Abstract
The cycle performance of refrigeration cycles depends not only on their configuration, but also on thermodynamic properties of working pairs regularly composed of refrigerant and absorbent. The commonly used working pairs in absorption cycles are aqueous solutions of either lithium bromide water or ammonia water. However, corrosion, crystallization, high working pressure, and toxicity are their major disadvantages in industrial applications. Therefore, seeking more advantageous working pairs with good thermal stability, with minimum corrosion, and without crystallization has become the research focus in the past two decades. Ionic liquids (ILs) are room-temperature melting salts that can remain in the liquid state at near or below room temperature. ILs have attracted considerable attention due to their unique properties, such as negligible vapor pressure, nonflammability, thermal stability, good solubility, low melting points, and staying in the liquid state over a wide temperature range from room temperature to about 300°C. The previously mentioned highly favorable properties of ILs motivated us for carrying out the present research and reviewing the available ILs found in the literature as the working fluids of absorption cycles. Absorption cycles contain absorption heat pumps, absorption chillers, and absorption transformers.
1. Introduction
With fast economic growth and constantly increasing energy consumption, the human kind is about to face a growing degradation of the environment if business continues as usual. For this reason, the utilization of low-grade energy has become one of the most attractive solutions to heating and cooling problems encountered in industrial and residential applications. It is possible to recover the low-grade heat wasted in many industries to use it in some processes in order to increase the energy efficiency.
Absorption cycles which include absorption heat pump (AHP), absorption chiller (AC), and absorption heat transformer (AHT) can use waste heat economically resulting in decreasing the consumption of primary energy and reducing the negative impact on the environment. The basic principle of an absorption cycle is shown in Figure 1. Since the invention of absorption cycles, the properties of working fluids have been a challenging issue, as performance of an absorption cycle critically depends on thermodynamic properties of working pairs composed of refrigerant and absorbent. Thus, searching for more beneficial working pairs with excellent thermal stability, no corrosion, and no crystallization has become the subject of research in recent years [1]. The basic components of the absorption cycles are the evaporator, the condenser, the generator, and the absorber, while the evaporator and generator are supplied with waste heat at the same temperature and the increased heat is delivered from absorber. The operating system of the basic absorption cycles is explained as follows.

The basic absorption cycle.
Refrigerant vapor is produced at state 4 in the evaporator, by low- medium-grade heat source. The refrigerant vapor dissolves and reacts with the strong refrigerant-absorbent solution that enters the absorber from state 10, and weak solution returns back to generator at state 5. In the generator some refrigerant vapor is removed from the weak solution to be sent to the condenser and consequently the strong solution from the generator is returned to the absorber. After condensing the vaporized refrigerant in the condenser, it is pumped to a higher pressure level as it enters the evaporator. The waste heat delivered to the evaporator causes its vaporization. Again the absorber absorbs the refrigerant vapor at a higher temperature. Therefore, the absorption cycles have the capability of raising the temperature of the solution above the temperature of the waste heat [2].
The requirements of working fluids of absorption cycles areas are as follows [3].
The difference in boiling point between the pure refrigerant and the mixture at the same pressure should be as large as possible.
Refrigerant should have high heat of vaporization and high concentration within the absorbent in order to maintain low circulation rate between the generator and the absorber per unit of cooling capacity.
Transport properties that influence heat and mass transfer, for example, viscosity, thermal conductivity, and diffusion coefficient, should be favorable.
Both refrigerant and absorbent should be noncorrosive, environmentally friendly, and of low cost.
Ionic liquids (ILs) are a class of low-temperature molten salts, which are composed of an organic cation and an inorganic anion. During recent years, ILs have been used as organic green solvent in catalysis, separation process, electrochemistry, and many other industries for their unique physical and chemical properties, such as negligible vapor pressure [4], negligible flammability and thermal stability [5], low melting temperature and liquid state over a wide temperature range, and good solubility [6]. In particular, the low volatility of the ILs enables easy separation of the volatile working fluid from the ILs by thermal stratification with the minimum harmful impacts on environment [7]. ILs can be a substitute for some of the most used toxic working fluids (such as ammonia/water) in absorption cycles. Since many of the ionic liquids have melting points below the lowest solution temperature in the absorption system (~300) [8, 9], they also wipe out the crystallization and metal-compatibility problems of water/LiBr system.
