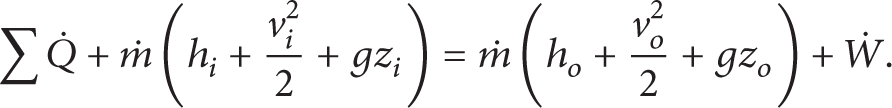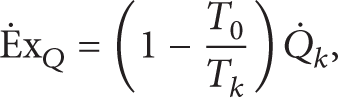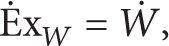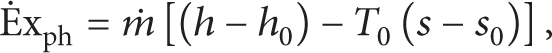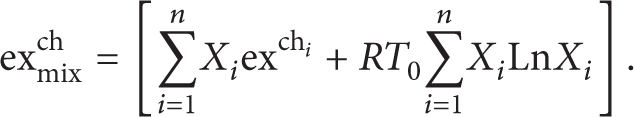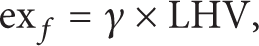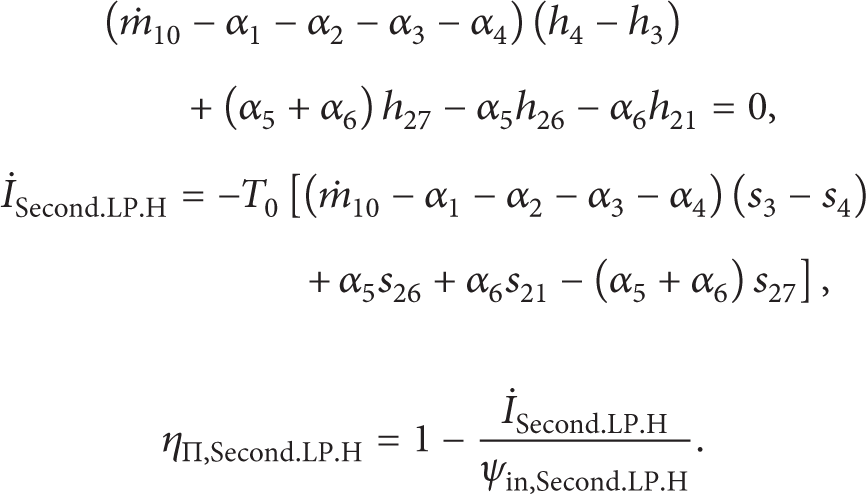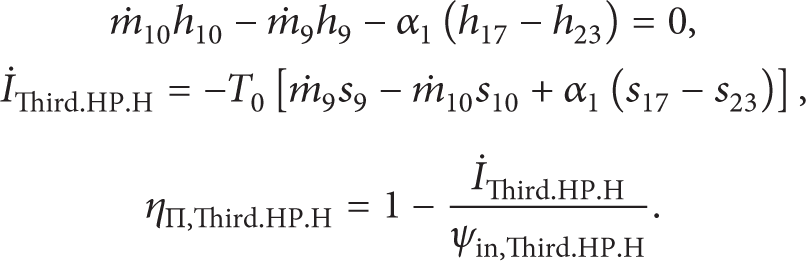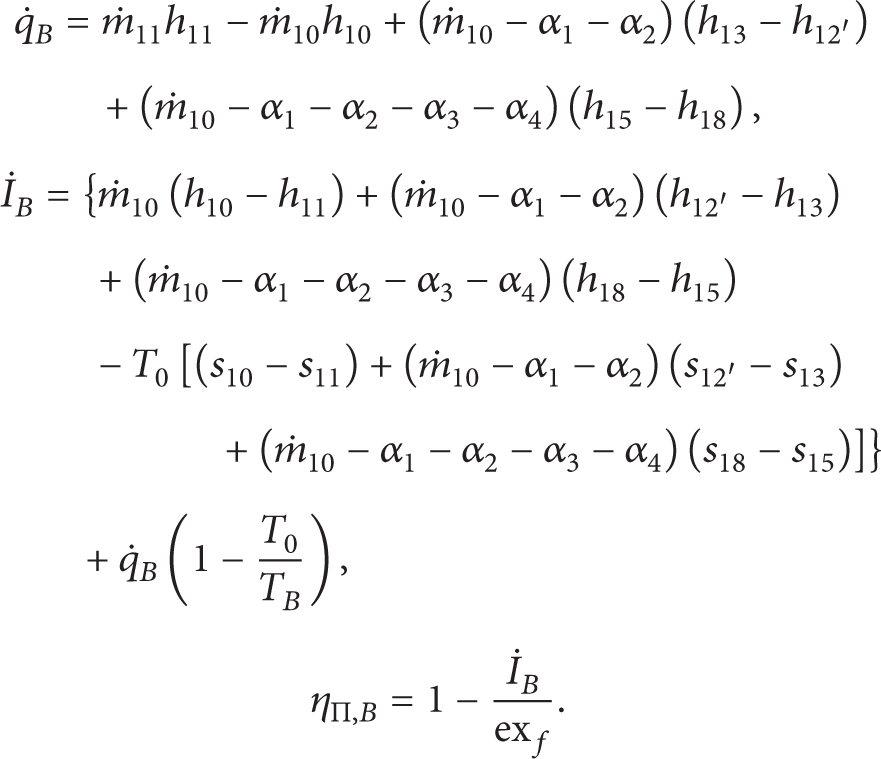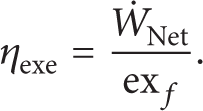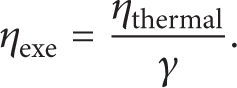Abstract
A steam cycle with double reheat and turbine extraction is presented. Six heaters are used, three of them at high pressure and the other three at low pressure with deaerator. The first and second law analysis for the cycle and optimization of the thermal and exergy efficiencies are investigated. An exergy analysis is performed to guide the thermodynamic improvement for this cycle. The exergy and irreversibility analyses of each component of the cycle are determined. Effects of turbine inlet pressure, boiler exit steam temperature, and condenser pressure on the first and second laws' efficiencies are investigated. Also the best turbine extraction pressure on the first law efficiency is obtained. The results show that the biggest exergy loss occurs in the boiler followed by the turbine. The results also show that the overall thermal efficiency and the second law efficiency decrease as the condenser pressure increases for any fixed outlet boiler temperature, however, they increase as the boiler temperature increases for any condenser pressure. Furthermore, the best values of extraction pressure from high, intermediate, and low pressure turbine which give the maximum first law efficiencies are obtained based on the required heat load corresponding to each exit boiler temperature.
