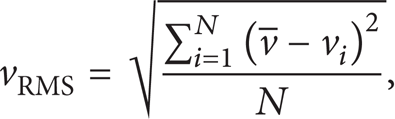Abstract
The Phase Doppler Particle Analyzer (PDPA) technique is utilized for characterizing the droplet sizes and velocity profiles in an electrostatic spray flow which is atomized using the aerodynamic forces assisted with electrostatic forces. An electrostatically-assisted twin-fluid atomization system is presented and spatial distributions of the droplet size and two-dimensional velocity components are measured simultaneously at each specified point. The droplets are firstly ejected from a liquid jet through the nozzle by aerodynamic shear forces, and the breakup mechanism of such an electrostatically-assisted twin-fluid atomization is investigated using the Rayleigh instability conditions through studying the specific surface charges on these droplets measured by a Faraday canister. The discrepancies between experiment data and Rayleigh limit for specific charges of droplets are presented and discussed due to energy transforming from electrical energy to surface energy, energy loss, Taylor instability breakup and nonexcess charge polarization. The measured average velocity profiles and their RMS (root mean square) velocity values for two different scale drops further reveal the effects of the aerodynamic and electrostatic forces on the atomization process.
1. Introduction
The twin-fluid atomization technology has been widely implemented in atomizing the highly viscous fluids such as the water-coal slurry, heavy oil, lime water, and pesticide liquids [1–5]. During atomization the excess energy is provided by a coflowing airstream which surrounds the liquid stream and causes the breakup and atomization of the liquid jet. In order to improve the atomization quality and to generate the desired sizes and spatial distribution of droplets, two different approaches have often been used. The first is to produce a large ratio of gas to liquid flow rates so that the aerodynamic shear forces applied by the gas stream are large enough to disintegrate the bulk liquid into small droplets. This method normally requires a very high gas pressure and hence has a stringent requirement of the mechanical properties of the gas supply line. The second method is to utilize the concept of electrostatic spraying through which the net charges are applied, through arranging an external electrical field, on the liquid surface and polarize it [4–7]. These charges, once accumulated, make the polarized structure very unstable and eventually break it up into smaller droplets. The electrostatic spraying process can be optimized through adjusting the ratio of the gas to liquid flow rates and the intensity of the external electrical field for achieving the best efficiency.
In the past 30 years, extensive studies and experiments have been carried out to avoid the disadvantages, improve the atomization quality, and expand the applications of the electrostatically assisted twin-fluid atomization for processing different nozzles, electrodes, and charging methods and using a variety of theoretical, numerical and experimental approaches [8–17]. Compared to conventional atomization techniques mainly by means of the aerodynamic forces, the electrostatically assisted twin-fluid atomization device generates a desired size distribution and sufficient surface charge droplets, which could be facilitated to improve deposition on various targets especially on the underside of the part and significantly reduce the drift of small inhalable droplets due to electrical forces. In addition, the agglomeration of highly charged droplets experiencing the Coulomb repulsion can be prevented, which could obtain the uniform spatial distribution, increase contacting opportunities, and enhance heat and mass transfer between droplets and surrounding media. The electrostatically assisted twin-fluid atomization is of great interest in many applications such as pesticide processing, industrial painting, fuel and water coal slurry atomization combustion, and flue gas treatment.