Although a large number of ILs as the working fluids in absorption cycles are given in the literature, there is still a need for a complete review with comparison of all ILs for achieving higher performance of theabsorption cycles. It seems to be necessary to have a review in this class of working pairs. So in the present work, a review of specific properties of ionic liquids as working fluids for absorption cycles has been carried out.
2. A Review of ILs as Working Fluids
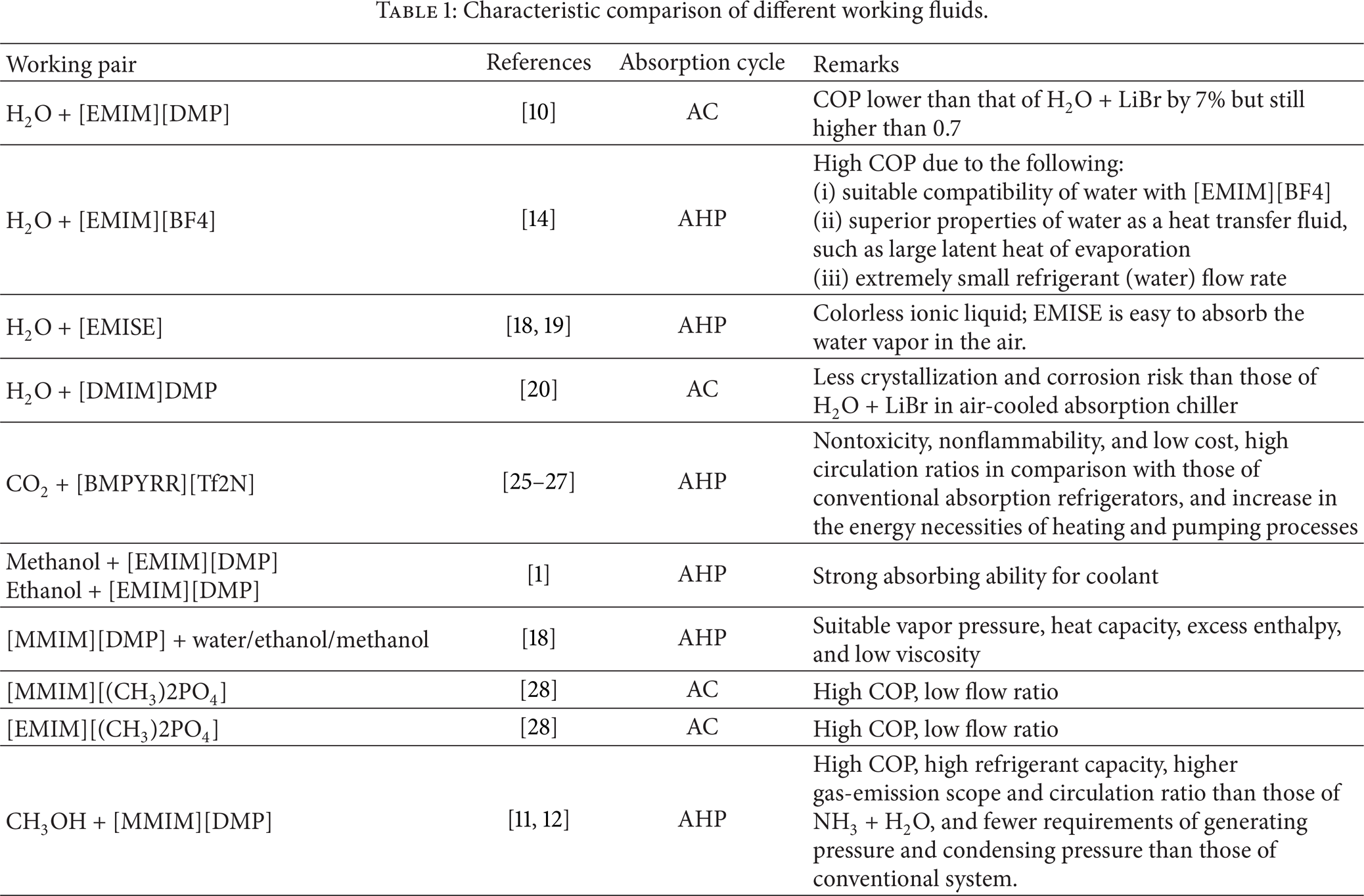
The thermodynamic performance of an absorption chiller operating with water + 1-ethyl-3-methylimidazolium dimethylphosphate [EMIM][DMP] and H2O + LiBr mixtures has been compared under the same operating conditions by Zhang and Hu [10]. The coefficient of performance of the absorption chiller for working fluid H2O + [EMIM][DMP] was lower than that of H2O + LiBr by 7% but still higher than 0.7. Also for the same condensation and absorption temperatures, the generation temperature of refrigeration cycle for H2O + [EMIM][DMP] is somewhat lower than that for H2O + LiBr, allowing the operation of the absorption chiller at lower temperatures of heat supply. These features indicate that the binary ionic liquid solution, H2O + [EMIM][DMP], has potential to be a new working pair for the absorption chiller driven by low-grade waste heat or hot water generated by common solar thermal collectors. The performance of absorption refrigeration cycle with methanol and 1,3-methylimidazolium dimethylphosphate (CH3OH + [MMIM][DMP]) is investigated, and a COP of 0.82 was found with condensing, absorption, and evaporation temperatures of 40°C, 30°C, and 5°C, respectively. The requirements of operating pressure, condensing pressure, and vacuum for the system using CH3OH + [MMIM][DMP] are lower, which makes the operation and the maintaining of the system more trouble free [11, 12]. Performance benchmarks and system-level