Abstract
A new method of near-liquidus squeeze casting (NLSC), which obtains globular crystal, has been developed. The research focuses on the formation and growth mechanism of globular crystals of ADC12 alloy by NLSC. Globular crystal transforms into rosette crystal, average grain size (AGS) increases, and average globular coefficient (ASC) decreases with the rise of pouring temperature. But AGS decreases to 17.8 μm directly and ASC increases directly to 0.46 when the pouring temperature is 650°C. NLSC globular crystal of different sizes (AGS is 22.5 μm; ASC is 0.73) distributes uniformly in matrix. Heterogeneous nucleation grows on the solid particles of metal walls and fusing grains. The formation and growth modes of globular crystal include normal grain growth, annexation of small globular crystal, and fracture and spherulization of rosette crystal. NLSC globular crystal critical radius is inversely proportional to degree of supercooling. The pressure has very little influence on critical radius by increase of melting point and thermal conductivity, but it affects critical radius greatly by changing the degree of supercooling. NLSC globular crystal has no enough time to exceed the critical radius.
1. Introduction
Semisolid processing is an advanced near-net shaping technology [1, 2]. The key of semisolid processing is to prepare semisolid slurries [3]. There are many methods to prepare semisolid slurries, for example, mechanical stirring [4], electromagnetic stirring [5], SIMA method [6], and near-liquidus method [7]. Near-liquidus casting is a method which obtains semisolid billet in which alloy billets need preserving heat at near-liquidus temperature [8]. But, with holding time prolongs, globular crystal grows unstably and transforms to dendrite crystal [9]. Only under the conditions of right and short time, semisolid slurries can be made via near-liquidus method.
Squeeze casting is an advanced process that fills the mold and solidifies under pressure [10–12]. For nonimpediment of pressure transmission, the pouring temperature of traditional squeeze casting is high. Sun et al. [12] researched the simulation of heat flow and solidification of squeeze casting A356 aluminum alloy (liquidus temperature was 615°C) and the pouring temperature 700°C was chosen. Youn et al. [13] researched the relationship between squeeze casting parameter and mechanical behavior of AZ91-Ca magnesium alloy (liquidus temperature was 595°C) and the pouring temperature was 700–800°C. However, the semisolid processing temperature is between the liquidus and solidus temperature. It is a blank of indirect squeeze casting at near-liquidus temperature. NLSC which combines the near-liquid method with the squeeze casting has been developed. The advantage of NLSC is that parts are composed by globular crystal with no heat preservation of molten metal and no preparation of semisolid slurries or billets. The growth rule of α phase has been researched by the support part of ADC12 aluminum alloy under different pouring temperature. The research focuses on the formation and growth mechanism of globular crystals under near-liquidus (590°C).
2. Experiment
The chemical composition of ADC12 aluminum alloy is shown in Table 1. The liquidus and solidus temperature of the alloy measured by DTA were 592°C and 539°C, respectively. ADC12 aluminum alloy was melt in the electrical resistance furnace. The temperature rose to 720°C and then held for 5 minute. The refining agent of 15% alloy weight was added into molten alloy. The molten alloy was poured into the holding furnace and was ready for using, after refining and deslagging. Temperature controlling accuracy of the holding furnace was ± 1°C. Indirect squeeze casting machine SCH-350 was used and the process of which is shown in Figure 1. Mold temperature controlled by mold temperature controller was 250 ± 10°C. The pressure was 150 MPa and the holding time was 15 s. The squeeze casting proceeded immediately when the holding temperature reduced to 680°C, 650°C, 620°C, and 590°C, respectively. The experiment part was the support, which is shown in Figure 2(a). The sample was chosen at the thinnest place (11 mm) of the support, which is shown in Figure 2(b). In order to compare the microstructure, the gravity casting was proceeded in the same mold. The sample was chosen at the same place as the squeeze casting. The pouring temperature and mold temperature of the gravity casting were 250 ± 10°C and 590°C, respectively.
Chemical composition of ADC12 aluminum alloy (wt%).

