Abstract
Custom-built implants manufacture has always presented difficulties which result in high cost and complex fabrication, mainly due to patients’ anatomical differences. The solution has been to produce prostheses with different sizes and use the one that best suits each patient. Additive manufacturing technology, incorporated into the medical field in the late 80's, has made it possible to obtain solid biomodels facilitating surgical procedures and reducing risks. Furthermore, this technology has been used to produce implants especially designed for a particular patient, with sizes, shapes, and mechanical properties optimized, for different areas of medicine such as craniomaxillofacial surgery. In this work, the microstructural and mechanical properties of Ti6Al4V samples produced by direct metal laser sintering (DMLS) are studied. The microstructural and mechanical characterizations have been made by optical and scanning electron microscopy, X-ray diffraction, and microhardness and tensile tests. Samples produced by DMLS have a microstructure constituted by hexagonal α′ martensite with acicular morphology. An average microhardness of 370 HV was obtained and the tensile tests showed ultimate strength of 1172 MPa, yield strength of 957 MPa, and elongation at rupture of 11%.
1. Introduction
The malfunction or loss of total or partial functions of an organ or tissue, resulting from trauma or disease, is currently one of the most important and troubling public health problems affecting a significant number of people around the world. Annually in the US, the use of pins, plates, and screws for fixation of fractures reaches 1.5 million procedures and it is estimated that more than 500,000 joint replacement surgeries (hip and knee) are performed [1]. To meet the great demand for orthopedic surgical procedures to repair or replace body parts, it is necessary to develop biomaterials and more advanced surgical techniques.
The combination of high mechanical resistance, high toughness, manufacturing ease, good resistance to degradation by corrosion, and low cost makes some metallic materials the preferred biomaterials in the manufacture of orthopedic implants subjected to severe mechanical stresses within the human body.
Nowadays, pure titanium and its alloys are some of the most used metallic biomaterials in temporary or permanent orthopedic applications [2–5]. The temporary implants perform their function of fixing the fractures by a predetermined period, until the member is completely recovered so that the implant can be removed. The fixing plates, screws, wires, and intramedullary pins to repair broken bones are examples of temporary implants. The permanent implants replace body parts and need to play their role for the rest of the patient's life, such as the hip, knee, shoulder, elbow, or wrist prosthetics.
These implants, as well as other structural components, may fail due to mechanic fracture, wear, or corrosion. The combination of the electrochemical process of corrosion and cyclical mechanical stress can speed up the release of particles and metal ions, leading to premature failure of the implant [1, 6]. As an example, the articulated implants that are exposed to high loads and severe wear due to the patient's movement can be mentioned. Moreover, the degradation of metallic implants inside the human body may not only impair the integrity of the material, but also generate biocompatibility problems such as infection or allergic reactions, leading to premature removal of the implant [7, 8]. The derived detritus are harmful to the tissues that are in contact with the implant and can be taken into the bloodstream, settling in organs and impairing their functions. Thus, the metallic biomaterials must have properties such as resistance to fatigue and wear, toughness to fracture, and mainly high corrosion resistance.
Titanium is particularly suitable for works in corrosive environments and for applications in which low density is essential. It has a high relation strength/weight, nonmagnetic properties, and high corrosion resistance due to the formation of a compact protective film on the metal surface, the titanium oxide (TiO2). Due to its highly reactive nature in the presence of oxygen, the casting must be conducted in vacuum furnaces [1, 9].
Two types of crystalline formation can occur in titanium. The first one is called alpha and has a hexagonal close-packed (hcp) crystalline structure, while the second is termed beta with a body-centered cubic (bcc) crystalline structure. In pure titanium the alpha phase is stable at room temperature. The alloying elements are added to pure titanium and tend to modify the temperature at which the phase transformation occurs and the amount of each phase present. The alloying additions to titanium tend to stabilize the alpha or beta phase. The alpha phase is stabilized at higher temperatures, by elements called alpha stabilizers, as aluminum, tin, and zirconium, while the beta phase is stabilized by beta stabilizers at lower temperatures, as vanadium, molybdenum, niobium, chromium, iron, and manganese. There are three structural types of titanium alloys: alpha alloys (α), alpha-beta alloys (α + β), and beta alloys (β) [10–14].
