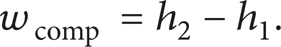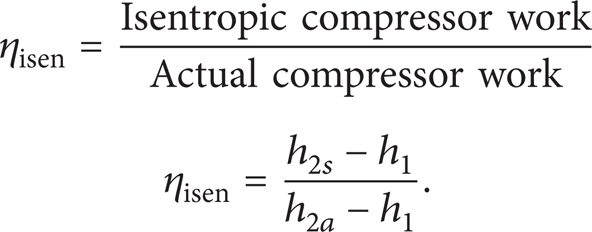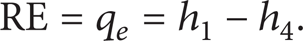Abstract
The topic of nanofluid heat transfer is certainly of interest to the heat transfer community. Nanorefrigerants are a type of nanofluids that are mixtures of nanoparticles and pure refrigerants. This paper focuses on five different nanorefrigerants with Al2O3 nanoparticles and their pure fluids: R12, R134a, R430a, R436a, and R600a. The coefficient of performance (COP) and compressor work for various evaporation and condensation temperatures are investigated. A method is developed to estimate the performance characteristics of nanorefrigerants in the refrigerant cycles for the nonsuperheating/subcooling case and superheating/subcooling case. The enthalpy of nanorefrigerants is obtained through the density. The validation process of the proposed method was accomplished with the available data in the literature. The results indicate that COP is enhanced by adding nanoparticles to the pure refrigerant and maximum values obtained using the R600a/Al2O3 mixture.
1. Introduction
Nanofluids as an advanced kind of working fluid have attracted special attention due to their capabilities in heat transfer enhancement. Nanofluids are mixtures of ultrafine particles (1–100 nm) and common working fluids such as water and oil. When a refrigerant is used as the base fluid, the suspension is usually called a “nanorefrigerant.” A considerable number of studies can be found on the applications or thermal properties of nanorefrigerants in the literature. Here, a brief review of some research on the application of nanorefrigerants in different systems is conducted.
Park and Jung [1] performed an analysis of nucleate boiling of carbon nanotubes (CNTs) with 1% volume fraction mixed with R134a and R123. They observed that at low heat fluxes, the heat transfer enhancement is greater than at high heat fluxes. Bartelt et al. [2] studied the flow boiling of CuO nanoparticles dispersed in a mixture of R134a/polyol-easter oil (POE). They found that a nanofluid with 0.5% weight concentration has no effect on heat transfer enhancement while 1% mass fraction displays 42–82% improvement in heat transfer. And a 2% mass fraction showed heat transfer enhancement as high as 50–101% Bi et al. [3] studied with the R134a/Al2O3 nanorefrigerant with a refrigerator and reported that energy savings of a 0.06 wt. % mixture was 23.24%, and enhancement in COP was 18.30%. Peng et al. [4] investigated the influence of the concentration of the flow boiling characteristics of the R113/CuO nanorefrigerant inside a horizontal tube at 0.1 wt.%, 0.2 wt.%, and 0.5 wt.% nanoparticle concentrations. A correlation predicted the heat transfer enhancement of the R113/CuO nanorefrigerant. It was found that dispersing nanoparticles in R113 refrigerant enhances the flow boiling heat transfer of the nanofluid, and the maximum heat transfer enhancement is 29.7%.
In another work, Peng et al. [5] investigated the influence of mass fraction on the pressure drop of R113/CuO nanofluid flow boiling inside a horizontal tube. They concluded that dispersing nanoparticles in R113 pure refrigerant increases the pressure drop; the maximum enhancement in pressure drop was 20.8%. Trisaksri and Wongwises [6] studied the nucleate pool boiling of R141b/TiO2 nanorefrigerant at low concentrations, including 0.01%, 0.03%, and 0.05% by volume. The results showed that increasing the particle volume concentration decreases the boiling heat transfer coefficient. Jwo et al. [7] studied the potential of Al2O3 nanoparticles as additives to R12/MO refrigerant. The results showed that using the nanorefrigerant at 0.1 wt.% displays a higher compression ratio compared to R12.
Henderson et al. [8] investigated the heat transfer performance of R134a and R134a/polyolester mixtures with nanoparticles during boiling flow conditions in a horizontal tube. The R134a/SiO2 nanorefrigerant having volume concentrations of 0.5% and 0.05% was tested to determine the effects of nanoparticles. It was found that using nanofluids leads to a decrease in convective boiling heat transfer coefficient (as much as 55%) compared to pure R134a. They also conducted experiments with R134a/POE mixtures having CuO nanoparticles with volume fractions of 0.02%, 0.04%, and 0.08%. It was observed that R134a/CuO/POE nanorefrigerant with a volume fraction of 0.02% displays no significant enhancement in heat transfer while the volume fractions of 0.04% and 0.08% caused considerable heat transfer enhancement as high as 52% and 76%, respectively.