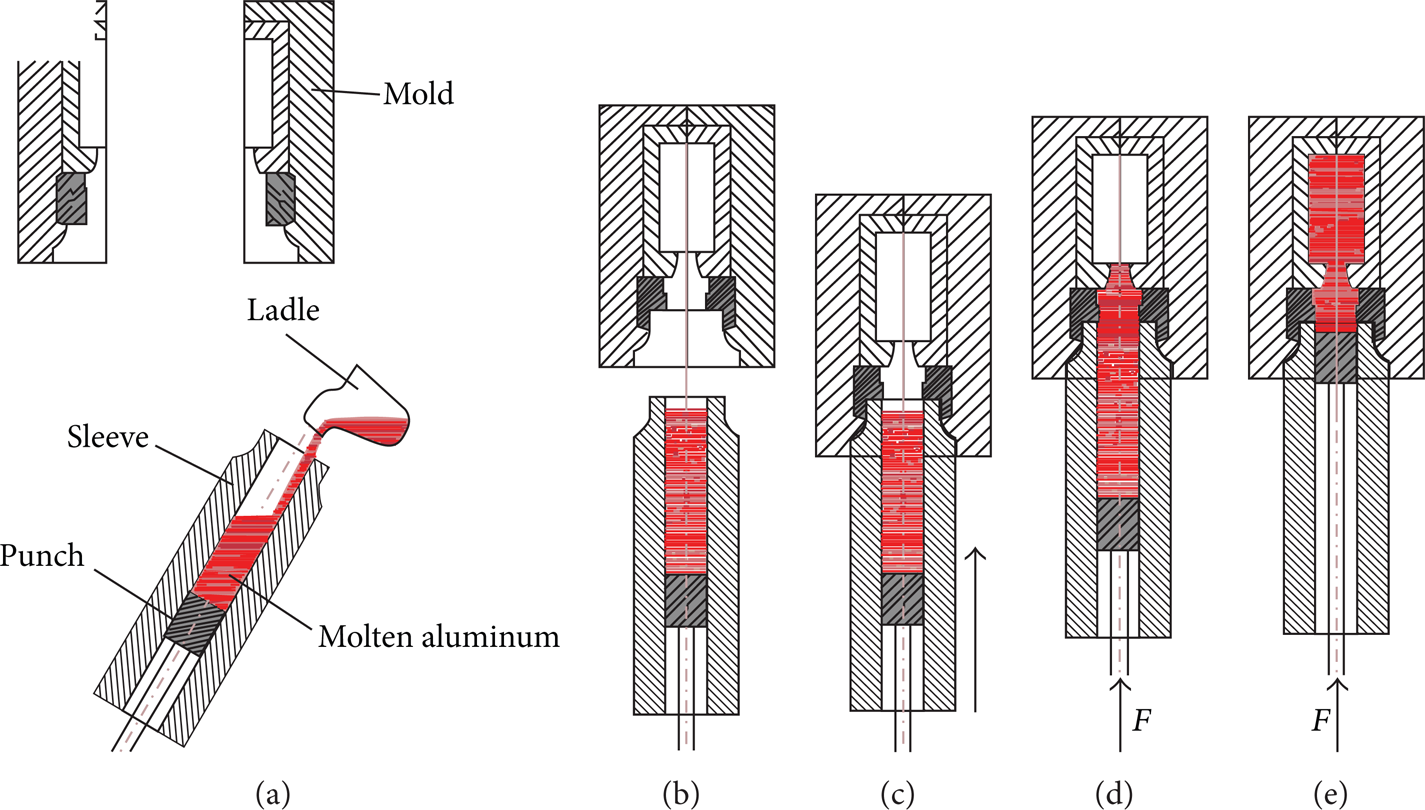
Schematic diagram of the process of indirect squeeze casting: (a) molten alloy was poured into heated sleeve after the sleeve has tilted 30°, (b)-(c) sleeve was moved back to its original position and rose to cooperate with mold, (d) punch rose fast until molten alloy got to flow gate, and (e) molten alloy was slowly filled to cavity and solidified under pressure.

The parts and samples location of indirect squeeze casting and gravity casting: (a) the support of indirect squeeze casting and gravity casting and (b) samples location of indirect squeeze casting and gravity casting.
Used for observing the microstructure, the samples were grinded and polished and then corroded by 0.5% HF for 10 s. The microstructure was observed by optical microscope. The image processing software Image-Pro Plus was used for calculating the AGS (d) and ASC (μ) of α (Al) phase. The equation [17] gives