Among the titanium alloys, the most employed in the manufacture of surgical implants is Ti6Al4V. The standard that describes the requirements for this alloy is ASTM F136 [15]. The structure of this alloy is α + β type, in which the α phase stabilizing element is aluminum and the one in phase β is vanadium [16]. In the same way as other materials, this titanium alloy can be wrought, quenched, or annealed and can be machined by conventional means [17].
The marketed products for medical use must be strictly tested as far as their chemical composition and mechanical and structural features are concerned to ensure that they are in accordance with the standards established for implants such as ASTM F136 [15], ASTM F1798 [18], and ASTM F1800 [19] among others. The manufacture of metallic implant can include a series of procedures that depend on some factors such as shape and size of the final product, metal features, and manufacturing cost, among others. The methods employed are casting, machining, forging, and powder metallurgy. Usually, after the melting and mechanical forming of the material, an annealing thermal treatment is performed in order to relieve residual stresses, to make the material more ductile and tough, and to produce a specific microstructure [20].
Following the manufacturing step, the implant may be subjected to surface modification procedures and finishing as ionic implantation, nitriding, porous or microporous coating application, polishing, chemical cleaning, and passivation. The surface features of the implants are essential in their biological performance.
Additive manufacturing (AM) or rapid prototyping (RP) is a fabrication technique by the additive method, which is based on successive addition of flat layers of material. This technology allows the production of physical components (prototypes, models, molds, etc.) with information obtained directly from a three-dimensional geometric model CAD (Computer Aided Design) system. The process starts with the 3D computer model of the part, obtained by a CAD system, electronically sliced. From this slicing, 2D contour lines are obtained which will define, in each layer, where material is going to be added. These layers are sequentially processed, generating the physical part through stacking and adhesion of them, beginning at the bottom and going up to the top part [21, 22].
The construction of parts with complex geometry and in the most varied materials, the use of only one piece of equipment to build the part from the beginning to the end, and less time and cost to obtain prototypes are some advantages that rapid prototyping offers when compared to other manufacturing processes [23–25].
In the medicine field, additive manufacturing was integrated to digital imaging techniques of computed tomography (CT) and magnetic resonance (MR), making it possible to obtain solid biomodels that reproduce the anatomical structures of the body. The internal structures images, acquired by these techniques, are handled in a system for medical imaging. From these images, specific algorithms of segmentation are applied to the separation of the interest structure (bone or tissue). From these image data, a 3D model is generated on the specific computer software (InVesalius) and exported to be made by additive manufacturing, originating the biomodels (biomedical prototypes) [26, 27].
These biomodels can be used for surgical planning, didactic purposes, the diagnosis and treatment of patients, and communication between professionals and patients. As a result, the biomodels facilitate surgery and reduce infection and rejection risks, complications, and length of surgery [28–31]. Nowadays, the additive manufacturing allows also the design of customized prosthetic implants, suiting them directly to the patient's needs. It is possible to produce implants with optimized sizes, shapes, and mechanical properties. For this, digital information obtained from computed tomography and magnetic resonance is used, making it possible to obtain directly the final implant and reduce the manufacturing time.
The use of these integrated techniques for manufacture of custom implants offers a significant potential of cost savings for health systems as well as the possibility to provide a decent life for a huge number of people [32–34]. Additive manufacturing can be used by health professionals who work in orthopedics, neurosurgery, maxillofacial and orthognathic surgery, traumatology, craniofacial and plastic surgery, implant dentistry, and oncology, among others.
An application in the medical field to be considered is the bone reconstruction surgery, for instance, the craniofacial reconstruction [23, 29]. Among the congenital defects, craniofacial anomalies are a group of highly diverse and complex that affects a significant proportion of people in the world [10]. Besides the cases of congenital deformities, there is an occurrence of acquired craniofacial defects due to other diseases, as tumors. In the last four decades, an increasing volume of cases of facial trauma was also observed, which is closely related to the increase of traffic accidents and urban violence [11].
Additive manufacturing can also be used in dental implants because the models provide information about size, direction, and location of the implants, as well as anatomical information, such as the path of mandibular canals. In cases of bone abnormalities, it is important to notice that the gain in patient's functional and psychological terms and the increased quality of life after surgery justify the costs of the application of new technologies [24].
There are more than 20 types of additive manufacturing systems on the market which, in spite of using different technologies of addition material, are based on the same principle of manufacturing by layer. The main additive manufacturing technologies used are stereolithography (SLA), selective laser sintering (SLS), direct metal laser sintering (DMLS), selective laser melting (SLM), fused deposition modeling (FDM), 3D inkjet printing (3DP), and electron beam melting (EBM). These systems are classified according to the initial state of raw material, which can be liquid, solid, or powder form [35, 36].
