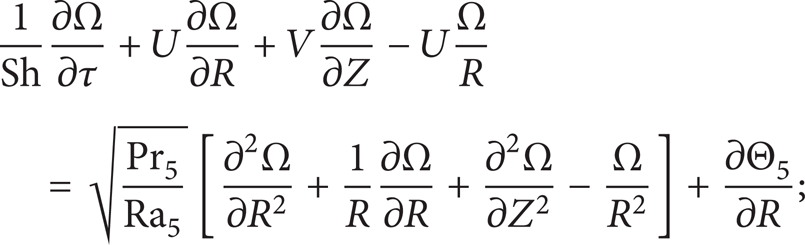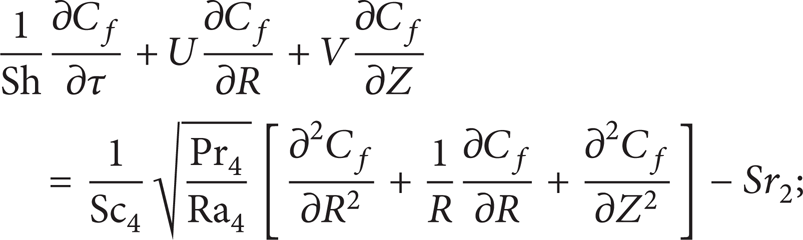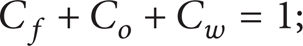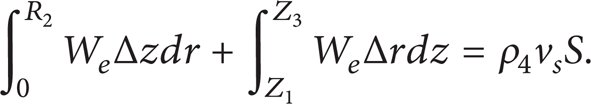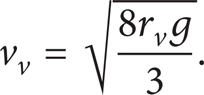Abstract
We carried out a numerical and experimental investigation of heat and mass transfer at the ignition condition of a liquid high-energy material by a typical immersed source with a limited energy capacity, being a small, intensely heated metallic particle. The numerical research is made on the basis of a model taking into account a group of interrelated physicochemical processes (thermal conductivity, diffusion, convection, mixing, and radiative heat transfer) with phase transitions (evaporation of the liquid and crystallization of the particle's material). We established such terminal conditions for the immersion energy source that prevent inflammation of the high-energy material.
1. Introduction
The processes of high-energy material (HEM) ignition caused by various sources of heat are interesting, first of all, due to a high risk of fire and explosion and due to application of such material in special technics. Recent years, a fairly comprehensive analysis of macroscopic regularities was done for ignition processes of solid [1–3] and liquid [4–9] HEM at local heating induced by typical sources with a limited energy capacity (in particular, by intensely heated metallic and nonmetallic particles of different physical nature). It has been shown [1–7] that inertial processes of heat and mass transfer play leading role rather than rapid oxidation reactions, as could have been supposed. The integral ignition characteristics have been found to be dependent on a large group of factors [1–7] that determine the conditions of warming and subsequent ignition of HEM. Possible regimes of igniting HEM by local heating have been determined [1–4], as well as the peculiarities of location of the leading oxidation zone with respect to the contact boundary between the HEM and the local energy source [4–6]. It was noticed that the basic consideration has been made for the conditions of an “ideal” contact (without roughness, unevenness, cavities of energy source) at the boundary “local energy source—HEM” [1–3] and for the conditions of partial embedding of a small heat source into the near-surface layer of HEM [4–7]. At the same time, there exist conditions that permit a complete immersion of local sources with a limited energy capacity into HEM (in particular, liquid and gelatinous combustibles). It is interesting to analyze the igniting conditions of a typical liquid HEM by an immersed (submerged) source of heat with limited enthalpy.
The purpose of this work is a numerical and experimental investigation of ignition processes macroscopic regularities under phase transitions at the interaction of a typical liquid HEM with an immersed high-temperature metallic particle and determination of parameters values of local energy source when ignition is impossible.
2. Problem Statement
The processes of heat and mass transfer are studied in the system “liquid HEM—small heat source—oxidant.” Its scheme is outlined in Figure 1.