where S is sectional area of α (Al) phase. C is perimeter of section of globular coefficient. μ is globular coefficient varied from 0 to 1 and with the value that tends to “1” it tends more to be round.
3. Experimental Results
3.1. The Effect of Pouring Temperature on Squeeze Casting Microstructure
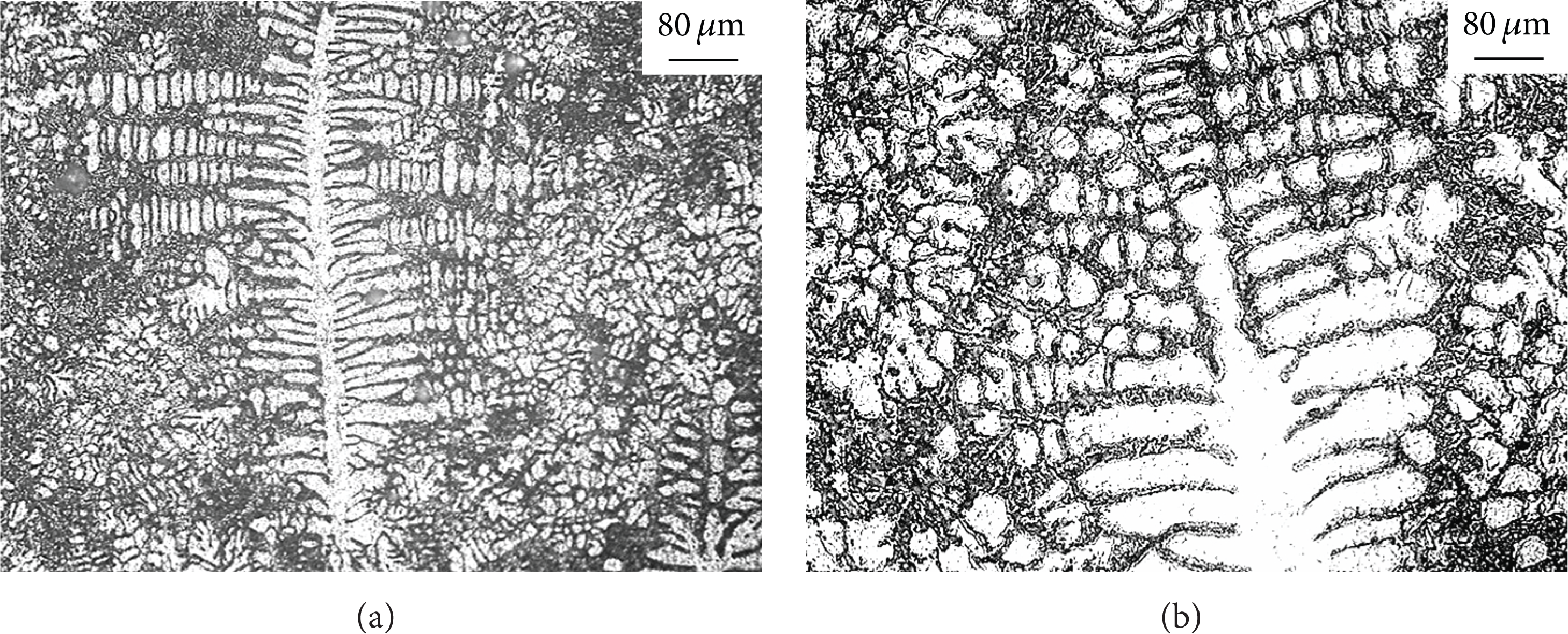
Figures 3(a) and 4 show that different sizes of globular crystal which have larger globular coefficient distribute uniformly in matrix, ASC and AGS of which are 0.73 and 22.5 μm, respectively. When the temperature is 620°C, globular crystal gradually grows into rosette or dendritic crystal. ASC decreases to 0.37 and AGS increases to 38.5 μm, as shown in Figures 3(b) and 4. When pouring temperature is 650°C, the grain of different shape distributes in the matrix which is fractured dendrite. ASC increases to 0.46 and AGS decreases to 17.8 μm, as shown in Figures 3(c) and 4. When pouring temperature is 680°C, the dendrite crystal and fractured dendrite are observed in Figure 3(d). ASC decreases to 0.26 and AGS increases to 28.7 μm, as shown in Figure 4.

The microstructure of the support of SC at different pouring temperature: (a) 590°C, (b) 620°C, (c) 650°C, and (d) 680°C.