The additive manufacturing technique used in this work is the direct metal laser sintering (DMLS). Among the advantages of this technique, there is the ability to process titanium and other metal powders directly on the machine, without the use of binders or the need for postcure. The manufacturing stage of a model and a mold for casting is not required, which reduces the cost and manufacture time of the implant. Furthermore, inadequate geometries to the casting process and hollow models can be produced. The sequence to obtain the prosthesis, from acquired data, to the custom-built design, until its construction using the DMLS technology, was carried out in a craniofacial reconstruction surgery (Figure 1).

Application of additive manufacturing in craniofacial reconstruction surgery.
The aim of this work is to study the mechanical and microstructural properties of samples produced by DMLS and compare them with the properties of a wrought and annealed commercial alloy.
2. Materials and Methods
The samples studied in this work were produced by the direct metal laser sintering (DMLS) technique using EOSINT M 270 machine (Figure 2(a)). In DMLS technique, the powder is spread and processed by the action of an Ytterbium fiber laser in an inert and thermally controlled environment inside a chamber. A system of scanning mirrors controls the laser beam describing the geometry of the layer on the surface of the spread material. With the incidence of the laser, the particles of material are heated and reach their melting point, joining each other and also to the previous layer. When the material solidifies, a new powder layer is added and the laser scans the desired areas once more; in other words, after the sintering of a layer, a new layer is deposited, and this process goes on until the construction of the part is finished. Thus, the solid model is built layer by layer [25, 27].
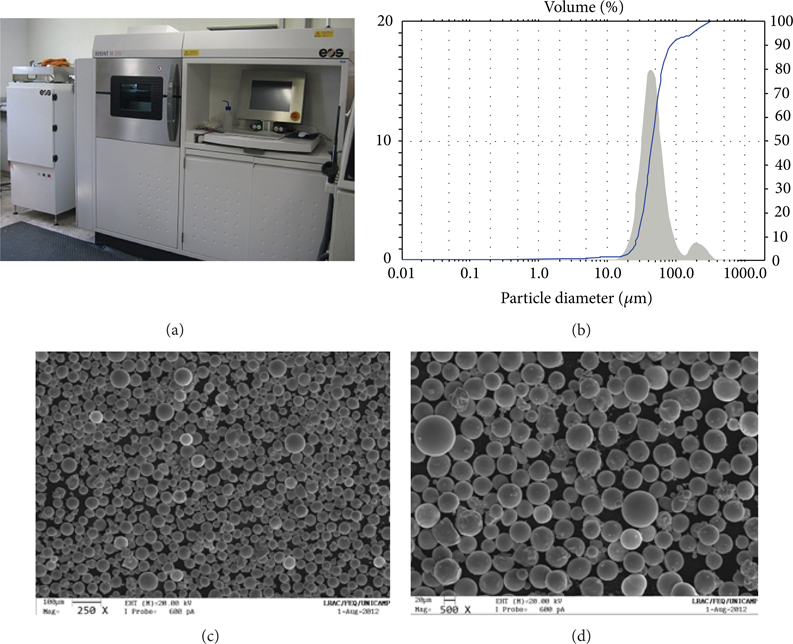
EOSINT M 270 machine (a), particle size distribution (b), and morphology of the commercial powder of the Ti6Al4V alloy used in the manufacturing of the samples ((c) and (d)).
The material used for production of specimens was the commercial powder of the Ti6Al4V alloy. This powder has spherical morphology with a particle mean size of 57 μm and a size distribution with D10 = 28 μm, D50 = 45 μm, and D90 = 85 μm (Figures 2(b), 2(c), and 2(d)). The processing parameters used to manufacture the specimens are shown in Table 1. During the processing, the argon gas was used to monitor the oxygen level within the chamber, acting also as protective gas.
Processing parameters used in the DMLS process.

For microstructural characterization and microhardness tests, three cylindrical samples measuring 12 mm diameter and 12 mm height were produced. The chemical composition of the samples was determined by the energy dispersive spectroscopy (EDS) technique using LEO-440i electron microscope with X-ray dispersive energy detector model 6070. The samples had their density measured according to the Archimedes principle and their porosity and microstructure analyzed by Olympus-BX60M optical microscope with Image Pro Plus 5.1 software and Zeiss-EVOMA15 scanning electron microscope with Smart SEM software, respectively.
