Abstract
Although several approaches have been explored for the removal of dyes and other toxic materials from water as well as the entire environment, notwithstanding, researchers/scientists are still pursuing novel, low-cost, and eco-friendly biosorbents for the effective removal of such contaminants. Herein, clove leaves (CL) were utilized as a biosorbent for the sequestration of malachite green (MG) from a water-soluble solution. The CL was subsequently activated using potassium hydroxide (KOH) and characterized using the FTIR and FESEM to determine the functional groups on the activated clove leaves (CL-KOH) and the morphology of the adsorbent. The adsorption of MG was observed to be relatively dependent on the dosage of sorbent utilized, initial MG concentration, and sorption process contact time. The adsorption process of MG to CL was ideally described using the Dubinin–Radushkevich and Elovich models with the determination of maximum sorption capacity of approximately 131.6 mg·g-1. Furthermore, the thermodynamic parameters calculated showed that the adsorption of MG to the adsorbent was exothermic with the process involving physical sorption as well as chemical sorption processes with negligible adsorption energy. In conclusion, the study has revealed that the CL is a cost-effective biosorbent with high adsorption efficiency for the sequestration of MG from a water-soluble solution and can be recycled for further usage.
1. Introduction
The obliteration of the quality of water, soil, and air due to the fast-growing global population and industrialization is critical for life sustenance on earth. Water quality has exceptionally declined owing to the untreated effluents discharged into streams, oceans, and lakes, by the processes of mining, textile, paint and dye productions, and metal plating [1]. Among these noxious and aromatic contaminants released into water bodies devoid of the preceding appropriate treatment, the textile industry is a primary user of synthetic or noxious dyes for product colouring [2].
Dye is a major source of serious contamination owing to fast industrial development and extensive utilization [3]. Major consideration has been drawn by pollution of water by dye discharge from paper production, food processing, textile dyeing, paints, and cosmetic industries owing to community health and ecosystem hazards. At present, the worldwide colourant market is about 32 billion United States Dollars (USD) and is projected to grow to 42 billion USD in 2021. Yearly, the total of synthetic dyes manufactured is estimated to be over 700,000 tons, and over 15% of these synthetic colourants are yearly discharged into water. Water polluted by these dyes is noxious, xenobiotic, and carcinogenic, and they are grouped according to their chromophore structure into the solvent, reactive, basic, vat, and direct dyes. Exclusively, 47% of synthetic dyes are decomposable [4].
MG is a cationic dye and is positively (+ve) charged in a water-soluble solution. It has a three-benzene ring structure compound as shown in Figure 1, with a molecular formula of C23H25CIN2 and a molecular weight of 364.917 g/mol [5, 6]. It is a synthetic triarylmethane dye and a natural compound utilized as a dyestuff and has appeared as a contentious agent in aquaculture as an ectoparasiticide and fungicide owing to its effectiveness and minimal cost. It is conventionally employed as a dye for materials like silk, wood, acrylic, paper, and leather. However, their intense colour causes a horrible colour to water bodies, and it is intractable to degrade by light and other oxidizing agents, heightening concerns for the environment and making its confiscation challenging. It is extremely noxious to animal cells, carcinogenic, and mutagenic and causes inflammation to the skin. Various health problems associated with the use of MG include a reduction in red blood cell (RBC) count (dyscrasia) and enhanced white blood cell count (leukocytosis), haemoglobin (anemia), and interruption of blood coagulation, eye blisters, wild breathing, and copious sweating. Also, associated with MG utilization is the impairment to the kidney, liver, spleen, and heart and infliction of lacerations on the skin, eyes, lungs, and bones which produces teratogenic impacts [1, 3, 5, 7]. Therefore, there is an urgent need for the adaptation of eco-friendly approaches for the treatment of MG effluents before their release into water bodies and streams to safeguard the ecosystem [1].