Peng et al. [9] investigated the nucleate pool boiling heat transfer characteristics of R113/CNT/oil nanorefrigerant. The results showed that the R113/oil mixture with CNTs increased nucleate pool boiling heat transfer coefficient up to 61% compared to the R113 oil mixture. Peng et al. [10] performed an experimental study to determine the nucleate pool boiling heat transfer characteristics of R113/VG68 oil with diamond nanoparticles. They observed that the usage of diamond nanoparticles in R113/VG68 oil mixture increased the nucleate pool boiling heat transfer coefficient up to 63.4%.
Peng et al. [11] investigated the effects of nanoparticle size (three different average diameters) on nucleate pool boiling heat transfer characteristics of the mixture of refrigerant R113, ester oil VG68, and Cu nanoparticles. The results showed that the nanoparticles with an average diameter of 20 nm give higher heat transfer enhancement (up to 23.8%) compared to other nanoparticle sizes—for example, 50 nm and 80 nm. Abdel-Hadi et al. [12] investigated the usage of the R134a/CuO nanorefrigerant in a vapor-compression system. They obtained an optimum size and concentration of CuO nanoparticles in which the heat transfer enhancement is maximized; the optimal values were 25 nm and 0.55%, respectively. Kedzierski [13] examined the pool-boiling performance of R134a/Al2O3/poly alkylene glycol (PAG) nanorefrigerant on a roughened, horizontal, flat surface. In the study, polyolester lubricant of three different mass fractions (0.5%, 1%, and 2%) and a 1.6% volume fraction of Al2O3 nanoparticles were considered to make the nanorefrigerant. For all mass fractions of lubricant, the heat transfer improvement was observed due to using Al2O3 nanoparticles instead of an R134a/polyolester mixture.
Subramani and Prakash [14] studied the R134a/Al2O3 nanorefrigerant and found that there is an enhancement in COP while nanoparticles were added to the system. They also determined the system's COP theoretically for both R134a and R134a/Al2O3. They found that the COP of cycles that use R134a increases by 10.11% while the R134a/Al2O3 mixture used in the actual refrigeration cycle and enhancement ratio was 9.74% when COP determined theoretically. Kumar and Elansezhian [15] conducted experimental studies to discover the effects of the R134a/Al2O3/PAG oil mixture on energy consumption and freeze capacity of a vapor-compression refrigeration system. The results showed that using the nanorefrigerant at a 0.2% concentration reduces the energy consumption by 10.32% compared to the R134a/PAG oil mixture. They also observed that the usage of nanorefrigerant in the refrigerant system increases COP and minimizes the length of the capillary tube; it is also cost effective.
Sabareesh et al. [16] studied the potential of R12/TiO2/mineral oil nanorefrigerant to improve the COP of a vapor-compression refrigeration system. According to their results, the usage of nanorefrigerant in the vapor-compression refrigeration system (instead of the R12/MO mixture) decreases compressor work by 11% while the COP increases it. Kedzierski [17] investigated the pool boiling of R134a/diamond/RL68H nanorefrigerant and reported a 98% enhancement in boiling heat transfer for the 0.5% nanolubricant mass fraction compared to R134a/polyolester while the enhancement of the 2% nanolubricant mass fraction was 19%. Kedzierski [18] used a rectangular finned surface to investigate the effects of Al2O3 nanoparticles on the pool boiling performance of the R134a/polyolester lubricant (RL68H) mixture. Kedzierski [18] found that the use of nanoparticles in R134a/polyolester lubricant enhances the boiling performance up to 113%. Javadi and Saidur [19] studied the use of nanorefrigerants in domestic refrigerators to reduce the energy consumption and emissions of greenhouse gasses. They used TiO2-mineral oil-R134a and Al2O3-mineral oil-R134a with mass fractions of 0.06% and 0.1% as nanorefrigerants. The nanorefrigerant containing TiO2 nanoparticles of 0.1 wt.% displayed the highest energy savings—up to 25%. Sun and Yang [20] studied the use of R141b/Cu, R141b/Al, R141b/Al2O3, and R141b/CuO nanorefrigerants in a computer-aided test section to investigate the effects of material types and vapor quality on flow boiling heat transfer inside a horizontal tube. For the same mass fraction, the Cu-R141b nanorefrigerant had the highest heat transfer coefficient, and R141b/CuO gives the minimum heat transfer coefficient.