Savage and Hieftje experimentally investigated the effect of the electrical field on the spatial dispersion and size distribution of droplets generated by electrostatically assisted pneumatic nebulization and revealed a 63% decrease in the volume mean droplet diameter in the field's presence [8]. Law utilized the electrostatic-induction nozzle to atomize the pneumatically spraying and found that electrsotatically charged spray increased the mass transfer of pesticides onto plant targets and could save 50% consumption than conventional spray with the same efficacy [4]. Johnson et al. developed an embedded-electrode, internal-mixing, two-fluid, spray charging nozzle which was used to atomize and charge spray droplets in a pilot-scale milk spray-drying unit [10]. Im et al. experimentally investigated detailed information on the electrostatic rotary-bell paint spray structure and transfer processes and provided qualitative information on the atomization mechanism [12]. Laryea and No developed an electrostatic pressure-swirl nozzle and the result proved to be relevant due to the minimum pesticide input, increase deposition, and reduction in drift [13]. Jahannama et al. experimentally and theoretically investigated a two-flow, concentric, internal-mixing induction-charging nozzle for electrostatic spraying and found a significant difference between the uncharged and charged cases [14]. Wang et al. designed an electrostatic spraying twin-fluid nozzle, experimentally studied the drops size and velocity distribution, and revealed the effect of surface charge and high voltage on spray characteristics [6]. Almuhanna et al. developed a prototype electrostatically assisted particulate wet scrubber for controlling dust in livestock buildings and experimental data showed that charged water spray had significantly higher particle removal efficiency (79%) than either of the uncharged wet scrubber (58%) [16]. Wang et al. developed electrostatic atomization utilizing a combined annular and needle electrode which was coupled with a twin-fluid atomization device and investigated the characteristics of atomization and motion of droplets of uncharged and charged spray [7]. To quantify the spray shape and measure the droplet size distribution during electrostatically assisted atomization, laser based techniques have shown their merits in avoiding probe sampling which inevitably disturbs continuous spraying and the charging field. These approved approaches include the laser based Malvern instruments [18], Laser Doppler Velocimetry (LDV) [19], and Particle Imagine Velocimetry (PIV) [7]. Meanwhile the Phase Doppler Particle Analyzer (PDPA) technique has been developed for simultaneously measuring the size distribution and velocity components of droplets, which helps reveal the insights of the charged gas-droplets flow. Moreover, PDPA was used in characterizing the droplets under a variety of flow conditions such as within a high-pressure swirling spray [20], in a gas-particle flow [21], during a fluid-particle dispersion process [22], and in electrostatic atomization and spraying [12, 23–25]. Note that, although some significant progress has been made in developing the PDPA technique for characterizing the pure twin-fluid atomization, pure electrostatic atomization and sprays, useful experimental data, especially these twin-fluid atomization patterns associated with electrostatic forces and gas-droplets trajectories, are still scarce due to the inherent complexity and difficulty in in situ mapping of droplet sizes and the gas droplets two-phase velocity field.
In this present work, a twin-fluid aerodynamic atomization device is integrated with a combined annular-needle electrode which produces an electrostatic field. The design of this system, through utilizing both the aerodynamic and electrostatic forces, allows for easier and effective controls of the ratio of gas to liquid flow rates and the droplets charging voltage. We have mainly concentrated our efforts on gaining some insight into the effects of an electrical field on the instabilities, which improve the breakup of the jet in droplets, and on the motion of droplets which could enhance the contacting chance between droplets and surrounding gas. The two different scales of drops parameters including the spatial distribution of the droplet size, droplet velocity, and their root mean square (RMS) velocity profiles are obtained from PDPA. We also discuss the effects on motion of drops by aerodynamic and electrostatic forces especially smaller scale drops. It is expected that the testing apparatus and experimental data would provide a platform for further investigating the characteristics of electrostatically assisted twin-fluid atomization for liquids and multiphase flows with different applications.
2. Experimental
The current experimental apparatus is shown in Figure 1. The electrostatically assisted twin-fluid atomization device connects to its gas supply and liquid supply systems, as shown in Figure 1(a). The gas supply system includes an air compressor, a buffer tank with a flow meter, a valve, and a manometer. The gas supply system includes a water tank, a pump, a water buffer tank, a flow meter, a valve, and a manometer. The gas and liquid enter into the atomization nozzle, as shown in Figure 1(b), and form a spray flow. In order to charge the droplets more efficiently, the corona charging method is applied and the annular electrode combined with needles are chose, which could generate a strong electrical field and charge the high-speed fluid relatively easily. The electrostatic spraying is achieved when the spray jet is ejected into an electrical field which is generated by the annular-needle electrode and the grounded atomizer. The PDPA diagnostic system measures the droplet sizes and velocity in a horizontal plane which is 300 mm away from spray nozzle. The electrode is made of copper wires and the vertical distance between the electrode and the nozzle is set to 20 mm. The diameter of this electrode is 120 mm and eight needles are welded with an annular electrode at homogeneous distribution. This electrode is connected to a high-voltage electrostatic generator (HVEG) which supplies a negative voltage ranging from 0 to 30 kV. A Faraday canister, shown in Figure 1(c) and consisting of a copper wire net, a current meter, and a collector, measures the specific charges of liquid droplets. For processing the PDPA data, the software BAS Flow V2.1 is used. The major characterization parameters are found in some references [6, 7, 22, 25].