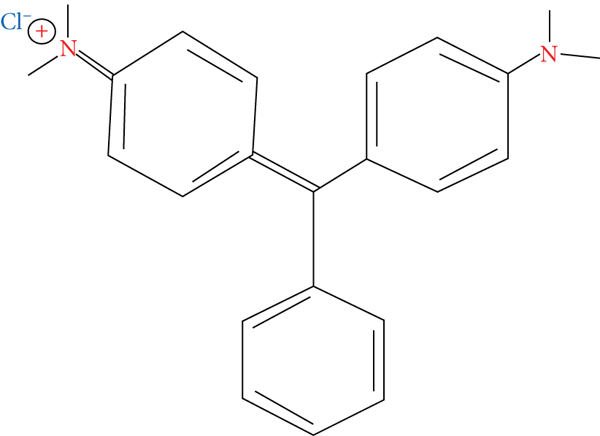
The molecular structure of MG was adopted and redrawn from [6].
Dye sequestration from water bodies has substantial importance to the ecosystem, and developing effective and cost-effective removal techniques is an ongoing and continuous effort. These methods comprise the adsorption of dye pollutants to mineral or carbon-based matrix [8], photocatalysis or oxidation processes [9], chemical precipitation [10], ion exchange [11], microbiological or enzymatic decomposition [12], coagulation [13], reverse osmosis [14], membrane filtration [15], biological treatment [16], etc.
Among these existing methods, adsorption is more efficient due to its effortlessness, operational ease, and the reality that it does not produce sludge, thus ensuring a safe environment [8]. The process of adsorption is a distinctive technique for the confiscation of contaminants from water-soluble solutions by putting them precisely on the surface of the adsorbent [17]. Adsorption methods are more cost-effective when compared to their substitutes if the adsorbent is local to the treatment area and needs no prior treatment before its application in the treatment of effluents. While various biosorbents such as sugarcane waste, neem sawdust, maize cob, Parthenium weed, jackfruit peel, freshwater algae, rice husks, fly ash, CL, and spent tea leaves have been employed for MG dye sequestration from aqueous solutions, the search for minimal cost waste materials for harmful dye removal from water-soluble solution before removal lingers. From a financial point of view, the utilization of these waste materials is crucial [5].
To confiscate various noxious metals, dyes, and contaminants, numerous nontraditional low-cost biosorbents from agriculture and forestry, plant biomass, and microorganisms are utilized. Agricultural leftover utilization as a biosorbent is due to the substantial benefit of easy application, minimal cost, no or low chemical utilization, and elevated efficiency [18, 19].
Cloves (
Supposedly, there are some existing studies on the removal of MG. For example, Reference [23] investigated the concurrent ultrasound-assisted adsorption of two dyes (MG and safranin O) from aqueous solutions onto copper nanowires laden on activated carbon (Cu-NWs-AC) with results revealing that 0.022 g of Cu-NWs-Ac led to the removal of 15 mg·L-1 of MG and 15 mg·L-1 of safranin within 6 min at pH of 5.5. Similarly, a study by [24] recently sequestered the
There are also other reported removal procedures of dyes such as crystal violet and a brilliant green from wastewater using various biosorbents such as live yeast
As reported in a recent review study by [31], there are several existing publications on the utilization of adsorbents for the elimination of dyes and heavy metals as well as other contaminants from wastewater and the environment at large. Notwithstanding, there is still a need for researchers/scientists to continue the pursuit of other novel and eco-friendly biosorbents for the effective removal of such contaminants. Under these recent situations, cheap and effective biosorbents are needed for the removal of dyes and other environmental contaminants. Hence, this present paper summarizes the exploration and application of biosorbent created from clove leaf (CL) agrowaste activated using potassium hydroxide (KOH) as a novel bioadsorbent with higher efficiency (removal capacity) as well as the cost-effectiveness trends for MG (Figure 2 shows an outline of the study).