X-ray diffraction tests were performed with radiation source of Cu (λ = 1.5406 Å) and scanning angle 2 theta from 20° to 80°, using the Philips Analytical X Ray diffractometer, model X’Pert-MPD, and the X’Pert Data Collector software. Vickers microhardness tests were conducted according to the ASTM E384 standard [37] using Shimadzu digital microdurometer, model HMV-2T. The applied load was 500 gf for 10 seconds. 20 measurements were made and the shown results are the mean values.
Three specimens measuring 120 mm × 10 mm × 4 mm were manufactured by DMLS for tensile tests. These tests were conducted according to the ASTM E8/E8M standard [38] using the equipment MTS-810 and an extensometer MTS-632.24C-50. The mean values of ultimate tensile strength (σ u ), yield strength (σ e ), Young's modulus (E), and elongation at rupture (Δl) of the material were determined. The microstructural and mechanical features of the samples produced by DMLS were compared to the samples features of the commercial Ti6Al4V alloy in the annealed condition.
3. Results and Discussion
The chemical composition of the samples produced by DMLS is according to the specification of the ASTM F136 standard [15], as shown in Table 2. The spectrum shown in Figure 3 confirms the presence of titanium, aluminum, and vanadium in the material. The processing parameters used in the process resulted in the production of samples at density 4.3785 g/cm3, in other words, about 98%. This value shows that the samples produced by DMLS are very dense, even in the presence of some pores.
Chemical composition of the samples produced by DMLS.

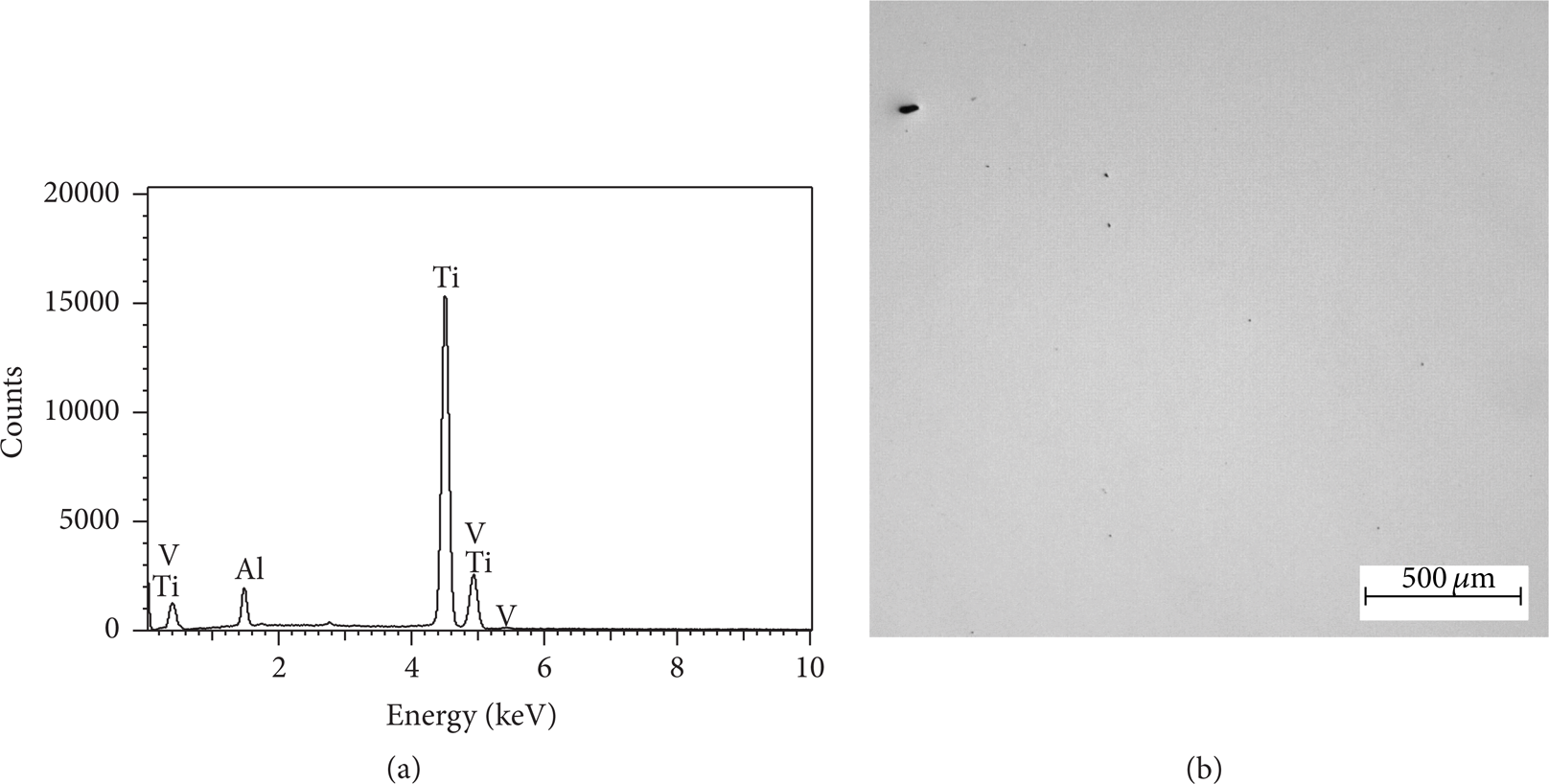
Chemical elements (a) and pores (b) present in the samples produced by the DMLS process.