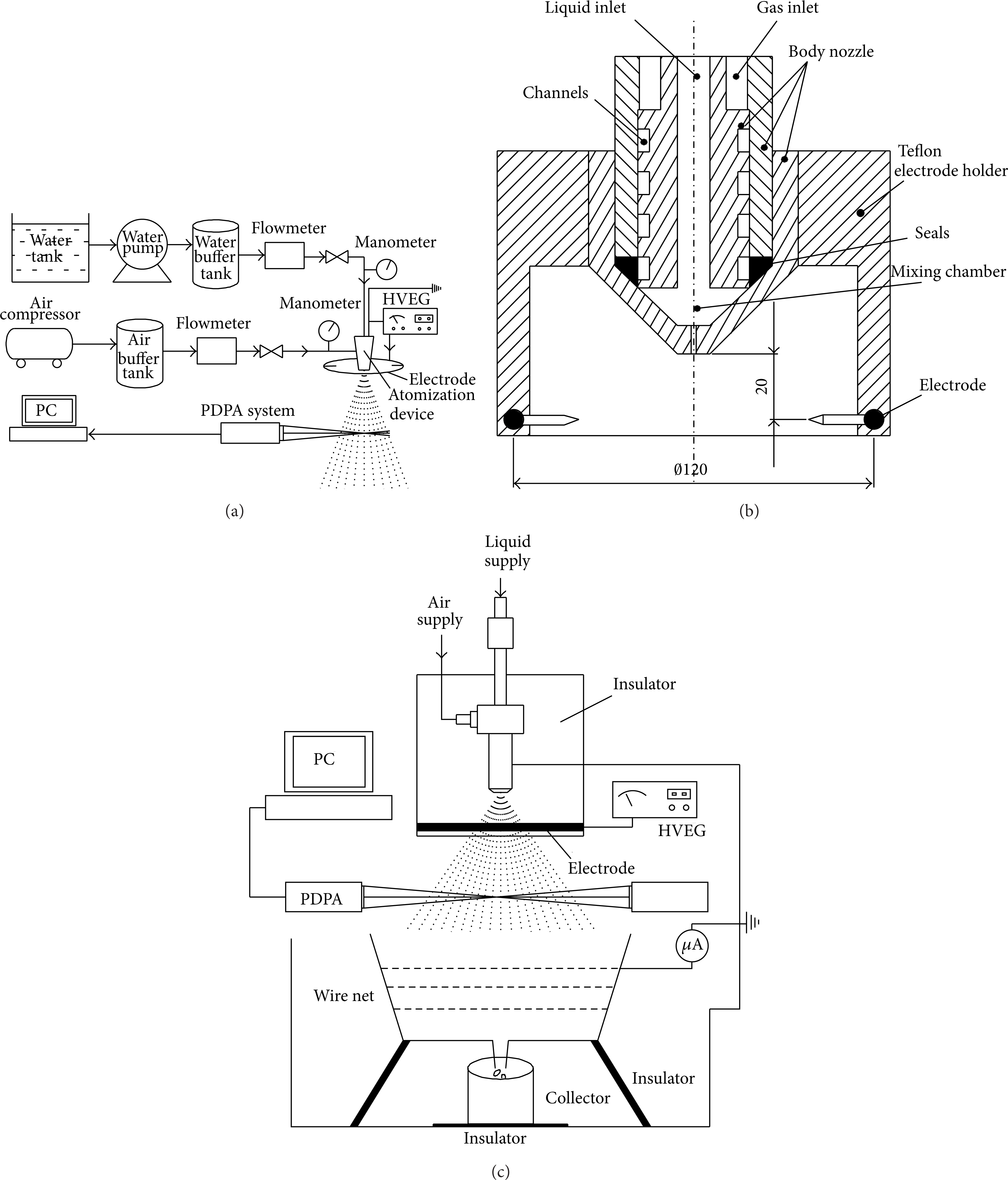
Schematic of the experimental apparatus. (a) The atomizer, its supply systems, electrode, and PDPA system; (b) schematic of the nozzle and electrode; and (c) atomization system, PDPA system, and the Faraday canister.