1. Introduction
Steam power plants are widely utilized throughout the world for electricity generation, and coal is often used to fuel these plants. Energy consumption is one of the most important indicators showing the development stages of countries and living standard of communities. Currently, 80% of electricity in the world is approximately produced from fossil fuels (coal, petroleum, fuel oil, and natural gas) fired thermal power plants, whereas 20% of the electricity is compensated from different sources such as hydraulic, nuclear, wind, solar, geothermal, and biogas [1]. Many techniques are being used to increase the efficiency of the steam cycle. The most outstanding of these techniques are reheating in order to reduce the irreversibility of the cycle. Reheating increases the efficiency by raising the mean temperature of the heat addition process, by increasing the steam temperature at the turbine inlet and also by increasing the efficiency of the expansion process in the steam turbine. With reheat, a power plant can take advantage of the increased efficiency that results in higher boiler pressure and yet avoid low-quality steam at the turbine exhaust [2]. Habib and Zubair [3] conducted a second law analysis of regenerative Rankine power plants with reheat. Chiesa and Macchi [4], Gambini et al. [5], Bhargava and Peretto [6], and Poullikkas [7] have studied and predicted the performance of reheat gas turbine using air as a coolant. Srinivas [8] showed that the deaerator placed in between the LP and IP heaters gives high efficiency compared to a deaerator-condenser arrangement.
