Abstract
In this study, fly ash (FA) was modified by sodium hydroxide to prepare a new adsorption material (IP) and treat Pb2+- and Cd2+-polluted wastewater. The effect of preparation parameters (mass ratio of FA/NaOH and modification temperature) on IP adsorption performance was investigated. The results indicated that the IP4 showed the highest adsorption capacity prepared at the FA/NaOH mass ratio of 1 : 2 and the roasting temperature of 250°C. The IP4 was characterized by SEM, EDX, XRD, and FTIR analyses. The results showed that the surface morphology and microstructure of FA were significantly changed. Furthermore, in order to study the adsorption performance of Pb2+ and Cd2+ on IP4, the different initial concentrations of Pb2+ and Cd2+, pH, and contact time were analyzed, and the results indicated that IP4 has excellent adsorption capacity for heavy metals. In addition, kinetic model results demonstrated that the adsorption behavior of Pb2+ and Cd2+ on IP4 was better described by a pseudo-second-order model.
1. Introduction
Among water resource problems, heavy metal ions have been considered the most serious pollutants [1], which can lead to tremendous damage to the ecosystem [2]. Lead (Pb) and cadmium (Cd) are considered the most dangerous heavy metals as they can induce some serious health problems [3]. Heavy metals accumulate in the organs of the human body through the food chain and further cause serious harm to human health [4].
Many technologies were introduced to treatment metal ions in wastewater, such as ion exchange [5], membrane separation [6], solvent extraction [7], and evaporation and chemical precipitation [8]. However, these technologies have limited applications due to their high cost and low efficiency [9]. Among these wastewater treatment methods polluted with heavy metals, the adsorption method was an effective and feasible method because of its low cost, simple operation, and high efficiency [10–12]. The key of adsorption technology is to prepare economical adsorbents with a large specific surface area and high adsorption efficiency. For example, activated carbon is used as heavy metal adsorbents, which presented excellent adsorption behavior due to its many macro- and micropores and large specific surface area [13]. However, those adsorbents perform escalating costs. In addition, there were many micropores on the activated carbon, which results in slow diffusion kinetics; thus, it is limited in wastewater treatment [14, 15]. Thereby, it is imperative to develop new economical and efficient adsorbents, such as modified fly ash.
Coal fly ash (FA), a secondary product of coal burning in thermal power plants, is one of the most accessible and abundant anthropogenic materials [16]. The results of previous studies demonstrated that alkali-modified FA was an effective method in the adsorption and remediation of heavy metals (HMs) in water and soils [17, 18]. Besides, FA is rich in silicon, which can improve the HM removal with NaOH addition [19, 20]. However, there are few reports on adsorption of HMs for modified FA by low-temperature roasting assisted with sodium hydroxide. Therefore, the purpose of this study is to use a low-temperature roasting method to modify FA with NaOH as an activator and use modified fly ash to remove metal ions in wastewater compared to fly ash.
This study is aimed at investigating the possibility of using alkali-modified coal fly ash to remove Pb and Cd. And the following key objectives were determined: (i) determine the best modification conditions of fly ash; (ii) characterize the modified fly ash with the best adsorption performance; (iii) study the influence of initial pH, temperature, absorbing time, and initial concentrations of Cd2+ and Pb2+; and (iv) analyze the kinetic models.
2. Methods and Materials
2.1. Materials
