Abstract
The thermodynamic performance of the absorption chiller using (H2O + LiCl) as the working pair was simulated and compared with the absorption chiller using (H2O + LiBr). The effects of evaporation temperature on the performance coefficient, COP, generation temperature, concentration of strong solution, and flow rate ratio were also analyzed. At the same condensing and absorbing temperature, the simulating results indicated that the performance coefficient for (H2O + LiCl) is approximately equal to (H2O + LiBr) and the generation temperature was lower than that for (H2O + LiBr). On the other hand, the exergetic efficiency, ECOP, which is based on the second law of thermodynamics, for the absorption chiller using (H2O + LiCl), was more than the system using (H2O + LiBr) under the same operating conditions. The absorption chiller cycle was then optimized based on the coefficient of performance. The results show that the coefficient of performance of the absorption chiller, using (H2O + LiBr) at the optimum conditions, was around 1.5–2% higher than that of (H2O + LiCl).
1. Introduction
Since cooling and heating systems of buildings cause between 30 and 50% of the global energy consumption, increased efficiency of such systems considerably reduces energy consumption [1]. This decade has witnessed an unprecedented research interest in vapor absorption cycle refrigeration and heat pumping in order to reduce CO2 emissions for a healthy environment and balanced ecology. To pursue the desire to reduce the greenhouse gas CO2 to levels of 1990s, the United Nation Framework Convention on Climate is urging a phase out of CFC and HCFC fluids. The phase out of CFC and HCFC fluids has encouraged research into environmental friendly refrigerants such as water and other binary solutions. These researches have led to the use of low grade energy systems like vapour-compression cycle systems for heating and cooling as compared to other heating and cooling systems. Aside from economic benefits, improving theefficiency of like vapour-compression cycle systems is effective means of curbing CO2 emissions. The vapour-absorption cycle is considered to be the best in terms of energy performance today. In addition, it has the potential to be improved among the several heat-powered cycles [2]. In comparison with other vapour-compression cycle, the absorption cycle has a low efficiency reputation although this is a result of an unfair comparison between them, but the environmental concern calls for high efficiency, pollution-free refrigerators, and heat pumps.
The essential condition in absorption chillers is the availability of an inexpensive or even free heat source such as waste (or rejected) heat. Several studies have been made using solar energy [3, 4]. Absorption cooling is based on the strong chemical affinity between two working fluids, the refrigerant and the absorbent, the former having a much lower vapour pressure than the latter. In a single-effect absorption chiller, at a lower pressure and temperature level, the refrigerant is evaporated using the heat removed from the conditioned space and absorbed by the absorbent solution. At a higher pressure and temperature level, a heat source provides the energy needed to extract the absorbed refrigerant vapour from the solution, which is subsequently regenerated and ready for the next cycle. Water, as a refrigerant and lithium-bromide as an absorbent, is one of the most used working fluid pairs in current absorption chillers [5].
One of the earliest dynamic simulations of absorption refrigeration systems has been performed by Jeong et al. [6] for a steam driven heat pump. The model assumes that solution mass storage in the vessels, thermal capacity heat storage, and flow rates (vapour and solution) are calculated according to the pressure differences between vessels. Later, Fu et al. [7] developed a library of elemental dynamic models for absorption refrigeration systems (CHP applications), in which the components are described as lumped processes involving two-phase equilibriums. In a series of two papers, Kohlenbach and Ziegler [8, 9] presented a simulation model and its experimental verification for a single-effect water/LiBr chiller. As a special feature, all of the thermal capacities have been divided into an external part (influenced by the temperature of the external heat carriers) and an internal part (influenced by the temperature of the refrigerant or the absorbent). Moreover, a transport delay time has been assumed in the solution cycle. Matsushima et al. [10] developed a program using object-oriented formulation and parallel processing to simulate the transient operation of a triple-effect absorption chiller. A special algorithm based on the pressure difference and flow resistance between the generators and the absorber has been used to calculate the flow rate of solution. Wu et al. [11] experimentally studied the equilibrium pressure, equilibrium temperature, and gas composition of NH3-H2O-LiBr ternary mixture for its application in industrial absorption chillers and heat pumps. Xie et al. [12] investigated the relationship of coefficient of performance (COP) of a lithium-bromide absorption chiller with solution concentration of LiBr/H2O. Papaefthimiou et al. [13] developed a mathematical model for analyzing the heat and mass transfer process of LiBr-H2O absorption on a horizontal tube, and a good agreement with the test data was obtained.de Lucaset al. [14] added formats and lithium-nitrates in the lithium bromide solution and analyzed the effect of generation temperature, condensing temperature, and evaporation temperature on system performance. This action was found to increase the COP by 30% and the solution circulation rate to decrease by 12%.
In recent years, finding ways to improve absorption-system efficiency has been a great challenge for researchers [2, 15, 16]. Works have mainly focused on inventing new or hybrid cycles, finding new working fluids, and improving the heat and mass transfers of the absorption refrigerator. The performance of absorption cycles is attributed to the thermodynamic properties of the working pairs which consist of the refrigerant and the absorbent. Most commonly used working pairs are ammonia + water solution (NH3 + H2O) and aqueous solution of lithium bromide (H2O + LiBr). However, Zhang and Hu [17] have identified corrosion, crystallization, and toxicity as inevitable weaknesses of these working pairs. The need for working pairs not susceptible to these weaknesses has become the focus of research. In this study, we have used LiCl + H2O as a working pair in the absorption cycle, for the aim of a comprehensive study and optimization. The studied working pairs have the advantages of availability and environmentally friendly and are suitable for moderate temperature applications [18]. LiCl/H2O is preferred over LiBr/H2O for the reason of cost and long-term stability, while it is preferred over CaCl2/water for its better hygroscopic properties leading to better cycle performance [19]. Furthermore, the smaller chemical potential of LiCl relative to LiBr leads to a larger absorber area and shows a larger performance reduction than LiBr [20].
A detailed review of thermodynamic properties of (LiCl/H2O) can be seen in the work of Pátek and Klomfar [21].
2. The Performance Simulation
Figure 1 presents the schematic of the single effect absorption refrigeration.