As mentioned earlier, the twin-fluid atomization can process a variety of high-viscosity liquids. In this study, the electrostatically assisted twin-fluid atomization system is used to process a charged spray flow passing through a controlled electrical field. After these steady-flow conditions are satisfied, the velocity and spatial distributions of droplet sizes are measured using PDPA under uncharged (i.e., without applying the external voltage) and charged conditions. Both aerodynamically atomized spray and electrostatically assisted dispersed spray exhibited the axial-symmetrical flow patterns. In order to characterize the flow along the r direction in the horizontal plane, eight measuring points are chosen, covering the maximum radius of 70 mm. The maximum vertical distance was 300 mm below the nozzle, as shown in Figure 2. Totally eight locations in r direction are characterized. At each measuring point, the average diameter of the droplets, the r and z components of the droplet velocity, and the corresponding RMS velocity are quantified using PDPA.

Schematic of the characterization locations of measuring points in the horizontal plane.
3. Results and Discussion
3.1. Spatial Distributions and Breakup of Drops
Using PDPA the average sizes and velocity components of droplets are measured at 8 specified points in the horizontal radial plane (z = 300 mm) when it moves from the r = 0 to r = 70 mm. At each point, the drops size and velocity components of the droplets are determined in both r and z directions. The liquid is limewater and the concentration of limewater is 3.0% Ca(OH)2 in tap water (the conductivity of limewater is around 0.2's/m in our experimental conditions). When these measurements are taken, a stable spray flow is obtained using the following fluid parameters: 0.25 MPa in gas and water tanks, the gas flow rate of 7.50 mL/s, and the liquid flow rate of 11.12 mL/s. Figure 3(a) shows the diameter and velocity frequency on the centerline with a distance of 300 mm to the nozzle under 15 kV. Figure 3(b) shows the PDPA measurements of the arithmetic mean droplet sizes and velocity vectors when the measuring plane moves from the nozzle to z = 300 mm. In Figure 3(b), the average sizes of droplets are visualized by the sizes of representative spheres while the lengths of the arrows represent the magnitudes of the velocity vectors. This represents a typical case of the aerodynamics driving atomization, which are the shear forces between the liquid stream and the surrounding gas stream in the mixing chamber of nozzle. When droplets move down and experience the electrostatic force, the large droplets break up and form finer ones (in fact, the droplets are produced in the nozzle by coupling aerodynamic and electrostatic forces). The further droplets move downwards; the more small ones are formed. Across the horizontal plane, however, more uniform droplets exist inside of the spray. Figure 3(b) shows the velocity