Although the nanorefrigerants have been extensively studied over the years, there still remains room to discuss, especially, application of nanorefrigerants in refrigeration cycle. This paper presents the effect of nanoparticles on the thermodynamic performance of a refrigeration system. The aim of the present theoretical study is to examine the COP and the compressor work of five different nanorefrigerants, including R12, R134a, R430a, R436a, and R600a and Al2O3 nanoparticles. The performance of these five nanorefrigerants in a vapor-compression refrigeration cycle is evaluated; Al2O3 nanoparticles with a mass fraction of 0.06% are added to the refrigerants. The effects of the main parameters of the refrigeration cycle, such as the refrigerant type and the effect of subcooling and superheating on the COP, are investigated for various evaporation and condensation temperatures.
2. Theoretical Model
The vapor-compression refrigeration is a variety of refrigeration. Generally, the system of vapor-compression refrigeration consists of a condenser, an expansion valve, an evaporator, and a compressor. The vapor-compression refrigeration cycle consists of four processes:
compressing refrigerant in compressor isentropically,
condensation at constant pressure,
adiabatic expansion in the expansion valve,
evaporation at constant pressure.
The pressure-enthalpy diagrams of nonsuperheating/subcooling and superheating/subcooling cases are given in Figures 1(a) and 1(b), respectively. For the nonsuperheating/subcooling case, the evaporation of the refrigerant occurs at a constant pressure during process (4-1) in the evaporator. The refrigerant at the inlet of the evaporator is vaporized by removing heat from the area desired to cool. Saturated vapor at point 1 goes into the compressor at low pressure and is exposed to a reversible adiabatic compression during the process from 1 to 2 in Figure 1. During process (2-3), while the heat is rejected in the condenser at a constant pressure, the working fluid changes to a saturated liquid when exiting the condenser. Refrigerant at point 1 in Figure 1(a) is saturated vapor at the evaporator temperature and at point 3 is saturated liquid at the condenser temperature. The working fluid at the exit of the expansion valve enters the evaporator, and the cycle is completed. Completely vaporized refrigerant enters the compressor, and its pressure and temperature increase during the compressing process. The working fluid at the condenser outlet should be completely liquid, which is achieved by using superheating/subcooling instead of the nonsuperheating/subcooling arrangement. COP improvement can be observed in superheating/subcooling compared to nonsuperheating/subcooling. Calculations of the comparison of pure and blended refrigerants during various refrigeration cycles can be seen in previous studies [21, 22].
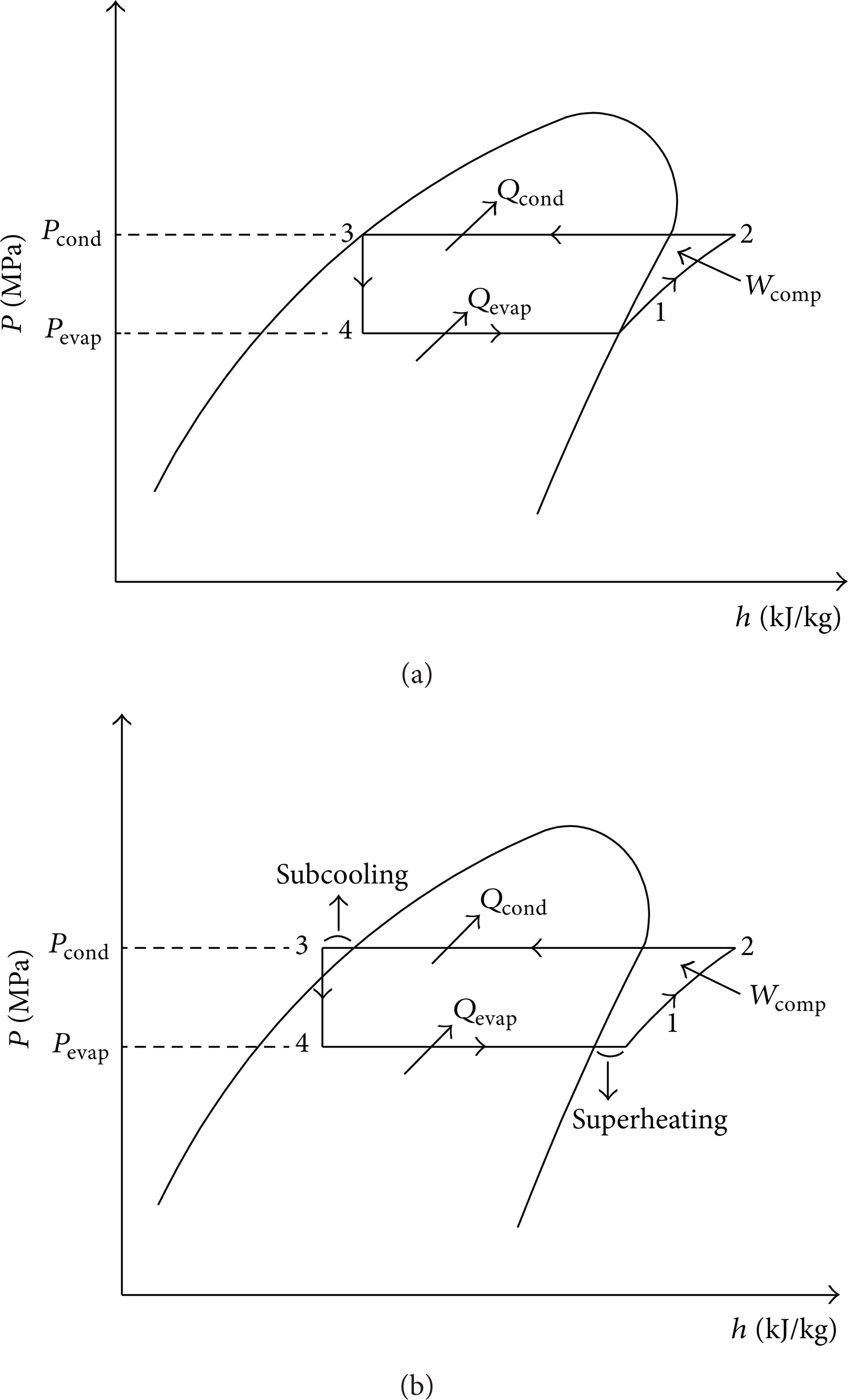
P-h diagrams of vapor-compression refrigeration cycle for (a) nonsuperheating/subcooling case and (b) superheating/subcooling case.
The heat transfer rate of the evaporator (q e ) is calculated as follows:
The heat transfer rate of the condenser (q c ) is calculated as follows:
The isentropic compression work of the compressor (wcomp) is expressed as follows:
The isentropic efficiency of the compressor is calculated as follows:
The performance of refrigerators is determined in terms of the COP. COP is defined as follows:
The refrigeration effect (RE), also called the heat transfer rate of the evaporator (q e ), is calculated as follows:
The density of a mixture of nanoparticles (ρNR) and the base fluid (ρPR) and the density of the base fluid can be determined based on Xuan and Roetzel [23]; however, it should be noted that the original equation is modified by using mass fraction instead of volume fraction in this study. Consider
3. Results and Discussion
The density of a nanorefrigerant can be a parameter to obtain the enthalpy of working fluid. Mollier charts of pure refrigerant can be used to obtain enthalpy values corresponding to the density and temperature or pressure at cycle point, so there is no chart or correlation to predict the properties of nanorefrigerants. The density of Al2O3 was accepted to be equal to 3,690 kg/m3 [24]. It should be noted that the enthalpy of point 3 is estimated as follows. First, temperature (TPR3) and saturation pressure (PPR3) corresponding to TPR3 are specified at 45°C and 11.6 bar, respectively. Then density is read from the Mollier chart of R134a. Density of nanorefrigerant (ρNR) is calculated by replacing nanoparticle mass fraction in nanorefrigerant (ω), ρNP and ρPR in (7). Then saturation temperature and pressure and enthalpy of nanorefrigerant are read from chart R134a for point 3. Enthalpy at point 3 decreases to 262.94 kJ/kg from 263.44 kJ/kg.
Similar to the determination of enthalpy of nanorefrigerant at point 3, enthalpy of nanorefrigerant at point 1 can be found. Temperature (TPR1) and saturation pressure (PPR1) corresponding to TPR1 is specified at −7°C and 2.25 bar, respectively. Then density is read from the chart of R134a. The density of nanorefrigerant (ρNR) is calculated by replacing nanoparticle mass fraction in nanorefrigerant (ω), ρNP, and ρPR in (7). Then saturation temperature and pressure and the enthalpy of the nanorefrigerant are read from chart R134a for point 1. Enthalpy at point 1 increases to 394.70 kJ/kg from 394.47 kJ/kg. Table 1(a) summarizes the properties of points 1 and 3.
Enthalpy of R134a/Al2O3 nanorefrigerant at points 1 and 3.

The enthalpy of points 2 and 4 in the refrigeration cycle can be determined by using specifications at point 1 and point 3 of the cycle, such as pressure and entropy. At the inlet of the evaporator, h4 must equal h3 because of adiabatic expansion in the expansion valve. And h1 is read from the chart by making use of s1 and saturation pressure at point 3. At the outlet of the compressor, if isentropic efficiency is 1 and the refrigeration cycle is ideal, s1 and s2 must be equal. For this calculation, isentropic efficiency is 0.51. COP enhancement is 14.41% when the density of Al2O3 nanoparticles is assumed to be 3690 kg/m3. Table 2(a) represents corresponding properties and results for the cycle working with R134a and R134a/Al2O3, which has a density of 3,690 kg/m3.
Cycle parameters and calculated values for Al2O3 density of 3690 kg/m3 (a) and 2200 kg/m3 (b).