Schematic diagram of absorption refrigeration. A—absorber, C—condenser, E—evaporator, G—generator, HX—solution heat exchanger, P—pump, and V—valve.
2.1. Assumptions
The following assumptions are made through simulating the thermodynamic performance of the absorption refrigeration cycle [17, 22].
The entire system runs under steady state conditions.
Solutions leaving the generator and the absorber are at a saturated liquid temperature.
Refrigerant leaving the condenser is in a saturated liquid phase.
In the whole cycle, pressure losses are neglected.
Throttling valves are an isenthalpic device.
Temperature difference between the cold and hot streams at the cold end of solution heat exchange (HX) is 10°C.
The consumption of mechanical energy by the pump is negligible.
The difference of the concentration between the strong and weak solutions is 5% (wt%, LiCl or LiBr).
2.2. Mass and Energy Balance Equations
Mass and energy balance equations for any component in the cycle for the aim of thermodynamic cycle performance simulation are given as follows:
Generator
Condenser
Evaporator
Absorber
Solution Heat Exchanger
The saturation pressure of water and working solutions (H2O + LiBr or H2O + LiCl) are shown, respectively, with P s (T) and P(T, x).
A computer program has been developed using Engineering Equation Solver (EES) [23] to carry out the thermodynamic analysis of the absorption chiller cycle.
For the previously mentioned properties, we have used the available date in the library of EES Software.
By applying the mass, energy, and phase balance equations around any element in the cycle and by utilizing the above stated assumptions, the heat load of the generator, absorber, evaporator, and condenser, the concentration of strong and weak solution, and flow rate ratio were obtained.
The coefficient of performance, COP, exergetic efficiency, ECOP, which is based on the second law of thermodynamics [24], and the flow rate ratio, F, are defined as follows:
2.3. Optimization Method
The results obtained from the mathematical model reveal that the optimum performance of the absorption chiller cycle depends on the following design/operating parameters:
condenser temperature,
evaporator temperature,
absorber temperature,
generator temperature.
Therefore, the optimum value of the coefficient of performance for the absorption chiller cycle can be expressed as a function of four design/operating parameters, as shown in the following equation:
subject to (10)
Using direct search method and applying the constraints on each variable by setting the bounds, the performance of the whole cycle is optimized by the EES software from the viewpoint of quantity of produced distilled water. The direct search method is based on a successive search intended to find an extremum by directly comparing function values at a sequence of trial points without involving derivatives. This method is deemed suitable for problems involving simulation-based optimization or optimizing nonnumerical functions, as well as, in practice, problems involving nonsmooth or discontinuous functions [25].
2.4. Model Validation
For validating the models developed for the single-effect absorption refrigeration cycle the theoretical data given by Kaushik and Arora [26] and Yari et al. [22] are used.
The results of comparisons are shown in Table 1, which indicate that there is a good agreement between the present work and numerical data reported in the literature.
Input parameter values: (Tgen = 87.8°C, Tevap = 7.2°C, Tabs = Tcond = 37.8°C, εHX = 0.7,
3. Results and Discussion
Figures 2–9 demonstrate the results of absorption chiller using (H2O + LiBr) and (H2O + LiCl). Figure 2 shows coefficients of performance versus evaporator temperature for both working pairs. (H2O + LiCl)'s performance is approximately equal to (H2O + LiBr). Moreover, for both pairs the coefficient of performance will increase with the increase of evaporation temperature.

