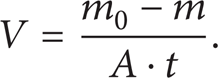Abstract
3-Fourteen alkoxy-2-hydroxypropyl triethyl ammonium chloride (TPAC) was synthesized by using tetradecyl alcohol, triethylamine, epichlorohydrin, and so on. The surface properties of product such as critical micelle concentration and surface tension were studied. The corrosion inhibition performance of product to A3 carbon steel was tested. Given the conditions of acidic medium, the immersion lasted for 72 h, under 25°C, and the concentration of product is 0.70 m mol/L; the corrosion inhibition rate is 95.21% over A3 steel. The result showed that TPAC has good surface activity and capacity in inhibiting corrosion.
1. Introduction
Water flooding oil is an important means of exploitation of petroleum. Many oilfields’ produced liquid have a rising water content, and the composition of sewage become more complicated. To minimise the environmental contamination caused by sewage efflux and also to reduce water wastage, we shall afflux the well-treated sewage back under ground for oil displacement. However, the reinjected water contains CO2, H2S, inorganic salts, and types of bacteria like sulphate-reducing bacteria, iron bacteria, and saprophytes which would still multiply at mass propagation raising the occurrence where water pipelines and drilling equipment are badly damaged and the oil additive liquid degraded to metamorphism [1–5]. In November 22, 2013, someplace petroleum pipeline explosion causes huge economic losses. The reason of the accident is that the petroleum pipeline was corroded and split and the oil spilt. To prevent that petroleum pipeline and equipment from corroding and splitting, research and development of high efficiency corrosion inhibitor have important significance. Adding corrosion inhibitor is one of the most effective approaches for sterilization and antisepsis which turns out to be usage saving; convenient; effective; cost saving; and safe [6–8]. We designed and synthesized a new surfactant. This paper takes tetradecyl alcohol, triethylamine, epichlorohydrin, and so on as raw materials while bringing hydroxyl groups and ether groups into the typical structure of quaternary ammonium molecules for the creation of 3-fourteen alkoxy-2-hydroxypropyl triethyl ammonium chloride (TPAC). One of the raw materials takes from renewable resources fatty alcohol. The synthesis process is simple with high yield and fewer by-products. The product has good thermal stability and biodegradability. In the laboratory, we analyzed the surface tension of the synthetic product and examined the corrosion inhibition performance of product to A3 carbon steel. The result showed that TPAC has good capacity in inhibiting corrosion.
2. Experiments
2.1. The Main Reagents and Instrument
Tetradecyl alcohol, triethylamine, epichlorohydrin, ethyl acetate, and potassium ferricyanide, A.R., were purchased from Tianjin Da Mao Chemical Reagent Factory in China; absolute ethanol, A.R., was purchased from Shenyang Hua Dong Chemistry Reagent Factory; triethyl benzyl ammonium chloride (TEBAC), A.R., was purchased from Tianjin Gung Fu Chemistry Research Institution; concentrated hydrochloric acid, C.P., was purchased from Daqing Yuanfu Chemistry Reagent Corp. Ltd.; sodium hydroxide, A.R., was purchased from Yi Xing Second Chemistry Reagent Factory; potassium chloride, A.R., was purchased from Tianjin Tian Da Purification materials Fined Chemical Factory; JC2000CI intravenous contact angle interfacial tension meter was purchased from Shanghai Zhong Chen Digital Instrument Co., Ltd., in China; DS-101S collector constant temperature heating magnetic stirrer was purchased from Gongyi Yu Hua Equipment Co., Ltd., in China; DZF-6030A vacuum drying oven was purchased from Shanghai Yiheng Technology Co., Ltd., in China; A3 steel was purchased from Jiangsu Gao You Mo Tian Digital Instrument Co., Ltd.
2.2. Synthesis
0.1 mol tetradecyl alcohol, 1.2 g TEBAC, 4.4 g NaOH, and 10 mL solvent are added into a four-necked flask with temperature control stirrer. Then, 0.2 mol epichlorohydrin is dropped into the flask dropwise at a speed of 10 S/D under 45°C. Stir strongly until the effect comes to an end before putting away the solvent. After washing and drying, the viscous intermediation alkylglycidyl ether in pale yellow is made.