Fly ash, used in the batch adsorption experiments, was collected from the Datong Power Plant, Shanxi Province. Chemical reagents used in this study include HCl, NaOH, Cd(NO3)2·4H2O, and Pb(NO3)2, and the above reagents were of analytical grade (GB/T 601-2016, Jinhuitaiya, China).
2.2. Preparation of IP
FA was dried in a constant temperature drying oven at 85°C. Then, the material was mixed with NaOH in 1 : 1 and 1 : 2 proportions and the mixture was put in the nickel crucible, respectively, and then the nickel crucible was put in the muffle furnace and fired at 200°C for 2 h. Finally, the samples were cooled to ambient temperature, and the pH of adsorbents was washed to neutral. The adsorbents prepared under the 1 : 1 and 1 : 2 proportions of FA : NaOH were marked IP1 and IP2, respectively.
Then, dried FA was mixed with NaOH in 1 : 2 proportion and the mixture was put in the nickel crucible, respectively. Then, the nickel crucible was put in the muffle furnace and fired at 150 and 250°C for 2 h, respectively. Finally, the samples were cooled to ambient temperature, and the pH of samples was washed to neutral. The adsorbents prepared at the roasting conditions of 150 and 250°C were marked IP3 and IP4, respectively.
Initial pH of the solution was determined by a Mettler Toledo digital pH meter (calibrated using buffers of pH 4.01, 7.00, and 9.21), and FA and IP4 were analyzed through energy-dispersive X-ray (EDX), X-ray diffraction (XRD, D8 Advance), Fourier transform infrared (FTIR) spectroscopy (Nicolet iS10, Thermo Fisher Scientific), and scanning electron microscopy (SEM, S-4800, Hitachi Limited).
2.3. Adsorption Trial
The influence of different adsorption parameters on the adsorption of two metal ions was analyzed by batch adsorption experiments. Heavy metal-polluted wastewater was obtained using Cd(NO3)2·4H2O and Pb(NO3)2. Dilute the 1000 mg/L Pb2+ and Cd2+ solution to 100 mg/L; then, 50 mL Pb2+ and Cd2+ solutions (the initial concentrations of two metal ions are both 100 mg/L) were put in 250 mL Erlenmeyer flasks and mixed with 0.1 g of IP1, IP2, IP3, and IP4, respectively. Then, Erlenmeyer flasks were carried out on a shaker at 180 rpm and continuously stirred for 60 min at 25°C. After adsorption, the residual concentration of Cd2+ and Pb2+ in the filtrate was determined through an atomic absorption spectrometer (PinAAcle 900, PerkinElmer). The material with the best adsorption performance was selected by comparing the adsorption capacity. The basic physicochemical properties of materials are shown in Table 1.
Basic physicochemical properties of materials.
The removal efficiency
3. Results and Discussions
3.1. Effect of Preparation Parameters on IP Adsorption Capacity
3.1.1. Effect of the FA/NaOH Mass Ratio
Figure 1 displays the effect of different FA/NaOH mass ratios on the adsorption of Pb2+ and Cd2+. Compared with IP1 and IP2, FA had the lowest adsorption capacity for Cd2+, which was only 27.59 mg/g, while the adsorption capacity for Pb2+ was 20% higher than that for Cd2+, indicating that the original fly ash had a certain adsorption capacity for Pb2+ and Cd2+. As can be seen, the adsorption amount of IP on both Pb2+ and Cd2+ was increased compared with that of fly ash, while the adsorption capacity of IP2 prepared at the FA/NaOH mass ratio of 1 : 2 is stronger, possibly because the OH- can easily combine with heavy metal ions to form a more stable salt complex under alkaline conditions. It is precisely because of the participation of NaOH that the grain size of some minerals in the raw ash gradually decreases. The silicon and aluminum oxides can fully react with NaOH and play a good activation effect on the fly ash. Compared with the raw ash and IP1, IP2 has a better adsorption effect. Therefore, in the following preexperiment, the gray-base ratio of 1 : 2 will have a better effect.