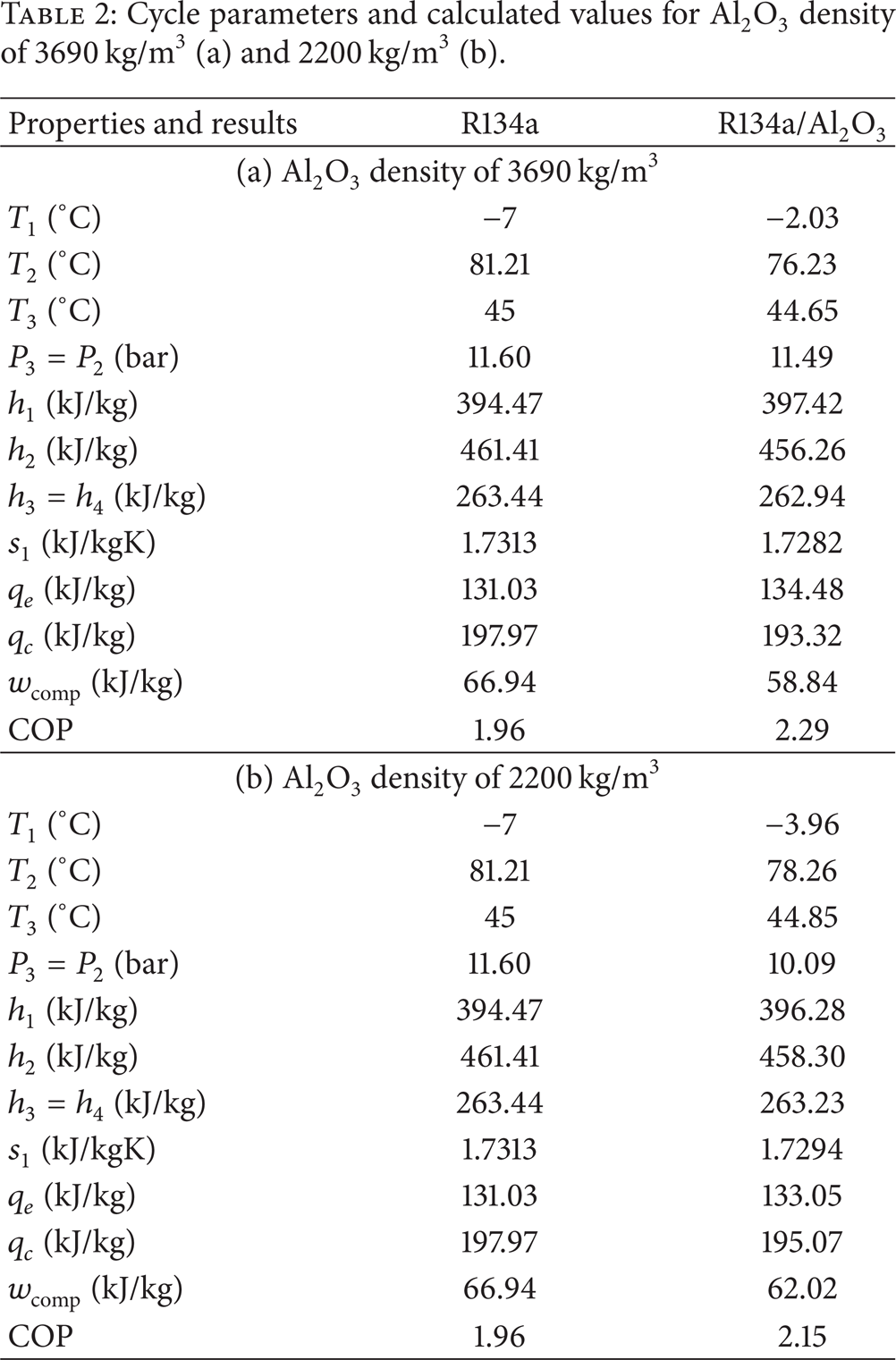
Because the Al2O3 nanoparticle can be produced in a wide range of densities using recent techniques, if we assume that the density of Al2O3 nanoparticles is 2,200 kg/m3 instead of 3,690 kg/m3, enhancement in COP is 9.6%. Table 1(b) summarizes the properties of points 1 and 3 while Table 2(b) depicts related properties and results for the cycle working with R134a and R134a/Al2O3, which has a density of 2,200 kg/m3. If the density of Al2O3 is 2,200 kg/m3, the results of the calculation approximate those of Subramani and Prakash [14] results with a 79.2% accuracy; accuracy was 71.4% for experimental COP enhancement with an assumed Al2O3 density of 3,690 kg/m3. Some results from Subramani and Prakash [14]'s experimental study for the validation process of the current study can be seen in Table 3. Due to the limited number of studies regarding the experimental study of nanorefrigerants, Subramani and Prakash [14]'s experimental database was used in the validation process of the proposed method.