The outline of the study.
From the research papers/publications reviewed, it is evident that there is a lack of adequate reports on utilizing/employing CL-KOH as a biosorbent for dye removal, particularly MG from water-soluble solutions; thus, this research will try to bridge the gap. Consequently, this work examines the feasibility of dye confiscation from water-soluble solution using CL synthesized at an elevated temperature and activated using KOH. The CL-KOH synthesized was characterized using various procedures such as Field Emission Scanning Electron Microscope (FESEM) and the Fourier Transform Infrared Spectroscopy (FTIR). Then, the adsorption studies were systematically examined by enhancing various adsorption parameters such as the adsorbent dosage, time of contact, and temperature as well as stirring rates. Finally, the results were fitted to isotherm, kinetic, and thermodynamic models.
2. Experimental
2.1. Materials and Methods
Analytical grade purified chemicals were used without further distillation. KOH and MG were purchased from UD, Sumber Ilmiah Persada, Surabaya, Indonesia.
2.2. Synthesis of Biosorbent
The CL (

Steps for the synthesis of CL-KOH biosorbent for this study.
2.3. Characterization of Biosorbent
The produced CL-KOH biosorbents were characterized utilizing the FESEM (Thermo Scientific Quattro S) to determine the particle size and morphology of the prepared biosorbent. While the FTIR spectra of the biosorbent were obtained using PerkinElmer UATR at a range of 400-4000 cm-1 wavenumber.
2.4. Preparation of Stock Solution of MG
A 1000 mg·L-1 of MG stock solution was produced by measuring 1 g of the adsorbate and dissolving it in DW (1000 mL) in a volumetric flask. To prepare appropriate initial MG concentrations of the adsorbate used for batch experiments, it was achieved by diluting the stock solution using DW.
2.5. Biosorption Batch Studies
The batch method was employed in this study to examine the uptake of MG from water-soluble solutions to CL-KOH. Determining the impact of the biosorbent dosage employed for the sequestration of MG was explored by contacting varying amounts of the biosorbent of 0.5-3.0 g/L at a constant temperature of 303.15 K, shaking speed of 200 rpm, initial MG concentration of 0.03 mg/L, pH value of 6, and contact time of 150 min. Determining the equilibrium concentration of MG after adsorption using the adsorbent was evaluated from a calibration curve using the GENESYS 10S UV/Vis spectrophotometer at a 617 nm wavelength. The removal efficiency of the CL was evaluated by computing the percentage removal using
To explore the effect of the contact time on the adsorption of MG to the biosorbent, 0.03 mg·L-1 of MG was treated with 0.015 g/L of the adsorbent at a temperature of 303.15 K, shaking speed of 200 rpm, and pH at 6 and under varying contact times of 0-160 min. The time-dependent quantity of the MG sorbed by a unit mass of CL at varying contact times was determined using
The impact of initial MG concentrations and the adsorption isotherm was determined by contacting 0.9 g/L of the adsorbent with varying initial MG concentrations of 0.005-0.05 mg/L at a temperature of 303.15 K, pH 6, sorption process time of contact of 150 min, and shaking speed of 200 rpm. The equilibrium sorption capacity (
2.6. Desorption and Regeneration
To clarify the nature of the adsorption process and assess the possibility to recover the sorbate and regenerate/recycle the biosorbent, desorption experiments were performed on the CL-KOH with preadsorbed MG dye using 0.1 M NaOH and HCI to treat MG of the 0.03 mg/L concentration dye for a time duration of one hour at a shaking speed of 200 rpm on a magnetic stirrer. After desorption, the biosorbents were isolated from the MG solution by centrifugation at 4000 rpm for 10 min, and the supernatant solution was analyzed using the UV-vis spectrophotometer. The percentage efficiency was determined using
3. Results and Discussions
3.1. Biosorbent Characterization
Figure 4 depicts the FTIR spectra of CL-KOH before and the adsorption of MG obtained at a wavenumber range of 400-4000 cm-1. The characteristic FTIR peaks appearing at 3333.43, 2866.21, 1686.53, 1594.05, 1456.36, 1316.37-1030.96, 1240.75, 912.14, and 433.13-535.95 cm-1 were due to hydroxyl stretching vibration, asymmetrical patterns of group CH2 of the alcoholic group in the essential oil, C=C stretching vibration of an aromatic moiety of eugenol, C-C aromatic ring vibrations of eugenol acetate, -C=C, C-O vibrations of ether and alcohol functional group of eugenol and eugenol acetate, C-O-C symmetric expansion of aromatic acid esters and C-OH vibrational stretching of phenolic compounds, bending vibration absorption of C-H groups, and C-C of the amine group (Figure 4(a)) [32–35]. After the adsorption of MG to the adsorption, there was a redshift of most of the functional groups with FTIR peak values of 3334.26, 2850.11, 1605,48, 1687.16, 1463.02, 1314.96-1031-03, 1268.15, 912.03, and 429.10-534.92 cm-1 (Figure 4(b)). The wavelength shift demonstrated that a dye-binding process took place at the surface of the biosorbent.