vectors as well. Generally the velocity profiles exhibit a boundary layer flow pattern in which the maximum velocity magnitude exists at the center of the spray. When droplets move down and experience the electrostatic forces, the maximum value decreases while the spray extends in r direction. Figure 4 shows the measured average droplet size (SMD) at the z = 300 mm plane for different voltages. First of all, all curves show the same shape with the maxima at both edges of the spray. The smallest droplet size is found in the centerline and increases gradually along the horizontal radial direction. As explained earlier, this shape is mainly caused by effects of the aerodynamic forces and is in accordance with twin-fluid atomization. Secondly, the curves become flatter, showing a more uniform size distribution which is caused by effects of the electrostatic forces across the vertical direction, when the applied voltage becomes larger. This confirms that electrostatic atomization helps generate monodispersed droplets in this situation. Meanwhile, the average droplets size is the largest at each horizontal radial position when the electrical field is not applied. With the external electrical field, the droplets size decreases with the increasing voltage. This suggests that the electrostatic force affects the average sizes of droplets. Figure 5 shows the measured average droplets size, specific charges on the droplets, and Rayleigh limit calculated for given experimental conditions. It clearly demonstrates that increasing the external voltage would generate more surface charges on the droplets and result in smaller droplets from better atomization. For the uncharged spray (with U = 0), the average droplets size at the center line (r = 0) is about 140 μm while at the same location for the charged sprays (U = 20.0 kV) the average droplet size decreases to around 108 μm. The effect of the electrical voltage on the droplets size distribution hardly changes any more when the voltage becomes larger and larger (e.g., when U>15.0) and two bottom curves are found close to each other. It is well known that, in pure electrostatic atomization, the formation of drops could be explained by Rayleigh instability, shown in Figure 6(a) [26]. In fact, these individually charged droplets usually break up at sub-Rayleigh limit [27]. The Rayleigh instability could be expressed as the follows:
where q is the surface charges of the droplet, η is the Rayleigh limit coefficient (η ≤ 1.0), ε0 is the permittivity of vacuum, σ is the liquid surface tension, and r is the original radius of the droplet.

The spatial distributions of droplet sizes and velocities at 15.0 kV. (a) Frequency of velocity and arithmetic mean diameter distribution at the centerline (z = 300 mm) and (b) representative spheres and velocity vectors.
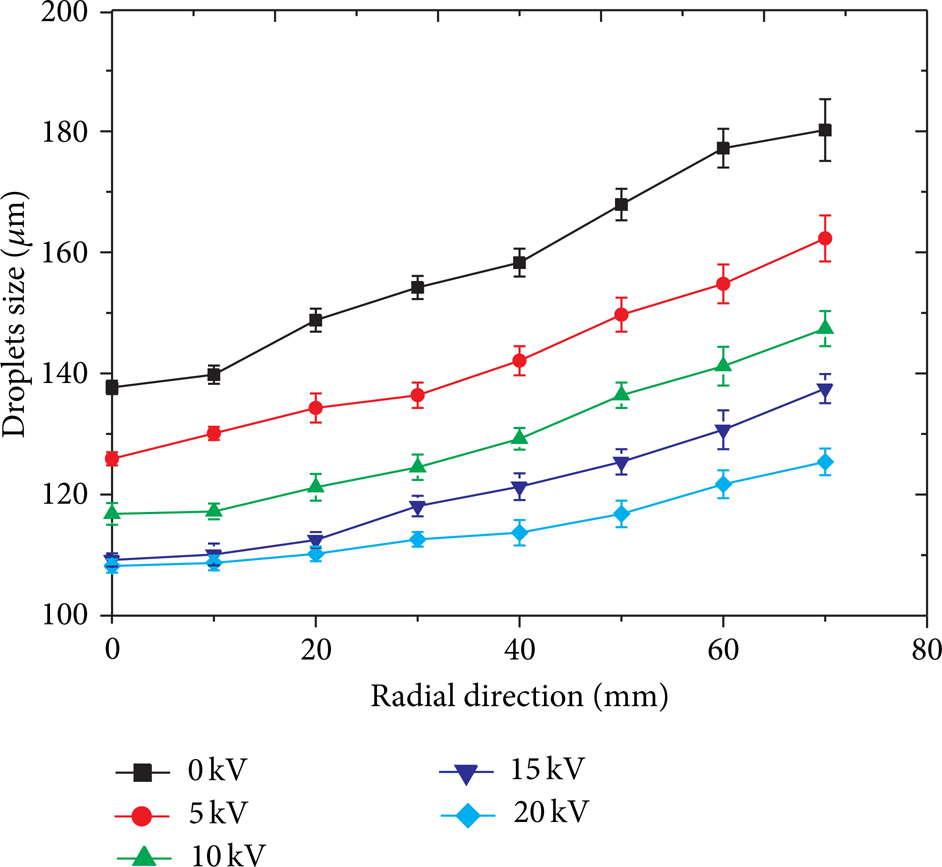
Measured average droplets size (SMD) at the z = 300 mm plane for different external voltages.