Analyses of power generation systems are of scientific interest and also essential for the efficient utilization of energy resources. The most commonly used method for analysis of an energy-conversion process is the first law of thermodynamics. Furthermore, exergetic analysis provides a tool for a clear distinction between energy losses to the environment and internal irreversibility in the process [9]. As energy analysis is based on the first law of thermodynamics, it has some inherent limitations like not accounting for properties of the system environment or degradation of the energy quality through dissipative processes. An energy analysis does not characterize the irreversibility of processes within the system. In contrast, exergy analysis will characterize the work potential of a system. Exergy is the maximum work that can be obtained from the system, when its state is brought to the reference or dead state [10]. Ozgener et al. [11] illustrated that the overall energy and exergy efficiencies of the SGDHS (Salihli geothermal district heating system) components were also studied to evaluate their individual performance and obtained to be 55.5 and 59.4%, respectively. Ameri et al. [12] showed that the greatest exergy loss in the gas turbine occurs in the combustion chamber due to its high irreversibility. Rosen [13] evaluated the performance of coal-fired and nuclear power plants via exergy analyses. Dincer and Muslim [14] performed a thermodynamic analysis of reheat cycle power plants. Medina-Flores and Picón-Núñez [15] proposed algorithms to predict power production for single and multiple extraction steam turbines. Aljundi [16] studied the energy and exergy analysis of Al-Hussein power plant in Jordan and reported that the chemical reaction is the most significant source of exergy destruction in the boiler system. Oktay [17] presented exergy loss and proposed improving methods for a fluidized bed power plant in Turkey. Ahmadi et al. [18] have shown the results of both exergy and exergoeconomic analyses. Their results showed that the largest exergy destruction occurs in the CCPP (combined cycle power plant) combustion chamber and that increasing the gas turbine inlet temperature decreases the CCPP cost of exergy destruction.
In the present study, the energy and exergy analysis are conducted to evaluate the performance of the above suggested steam power plant cycle using 6 closed feed water heaters with double reheat and with different extracting pressure. Sources of exergy destruction in the power plant are identified and a parametric study is performed to determine the first and second law efficiencies with different operating conditions. Maximum first law efficiencies are obtained at the optimal extraction pressure.