Alkylglycidyl ether, 0.1 mol triethylamine, and 50 mL absolute ethanol are added into the 250 mL dried four-necked flask. Let it react under 50°C for 3 h before steaming ethanol out. The target product TPAC is gotten by recrystallizing.
2.3. Evaluation of Inhibition Performance Method
Referring to the standard SY/T5273-2000 set by the oil and gas industry, we choose the rotating corrosion coupon weight loss method and examined the corrosion inhibition performance of product to A3 carbon steel.
2.3.1. Preparation of Coupon
Put all A3 coupons successively into acetone and absolute ethanol. After take out, and clean them up with absorbent cotton, and dry them with filter paper. Then, measure and calculate the surface A and weigh them accurately (0.0001 g) and preserve them in dry containers for further usage.
2.3.2. Test of Coupon
Immerse the prepared coupon into the corrosion inhibition solution which is a mixture of 10% hydrochloric acid and the product of different concentration of aqueous solution. The temperature of the test solution is 25°C and the whole test is set for 72 hours. Then, we started running the equipment and let the coupon spin rotate at 80 r/min and last for 72 hours. The blank test meanwhile was carried out.
2.3.3. Deal with Coupon
After the test, the coupons are taken out immediately and immersed into the pickling solution for 3–5 minutes after being cleaned up with brush. Then, take them out and wash them with tap water and again immerse them into sodium hydroxide solution for around 30 seconds before being taken out and washed with distilled water. And then clean and dry them up with filter paper. Again immerse them into absolute ethanol for around 3 minutes and dry them up with filter paper. Put them all on dry containers for up to 4 hours and weigh them accurately (0.0001 g). So we may calculate the corrosion rate and corrosion inhibition rate with the formulas (1) and (2). Consider
In the formula, V is the corrosion rate, g/m2·h; m0 is the initial weight of the steel coupon, g; m is the weight of the steel after being taken away from those corrosion products, g; A is the superficial area of the steel, m2; t is the corrosion time period, h.
Corrosion inhibition rate η formula is as follows:
In the formula, η is the corrosion inhibition rate, %; V0 is the corrosion rate for the blank sample, g/m2·h; V is the rate after the addition of the product solution, g/m2·h.
3. Results and Discussion
3.1. Characterization
The infrared spectrum (Figure 1) showed that spectral absorption peak at 3306 cm−1 may be the group of –OH. The special absorption peak at 2923 cm−1, 2853 cm−1, and 1463 cm−1 may be the group of CH3– (or –CH2–). The special absorption peak at 1633 cm−1 may be the group of C–N. The special absorption peak at 1118 cm−1 may be the group of C–O. The special absorption peak at 721 cm−1 may be the group of –(CH2) n –. The data of FTIR proved that the synthetic product was the target product.
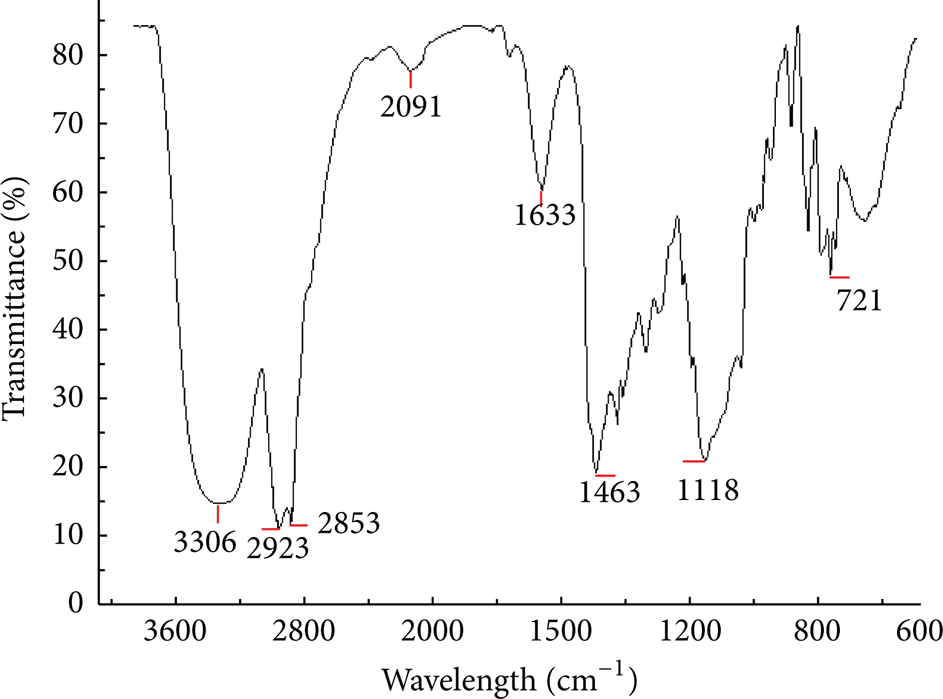
FTIR absorption spectrum of the product.