The average droplets size, measured specific charges, and calculated specific charges based on Rayleigh limit.
Rayleigh and Taylor instability and breakup mechanism of the droplets during electrostatic atomization. (a) Rayleigh instability breakup and (b) Taylor instability breakup.
The breakup mechanism of liquid jet and droplets could be interpreted by aerodynamic shear forces and Rayleigh instability conditions. In an electrostatically assisted twin-fluid atomization, two-stage atomization mechanism and processes could be assumed. In fact, the aerodynamic and electrostatic forces usually couple each other in the process of the electrostatically-assisted twin fluid atomization. The first stage is the aerodynamic forces dominant atomization which are ejected from the nozzle and assisted by electrostatic forces. At this stage the kinetic energy of the droplet flow is initially carried by both gas and liquid streams and its intensity increases with increasing flow rates. The shear forces exist in the mixing chamber of nozzle between the gas and liquid streams and their value increases when the ratio of gas to liquid flow rates increases. These shear forces cause disintegration of the liquid jet stream into space with droplets of relatively larger sizes with the assistance by perturbation due to external electrostatic field. During this first stage of breakup (or the aerodynamic atomization), the total energy and momentum of the twin-fluid flow are conserved. After the droplets enter into the electrical field, net charges are applied when produced at the outlet of nozzle and accumulated on the whole or part surface of droplets. When external electrostatic field is applied, these charges could polarize the droplets and generate the localized electrostatic stresses on the surface, which tend to expand and flatten the surface. Meanwhile the surface tension holds and minimizes the surface area. When the latter cannot balance the electrostatic stresses, the large droplets disintegrate and smaller ones form. During this second stage the breakup of droplets is mainly caused by the electrostatic forces and the total energy and momentum of the spray flow are still conserved by taking into account the additional contributions from the external electrical field. The newly generated small droplets, if flowing through the electrical field, repeat the second-stage atomization procedure until certain conditions of their stability are met.
In the abovementioned explanations, Rayleigh limit is used to examine the relationship between the surface charges and the breakup mechanism. Note that the specific charges are measured using a Faraday canister and for existing droplets in the field [6]. If the Rayleigh limit of a large droplet was reached, this droplet would break up and form a few smaller droplets which have lower specific surface charges than the previous droplet. On the other hand, the diameter of existing droplets should be smaller than (but close to) their critical diameter corresponding to the Rayleigh limit. The measured specific charges are lower than the critical values calculated from the Rayleigh limit (about 32%). This would argue about the validity of the Rayleigh instability conditions in describing breakup mechanism. There exist several explanations for the discrepancies between experimental and theoretical results. The specific charges are measured after disintegration of droplets. In electrostatic atomization, droplets formed from breakup would get much more surface tension energy and this energy is come from electrical energy which the original droplet holds. The electrical energy would reduce because of increasing the surface and total energy loss due to discharging towards surrounding media. In a nonuniform electrical field, the droplets would be polarized and hold negative and positive charges on the opposite poles. Consequently, on the pole area, the surface charge density is higher than droplets equator area. And surface tension may be less than the electrostatic stresses, because specific charges on this area may be higher than critical charges or measured values. And a droplet may be disintegrated into two or more finer drops beginning from one of the pole areas. On the other hand, the shape of the droplet tends to deform into a flat and long ellipsoid due to polarization force which is generated by nonsuperfluous (nonexcess) polarization phenomenon. This process is named as the Taylor instability condition and is shown in Figure 6(b) [28]. Obviously, the measured specific charges are net charges, which could not take this Taylor effect into account. Note that the Taylor effect becomes much more significant when the external electrical field becomes stronger. When the length of the semimajor axis of the ellipsoid is longer than 1.9 times that of the semiminor axis, the breakup of the droplet would happen. Obviously, the shear stresses existing between droplets and surrounding gas also influence the disintegration of drops in spray flow, and this is a very important factor. Shrimpton pointed out that these individually charged droplets usually break up between 70% and 80% of Rayleigh limit, while the charged droplets within a spray plume could break up at 55% of this limit [27]. So, the results shown in Figure 5 do not conflict with this previous finding.