2. Energy and Exergy Analysis
This cycle consists of boiler, turbine, condenser, feed water heaters, deaerator, feed water pump, and the condensate extraction pump. The schematic of cycle and the T-s diagram are shown in Figures 1 and 2, respectively.

The schematic of steam power plant cycle.
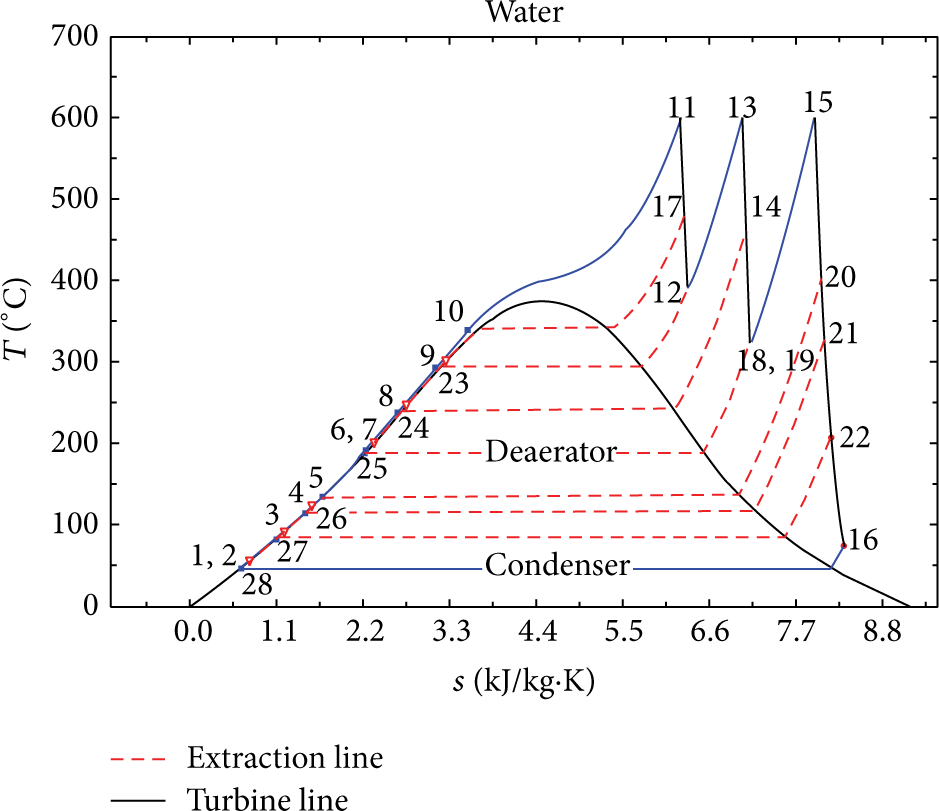
The T-s diagram of the cycle.
For an open system at steady state, the first law of thermodynamics can be written as [19–21]
Exergy is defined as the maximum work that can be achieved by bringing a system into equilibrium with its environment. The concept of exergy is supported by the consideration of the temperature level, which is based on the energy conversion from thermal to power. Exergy analysis is a useful method for complementing but not for replacing the energy analysis. It is becoming the most appropriate tool for thermodynamic analysis. The exergy efficiency of the power cycle may be defined in two ways. The first one is the physical exergy and the second one is the chemical exergy. In this study, the kinetic and potential parts of exergy are considered negligible. The chemical exergy of gaseous fuels is computed from the stoichiometric combustion chemical reactions. The chemical exergy is associated with the departure of the chemical composition of a system from its chemical equilibrium which is important in processes involving combustion.
Equations (2) and (6) show in general the exergy destroyed rate and the exergy at any state of an open system:
where
The physical exergy of the fuels is negligible when compared to the chemical exergy. For the evaluation of the fuel exergy, the above equation cannot be used [22, 23]. Thus, the corresponding ratio of simplified exergy is defined as follows [24]:
where γ stands for the exergy factor and LHV denotes the lower heating value of the fuel material. It should be noted that γ can be estimated for any fuel C x H y by [23, 25, 26]
The energy and exergy destroyed equations for all components of the cycle are given as follows.
The First Adiabatic Low Pressure Feed Water Heater.
where α is the extraction mass flow rate.
The Second Adiabatic Low Pressure Feed Water Heater.
The Third Adiabatic Low Pressure Feed Water Heater.
The Adiabatic Deaerator.
The First Adiabatic High Pressure Feed Water Heater.
The Second Adiabatic High Pressure Feed Water Heater.
The Third Adiabatic High Pressure Feed Water Heater.
The Condenser.
The Adiabatic Feed Water Pump.
The Adiabatic Condensate Extraction Pump.
The Adiabatic Turbine.
The Boiler.