Effect of evaporator temperature on COP.

Effect of evaporator temperature on ECOP.

Effect of evaporator temperature on generator temperature.

Effect of evaporator temperature on concentration of strong solution.

Effect of evaporator temperature on flow rate ratio.

Flow ratios of the absorption system with respect to the generator outlet temperature using (H2O + LiBr) and (H2O + LiCl).

Effect of flow ratio on COP for different conditions.

Relationship between COP and generator temperature under different condenser temperature and equal evaporator temperature.
The variation of ECOP with the evaporation temperature for two working pairs is shown in Figure 3. The evaporation temperature's effect on COP and ECOP is completely different from each other which are very interesting and are in agreement with the results available in the literature [3, 24]. The COP will increase with the increase of Teva while ECOP will decrease with the increase of Teva. The reason for the difference between COP and ECOP is that the former only takes account of the quantity of the heat, but the latter not only takes account of the heat but also of quality of the heat. In other words, the cooling load of Qeva and the temperature level of Teva, are all important for the refrigeration process, namely, the lower the Teva, the higher the quality of cooling load Qeva. This was why the ECOP will decrease with the increase of Teva. It was also discovered that the ECOP for working pair H2O + LiCl is slightly higher than that for H2O + LiBr (5–6%). This is an advantage for (H2O + LiCl).
Figure 4 demonstrates that the generation temperature for (H2O + LiCl) is lower than that for (H2O + LiBr), which designates that absorption chiller using (H2O + LiCl) as working fluid can be driven by lower temperature level waste heat under the same operating conditions. Moreover, the generation temperature will decrease with the increase of the evaporation temperature.
According to Figures 5 and 6, the concentration of strong solution and flow rate ratio, f, for (H2O + LiBr), are both higher than those for (H2O + LiCl), and they both decreased with the increase of the evaporation temperature. It is obvious that having smaller flow ratio for a working pair is a major advantage from the view point of performance and lesser amount of mechanical power losses [15, 16, 27].
Figure 7 shows the flow ratio variation with respect to the generator outlet temperature. The generator outlet temperature rises with the increment of flow ratio due to a decrease in the mass fraction of the weak solution. Equation (4) can be rewritten to yield the following:

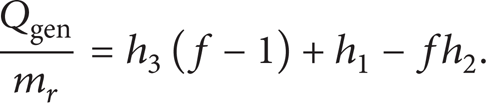
Equation (11a) shows that a smaller flow ratio results in less heat input to the generator for the same amount of cooling at the evaporator, which would increase COP [27] (far ahead shown in fig f-COP). These results are in agreement with that shown Figures 4 and 6.
Figure 8 examines the COP trends against the recirculation flow ratio for different condenser and evaporator temperatures and for equal evaporator temperature ranges of 5–15°C. As is seen, the COP decreases in all cases as the recirculation flow ratio increases. This is because when the evaporator temperature increases, the minimum system pressure will increase and the strong solution concentration will decrease by decreasing the flow ratio (according to Figure 5). The lower flow ratio results in a higher absorption heat capacity and a higher COP.
Coefficients of performance for both working pairs are approximately equal to each other while, (H2O + LiCl) owns less magnitude of flow ratio.
It can be seen in Figure 8 that as the absorber, temperature is increased, the COPs of both pairs decrease and flow ratio increase very rapidly. A similar behavior is observed as condenser temperature is decreased.
Figure 9 shows the variation of COP with the generator temperature under different condenser temperature and equal evaporator temperature ranges of (5–15)°C conditions. The COP of the system decreases as Tcon increases for both working pairs. Increasing the generator temperature Tgen tends to decrease the COP at constant condenser temperature. At the same working conditions, COP of the system utilizing H2O + LiBr is (0.6–0.8%) more than that of H2O + LiCl. But on the other hand the desirable generator temperature for the H2O + LiCl is (2–2.5)°C less than that of H2O + LiBr. Our results for H2O + LiBr are in agreement with those reported in the literature [28].
4. Optimization
Using the direct search function in the EES software, the coefficient of performance of the absorption chiller cycle is optimized with respect to the condenser, evaporator, absorber and generator temperatures for the two cases using (LiBr + H2O) and (LiCl + H2O) working pairs. The results are outlined in Tables 2 and 3.
The results of optimization for maximum coefficient of performance of the absorption chiller cycle using (LiBr-H2O).