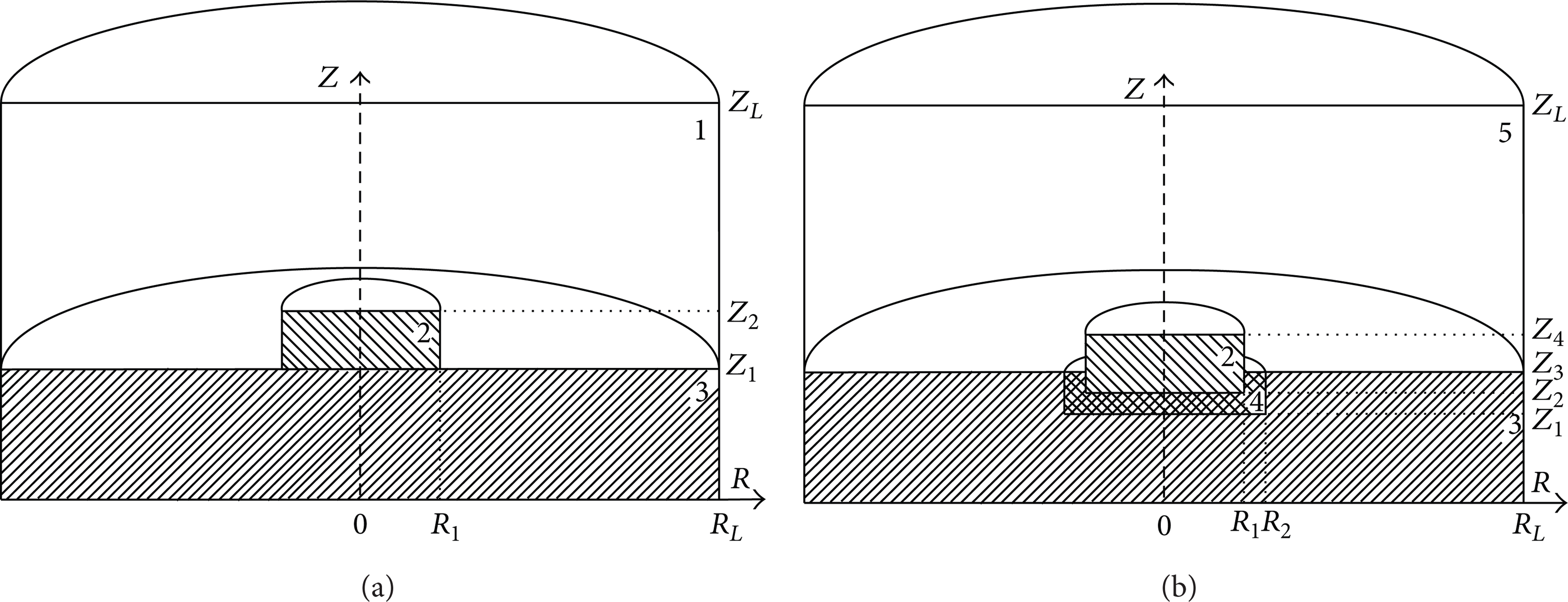
A scheme of the solution domain at (a) τ = 0 and (b) 0 < τ < τ d : 1: oxidant (air), 2: particle, 3: liquid HEM, 4: vapor gap, and 5: vapor-gas mixture.
As a local heat source with limited enthalpy, we examine a high-temperature metallic (steel) particle of small sizes. Mathematical modeling of ignition of a liquid is made for a typical high-energetic material with well-known thermophysical and thermochemical characteristics: kerosene.
It is assumed that a hot particle is slowly (at 0.5 m/s) deposited on the surface of a liquid HEM (Figure 1(a)) and immersed into it (Figure 1(b)). Different conditions of immersion of the particle, involving the formation of a vapor gap between energy source and liquid during the ignition time delay, are examined. It is taken into account that the liquid warms up as energy is supplied from a local heat source. The conditions of vapor formation are reached near the frontal and lateral surfaces of the particle. In these areas, the evaporation process of liquid begins. As a consequence, there emerges a vapor gap between the immersed source and the liquid. The vapor is injected into the oxidant (air) and, due to diffusion and convection, becomes mixed with it. There arises a vapor-gas mixture. It includes initial component (oxidant) and vapors of water and combustible, involved in a chemical reaction. When terminal conditions are reached (for the concentration of combustible and for the temperature of vapor-gas mixture), the process of oxidation accelerates and becomes irreversible, thereby causing ignition.