Figures 4(a) and 4(b) show that the microstructure of the annealed Ti6Al4V alloy consists of two phases, the α phase (dark phase) and the β phase (lighter phase), homogeneously distributed in the matrix α. The microstructure of the samples produced by DMLS is refined and constituted by hexagonal α′ martensite with acicular morphology (Figures 4(c) and 4(d)). According to some authors [39, 40], the transformation of the bcc β phase to the α phase in the titanium alloys can occur martensitically or by nucleation and growth process, depending on the cooling rate and the alloy composition. During the fast cooling, the β phase can be transformed martensitically to the hexagonal α′ phase. On slower cooling, the β phase can be transformed by nucleation and growth to the Widmanstätten α phase. In Figures 4(c) and 4(d), it is possible to observe the presence of orthogonally oriented martensite plates. The formation of martensite plates in the material means that the cooling rate during the laser sintering was high enough to induce the martensitic transformation. Since the DMLS process involves the melting of thin layers, with approximately 30 μm, the fast cooling favors this type of transformation [41].

Microstructure of the Ti6Al4V samples in the annealed condition ((a) and (b)) and produced by DMLS ((c) and (d)).
The X-ray diffractograms shown in Figure 5 confirm the above described microstructural analysis. Concerning the annealed Ti6Al4V alloy, it is possible to observe the presence of not only the α phase but also the β phase in the 〈110〉 direction. For the DMLS sample, the only present phase is the α′ phase. Since the α and α′ martensite phases have the same crystalline structure, these phases show the same pattern in the diffractogram [42]. However, it is possible to observe in the sample produced by DMLS that the relative intensity of the peak of α′ in the 〈110〉 direction is higher than the intensity of the other peaks, indicating a change in the preferential crystallographic orientation when compared to the annealed alloy.

Diffractograms of the annealed and DMLS samples.
Figure 6 and Table 3 show the results of the mechanical tests performed on both annealed and produced DMLS samples. There was no significant difference in the values of σ e and E; however, the samples produced by DMLS showed values of σ u and microhardness higher than the samples in the annealed condition. On the other hand, the values of elongation at rupture show a lower ductility than the DMLS samples, which can be explained by a slight embrittlement due to the laser melting [43].
Tensile and microhardness tests results.


Stress-strain graphs of the annealed and DMLS samples.
4. Conclusions
Additive manufacturing techniques have been applied in the production of custom-built implants that meet the physical characteristics of each patient. The processing parameters used in the DMLS process resulted in samples with refined microstructure, composed by hexagonal α′ martensite, and acicular morphology, having a relative density of about 98%. The chemical composition of the samples is in accordance with the specification of the ASTM F136 standard. The X-ray diffraction confirmed that the only phase present in the DMLS samples is the α′ phase. The ultimate tensile strength and microhardness values were superior to the ones obtained for the alloy in the annealed condition. There was a loss in ductility of the material, which can be explained by a slight embrittlement due to melting by laser.
Conflict of Interests
The authors declare that there is no conflict of interests regarding the publication of this paper.
Footnotes
Acknowledgments
The authors wish to acknowledge the financial support provided by the Scientific Research Foundation for the State of São Paulo (FAPESP-Process 2008/57860-3 and 2010/05321-1) and National Council for Scientific and Technological Development (CNPq-Process 573661/2008-1).