The FTIR spectra (a) prior to adsorption onto CL-KOH (b) following MG adsorption onto CL-KOH.

The FESEM images of the biosorbent prior to and subsequently after adsorption are shown in Figures 5(a) and 5(b). A porous, rough, and irregular surface morphology is observed. It was observed that there was no significant difference in the biosorbent surface form before and after the biosorption of MG. Images show clearly the action of biosorption on the surface morphology of the biosorbent, which showed a decreased pore size of the biosorbent owing to MG impregnation onto the surface of the adsorbent.

The SEM images of biosorbent (a) before adsorption of MG and (b) after adsorption of MG.

3.2. The Contact Time Effect on the Adsorption of MG to CL-KOH
The adsorption of MG to CL-KOH at various contact times is displayed in Figure 6. It was noticed that the sorption capacity was improved with an upsurge in the time of contact for the sorption process and that equilibrium was attained after 120 min for MG. At low MG concentrations, the sorbent surface area available to the ratio of the initial MG concentration was higher than that of the adsorbate molecules at low concentration, leading to improved adsorption capacity. Also, the high driving force of the mass of adsorbent transfer at high concentrations leads to an expansion in the sorption capacity as the initial MG concentration is enhanced [8, 19].

The contact time effect (condition:
3.3. The Impact of Dosage of Adsorbent Used on the Sorption of MG to CL-KOH
The sum of active sites accessible and ions exchanged for sorption varies with the number of adsorbent sites in the biosorption process [36]. It was noticed in Figure 7 that the percentage of MG removed onto the adsorbent increased with increasing adsorbent dosage. At a low adsorbent dosage, there were insufficient active sorption sites to inhabit by the adsorbate and while at a high adsorbent dosage, there were sufficient sorption sites for occupancy by the adsorbate. The increase in the percentage of MG removed was attributed to the enhanced amount of active sites accessible and the enhanced surface area. Above the dosage of 0.6 g/L that adsorbents used, no further remarkable change was observed in the removal percentage of MG and was ascribed to the overlapping of the active sites in the biosorbent. Also, with regard to the extent of adsorbate sorbed at equilibrium, the sorption capacity decreased with increasing adsorbent dosage due to the split in the concentration rise between the surface of the adsorbent and the solution [37]. The reduction in the sorption capacity with enhanced biosorbent dosage at a constant dye concentration and volume will lead to the unsaturation of a specific number of the active sites on the biosorbent. Also, the decrease of the sorption capacity may be associated with the particle aggregation resulting from increased biosorbent dosage. Such aggregation will result in reduced total surface area and improved diffusion path length to be travelled by the sorbate inside the biosorbent [38, 39].

The impact of dosage of adsorbent employed in this study (condition:
3.4. The Impact of Initial MG Concentration on the Adsorption Process
The effect of the initial MG concentration sorption onto the CL activated using KOH was examined by setting a time of adsorption contact of 150 min and pH value of 6, and the dosage of the adsorbent used was 0.09 g/L. With an intensification in the initial MG concentration, the sorption capacity was observed to be enhanced (Figure 8). The biosorbent sorption capacity improved from 7.675 to 67.102 mg·g-1 by increasing the initial MG concentration from 0.005 to 0.05 mg·L-1. This increase was ascribed to the high collision between the molecules of the adsorbate (MG) and the active adsorbent sites, hence improving the driving force of the adsorbate to the adsorbent surface [40].