Energy efficiency (thermal efficiency) is defined as [27]
It is obvious that
whereas exergy efficiency (the second law efficiency) is defined as
It is obvious that [28]
3. Results and Discussion
The main parameters for analyzing this steam cycle are shown in Table 1. It should be noted that the main parameters in Table 1 are chosen following some references as shown in Table 1. Furthermore, low values of both pinch and approach points are chosen following [29] such that the thermal efficiency of the cycle has a maximum. The thermodynamic properties of water were calculated using EES software [30] and summarized in Table 2. The energy consumption based on the dead state of P0 = 101 (kPa) and T0 = 25 (°C) for each component is shown in Tables 3 and 4, respectively. Table 3 shows the power balance of the power plant components at two states. One of these is based on the data from Table 2 and the other is from the power plant at the optimal first law efficiency. In other words, to obtain the optimum first law efficiency and then the power balance of other parts of cycle such as boiler and turbine, should vary as seen in Table 3. The optimum first law efficiency is shown, for example, in Figures 9, 11, 12, and 13 for some different extraction pressure. The exergy destruction, exergy efficiency of each component, and percent exergy destruction rate are also shown in Table 4 following the equations defined earlier for each component. The thermal efficiencies for boiler, heaters, and condenser are 100%, since the heat losses for these components are neglected. The efficiencies in the case of the turbine, feed water pump and, condensate extraction pump are 92.83%, 80%, and 80%, respectively. The first and second law efficiencies of this power cycle based on (23) and (26) are 48.9% and 46.13%, respectively.
The main parameters for analyzing the steam cycle.

Process parameters of the cycle.

Power balance of the power plant components.

Exergy destruction of the power plant components.

3.1. Effect of Condenser Pressure
The effect of condenser pressure on the boiler and condenser heat load and also on the turbine power for three boiler exit steam temperatures is shown in Figures 3 and 4, respectively. It is clear that as the condenser pressure increases, the condenser saturation temperature increases too which means that more heat has to be removed by the condenser for fixed boiler exit steam temperature as seen in Figure 3. Furthermore, the condenser heat load increases for fixed condenser pressure as the boiler exit steam temperature increases. However, the boiler heat load is independent of the condenser pressure. Figure 4 shows that the turbine power is affected by both the boiler exit steam temperature and the condenser pressure. Therefore, as the condenser pressure increases, more heat load will be removed through the condenser which leads to a decrease in the turbine power for fixed T B and vice versa. It is also noted that as T B increases the turbine power increases for fixed condenser pressure.
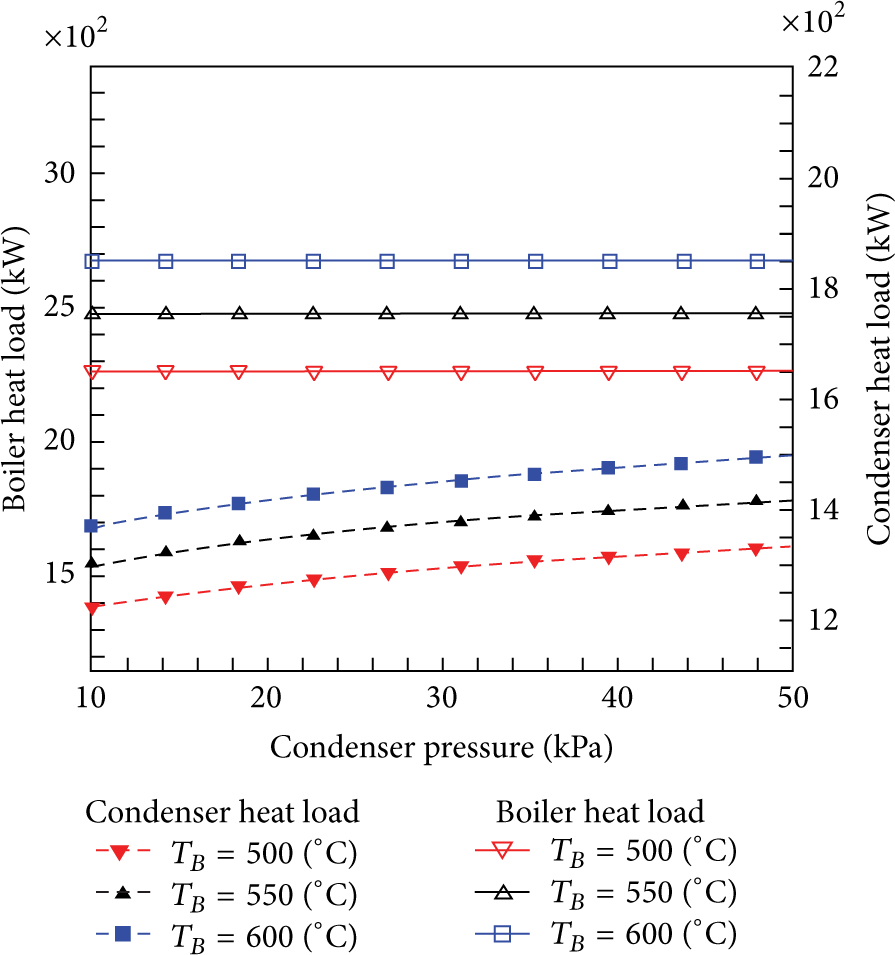
The effect of condenser pressure on the boiler and condenser heat loads at T B = 500°C, 550°C, and 600°C.