Some results from Subramani and Prakash (2011)'s experimental study [14] for the validation process of current study.

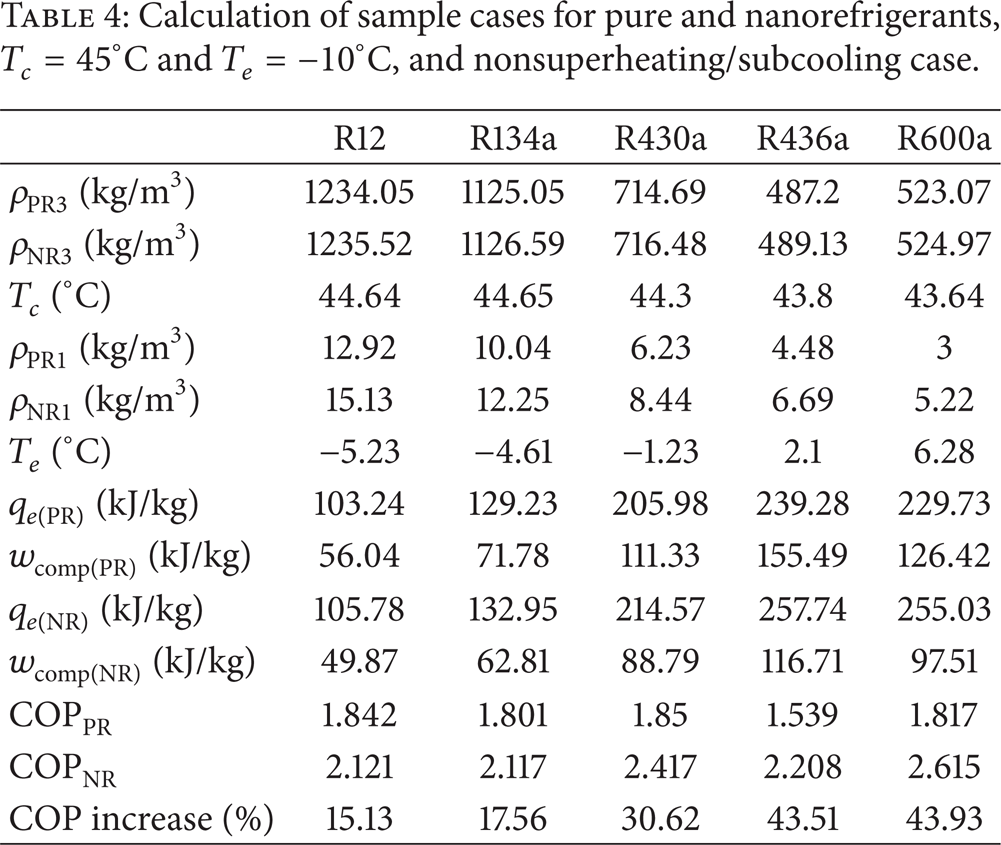
COP enhancement at various evaporating and condensing temperatures was calculated using equations presented in the previous section. Table 4 provides some data for the nonsuperheating/subcooling case for Al2O3 with a density of 3,690 kg/m3. For the nonsuperheating/subcooling case, the variation of COP, the compressor work per unit mass (wcomp), the evaporator heat per unit mass (q e ), and the condenser heat per unit mass (q c ) with condensation temperature (T c ) are presented in Figures 2, 3, and 4 at evaporation temperatures of 0°C, −10°C, and −20°C, respectively. At 5°C in the superheating/subcooling case, the variation of COP, the compressor work per unit mass (wcomp), the evaporator heat per unit mass (q e ), and the condenser heat per unit mass (q c ) with the condensation temperature (T c ) are presented in Figures 5, 6, and 7 according to the evaporation temperatures of 0°C, −10°C, and −20°C, respectively. It was observed that compressor work per unit mass (wcomp) decreased, and heat transfer rate per unit mass of evaporator (q e ) and heat transfer rate per unit mass of condenser (q c ) increased by adding alumina (Al2O3) nanoparticles to the base refrigerants. For all refrigerants and nanorefrigerants, an increase in the difference between T e and T c made q c decrease and q e and wcomp increase. According to COP equation (5), the increase in q e and the decrease in wcomp meant a rise in COP. This implies less energy consumption for the same refrigeration capacity.
Calculation of sample cases for pure and nanorefrigerants, T c = 45°C and T e = − 10°C, and nonsuperheating/subcooling case.

Alteration of COP (a), wcomp (b), q e (c), and q c (d) with evaporation temperature of 0°C for nonsuperheating/subcooling case.