The effect of initial concentration of MG (condition: the
3.5. Kinetic Studies
Various adsorption processes occur via a multistep mechanism which includes the dispersion across the liquid film bordering the particle of a solid (an external mass transfer coefficient controls this process), the dispersion within the individual particles assumes a mechanism of pore diffusion (intraparticle diffusion), and the chemical and physical sorption occurs at a sorption site. The transitory behaviour of a batch sorption process at various temperatures and initial adsorbate concentrations is generally studied using the Lagergren first-order (also called pseudo-first-order, PFO) and the pseudo-second-order (PSO) models [41]. Kinetic research is generally employed by several scientists to examine the mechanism of the sorption process taking place. Preexisting models are applied in most sorption kinetic research, and this acts as a guide in demonstrating the mechanism of sorption. An extensive range of standard models exists to rationalize the sorption at various requirements, and the most appropriate is selected for the provided experimental terms. The linearized forms and calculated parameters of each kinetic model are presented in Table 1 [42]. The various linear plots of the individual kinetic model are displayed in Figure 9. From each model plot, the rate constants and the calculated correlation coefficients (
The sorption kinetic models employed for this study.
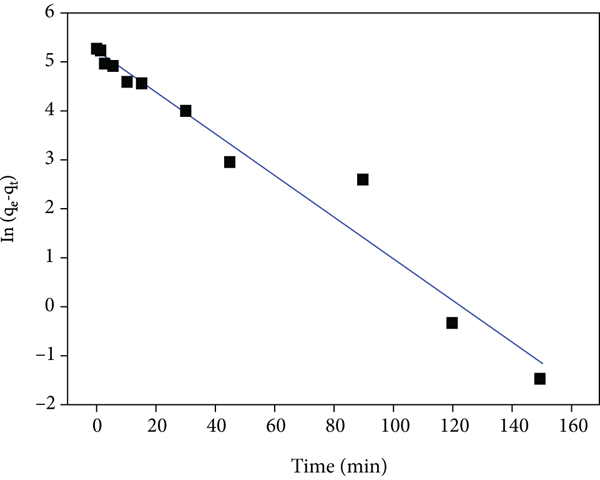
The plots of (a) PFO, (b) PSO (type 1), (c) intraparticle diffusion, and (d) Elovich and PSO (type 4) models.
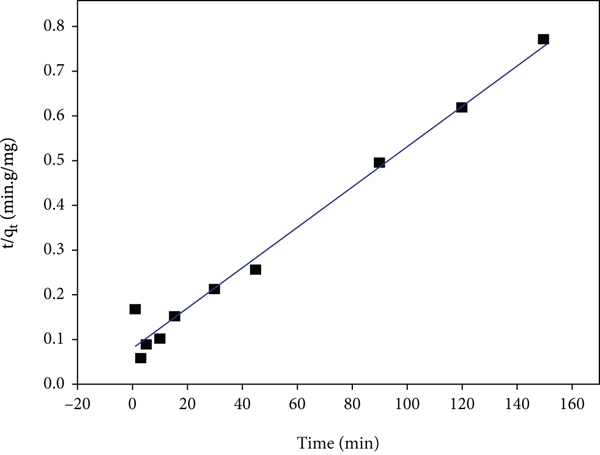