Adsorption of lead and cadmium by different fly ash-alkali ratios of modified materials.
3.1.2. Influence of Calcination Temperature
Figure 2 shows the adsorption amount and removal rate of two metal ions on FA and IPs prepared at different temperatures. From Figure 2(a), the adsorption capacity of two metal ions is higher on the three modified materials than FA, and the removal rate and adsorption capacity of adsorbents for Pb2+ and Cd2+ followed IP4>IP3>IP2>FA. The results indicated that increasing the modification temperature is conducive to the adsorption of the modified materials, which may be due to the reaction between the original fly ash and the NaOH being more adequate at 250°C. For IP4, the adsorption capacities of Pb2+ and Cd2+ were 59.51 and 45.91 mg/g, respectively, when the modification temperature is raised to 250°C, which are 13.5% and 23.48% higher than that for IP2, respectively. As shown in Figure 2(b), the removal rate of Pb2+ reached 97.5% when the temperature rose to 250°C, while the removal rate of Cd2+ was 78.21% under this condition, which was significantly higher than that of raw ash and IP3.

Adsorption amount of lead and cadmium (a) and removal efficiency of modified materials at different temperatures (b).
3.2. Characteristics of Adsorbents
3.2.1. SEM-EDX Analysis
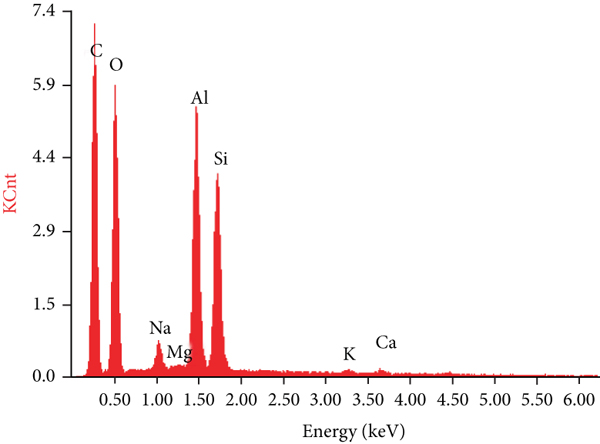
The SEM-EDX images of FA and IP4 are displayed in Figure 3. As we can see from Figure 3(a), FA was characterized by a spherical structure with a smooth surface. However, after the modification, many fine particles appeared on the surface of the sample, IP4 had an irregularly shaped and amorphous particle structure, and the surface of the adsorbent becomes loose and porous. The results may be due to the fact that both SiO2 and Al2O3 were dissolved from the surface of FA with the NaOH treatment; the vitreous structure on the FA surface was also destroyed, which is beneficial to increase the specific surface area (BET) of the sample [21] and further improve the adsorption ability of metal ions. The BET and pore volume of the IP4 were 26.37 m2/g and 0.0017 cm3/g, respectively, and its surface area was nearly 18 times larger than that of FA. Larger BET is more conducive to the improvement of adsorption capacity. This is mainly due to the rougher surface of IP4 with more vacancies for adsorption of heavy metals in case of producing more high-energy active sites on the surface. For the alkali roasting method making Ca and Al2O3 leach, its new chemical composition and surface structure got exposed, and the ability of being a heavy metal adsorbent is enhanced. While the size of particles got smaller, IP4 then had more extensive contact with heavy metal ions, which enhances the adsorption performance as well. Visa [22] reported a similar observation. As we can see from Figures 3(c) and 3(d), the main elements in the FA were Al, Si, and O; after modification, Al, Si, and O were also the main elements of the IP4.

The SEM-EDX diagram of FA (a, c) and IP4 (b, d).


3.2.2. FTIR Analysis
The FTIR spectrum of FA and IP4 is performed in Figure 4. Four obvious intense and wide adsorption bands were found, which were located at 410~500 cm-1, 530~570 cm-1, 800~910 cm-1, and 950~1200 cm-1, respectively. The adsorption peak that appeared at 457.13 cm-1 was ascribed to bending vibrations of T-O (

Infrared light map of FA and IP4.
3.2.3. XRD Analysis
Figure 5 presents the XRD spectra for the FA and IP4, which suggested that the position of diffraction peaks was similar to that reported by Chen et al. [28]. The XRD results indicated that the mullite and quartz were the main components of FA, and the diffraction peak of mullite was mostly at around

The XRD pattern of FA and IP4.
The excellent performance of IP4 is mainly due to containing Na2O and OH-, both of which dissociate the Si-O and Al-O peaks in the network structure with a higher degree of polymerization in FA; new unsaturated active bonds were constructed [21]. Under the action of the low-temperature roasting method with NaOH, the crystal structure in FA not only was destroyed but also led to the dissociation reaction of Al2O3, SiO2, etc. To form active negatively charged groups, the acidic ions on the surface of FA (H+) will also be dissociated under alkaline conditions. In addition, the surface of the modified material is electronegative, which enhances the adsorption performance.
3.3. Influence of Adsorption Time
The influence of contact time on the removal efficiency of Pb2+ and Cd2+ is shown in Figure 6. As we can see from Figure 6, the removal rate of two metal ions was increased rapidly in the initial adsorption period of 0~30 min, and the adsorption equilibrium was basically reached at around 30 min; after that, with the extension of time, the removal efficiency increased only slightly. When the adsorption time reached 120 min, the removal rate of Pb2+ and Cd2+ was 82.71% and 74.37%, respectively. This phenomenon may be due to the fact that there were many adsorption sites on the IP4 surface at the first period of adsorption; thus, the adsorption rate was fast [29]. With the reaction of adsorption sites and metal ions, available adsorption sites on the adsorbent surface were occupied, and the removal rate no longer changes significantly [30, 31].

The influence of adsorption time on the removal rate of Pb2+ (a) and Cd2+ (b). Adsorption conditions: IP4 dosage: 0.05 g; adsorption temperature: 25°C; agitation speed: 180 rpm; initial Cd2+ concentration: 100 mg/L; initial Pb2+ concentration: 150 mg/L; and adsorption time: 0-120 min.
The pseudo-first-order and pseudo-second-order models (Equations (3) and (4), respectively) were introduced to analyze the rate-controlling mechanism of the adsorption process [32].
The fitting results of adsorption kinetics are listed in Table 2. Obviously, the pseudo-second-order adsorption model showed higher correlation coefficients (
Kinetic model fitting parameters for Cd2+ and Pb2+ adsorption.
Comparison of previously reported Cd2+ and Pb2+ adsorption capacities of different adsorbents.
3.4. Influence of pH on Adsorption
The pH is a critical factor that influences the metal ion removal on the adsorbent. The effect of initial pH on the removal rate and adsorption amount is displayed in Figure 7. As we can see from Figures 7(a) and 7(b), the removal rate and adsorption capacity of Pb2+ and Cd2+ on IP4 significantly increased with the increasing pH from 2 to 6 and reached a plateau region at