3.2. Velocity Measurements of Droplets
In addition to the spatial distribution of droplets size, PDPA measurements simultaneously characterize the spatial distribution of droplets velocity. Figure 7 shows the measured magnitudes of velocity components in the horizontal radial and vertical directions (i.e., r and z), respectively. The measurements are done at the z = 300 mm plane with different external voltages. Note that the dataset for U = 0 refers to the uncharged spray which is driven by the aerodynamic forces solely. Generally speaking, applying the external electrical field does not bring in dramatic changes in the velocity distribution. This is confirmed by the difficult distinction of five data being set in both horizontal radial and vertical directions. Some interesting characteristics do exist however. For these profiles of the z-component velocity, most curves show a Gaussian shape with the maximum at the center line. Close to the edge of the spray, droplets in the uncharged spray possess slightly larger magnitudes. For the r-component velocity, their magnitudes are much smaller than the z-component velocity. This indicates the aerodynamic forces with fewer contributions of the electrostatic forces on the velocity profile. However, the velocity of droplets should be indeed size dependent and affected by the electrostatic field. When the droplet diameter is small enough, its motion will be influenced by the electrostatic force (through the Coulomb and polarization forces) and this effect could be quantified using a nondimensional number γ:
where E max is the intensity of the external electrical field, ρ is the liquid density, dp is the droplet diameter, ε0 is the vacuum permittivity, χ is a constant and usually set to 1/3, and g is the acceleration of gravity. Wang et al. indicated that if γ is greater than 10, the droplet motion will be significantly affected by the electrostatic forces [6]. Using (2) and γ = 10, the critical droplet diameter is calculated as 15.5 μm under the experimental conditions. As shown in Figures 4 and 5, the detected average droplets in this present work are much larger than 15.5 μm, which suggests that the effects of the electrostatic force on the bigger droplets motion are negligible, while smaller droplets motion is very important.

Velocity profiles of droplets measured for different voltages (z, r denote two different directions, resp.).
Although the droplet velocity is found to be dominantly controlled by the aerodynamic force, there still exist some important effects from the electrostatic force. These effects can be presented using the RMS velocity of a droplet. In this study, the RMS velocity is derived from the PDPA measured velocity profiles using the following:
where vRMS is the RMS velocity, vi is the measured velocity component for the droplet i at a location,

RMS velocity derived from the droplet velocity (z, r denote two different directions, resp.).
3.3. Velocity and RMS Velocity Comparisons of Two Different Scales Droplets
The twin-fluid electrostatic atomization system produces a two-phase flow which consists of moving gas and liquid droplets. In fact, the gas velocity could not be detected by PDPA. In the present work, we use the smaller scale drops to characterize the gas phase when the droplet sizes are smaller than 10 μm, and the larger scale drops would be considered as a dispersed flow of droplets when the droplet sizes are larger than 10 μm. The voltage is set to 10 kV. Figure 9 shows the average velocity profiles of the smaller and larger scale drops in z and r directions, respectively. The comparisons shown in these figures on the velocity profiles of the smaller and larger scale drops demonstrate that, inside of the spray and close to its core, the smaller scale drops move with larger scale drops, and the velocity is shown in both r and z directions. This suggests that the aerodynamic forces play a dominant role in determining the spray flow characteristics inside of the spray. In the outer area of the spray plume, the velocity of smaller scale drops is slightly smaller than the larger scale drops, which shows the effects of the electrostatic force on the motion of smaller scale drops in r direction. The difference between the z-component velocity of smaller and larger scale drops can be explained by the greater inertia of larger sale drops as well. In z direction, larger scale drops in the outer area of the spray should have larger velocity magnitude than smaller scale drops due to larger mass, while at the core area larger scale drops have the same level with the smaller scale drops because of the aerodynamic forces dominant motion of all the drops. In r direction, two different scale drops also have the same level velocity in the measurement points. The r-component is less than 0.6 m/s. The velocity difference in absolute value between smaller and larger scale drops is not apparent, while in relative value it is about 25% at the point of z = 300 mm and r = 60 mm. So, the effect on the motion of drops in charged spray flow due to electrostatic forces is obvious.

