removal process of MB by δ-MnO2 is as follows: Under low pH conditions, the δ-MnO2 with a small amount of positive charge on the surface has a high redox potential, so it is strongly oxidizing, and the positively charged MB will be adsorbed on its surface, and then, oxidation reaction occurs. With the consumption of H+ and the release of Mn2+, part of MB is completely degraded into CO2, H2O, and other inorganic substances, and part of it has not been completely degraded. It is accompanied by the generation of intermediate products, including MB parent ion and an MB-analogous compound in which one methyl group is replaced by a proton, Azure B, Azure A, Azure C, Thionin, and other substances.
As the pH of the reaction system increases, the oxidation-reduction potential of δ-MnO2 decreases [17, 33]; the oxidation removal rate of MB decreases significantly with the increase of pH. After the reaction, the UV-Vis band scanning spectrum of the solution can confirm that the oxidation degree of MB gradually decreases with the increase of the pH value. The oxidation of MB by δ-MnO2 is a process that consumes H+, is accompanied by the conversion of high-valence manganese to Mn2+, and is accompanied by the formation of intermediate products. When the initial pH is 2.00, the oxidation removal rate of δ-MnO2 to MB is as high as 98.85%. The more H+ consumed and Mn2+ released, the amount of H+ consumed is less than the original system.
As the initial pH increases to 4.01 and 5.53, the oxidation removal rate of MB decreases to 74.49% and 47.43%, and the H+ consumption and Mn2+ release in the reaction system gradually decrease. Then, when the initial pH is 7.35, the surface of δ-MnO2 has a large number of negative charges, and the added positively charged dye MB quickly replaces the H+ in the hydroxyl group on δ-MnO2, H+ is released into the solution, so that the H+ in the system increases rapidly, and the pH shows a downward trend. As the oxidation dye reaction progresses, after H+ is gradually consumed, the pH gradually increases, due to the change of pH 7.35 to H+. As the pH of the system increases, the surface of δ-MnO2 becomes electronegative. Through the interaction between charges, Mn2+ can be adsorbed to the surface of δ-MnO2, causing the released Mn2+ to be readsorbed on the surface of minerals [18, 34], so at high pH, Mn2+ is not detected in the system.
The results of FTIR (Figure 6) can prove the adsorption effect of δ-MnO2 on MB. In the experiment, the concentration of the dye after the reaction is measured by using the UV spectrophotometer to measure the absorbance at 664 nm to calculate the concentration of MB. The experiment measured the adsorption and removal of MB by δ-MnO2 as the pH increases. The rate gradually increases, but XPS (Figure 7) shows that under low pH conditions, the number of adsorbed substances is greater. From this, it can be inferred that the adsorbed substances of δ-MnO2 are not only MB. It can be judged by the results of UV-Vis band scanning that MB produced certain intermediate products during the decolorization process, namely, Azure A, Azure B, Azure C, and Thionin. These four intermediate products are obtained by the gradual demethylation of MB. The heterocyclic ring and benzene ring in the MB structure are still undamaged, so the adsorbed substances detected by FTIR and XPS may also include intermediate products in the reaction process, and the molecular weight of these oxidized intermediate products is less than that of MB, which is easier adsorbed on the surface of δ-MnO2. It can be inferred that as the pH of the reaction system increases, the amount of MB adsorbed on the surface of δ-MnO2 gradually increases, but the adsorbed intermediate product decreases.
5. Conclusions
The removal of MB by δ-MnO2 has both oxidation and adsorption mechanisms, and the change of pH significantly affects the oxidation and adsorption of MB by δ-MnO2. We believe that the removal of MB by δ-MnO2 should be divided into two stages: first adsorption and then oxidation. First of all, MB molecules migrate to the vicinity of the mineral surface and then diffuse to the surface of δ-MnO2 through the boundary layer, and adsorbed on the hydroxyl active sites on the surface of δ-MnO2 to replace the H+ in the hydroxyl group, and continue to diffuse into the micropores inside the adsorbent. Secondly, the adsorbed MB reacts with the high-valence manganese in δ-MnO2, and the high-valence manganese (Mn4+/Mn3+) is reduced to Mn2+. Part of Mn2+ is adsorbed by manganese oxide again due to the Coulomb effect. After the adsorption is saturated, the remaining Mn2+ is free in the solution. At the same time, the MB oxidation reaction produces organic intermediate products and some inorganic compounds, and some intermediate products after the MB reaction can also be adsorbed on the surface of δ-MnO2. Therefore, the research results show that δ-MnO2 is a combined effect of oxidation and adsorption on the removal of MB.
Footnotes
Data Availability
The data used to support the findings of this study are included within the article.
Conflicts of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Acknowledgments
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work is financially supported by the National Natural Science Foundation of China (41867004) and Scientific Research Project of Jiangxi Provincial Education Department (GJJ160408).