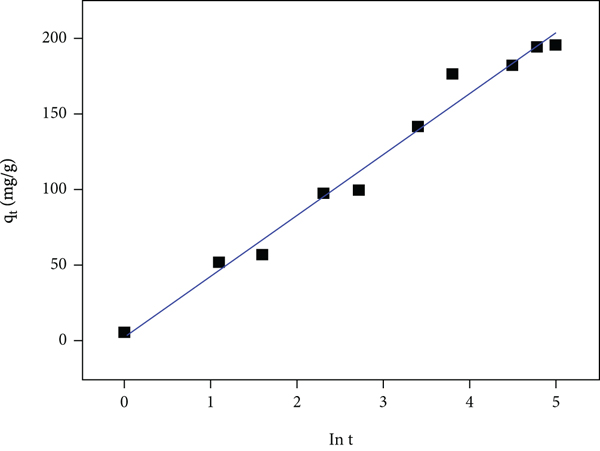

The least-square technique was utilized for finding the parameters for kinetic models. The fourth form of the linearized forms of the PSO model simple linear regression could result in various parameter estimates as observed from the fourth form of the PSO model [43]. Results using this type 4 PSO model are shown in Table 1. It was observed from Figure 9(e) that the data substantially deviated from linearity. Hence, the PSO model could not intensively describe the trend of the experimental data, which was confirmed by the low value of the coefficient of correlation (
3.6. Isotherm Studies
The association between the solute equilibrium concentration in the adsorbent and the solute equilibrium concentration in the solution at a steady temperature is defined by the isotherm [34]. Selecting the ideal model of isotherm for the process of sorption, the resulting conditions must be satisfied: (i) the model of the isotherm and the determined equilibrium data must have a superlative fit, (ii) the isotherm function must be thermodynamically accurate, and (iii) in ideal situations, the systematic calculation of concentration is from the capacity and the other way around [42]. The sorption equilibrium data in this study was assessed using the LNR, Freundlich (FRH), Temkin (TMN), and D-B isotherm models.
The earliest far-famed equation which described the sorption technique was the FRH model. It is linked with an empirical equation and employed for a nonideal natural process that comprises heterogeneous surface sorption. The FRH model’s expression is given in Table 2, where
The sorption isotherm models employed for this study.
The TMN model predicts that the energy of sorption is reduced linearly with the surface coverage owing to the interaction between the adsorbent and adsorbate. Sorption is considered by the bonding energies up to the optimum binding energy regular distribution [46], while the D-R model predicts that the sorption is multilayered and comprises the van der Waals forces which are related to the processes of physical sorption [47]. The linearized forms of the models are illustrated in Table 2.
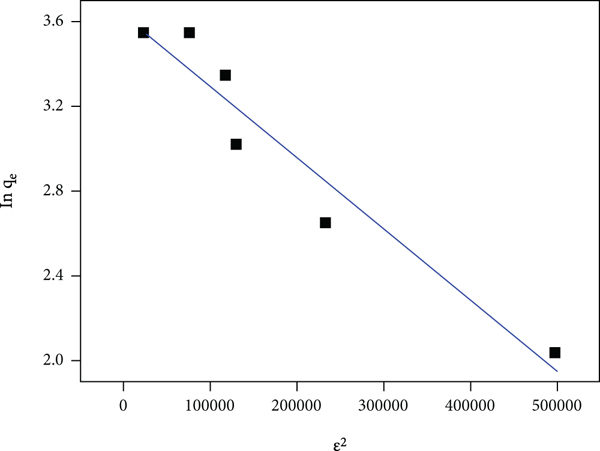
The various plots for each isotherm using their linearized forms are shown in Figure 10, and based on their correlation coefficient values for all models, the highest calculated correlation coefficient value was for the D-R model, which was 0.9332 when compared to other models (LNR -0.1567, FRH -0.8117, and TMN -0.8318). The magnitude of

The sorption of MG to CL-KOH using (a) LNR, (b) FRH, (c) TMN, and (d) D-R isotherm models.