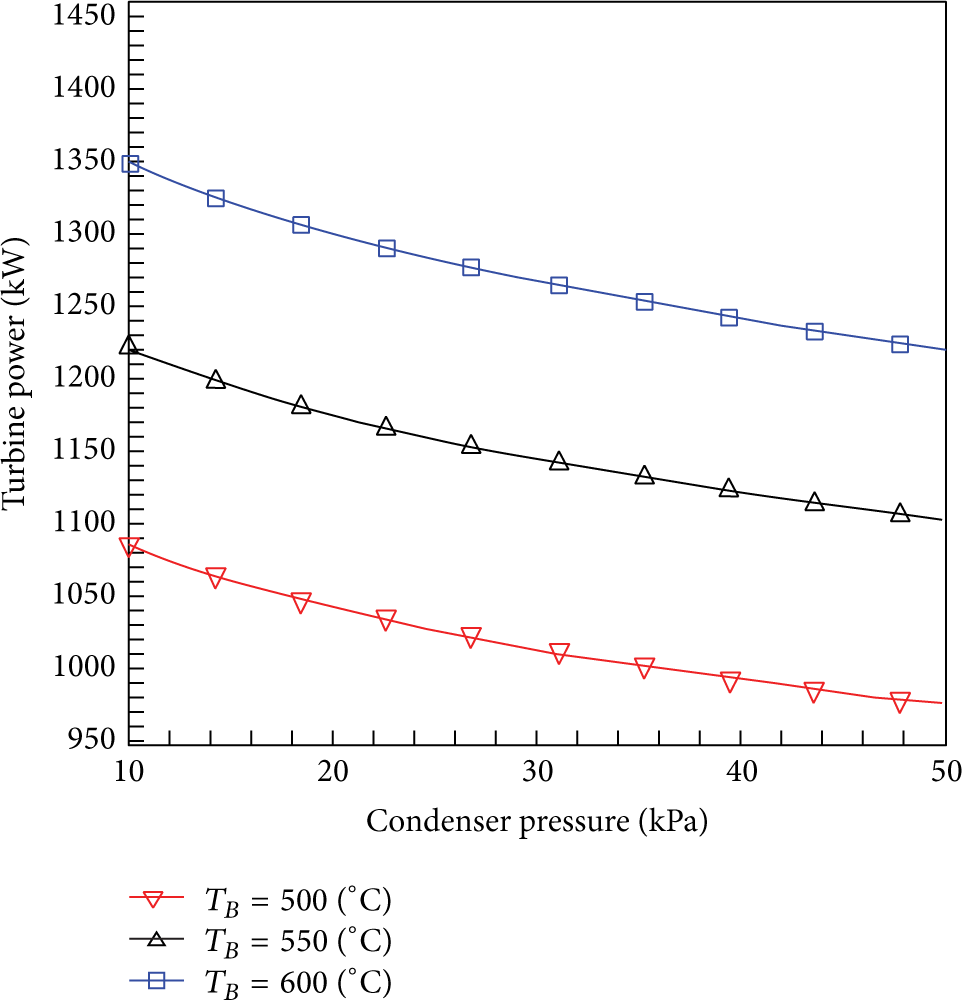
The effect of condenser pressure on the turbine power at T B = 500°C, 550°C, and 600°C.