Relationship between pouring temperature and grain size and globular coefficient of the ADC12 aluminum alloy support.
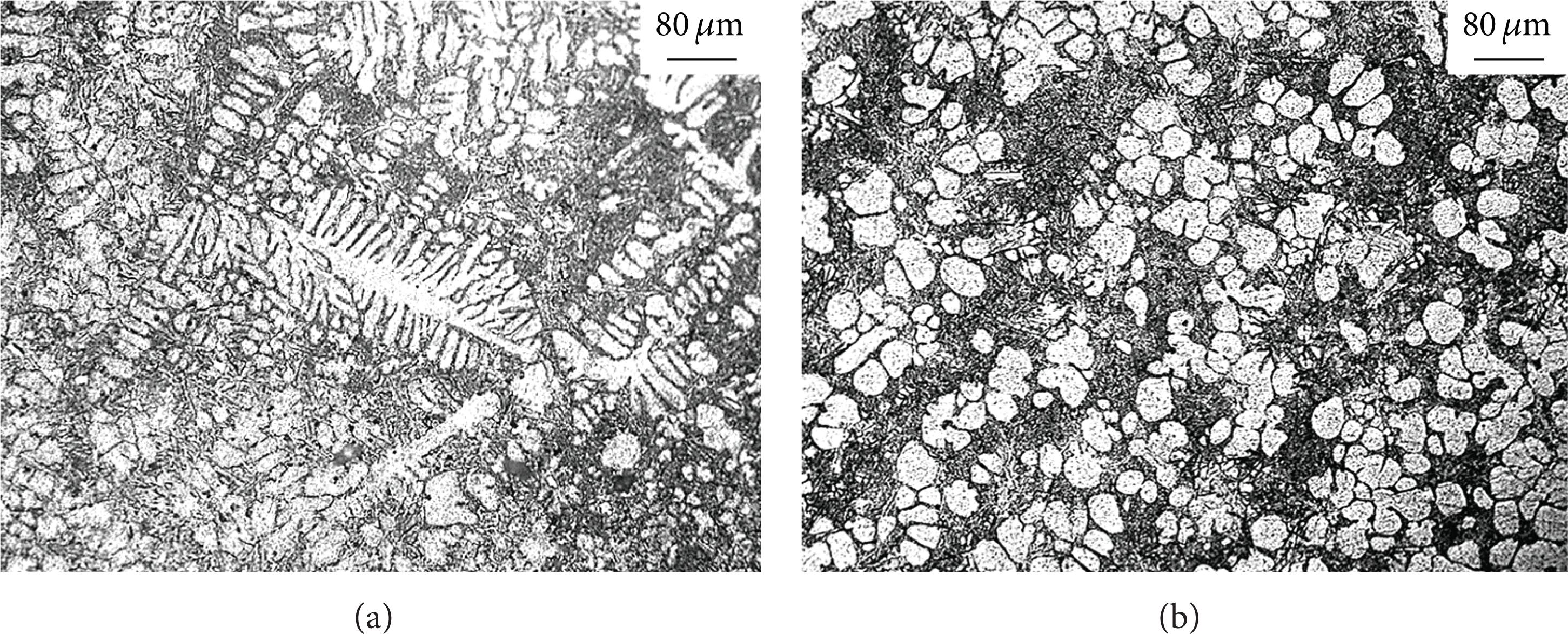
Figure 5 shows the microstructure of remaining fractured dendrite of squeeze casting at pouring temperature 650°C. The grain grows to dendrite first and then to fractures, which contains the facture of the secondary dendrite arm and the primary dendrite, as shown in Figures 5(a) and 5(b). When the pouring temperature is 650°C beyond the liquidus temperature 60°C, molten alloy flows into sleeve and runner and has no crystal nucleus, as molten alloy does not decrease below liquidus temperature. The molten alloy begins to solidify after the molten alloy is full of cavity. The crystal nucleus has enough time to grow to dendrite crystal with the increase of pouring temperature. The bigger pressure acts on the solidifying molten alloy, for there is no loss of pressure in runner at pouring temperature 650°C. The bigger pressure and liquid flowing of feeding promote the fracture of dendrite. As the flow of the feeding action is not severe, the fractured grains cannot distribute uniformly to the matrix but stay near the dendrite crystal. The outline of the dendrite crystal is observed in Figure 3(c).
The microstructure of remaining fractured dendrite of squeeze casting at pouring temperature 650°C: (a) fusing of the secondary dendrite arm and (b) fusing of the secondary dendrite arm and the primary dendrite.
From the above analysis, it can be obtained that, with the increase of pouring temperature and solidification time, the AGS increases and the ASC decreases. At 650°C, as there is no primary nuclei generation and due to the dendrites fracture with higher pressure, the grain size decreases bluffly while the globular coefficient increases slightly. As the pouring temperature continues rising, the fractured grain continues to grow into dendrite. The phenomenon appeared again that AGS increases and ASC decreases with the rise of pouring temperature.
3.2. The Microstructure of NLSC
Figure 6(a) shows the microstructure of gravity casting composed by thick dendrite crystal. Globular crystal of different sizes distributes uniformly in matrix in NLSC, as shown in Figure 6(b). In order to distinguish between the dendrite crystal and globular crystal at microstructure of two dimensions, three vertical planes of NLSC sample were observed by metallographic microscope. Figure 7 was acquired by combining the metallographs of three vertical planes. The globular crystal is not the section of the dendrite crystal and distributes in the microstructure of three vertical planes as shown in Figure 7.
Microstructure of gravity casting and NLSC of ADC12 alloy: (a) gravity casting and (b) NLSC.