Velocity of two different scales of drops (z, r denote two different directions, resp.).
Figure 10 shows the calculated RMS velocity for the two different scale drop spray plumes. The experimental conditions are set to the same as Figure 9. It can be easily observed that the RMS profiles of larger scale droplets do not overlap the RMS profiles of the smaller scale drops in both r and z directions. And in z direction, as shown in Figure 10, the RMS velocity of larger scale drops is larger than that of the smaller scale drops, while in r direction the RMS velocity of larger scale drops is smaller than that of the smaller scale drops. These observations can be explained by the pure aerodynamic behavior of the smaller scale drops but the combined aerodynamic and electrostatic phenomena associated with droplets. As mentioned earlier, droplets endure both the aerodynamic and electrostatic forces and their motions are controlled by the combination of two forces. In z direction, the RMS velocities of larger scale drops are uniform across the z = 300 plane, which confirms a better dispersion of finer droplets. In the same direction, the RMS velocity of the smaller scale drops presents much more fluctuations at the edge of spray plume, mainly due to electrostatic forces, but has no much more fluctuations at the core area of spray due to aerodynamic forces. In the r direction, the RMS velocity of larger scale drops is lighter than the smaller scale drops, which result from the extra electrostatic forces applied on those droplets.

RMS velocity of two different scales of drops (z, r denote two different directions, resp.).
4. Conclusions
An electrostatically assisted twin-fluid atomization system is developed using a gas and liquid supplied nozzle integrated with a combined annular and needle electrode. PDPA measurements are utilized to characterize the spatial distributions of droplets size and velocity. Experimental data reveals that the droplets size is influenced by the external electrical field, while the droplet velocity is controlled by the aerodynamic force. The breakup mechanism of jet is explained by aerodynamic shear forces and the secondary breakup of droplets due to external electrostatic field is illustrated using Rayleigh instability conditions and supported by the measurement of specific surface charges on droplets. During the assumption of two-stage atomization process, the aerodynamic force disintegrates the liquid jet into larger droplets which are further ruptured by the applied electrostatic force into monodispersed droplets in the downstream of the spray. There are some discrepancies between experiment data and Rayleigh limit for specific charges of droplets, and some reasons which are gas shear stress, energy transforming from electrical energy to surface energy, energy loss, Taylor instability breakup mode, and nonexcess polarization are analyzed to meet these discrepancies. The measured velocity data further explains the roles of the aerodynamic and electrostatic forces. First, the motion of smaller scale drops is affected by electrostatic forces in electrostatic field. Secondly, the average velocity of larger scale drops overlaps with smaller scale drops, which show that the aerodynamic forces are dominant factors for velocity of drops. Thirdly, the RMS velocities of the smaller scale drops do not overlap those larger scale drops, thanks to electrostatic forces on smaller scale drops.
Footnotes
Nomenclature
Conflict of Interests
The authors declare that there is no conflict of interests regarding the publication of this paper.
Acknowledgments
This work is supported by National Natural Science Foundation of China (nos. 51106064 and 51006047), China Postdoctoral Science Foundation funded project (2012M511214), Jiangsu Postdoctoral Science Foundation (no. 1101149C), and Jiangsu Government Scholarship for Overseas Studies. The authors would like to express their gratitude to anonymous reviewers and also appreciate Dr. Changfeng Li help in revising this paper at Jiangsu University.