Numerical modeling is made under the following assumptions, which do not impose any essential restrictions on the generality of the problem statement.
As a result of evaporation of the liquid, there emerges one substance with known characteristics. In the course of an experimental determination of kinetic parameters of the oxidation of gas mixtures and vapors produced by liquid combustibles and highly inflammable substances, one determines, as a rule, the “effective” values of the activation energy E and of the preexponential multiplier k0 [10, 11]. Consequently, we have assumed a realization of one “effective” reaction, involving only one combustible.
A possible burn-out of the liquid is not considered. Previously, it has been established [6], as one takes into account the burn-out of a liquid HEM, that the values of the integral characteristics of ignition can change only in the region of relatively low (less than 1000 K) temperatures of the hot particle (near the terminal conditions of inflammation). However, even in the regimes of minimally possible temperatures of ignition, the burn-out of a liquid HEM results in a change of τ d being no higher than 4% [6].
Convective flows emerging in the liquid due to the warming of its near-surface layer is not considered. The results [7] of numerical research of conductive and convective heat transfer in a liquid HEM ignited by local energy sources demonstrate that the characteristic time of convection in the near-surface layer of a hot liquid is several times larger than the duration of ignition delay.
Besides, it was quite difficult to give an exact description of real particles shape in the problem statement. As a rule, metallic and nonmetallic particles formed during different technological processes (welding, cutting, polishing of metals, etc.) or combustion of typical natural fuels (wood, coal, etc.) have shape of irregular polyhedron with characteristic rate (radius or width, high) ranging from 10−6m to 10−2m. Besides, it usually has a liquid state of material when initial temperature is more than 1600 K or solid state when its temperature is less than 1400 K. Exact temperature value of metal melting depends on a type of particle material. Quite often, “hot” particles are similar in shape to nonsymmetrical bodies of revolution, having one or two flat sides. Consequently, in numerical and experimental research, model sources of ignition are regarded in the form of single metallic particles shaped as small disks. Such form makes it possible to give a more simple description of the thermal contact condition between the energy source and the liquid HEM (Figure 1). Besides, ignition process is characterized by average values of integrated characteristics (e.g., ignition delay time) [8] at the interaction cylindrical particle with liquid fuel.
Results gained from the study of ignition of liquid combustibles and highly inflammable substances caused by hot melted particles [7] allow one to conclude that, during the crystallization of energy source, an additional amount of heat arises. According to that process, an intensive heat transfer into the liquid and into the emerging vapor-gas mixture happens near the border of endothermic phase transition (evaporation). It leads to a certain reduction of the ignition time delay. Given this, the shape of the particle, due to its crystallization during a sufficiently small time interval (τ d < 1÷2), changes quite insignificantly. The results [7] confirm that it is expedient to take into account a phase transition in the melted metallic particle at its crystallization. It will let us research the ignition problem (Figure 1) closer to real processes.
Research results of interrelated processes of heat and mass transfer with phase transitions and chemical reactions of oxidation at the ignition of liquid HEM by typical sources with a limited energy capacity [4–8] show that numerical simulation at using only one of the known ignition criteria [12] Zel'dovich, Frank-Kamenetskii, Kovalskii and Vilyunov does not allow one to take into account such specific characteristics of researched processes that are related to the cooling down of energy source and to the phase transitions (crystallization and evaporation).
Consequently, in our numerical modeling, we have used two ignition criteria, considering in the most complete manner the macroscopic features of heat and mass transfer processes in the system (Figure 1). Consider the following.
The heat emerging due to the chemical reaction of oxidation of combustible vapors in the air is greater than the heat transferred from the energy source to the liquid HEM and vapor-gas mixture.
The temperature of the mixture of the HEM vapors and the oxidant is higher than the initial temperature of the heat source.