Figures 5 and 6 show the effect of condenser pressure on the first and second law efficiencies, respectively. Profiles shown in these figures are confirmed by (23) and (26), since the first law efficiency depends on both the net power and the heat added at the boiler but since the boiler heat load is constant then increasing the condenser pressure will reduce the net power which in turn will reduce the thermal efficiency. Similar effects are obtained for the exergy since it depends on the thermal efficiency and the constant value of the exergy factor as seen in Figure 6.

The effect of condenser pressure on the first law efficiency at T B = 500°C, 550°C, and 600°C.

The effect of condenser pressure on the second law efficiency at T B = 500°C, 550°C, and 600°C.
3.2. Effect of High Pressure Turbine
Figures 7 and 8 show the effect of high pressure turbine on the boiler and condenser heat load and also on the turbine power for three boiler exit steam temperatures, respectively. When the HP turbine pressure increases, the turbine exit enthalpy decreases and the condenser heat load decreases because the inlet and the outlet enthalpies at condenser are constant and the mass flow rate decreases. Also it can be seen that the turbine power and boiler heat increase first to maximum and then decrease as the HP turbine inlet pressure increases. It should be noticed that the enthalpy drop across the turbine and extraction mass flow rate increase as HP turbine inlet pressure increases, so the turbine power output increases. However, due to the change of gradient of the saturated vapor line, the turbine power decreases and this explains the behavior of the thermal efficiency which increases and then decreases as the turbine extraction pressure increases as shown in Figures 11, 12, and 13.

The effect of HP turbine inlet pressure on the boiler and condenser heat loads at T B = 500°C, 550°C, and 600°C.

The effect of HP turbine inlet pressure on the turbine power at T B = 500°C, 550°C, and 600°C.

The effect of HP turbine inlet pressure on the first law efficiency at T B = 500°C, 550°C, and 600°C.
Figures 9 and 10 show the effect of HP turbine pressure on the first and second law efficiencies. Since the first law efficiency depends on both the turbine power and the boiler heat and since both of them (as shown in Figures 7 and 8) reach a maximum then decrease, therefore the thermal efficiency profiles follow them as seen in Figure 9. Similar effects are obtained for the second law efficiency since it is a function of the first law efficiency so it should follow the same trend as seen in Figure 8.