Microstructure of three vertical surfaces of the support by NLSC.
4. Discussion
4.1. The Mode of Nucleation of NLSC
NLSC is a complex process. The nucleation contains heterogeneous nucleation and homogeneous nucleation. Homogeneous nucleation includes small grain of homogeneous nucleation that exists at matrix of the solidification end, as shown in Figure 8. The grain which grows by homogeneous nucleation tends to aggregation. The temperature instability of solid-liquid interphase is caused by degree of supercooling, during the nucleation forming. The effect of pressure on degree of supercooling may roughly be given as follows [18]:
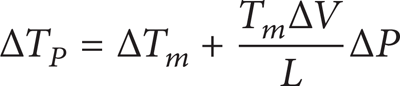
where ΔT m is the degree of supercooling under ordinary pressure. ΔT P is the degree of supercooling under pressure P. T m is the melting point under ordinary pressure. L is the latent heat of crystallization per mass. ΔP and ΔV are the increment of pressure and volume. ΔT P is bigger than the degree of supercooling under ordinary pressure ΔT m . The degree of supercooling of NLSC supplies greater force for nucleation. As molten metal directly contacted with mold under the pressure, greater heat exchange coefficient is obtained [19]. Molten metal flows fast and has self-stirring effect. It promoted emergence of the heat fluctuation and the phase fluctuation which are benefits to emergence of crystal clusters.
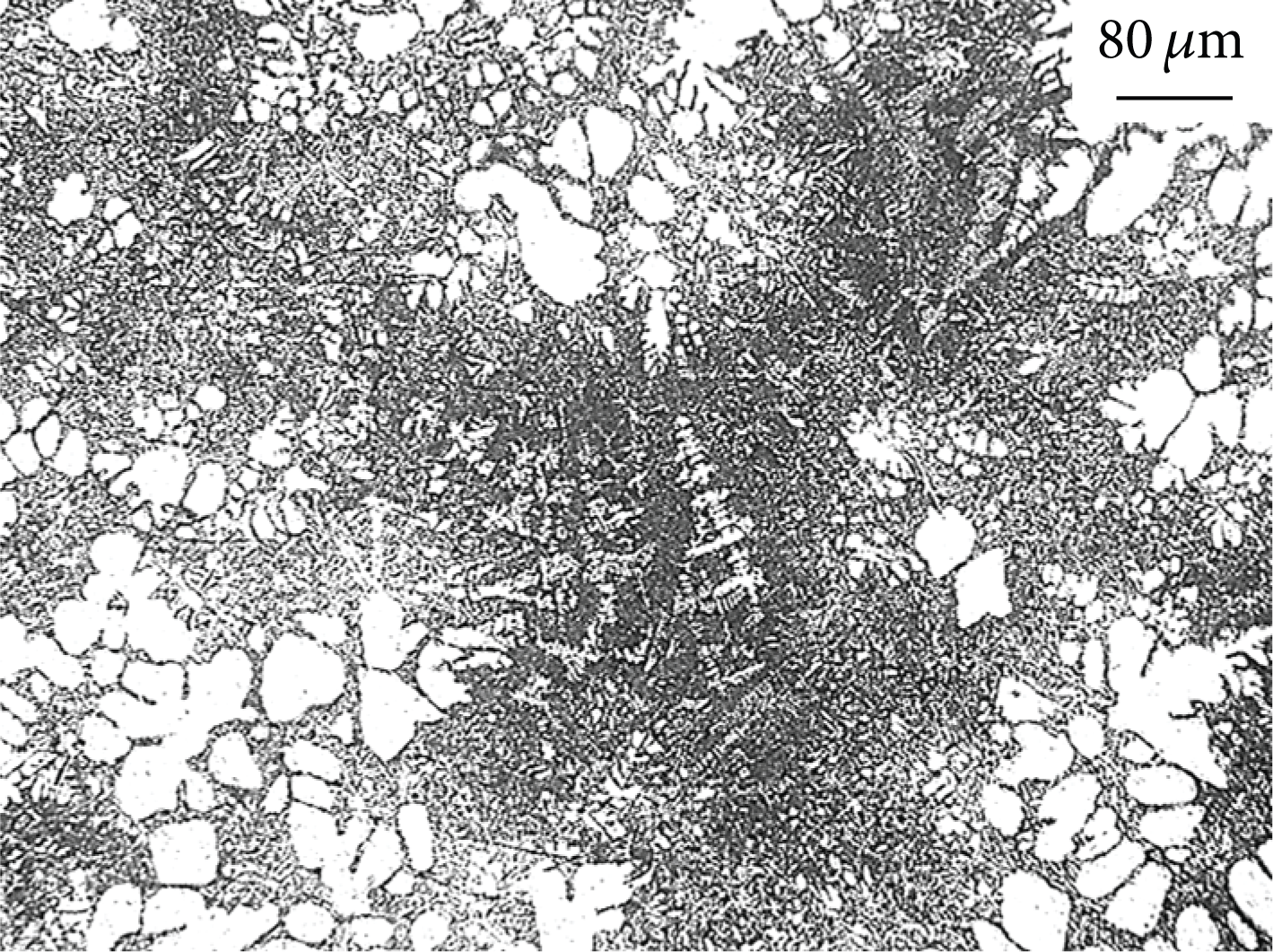
Small grains at the solidification end of NLSC.
Heterogeneous nucleation depends on the surface of solid particles in the molten metal. There are globular crystals of different size in the part as shown in Figure 6(b). The earliest nucleation grows to the biggest globular crystal, which depends on the wall of sleeve and runner. Flow and convection of molten metal lead to dissociation of crystal nucleuses which has grown [20]. The free grains will remelt and lessen if the temperature of molten metal exceeds the liquidus temperature. For NLSC, crystal nucleuses grow up, because the temperature of molten metal is between liquidus temperature and solidus temperature. The crystal nucleuses dissociate from the wall of sleeve. Due to time difference of flowing, globular crystals grow up and form different size. There is no heterogeneous nucleation depending on the surface of sleeve and runner, when the pouring temperature exceeds 650°C.
Figure 9 shows that crystal nucleuses form first and grow up to rosette crystal. Heterogeneous nucleation occurs, which depends on the crystal nucleuses that fuse from rosette crystal as the solid particle in NLSC. Different parts of rosette crystals have different radius. The interface of different radius has different equilibrium melting point of solid phase. The relationship between equilibrium melting point and the radius of solid phase is given as follows [21]:
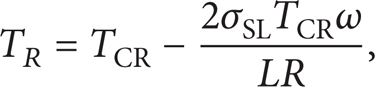
where T R is the equilibrium melting point of radius R interface and TCR is the melting point of solid-liquid planar interface. σSL is the interface energy of solid-liquid interface. L is the molar enthalpy of reaction of solid-liquid transform. ω is the molar volume of solid phase. R is the radius of solid-liquid interface.
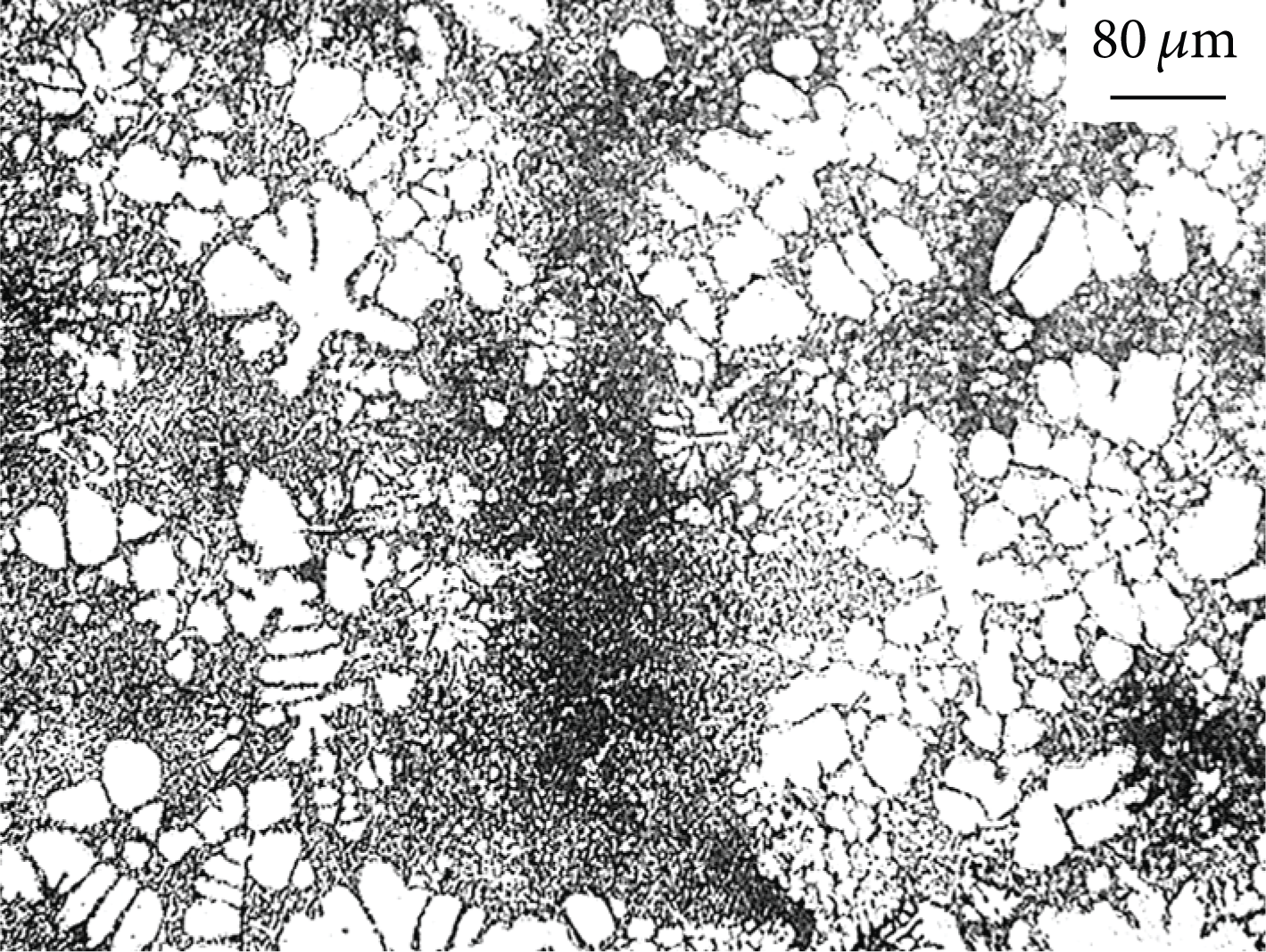
Fusing of rosette grains of NLSC.
The pressure can affect the interface energy of solid-liquid interface. The equation of the interface energy under the pressure is given as follows [22]:
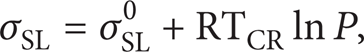
where P is the pressure and σSL0 is the interface energy of the solid-liquid interface under ordinary pressure. Equation (3) can be written as follows based on (4):