3. Mathematical Model
The numerical modeling of ignition of the liquid HEM in the system (Figure 1) at 0 < τ < τ d reduces to the solution of a set of nonlinear nonstationary differential equations with partial derivatives, presented in accordance with the basic principles of the general theory of diffusion and heat transfer in chemical kinetics [13–15], 0 < R < R2,Z1 < Z < Z2;R1 < R < R2,Z2 < Z < Z3;R1 < R < R L , Z3 < Z < Z4; 0 < R < R L , and Z4 < Z < Z L as follows:
the continuity equation:
the equation of motion for the vapor-gas mixture:
the energy equation for the vapor-gas mixture:
the diffusion equation for the combustible vapors in the air:
the diffusion equation for the water vapors in the air:
the balance equation for the vapor-gas mixture:
the heat transfer equation for the metallic particle, 0 < R < R1 and % Z2 < Z < Z4:
the heat transfer equation for the liquid, 0 < R < R1, 0 < Z < Z1; R2 < R < RL, Z1 < Z < Z3:
Dimensionless complexes are
The equation of motion for a metallic particle considers the forces of resistance at particle circumfluence by vapors of liquid HEM and gravity under the conditions of its immersion (Figure 1(b)) as follows:
The dimensionless parameter of resistance, cχ, depending in general on the surface shape of a particle and its locations with respect to the direction of motion is presented [16].
The initial conditions (Figure 1(a)) are v p = 0.5 m/s,v = 0 m/s;Θ = Θ p at 0 < R < R1,Z1 < Z < Z2;Θ = Θ0 at 0 < R < R L ,0 < Z < Z1;Ω = 0,Ψ = 0,С f = 0,C w = 0, Θ = Θ0 at R1 < R < R L , Z1 < Z < Z2;0 < R < R L , and Z2 < Z < Z L .
The boundary conditions (Figure 1(b)):
Heat dissipated from the source due to radiant heat exchange:
It is supposed that the vapor gap around the immersing particle has the form of a hollow cylinder (Figure 1(b)). At the same time, we take into account a dynamical change of cylinder parameters (ΔZ = Z2 − Z1 and ΔR = R2 − R1) during warming of the near-surface layer of liquid and its evaporation. We model a proportional shift of the boundaries Z1 and R2 concerning boundaries Z2 and R1, respectively. Given this, it has been assumed that ΔZ = ΔR in a first approximation.
At every time step, we calculate the values ΔZ and ΔR, using the condition of mass balance for the vapors of the liquid in the gap (Figure 1(b)):
The first term in the left-hand side of (13) determines the arrival of vapor mass of the liquid substance near the frontal surface of the immersing particle (Z = Z1 and 0 < R < R2); the second one determines the same characteristics for the lateral surface of the local heat source (R = R2 and Z1 < Z < Z3). The term in the right-hand side of (13) generally depends on the linear velocity of vapors of the liquid substance v s . Its velocity is determined at the solution of the equations of continuity (1) and motion (2) on each time step.
In order to determine the mass velocity of evaporation of a liquid HEM, we use the following expression:
The accommodation coefficient is computed by the formula
At the complete immersion of the energy source in liquid, the sizes of the vapor gap between its upper facet and the incoming layer of the liquid HEM are computed by analogy with ΔZ. At the same time, the energy equation for a thin layer of HEM above the submerged particle is presented with allowance for the passage of vapor bubbles of the liquid:
The velocity of ascending (surfacing) vapor bubbles v v depends on the regime of flow as well as on their size r v . Analyzing the model in question, we concluded that, in a first approximation, we can use an expression that characterizes turbulent surfacing:
It is known that the characteristic size of surfacing bubbles for a layer of liquid less than 5·10−3m thick under the condition of intense evaporation changes in the rage r v = (0.01÷0.1)·10−3m. Therefore, the average velocity of surfacing for the problem statement in question (Figure 1) can be set equal to v v = 0.05 m/s.
In order to make a passage to dimensionless variables, we use the following scale quantities: z L is the characteristic size of the solution domain (z L = 0.02 m), V m is the scale of convection velocity of the combustible vapors near the surface of the liquid, m/s, t m is the time scale (t m = 1 s), and T m is the temperature scale (T m = 1000 K).
The set of (1)–(8) is solved by the method of finite differences. At the same time, we solve difference analogues of the differential equations (3)–(8) by the locally one-dimensional method. The equations of elliptic type, that of continuity (1) and that of motion (2), are solved by using the method of alternating directions. In order to solve one-dimensional difference equations, we use the sweep method with an implicit four-point scheme. Nonlinear equations are solved by the iteration method. In order to increase the accuracy of solutions of the set of differential equations (1)–(8), we select no less that 400 knots of the difference net for each of the coordinates (near the borders of phase transition, the difference net is made denser) and use time step 10−6 s.