The comparison of sorption capacity of CL-KOH with other described/reported biosorbents on MG.
3.7. Thermodynamic Studies
The assessment of the three key thermodynamic parameters (change in enthalpy (
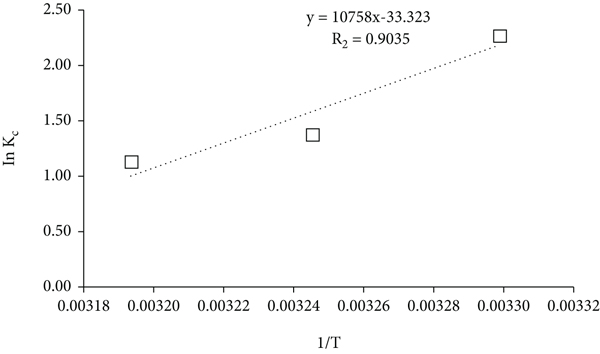
The plot of
The thermodynamic parameters for this study.
3.8. Desorption and Regeneration
Extreme desorption efficiency and excellent reusability after sorption are required for a sorbent; hence, the desorption studies are essential to describe the nature of sorption and evaluate the spent sorbent and dye recycling [60]. The desorption and regeneration processes of the spent biosorbent using 0.1 M of NaOH and HCl are shown in Figure 12. It was observed that 0.1 M NaOH was more effective in the regeneration of the biosorbent when compared to 0.1 M HCl. Three cycles of adsorption-desorption were carried with a maximum desorption efficiency of 94% and a minimum desorption efficiency of 80% observed in the first and last cycles. Hence, the CL-KOH biosorbent showed excellent sorption performance and regeneration, and its utilization can be extended to environmental applications for effluent treatment.
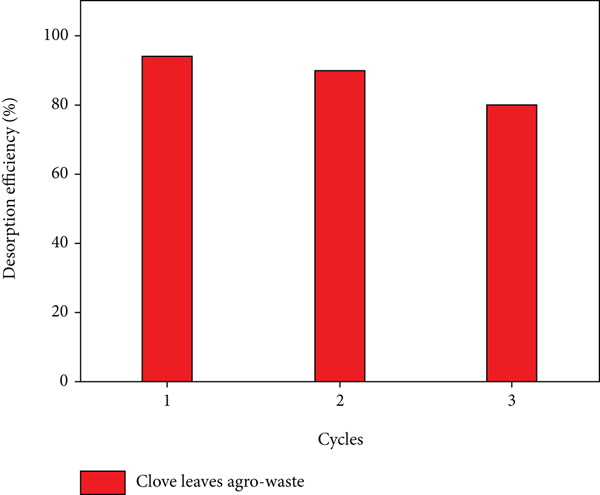
The cycles of adsorption-desorption of MG.
3.9. Proposed Mechanism of Adsorption of MG to the Biosorbent
The selection of the most promising biosorbent types from an exceptionally sizeable collection of freely accessible materials is a major task for the adsorption field. The subsequent real task is the recognition of the mechanism of adsorption [61]. The biosorption of MG to the biosorbent (CL-KOH) was sturdily dependent on different polar functional groups on the sorbent surface like hydroxyl, phenol, amine, carboxyl, and aromatic groups which are supported by the FTIR spectra in Figure 4. As observed from Figure 4, there was a small shift in the FTIR spectral position and a change in its intensity. Various active functional groups that show essential impacts on the sorption of MG were on the surface of lignin, pectin, cellulose, and hemicellulose. These results were indicative of the binding of the MG dye to potential active accessible sites of the functional groups like hydroxyl, amine, C=O, C=C, C–O, C-C, C-O-C, and phenols via a weak electrostatic interaction or van der Waals forces,

Proposed mechanism of MG adsorption to the biosorbent.
4. Conclusion
In this study, the ability of CL-KOH to confiscate MG from water-soluble solutions was investigated. Working parameters like initial MG concentration, adsorbent dosage, and contact time were determined to affect the sorption efficiency of CL-KOH. The D-R isotherm and Elovich kinetic models were found to ideally explain the sorption process of MG to CL-KOH and the established recorded sorption capacity was 131.5789 mg·g-1. Also, the adsorption of MG to CL-KOH was found to be exothermic. The main mechanism of MG sorption to the biosorbent was attributed to chemisorption and physisorption processes according to results from the D-R isotherm and thermodynamic parameters. The present study shows that the synthesized CL-KOH biosorbent could be applied as an efficient and inexpensive adsorbent for the sequestration of dye molecules from industrial dye effluents and the treatment of real wastewater contaminated with MG. Consequently, this recent study has further demonstrated the use of CL-KOH as an effective biosorbent for the removal of dyes, and it can be recycled/regenerated and reused, thus lowering operational costs and safeguarding the environment. Therefore, recommendations on further research studies and progression towards enhancing adsorption capacity should be encouraged.
Footnotes
Data Availability
Completely, the data produced or investigated during this work are involved in this submitted article.
Conflicts of Interest
There is no conflict whatsoever to declare.
Acknowledgments
The authors are honestly appreciative of their respective institutions.