Equation (5) shows that T R of the root of rosette crystal has small radius decreased obviously under the pressure in NLSC. The root fuses at the higher temperature around. Self-stirring of molten metal speeds up the solute transmission, and the pressure fractures the dendrite arms. So it is easy to fracture the rosette crystal in NLSC. The grain which fractures from rosette crystal becomes new crystal nucleus and grows up in NLSC.
Figure 10 shows three nucleation models of NLSC. Figure 10(a) is the homogeneous nucleation. Figure 10(b) is the heterogeneous nucleation depending on the metal wall as the solid particle. Figure 10(c) is the heterogeneous nucleation depending on the fractured rosette crystal as the solid particle.

Three nucleation models of NLSC: (a) homogeneous nucleation, (b) heterogeneous nucleation depending on the metal wall, and (c) heterogeneous nucleation depending on the fractured rosette crystal.
4.2. The Growth of Globular Crystal of NLSC
4.2.1. Globular Crystal Formation Method of NLSC
Once the nuclei formed, nuclei will start to grow under small undercooling. Except dendrites crystal, small globular crystal, large globular crystal, and rosette crystal are observed in the support of NLSC, as shown in Figure 6(b). The grain growth order of NLSC is nuclei, small globular crystal, large globular crystal, and the rosette crystal. As shown in Figures 11 and 12, there are other two formation modes of globular crystal growth in NLSC besides normal crystallization.