In solving the equations of elliptic type, those of continuity and motion, in the variables “current function-vector of velocity vortex,” we use the numerical algorithms [17, 18].
The reliability of the obtained results has been verified by the comparison with the experimental data [8]. Testing of numerical methods and elements of algorithm for differential equations system decision with corresponding initial and boundary conditions is carried out on the example of nonlinear processes of heat conductivity [19] and chemical kinetics [20]. Besides, check of the energy conservation law in the solution field was carried out. The error of the energy conservation law at change of initial temperature and the rates of a hot particle did not exceed 2.5%.
4. Results and Discussion
Our research of the processes of heat and mass transfer in the conditions of physicochemical transmutations has been carried out for the following values of parameters [4–8]: the initial temperature of the liquid HEM and oxidant being Θ0 = 0.3, that of the metallic particle being Θ p = 0.8÷2, the thermal effect of oxidation of the combustible vapors in the air being Qо = 42·106 J/kg, the thermal effect of evaporation of the liquid being Q e = 0.025·106 J/kg, the activation energy and preexponent of the oxidation reaction being E = 0.193·106 J/mol and k0 = 9·108 s−1, the melting temperature of the particle's material being Θ c = 1.4, the thermal effect of crystallization being Q c = 0.205·106 J/kg, the blackness degree of a steel particle being ε = 0.6, the particle sizes being Z p = 0.15 and R p = 0.05÷0.3, the thickness of the layer of liquid HEM being Zliq = 0.5, and the sizes of the solution domain being R L = 0.5, Z L = 1. Thermophysical characteristics of substances are the same as [4–7].
As a result of numerical (using the models (1)–(12)) and experimental (using the method [8]) research in the system “liquid HEM-local source with a limited energy capacity-oxidant,” possibility of realization several ignition regimes has been established. Its regimes differ by the ignition time delay τ d and the location of oxidation zone that depend on the enthalpy of the energy source. It has been revealed that oxidation zone can be located in the gas region above the source with limited enthalpy, being a submerging particle near the lateral facet of the heat source partially immersed into the HEM, in the vapor gap between the particle and liquid near the lateral surface of the source. Besides, it has been established that, under no circumstances, does ignition occur in the vapor gap below the particle (Figure 1). The discovered feature was explained by the deficit of oxidant in this region, the substantial absorption of energy during the evaporation of the liquid combustible HEM, and considerable cooling down of the hot particle near the border “particle-liquid.”
The most typical regime, as shown by experiment in the course of changing the temperature and size of hot particles within quite a large range (0.9 < Θ p < 1.4, 0.1 < R p < 0.2), is the regime for which the inflammation zone is formed in the gas region above the immerging particle (Figure 2). It can be explained by the fact that the temperatures and concentrations of components of the vapor-gas mixture being sufficient for inflammation are reached only inside this region. In the course of evaporation, a large quantity of energy accumulated in the source is expended for the warming of the near-surface layer and for the phase transition. Consequently, in the vicinity of the border of the injection of HEM vapors, the concentrations of combustible are maximal; however, the temperature is insufficiently high for inflammation. Vapors temperature rose only after an additional warming due to heat dissipation from the particle. So, the oxidation acquires an irreversible character, causing ignition (Figure 2).

A frame of kerosene ignition by a single steel particle at the moment of flame emergence (τ d = 0.386) at Θ p = 1.2, R p = 0.15, and Z p = 0.15: 1: liquid HEM, 2: metallic particle, 3: flame, and 4: glass vessel.
It has been established that the higher the temperature of the particle (Θ p ), the larger the size of the vapor gap between surfaces of the particle and the liquid due to a large energy supply and, therefore, due to a more intense evaporation of the combustible. As Θ p increases, the depth of particle immersion into the combustible also increases. The size of the region in which the liquid combustible warms up becomes larger. Under these conditions, the most part of particle's energy is expended for the warming and evaporation of the liquid. The vapor-gas mixture warms up of the remaining energy. Therefore, the lower power consumption of hot particle the higher probability of inflammation absence of liquid. For instance, it has been established that, in the case of Θ p < 0.9, with the remaining conditions of the process being adequate, hot particles of metal become entirely immersed in a liquid HEM without reaching the inflammation conditions (Figure 3).