simulations for refrigerant/[BMIM][PF6] pairs are carried out by Kim et al. [13]. A nonrandom two-liquid model is built and used to predict the solubility of the mixture as well as the mixtures properties such as enthalpy and entropy. The evaporator and condenser saturation temperatures are 25°C and 50°C, respectively, with the operating temperature being set at 85°C. As R32, R134a, and R152a are compared with each other, and it is observed that R32 produces the highest performance with a maximum COP of 0.55, while R134a and R152a returned similar COPs of 0.4 at a generator outlet temperature of 80°C.
Kim et al. [14] in one of their other studies have obtained the thermo physical properties of H2O + [EMIM][BF4] with the saturation temperatures at the evaporator and condenser being 25°C and 50°C, respectively. A power dissipation of 100 W is estimated, while COP value of the system reaches 0.91. The suitable compatibility of water with [EMIM][BF4] and the superior properties of water as a heat transfer fluid, such as large latent heat of evaporation, followed by extremely small refrigerant (water) flow rate, resulted in its high performance. Higher viscosity ILs cause an increased pressure drop in the compression loop, which would result in larger pumping power or larger pipes and system volume. The viscosity increases with cation mass: EMIM < BMIM < HMIM. The viscosity is more dependent on the anion with the following order: Tf2N < BF4 < PF6. The viscosity of [EMIM][Tf2N] is only 31.3 MPa s at 294 K, which is 10 times smaller than that of [HMIM][PF6] [14]. The COP of the [MMIM]DMP/methanol absorption refrigeration is lower than LiBr + H2O in absorption refrigeration under the same temperature conditions, while higher than that of H2O + NH3 absorption refrigeration under most temperature conditions. [MMIM] DMP+methanol has excellent potential to be applied as the working pair of absorption refrigeration [15].