The results of optimization for maximum coefficient of performance of the absorption chiller cycle using (LiCl-H2O).
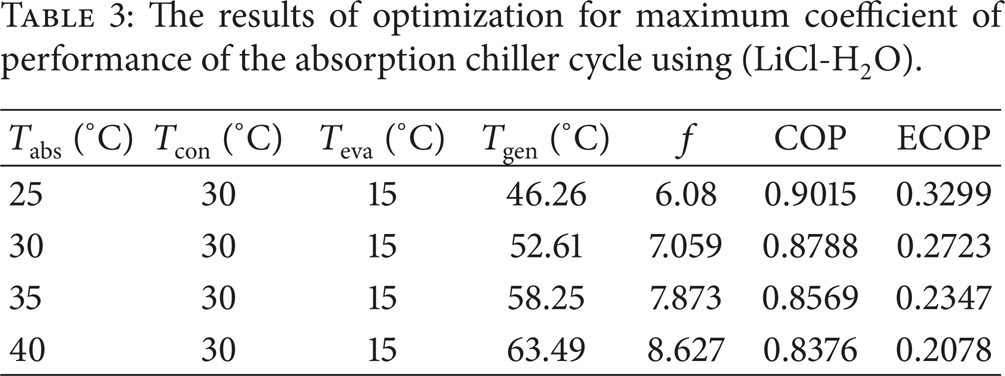
As the tables indicate the optimum mass flow ratio for maximum coefficient of performance is higher for the case of (LiBr + H2O) compared to that for the (LiCl + H2O). Also, with increasing Tabs, the coefficient of performance and exergetic efficiency decreases as expected. A comparison between Tables 1 and 2 reveals that when the (LiCl + H2O) is employed as a working fluid, the maximum coefficient of performance is slightly less than (1.5–2%) that when (LiBr + H2O) is used. However, regarding the smaller crystallization risk, the former maybe preferred in spite of the difference in the performance criterion.
5. Conclusion
The major conclusions drawn from the simulation are as follows.
Under the same condensation and absorption temperatures, the generation temperature of refrigeration cycle for H2O + LiCl was somewhat lower than that for H2O + LiBr, so it was possible that the absorption chiller was driven by lower temperature level waste heat by means of adopting H2O + LiCl as a working fluid.
At the same condensing and absorbing temperature, the simulating results indicated that the performance coefficient for the (H2O + LiCl) is approximately equal to (H2O + LiBr), while the generation temperature was lower than that for H2O + LiBr.
The exergetic efficiency ECOP for H2O + LiCl was somewhat higher thanthatfor H2O + LiBr and they will all decrease with the evaporation temperature Teva.
The concentration of strong solution and flow rate ratio, f, for (H2O + LiBr), are both higher than those for (H2O + LiCl), and they both decreased with the increase of evaporation temperature.
The results show that the coefficient of performance of the absorption chiller, using (H2O + LiBr) at the optimum conditions, is around 1.5–2% higher than that of (H2O + LiCl).
As the absorber temperature is increased, the COPs of both pairs decrease and flow ratio increases very rapidly. A similar behavior is observed as condenser temperature is decreased.
At the same working conditions, COP of the system utilizing H2O + LiBr is (0.6–0.8%) more than that of H2O + LiCl. But on the other hand the desirable generator temperature for the H2O + LiCl is (2–2.5)°C less than that of H2O + LiBr.
Footnotes
Acknowledgment
The authors gratefully acknowledge Mr. Mehrdad Khamooshi, the graduate student of Mechanical Engineering Department in Eastern Mediterranean University, for his valuable help searching the literature.