A frame of absence of kerosene ignition by a single steel particle (Θ p = 0.85,R p = 0.15, and Z p = 0.15): 1: liquid HEM, 2; metallic particle, 3: vapors of liquid HEM, and 4: glass vessel.
It can be explained by the fact that the course of immersion increases the contact area between the particle and the liquid and also increases the energy expended for the warming of the near-surface layer of the substance. Under these conditions, the source with a limited energy capacity cools down intensely and fails to heat the emerging vapors up to temperatures sufficient for inflammation (Figure 4). Besides, once the particle is entirely immersed in the liquid, the intensely heated combustible vapors become isolated from the oxidant by a layer of the HEM. Due to a sufficiently high dynamical pressure, these vapors pass through the layer of liquid and become involved in a reaction with the oxidant; however, the temperature in the reaction zone is sufficiently smaller than the temperature near the hot particle (Figure 4). Under the conditions of a partial immersion, however, both the emerging vapors and the oxidant warm up in direct contact with the hot particle (Figure 1). Therefore, the energy accumulated in the particle is sufficient for warming of both the liquid and the mixture of the combustible vapors with the oxidant. The established regularity makes it possible to conclude that the minimal durations of ignition delay can be reached by creating a contact with local energy sources being close to the “ideal” one, without their embedding into the near-surface layer of HEM.
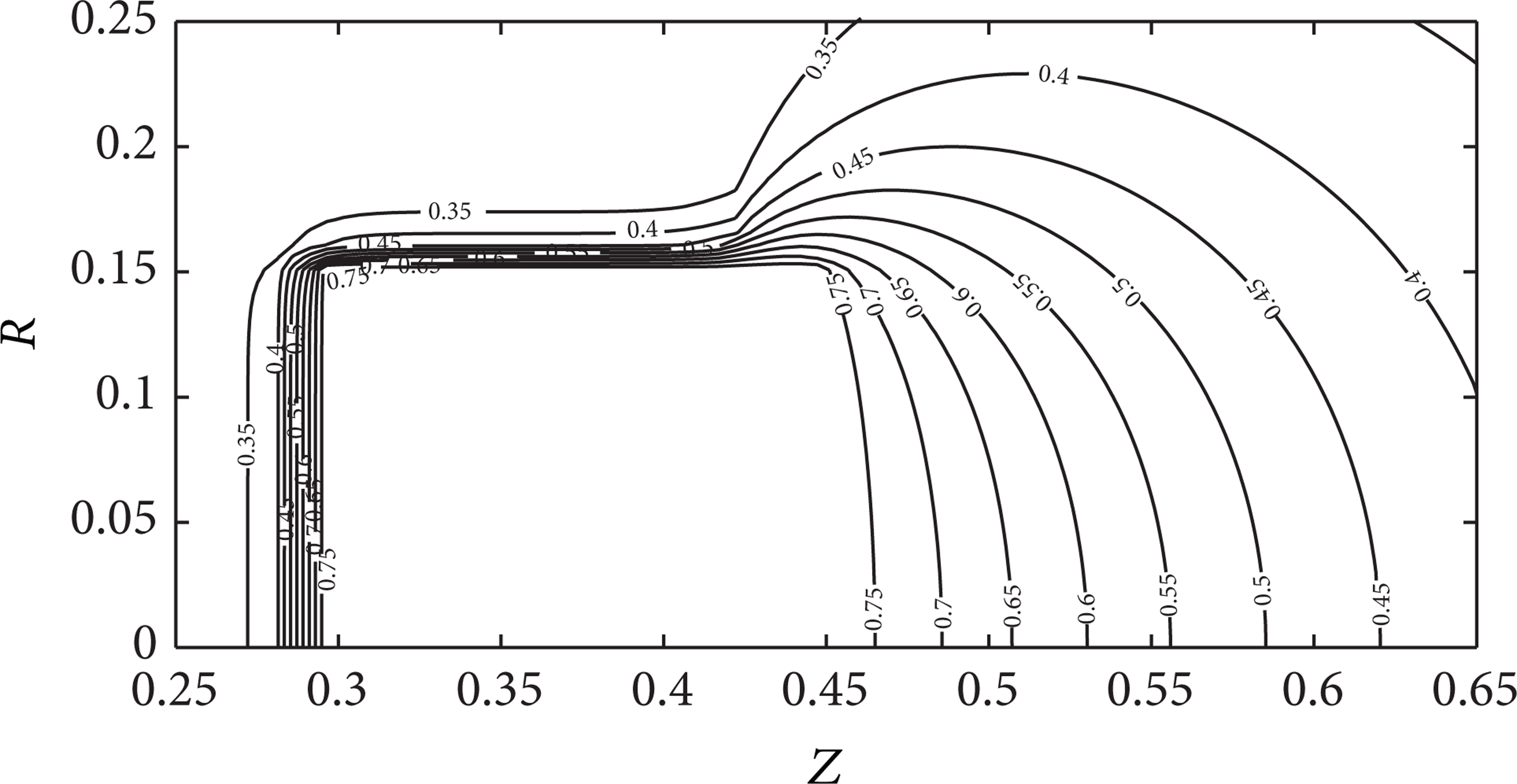
Isotherms (Θ) of the system “liquid HEM-metallic particle-air” at the complete immersion of the particle (τ = 3,Θ p = 0.85,R p = 0.15, and Z p = 0.15).