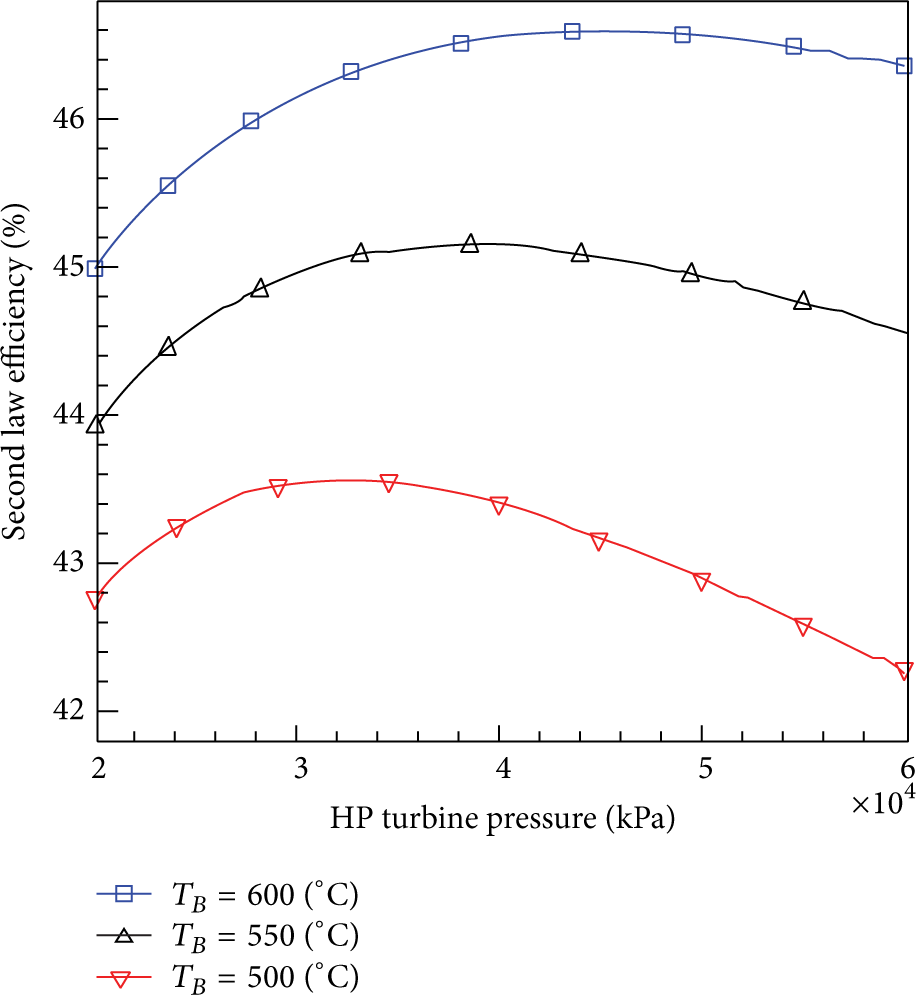
The effect of HP turbine inlet pressure on the second law efficiency at T B = 500°C, 550°C, and 600°C.

The best values of extraction pressure from HP turbine.

The best values of extraction pressure from IP turbine.

The best values of extraction pressure from LP turbine.
It is also clear that for both cases as the boiler exit steam temperature increases the boiler and condenser heat load, turbine power, and first and second law efficiencies increase for any fixed pressure. Figures 11, 12, and 13 show the best values of extraction pressure from high, intermediate, and low pressure turbine on the first law efficiency at T B = 600°C, respectively.
The maximum first law efficiency at optimum extraction pressure (P17 = 15639 kPa, P12 = 10000 kPa, P14 = 4540 kPa, P6 = 2073 kPa, P20 = 472.6 kPa, P21 = 171.7 kPa, and P22 = 50 kPa) is 49.16%.
4. Conclusion
In this study, a thermodynamic analysis of a reheat Rankine cycle steam power plant with three HP feed water heaters and three LP feed water heaters with deaerator is conducted, in terms of the first law of thermodynamic analysis and the second law analysis. The main conclusions drawn from the present study are summarized as follows.
When condenser pressure increases, the condenser rejected heat increases and the turbine power and first and second law efficiencies decrease but boiler heat transfer is independent of the condenser pressure.
When the boiler exit steam temperature increases, condenser rejected heat load, boiler heat load, turbine power, and first and second laws efficiencies increase.
As the high pressure turbine increases, the first and second laws’ efficiencies have similar profiles with maximum values depending on the HP turbine and the boiler temperature.
The maximum first law efficiencies are obtained for the optimal extraction pressure at high, intermediate, and low pressure turbine as 49.15%.
The maximum value of exergy loss occurs in the boiler followed by the turbine and the minimum value of exergy loss occurs in the condensate extraction pump.
Footnotes
Nomenclature
Conflict of Interests
The authors declare that there is no conflict of interests regarding the publication of this paper.
Acknowledgments
The authors extend their appreciation to the Deanship of Scientific Research at King Saud University for funding this work through the research group Project no. RGP-VPP-080. They also express their gratitude to the anonymous referees for their constructive review of the paper and helpful comments.