Alteration of COP (a), wcomp (b), q e (c), and q c (d) with evaporation temperature of −10°C for nonsuperheating/subcooling case.

Alteration of COP (a), wcomp (b), q e (c), and q c (d) with evaporation temperature of −20°C for nonsuperheating/subcooling case.

Alteration of COP (a), wcomp (b), q e (c), and q c (d) with evaporation temperature of 0°C for 5°C superheating/subcooling case.

Alteration of COP (a), wcomp (b), q e (c), and q c (d) with evaporation temperature of −10°C for 5°C superheating/subcooling case.
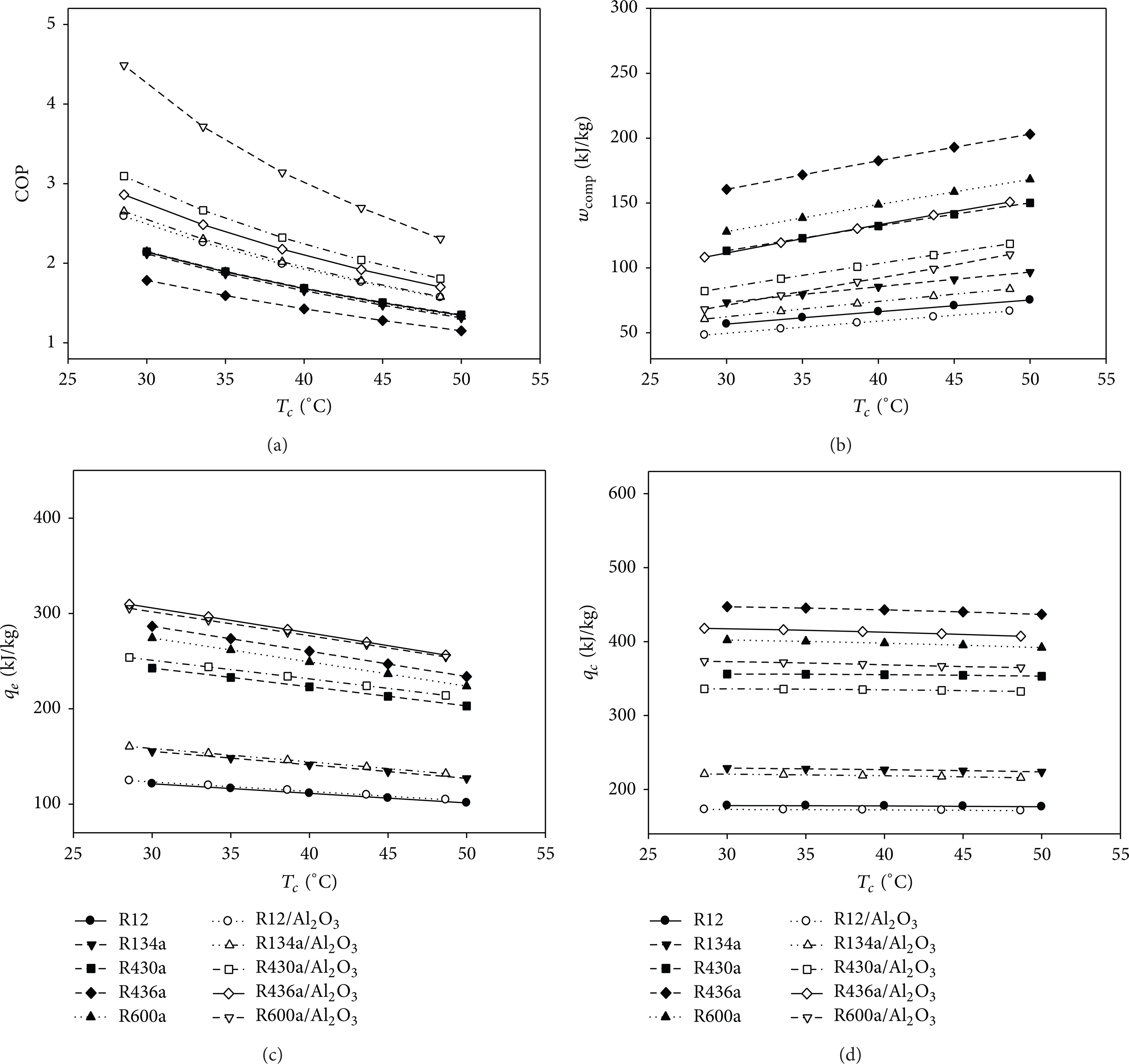
Alteration of COP (a), wcomp (b), q e (c), and q c (d) with evaporation temperature of −20°C for 5°C superheating/subcooling case.