Figure 5 shows the surface τ d = τ(R p ,Θ p ) that symbolically separates the regions of inflammation (below the surface) and noninflammation (above the surface). We indicate the terminal values of the basic parameters (temperature and sizes) of a hot particle that permit inflammation. Thus, for instance, it has been discovered that, in the system under consideration (Figure 1), the temperature Θ p = 0.9 (at R p = 0.15 and Z p = 0.15) is the low boundary of the range of temperatures of a particle that permit the inflammation of a liquid HEM. We have also established, both experimentally and numerically, a similar boundary for the range of the particle's sizes at a fixed temperature. Thus, for instance, at Θ p = 1, ignition occurs only at R p ≥ 0.05 and Z p ≥ 0.15. The previous study of [21] has determined the configurations of energy sources for which the durations of delay are minimal (particles shaped as a parallelepiped) and for which the processes of ignition are characterized by the maximal sluggishness (particles shaped as spheres and hemispheres). The statement of the problem of heat and mass transfer with particles shaped as cylindrical disks is specified by average [21] values of the integral characteristics of ignition in comparison with particles shaped as a parallelepiped or a sphere. Consequently, in the model under consideration (Figure 1), the dependencies of τ d on Θ p and R p (Figure 5) characterize the upper and lower estimations of the ignition time delay for systems of particles shaped as a parallelepiped and a sphere, respectively.

The dependence of the ignition time delay τ d on Θ p and R p .
Figure 6 shows the dependence between the ignition time delay of the HEM and the temperature of the particle in comparison with the experimental data, obtained by the methods [8].
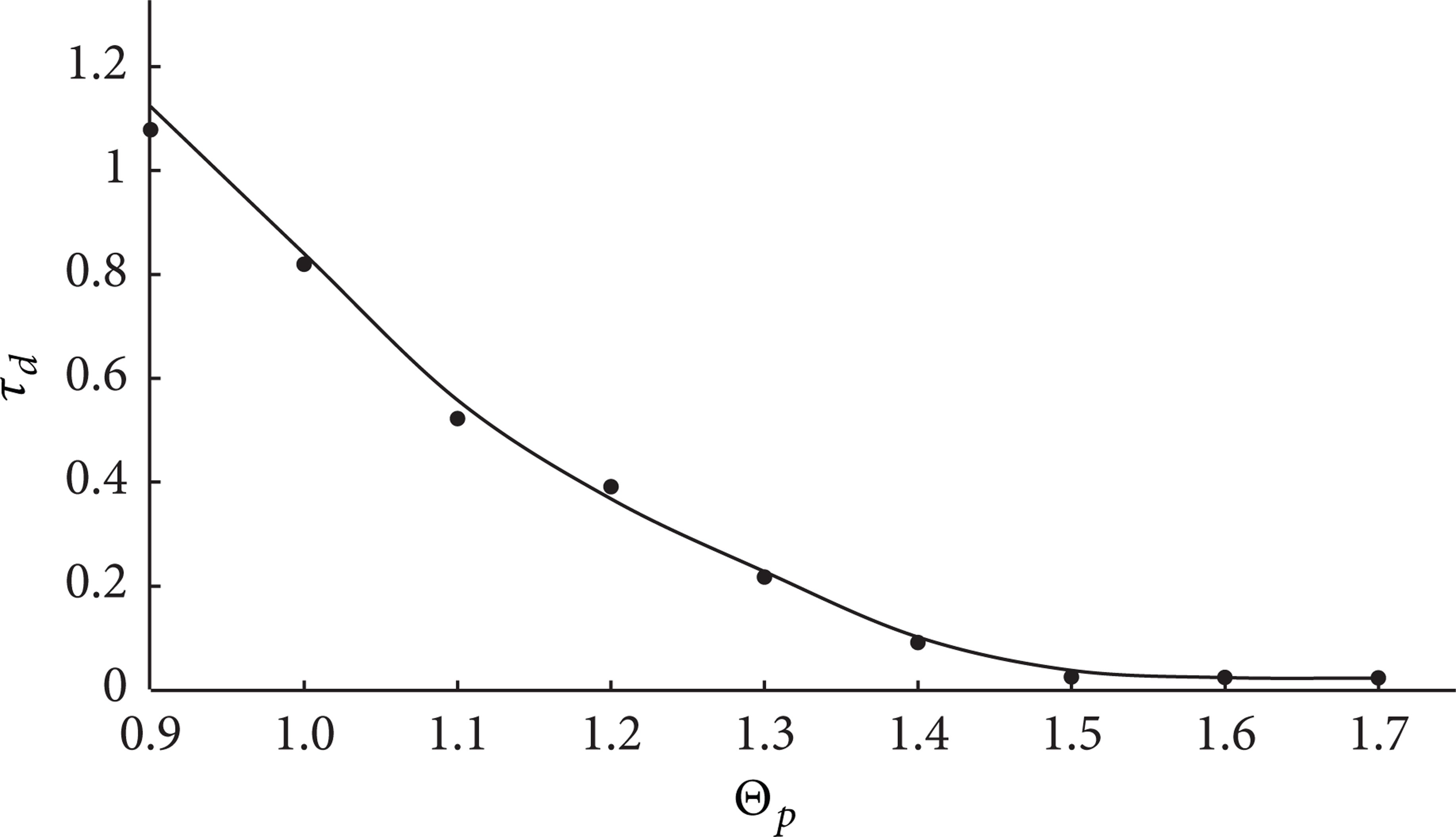
The dependence of the ignition time delay τ d on Θ at R p = 0.15 and Z p = 0.15 (•—experimental data).