3.2. Surface Properties
Critical micelle concentration (cmc) is the important parameter of surfactant performance. Surface tension (γ) of TPAC of different concentration of aqueous solution at 25°C is measured. We drew γ-log c curves and get the result that when cmc is 0.8 m mol/L, the surface tension (γ) is 34.1 mN/m; the curves are shown in Figure 2.

γ-lg c curve of the product.
Data showed that the product is the lower cmc. The product has a better surface activity. The main reason is that in the product structure, the hydrophilic group –OH, the ether group –O– and the three ethyl groups that connect to the nitrogen atom make it arranged tightly in the absorption layer. Its molecular structure decides the low level of interfacial tension.
3.3. Inhibition Performance
By using the weight loss method, we measured and calculated the corrosion rate and the corrosion inhibition rate of A3 steel affected by synthetic products with different concentration under 25°C. In the experiment, we observed that surface of the blank steel was attached with large quantities of air bladders, when there are bubbles escaping from time to time and causing the surface of steel to turn into black.
While the surface was bright after some quantity addition of synthetic products solution, only with few cloud points emerged, the data of the corrosion rate and the corrosion inhibition rate are listed in Table 1.
The corrosion rate and corrosion inhibition of the product.

As shown in Table 1, when the concentration is at low level, the rate of corrosion inhibition is low as well. While the concentration of the synthetic product goes up, the corrosion rate goes down gradually but the corrosion inhibition rate goes up. When the concentration of TPAC is 0.70 m mol/L, the corrosion inhibition rate reaches 95.21%, which means that the synthetic product has a good performance in corrosion inhibition. Then, the corrosion inhibition rate tends to stabilize. The reason is that hydrophilic groups of the alcohol ether quaternary ammonium surfactants are attached to the surface of the steel through either physical attachment or chemistry attachment in the acidic medium. While the hydrophobic groups stay away from the metal surface and directionally arranged, which means there is a hydrophobic protective film on the metal surface. The protective film prevents water molecules from penetrating into metal surface. Moreover, the metal ions are restricted diffusion in the aqueous solution. It also prevents other corrosive media immersion. When the concentration is rather low, surfactants have a low coverage rate on the surface of metal, so the corrosion inhibition rate is lower. As the concentration goes up, surfactants have a higher coverage rate on the surface of metal. The adsorbed surfactants are gradually increasing. The structure of hydrophobic groups turns into vertical state from bending form. This change makes the protective film denser which enhances the rate of corrosion inhibition [9].
4. Conclusion
TPAC was synthesized by using tetradecyl alcohol, triethylamine, epichlorohydrin, and so on as raw materials. The surface properties of product were studied, such as critical micelle concentration and surface tension. The product shows good surface activity. The corrosion inhibition performance of product to A3 carbon steel was studied in acidic medium. Given the conditions of acidic medium, the immersion lasted for 72 h, under 25°C, and the concentration of product is 0.70 m mol/L; the corrosion inhibition rate is 95.21% over A3 steel. The result showed that TPAC has good capacity in inhibiting corrosion.
Conflict of Interests
The authors declare that there is no conflict of interests regarding the publication of this paper.