For two cases at evaporation temperatures, R436a had the highest compressor work, and R12 had the smallest compressor work among the pure refrigerants considered. Moreover, the evaporator heat had the highest value with the usage of R436a and lowest for R12. However, the R430 nanorefrigerant had the highest COP, and R436a had the lowest COP. The condenser heat was the highest for R436a and lowest for R12.
R436a/Al2O3 had the highest compressor work and R12/Al2O3 had the lowest compressor work among the nanorefrigerants considered. Moreover, the evaporator heat had the highest value with the usage of R134a/Al2O3 and the lowest value for R12/Al2O3. However, the R600a/Al2O3 nanorefrigerant had the highest COP, and R436a/Al2O3 had the lowest COP. The condenser heat was the highest for R436a/Al2O3 and the lowest for R12/Al2O3.
The highest enhancement in COP of vapor-compression refrigeration cycles using the refrigerants and nanorefrigerants was observed for the lowest condensation and evaporation temperatures, and for all cases, the refrigerants’ heat of evaporation increases with decreasing temperature.
Outcome of the calculations are as follows: the enhancement of COP of the cycle using R600a is the greatest and the enhancement of COP of the cycle using R410a is the least one. It can be noted that the enhancements in COP by means of the addition of nanoparticles into alternative refrigerants of R12 are greater than those for the enhancement value of R12. This situation is not the same for R22 and its substitute refrigerants while some of the alternative refrigerants have larger and some of them have smaller COP values with the addition of nanoparticles than those for R22 enhancement ratios. The two main reasons for this are that density was the starting point of the prediction model and the thermophysical specification of the refrigerants.
Originally, it can be thought that the decreasing of the evaporator temperature corrupts the heat transfer and refrigeration. However, the refrigerant having higher temperature has higher evaporation enthalpy. Therefore, when it is considered that decreasing in evaporation temperature is little, decrease in the evaporator temperature can enhance the refrigeration effect.
In an actual vapor-compression refrigeration cycle, the enhancement in COP and energy-saving arising from the use of nanorefrigerant can be seen as decreasing in on-time ratio and electricity consumption.
The current model is developed to predict enhancement ratios in COP of vapor-compression refrigeration cycle. As there is not enough experimental data about the system parameters working with nanorefrigerant, the correlations available could be used for the model.
4. Conclusions
In this paper, the effect of adding the Al2O3 nanoparticle to different refrigerants was investigated by means of COP, the compressor work, evaporation heat, and condenser heat comparisons. The determination of enthalpy of the nanorefrigerant was the main challenge because of the lack of any correlation or experimental data in this subject. So the prediction method based on nanorefrigerant density was taken into consideration in the calculations to define nanorefrigerant enthalpies in the refrigerant cycle.
COP results were found to be higher at 5°C in the superheating/subcooling case compared to the nonsuperheating/subcooling case. However, cycles working with nanorefrigerants had a higher COP improvement in superheating/subcooling conditions over nonsuperheating/subcooling. R600 and R600a/Al2O3 nanorefrigerants had the highest COP under all conditions considered in the study. Although R12 showed the lowest compressor work, it cannot be used in the applications anymore because of its ODP and GWP values. Instead of this refrigerant, R134a can be used as it has the second lowest compressor work in the analysis.
Results obtained using theoretical analysis were compared with the results available in the literature, and deviations were 22.5% and 28.6% for pure R134a and R134a/Al2O3 mixtures (when the density of particles is 3,690 kg/m3), respectively. The deviation was 20.8% for the R134a/Al2O3 mixture when the nanoparticle density was 2,200 kg/m3.
The performance improvement of the refrigeration cycle by applying a nanoparticle is mainly due to heat transfer enhancement in heat exchangers and reduction of power consumption of the compressor by improvement of lubrication. These effects are included in the theoretical analysis as well.
Moreover, the nanoparticle is likely dispersed in the lubricant and is not dispersed in the gas phase. Therefore, the influence of the nanoparticle on heat transfer in a heat exchange depends on how much oil is discharged from the compressor and circulates with the refrigerant in the cycle. It is, therefore, necessary to consider the oil circulation ratio and the concentration of the nanoparticle in the cycle. However, this study combines various approximate aspects and proposes a practical method to the researchers. In addition, the term “nanorefrigerant” still has a problematic definition in the literature because the nanoparticle is not dispersed in the refrigerant. This topic is novel and is of increasing importance in the use of cooling applications.
Conflict of Interests
The authors declare that there is no conflict of interests regarding the publication of this paper.
Footnotes
Nomenclature
Acknowledgments
Ahmet Selim Dalkilic and Omid Mahian would like to thank King Mongkut's University of Technology Thonburi for the support during their research in Thailand. Somchai Wongwises would like to thank the Thailand Research Fund, the National Science and Technology Development Agency, and the National Research University Project for their support.