The effect of pH on the removal rate and adsorption capacity of Cd2+ (a) and Pb2+ (b). Adsorption conditions: IP4 dosage: 0.05 g; adsorption time: 60 min; adsorption temperature: 25°C; agitation speed: 180 rpm; initial concentration of Cd2+: 100 mg/L; initial concentration of Pb2+: 150 mg/L; and solution pH: 2-8.

3.5. Effect of Initial Concentration
Figure 8 shows the influence of the initial concentration of Pb2+ and Cd2+ on the removal efficiency and adsorption capacity. As we can see from Figure 8, with the increase of the initial concentration of Pb2+ and Cd2+, the adsorption percentage of the two metal ions decreased. When the initial concentration of Pb2+ increased from 50 to 250 mg/L and that of Cd2+ increased from 25 to 200 mg/L, the adsorption efficiency of Pb2+ and Cd2+ decreased from 89.47% to 63.49% and 95.23% to 48.72%, respectively. This may be attributed to the fact that in the case of a fixed dosage, there were a large number of adsorption sites on the IP4 surface when the initial concentration of metal ions was low, and metal ions can fully react with the adsorption sites. Therefore, the removal rate is higher [41]. The adsorption sites were decreased with the increase of the concentration of two metal ions, and the adsorption rate is reduced. In addition, the adsorption amounts of two metal ions increased with the increase of the initial concentration. Besides, Ca2+ and Mg2+ contained in IP4 can be ion-exchanged with Cd2+, and the Si-O functional group on the surface of the material can also have a surface complexation reaction with metal ions [28].

The influence of the initial concentration of heavy metals on the removal rate and adsorption amount of Pb2+ (a) and Cd2+ (b). Adsorption conditions: IP4 dosage: 0.05 g; adsorption time: 60 min; adsorption temperature: 25°C; agitation speed: 180 rpm; initial Cd2+ concentration: 25-200 mg/L; and initial Pb2+ concentration: 50-250 mg/L.

4. Conclusion
In this study, the mass ratio of FA/NaOH and the calcination temperature play a key role in the modification of FA. After low-temperature calcination modification of NaOH, the new silicon-aluminum composite material IP obtains higher Pb2+ and Cd2+ adsorption capacity. Among them, IP4 (the FA/NaOH mass ratio was 1 : 2; the calcination temperature was 250°C) can be favored for heavy metal adsorption, compared with the other materials. The BET and pore volume of IP4 are increased, and the specific surface area is 18 times that of the original FA. The results of the characterization of IP4 show that IP4 mainly contains Al, Si, O, and Na elements. SiO2 and Al2O3 on the surface of the FA are attacked, the glass phase on the surface is dissolved and destroyed, and then many new adsorption sites remain. The adsorption of two metal ions by IP4 is affected by initial concentration, solution pH, and reaction time. In addition, adsorption results indicated that when the dosage was 0.05 g, adsorption temperature was 25°C, and the initial concentration of Pb2+ and Cd2+ was 150 and 100 mg/L, respectively; the adsorption reached adsorption equilibrium at 30 min, and the removal efficiency of Pb2+ and Cd2+ was 74.37 and 82.71%, respectively. The adsorption capacity of IP4 for two metal ions followed Pb2+>Cd2+. The results of adsorption kinetic models demonstrated that the adsorption process of Pb2+ and Cd2+ by IP4 can be well described by a pseudo-second-order model and the adsorption process was chemical adsorption based on ion exchange.
Footnotes
Data Availability
The basic mapping data used to support the findings of this study are available from the corresponding author upon request.
Additional Points
Highlights. (i) A new adsorbent has been successfully synthesized from fly ash. (ii) IP presents excellent adsorption capacity for heavy metals. (iii) Both the mass ratio and the temperature significantly affect the IP preparation. (iv) The adsorption efficiency of Pb2+ and Cd2+ was 82.71% and 74.37%, respectively.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Acknowledgments
This work was supported by the Key Laboratory of Original Agro-environmental Pollution Prevention and Control, Ministry of Agriculture/Tianjin Key Laboratory of Agro-environment and Safe-product Open Fund Project (Grant No. 17nybcdgj-2).