One can observe that the difference of the values of ignition delay from the experimental data τ d is less than 5% in quite a large range of temperatures of the hot particle (0.9 < Θ p < 1.9). This result makes it possible to confirm the adequacy of the developed model of heat and mass transfer (Figure 1), as well as the correctness of the adopted assumptions. At the same time, besides the integral characteristics of ignition, a good correspondence with the experimental data has also been reached as regards the location of the inflammation zone, as well as the terminal temperatures and the sizes of local heat sources with a limited energy capacity.
5. Conclusion
We have shown both experimentally and numerically that inflammation of a liquid HEM is impossible in the case of a complete immersion of a typical source into it with a limited energy capacity, being an intensely heated metallic particle. It has been established that the complete immersion of a particle causes an intense evaporation of the liquid. However, the temperature of the injected vapors of HEM and that of the oxidant isolated by a layer of liquid are insufficient to accelerate the oxidation reaction. In the case of further immersion of the local energy source, its temperature decreases, and the velocity of evaporation too decreases, as well as the injection of the HEM vapors into the gas region. As a consequence, there occur a slow cooling down and immersion of the particle. It is possible to conclude that, once ignition has not occurred as a result of depositing onto the surface of a combustible liquid of an intensely heated particle, accompanied by its subsequent partial immersion, the conditions of inflammation will not be reached in the case of a complete immersion of the local energy source.
Footnotes
Nomenclature and Units
Conflict of Interests
The authors declare that there is no conflict of interests regarding the publication of this paper.
Acknowledgment
This work was supported by the Grant of the President of the Russian Federation (MK-620.2012.8).