Fracture, ripening, and spheroidizing process of NLSC under self-stirring: (A) two crystal arms constantly were close to each other and (B) residual liquid phase.

Process of annexation of small globular crystal of NLSC.
Figure 11 shows the formation of globular crystals by the effect of the self-mixing. The rosette crystal growing from large globular crystals shows shear and tear under effect of self-stirring when it flows across the runner and the cavity. Two crystal arms constantly are close to each other, as shown in Figure 11(A). With the further self-stirring, the interface of two crystal arms merges. Rosette crystal constantly ripens and spheroidizes. Then the large globular crystal forms. Residual liquid phase is observed in the globular grain under the effect of the self-mixing, which is shown in Figure 11(B).
Figure 12 shows that the globular crystal grows by the means of large globular crystal annexing small globular crystal. Affected by the turbulence and convection of molten metal, the small globular crystal which formed in supercooled molten metal was constantly moving. The small globular crystal stays around the large crystal as single or cluster. The interfaces of the two globular crystals contact with each other and fuse. Then the large globular crystal grows up in the way of annexing small globular crystal. The microscopic pressure exists around different radius globular crystals. The Gibbs-Thompson equation [22] gives the relationship between two different radius globular crystal and pressure as follows:

where p is the pressure of the plane interface and p1 is the pressure of the radius r1 interface. p2 is the pressure of the radius r2 interface. M is the atomic weight. ρ is the density. T is the temperature of molten metal. a is the constant. The higher pressure exists around the globular crystal of smaller radius while the smaller pressure exists around the globular crystal of higher radius. The pressure difference exists around different radius globular crystal. The pressure difference overcomes the interfacial energy of the different radius globular crystal. Then fusing and annexing of the different radius globular crystal happen.
Figure 13 shows the forming process of globular crystal of NLSC: normal growth, annexation of small globular crystal, and fracture and spheroidizing of rosette crystal.
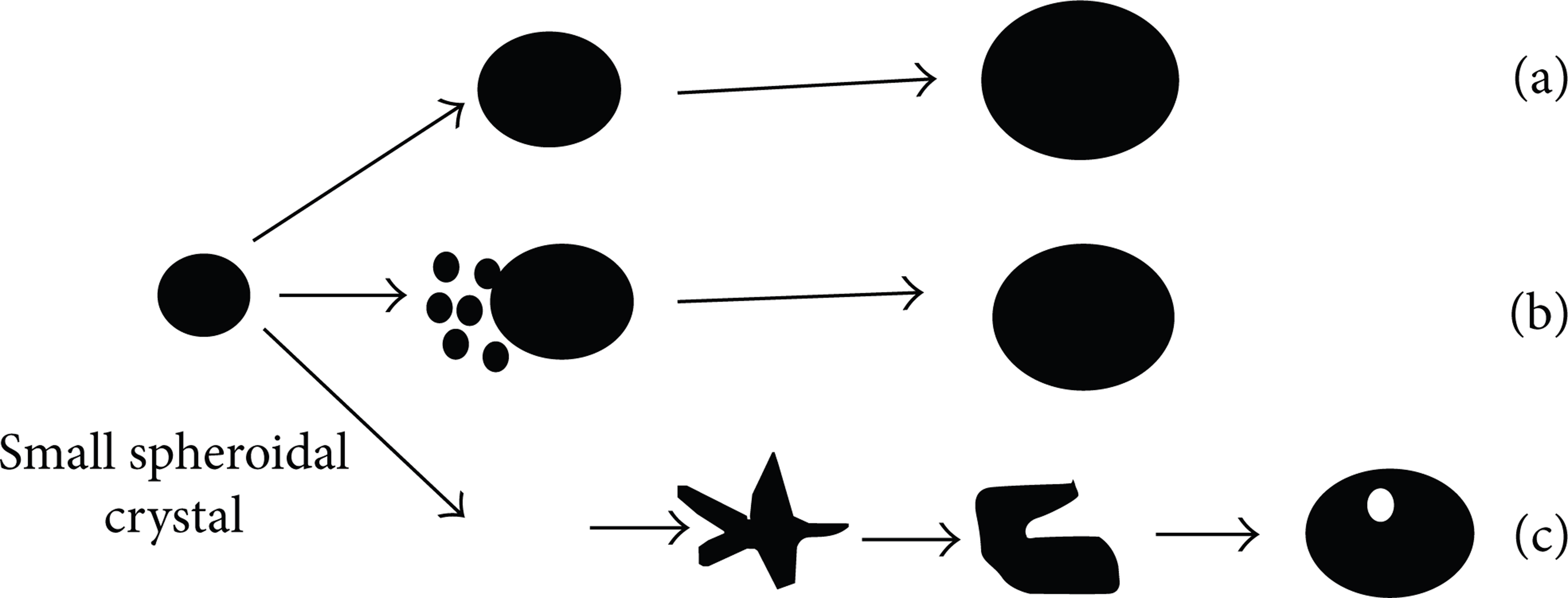
Forming process of globular crystal of NLSC: (a) normal growth, (b) annexation of small globular crystal, and (c) fracture and spheroidizing of rosette crystal.
4.2.2. Critical Radius of Globular Crystal of NLSC
In the NLSC, the grain first grows up by means of steady globular crystal. When the globular radius exceeds the critical radius, the globular crystal grows up into rosette crystal. Huang and Glicksman [23] concluded that the critical radius R C can strain globular growth, which is given as follows:

And Zhou et al. [24] give the r* as follows:

where

From (7), (8), and (9), the critical radius R C is obtained:

where T m is the melting temperature and k S is the heat conductivity of liquid metals under T m . k L is the heat conductivity of solid metal under T m . γSL is the liquid-solid interfacial free energy. L v is the latent heat of crystallization per volume under T m . L m is the latent heat of crystallization per mole under T m . ρ is the density. ΔT is the degree of supercooling under T m .
The thermal conductivity of solid metal k S does not change at different temperatures. However, the thermal conductivity of liquid metal k L increases as the temperature rising. According to the value of thermal conductivity liquid aluminum k L at different temperatures [25], the relation between temperature and the liquid thermal conductivity is given as follows:

The variable of the melting temperature T m is determined by Clausius-Clapeyron equation:

where T P is the melting temperature under pressure and ΔV is the volume difference of liquid and solid phase. T P is indirect proportion to ΔV. The pressure can increase the melting temperature because of metal volume contraction of solidification. In this experiment the pressure is 150 MPa and the melting point increases 9 K [11]. The melting temperatures under normal pressure and experiment pressure are 812 K and 821 K, respectively. The thermal conductivity of liquid metal under T m of normal pressure and experiment pressure is 99.1 and 99.4 W/mK, respectively. The other value of (10) is given in Table 2.
Physical and thermochemical properties of aluminium alloy at the melting point.

The values are substituted into (10):

where R C P is the critical radius under the pressure of 150 MPa. Figure 14 that depends on (13) shows the relationship between critical radius and ΔT. The critical radius increases with the ΔT decreasing. Globular crystal critical radius is inversely proportional to degree of supercooling. The pressure has very little influence on critical radius by increase of melting point and thermal conductivity, but it affects critical radius greatly by changing the degree of supercooling. In NLSC, the molten alloy has smaller degree of supercooling and bigger critical radius, as there is no pressure in the sleeve and runner. During solidifying of molten alloy under the pressure, the degree of supercooling increases, while the critical radius decreases. So when the pouring temperature exceeds 650°C, the globular crystal has smaller critical radius and grows up to dendrite crystal easily.

The relationship between critical radius for globular growth of aluminium alloy and ΔT.
4.3. The Growth Time of Globular Crystal of NLSC
Figure 6(b) shows that there is little rosette crystal in the matrix. The globular crystal has no enough growth time to exceed the critical radius. The growth time is given as follows:

where T P is the pouring temperature and T S is the solidus temperature. Vα is the average cooling rate. The NLSC pouring temperature is nearby the liquidus temperature. The temperature is under the liquidus temperature, during molten metal pouring into the sleeve. Equation (12) shows that the solidus temperature (melting point) of molten metal increases under the pressure. The value of (T P − T S P ) decreases. As molten metal directly contacts with mold under the pressure, greater heat exchange coefficient is obtained. The average cooling rate increases greatly. So the growth time decreases greatly, as shown in Figure 15.

Relationship between growth time and solidification temperature.
5. Conclusion
NLSC globular crystal transformed into rosette crystal, AGS increased, and ASC decreased with the rise of pouring temperature. But AGS decreased to 17.8 μm directly and ASC increased directly to 0.46 when the pouring temperature was 650°C. The phenomenon of increase of AGS and decrease of ASC with the rise of pouring temperature appeared again.
NLSC globular crystal of different sizes (AGS was 22.5 μm; ASC was 0.73) distributed uniformly in matrix.
The pressure and self-stirring promoted homogeneous nucleation. The equilibrium melting point of the root of rosette crystal had small radius decreased obviously under the pressure in NLSC; the arm of rosette crystal fused. The heterogeneous nucleation depended on the fractured rosette crystal and metal wall as the solid particle.
Forming process of globular crystal of NLSC contained normal growth, annexation of small globular crystal, and fracture and balling of rosette crystal.
NLSC globular crystal critical radius was inversely proportional to degree of supercooling. The pressure had very little influence on critical radius by increase of melting point and thermal conductivity, but it affected critical radius greatly by changing the degree of supercooling.
The growth time decreased greatly in NLSC, because solidus temperature increased and the average cooling rate increased under the pressure. The globular crystal had no enough growth time to exceed the critical radius.
When the pouring temperature exceeded 650°C, the globular crystal had smaller critical radius and grew up to dendrite crystal easily.
Conflict of Interests
The authors declare that there is no conflict of interests regarding the publication of this paper.
Footnotes
Acknowledgments
The authors are grateful to the support of Science and Technology Special Project of International Cooperation (no. 0102013DFA502120) and HLJ Province Graduate Student Innovation Research Project (no. YJSCX2012-079HLJ).