Ionic liquids with longer cation alkyl chain length cause a larger solubility but lower dependence of the solubility on temperature [16, 17]. Thus, Ionic liquids with shorter alkyl chains in cation are preferred ([EMIM] > [BMIM] > [HMIM]) due to more sensitive dependence of the solubility on temperature. R143a+[BMIM][PF6] and R134a+[HMIM][PF6] are remarkably less sensitive to temperature and show relatively low COPs [14]. The working pair H2O + EMISE was considered as a potential working pair [18, 19]. The vapor pressure, heat capacity, and density of the H2O + EMISE system were measured and correlated which verified the availability of being used in absorption cycles. Dong et al. [20] recommend H2O + ([DMIM]DMP) as an alternative to H2O + LiBr by comparing the coefficient of performance (COP). To predict the cycle performance, a single-effect absorption refrigeration cycle was simulated based on the models obtained from the studies of vapor pressure and heat capacity of the H2O + ([DMIM]DMP) system. The simulation results show that the cycle performance of H2O + ([DMIM]DMP) is close to that of conventional working pair H2O + LiBr. However, for the cycle using alternative working pair of the H2O + ([DMIM]DMP) system, the operating temperature range has been extended and the disadvantages of crystallization and corrosion caused by H2O + LiBr can be relieved. Ionic liquids have a large capacity of dissolving CO2, so that the [BMIM][PF6] can be used as a refrigerant in an absorption cycle. The COP of a system utilizing CO2/bmimPF6 is much lower than that of a traditional system using the NH3 + H2O pair. However, it should be noted that serious questions remain regarding the accuracy of the ideal-gas heat capacity coefficients for the CO2 + bmim PF6 mixture [21].
The permanent ion-dipole interaction between the ionic liquid and TFE evokes a considerable negative deviation from Raoult's law, which is normal in absorption pairs [22]. The Br anion shows a stronger interface with TFE than the BF4 anion. This means that the required temperature for regeneration will be smaller and the vapor pressure lines will be steeper [23]. Novel working fluids for an absorption heat pump have been proposed by investigating ionic liquids and fluoroalcohol. In this study [BMIM][Br]+TFE and [BMIM][BF4]+TFE have been considered as potential working pairs for the first time. The [BMIM][Br]+TFE system was found to be more favorable than the [BMIM][BF4]+TFE from the results of vapor pressure. The excess volume, apparent molar volume, partial molar volume, and apparent molar expansibility of [MMIM] Cl aqueous solutions were investigated. Based on the results, it was found out that the water content has significant impact on the volumetric properties and the temperature dependence of the density is significantly less than the water content. It was proposed here that [MMIM] Cl has the potential to be used as a novel absorbent species of an absorption cycle working fluid [24].
High circulation ratio increases the energy requirements of heating and pumping processes. Circulation ratios in H2O + LiBr and NH3 + H2O systems usually are smaller, with typical values being around f = 10 [25, 26]. ILs circulation ratios compared to conventional absorption refrigerators are high. The lowest and therefore the best circulation ratios observed ranged from 20 to 25 [27]. The estimated energetic efficiency of the cycle with ionic liquids as absorbents with COP of 0.21 is lower than that of conventional pairs (NH3 + H2O and LiBr + H2O) in absorption refrigerators. However, CO2 + [BMPYRR][Tf2N] with COP of 0.55 is compared with NH3 + H2O pairs operating in equivalent conditions. This is due to the necessity of operating the cycle with a relatively high circulation ratio (24 in the case of CO2 + [BMPYRR/Tf2N] pairs compared to 10 in conventional NH3 + H2O systems), which increases the energy necessities of heating and pumping processes. The content of the ILs has a direct relation with the deviation from the Raoult's law meaning that these binary solutions have a negative deviation from Raoult's law. Solutions such as water, ethanol, and methanol containing [EMIM][DMP] have strong absorbing ability for coolant, which is a very important property of a working pair for absorption heat pump or absorption refrigeration [1].
The processes of [EMIM][DMP] mixed with water, alcohol, and methanol are exothermic under the temperature of the 298.15 K and pressure of 1 atm. They are ranked in the order of water, methanol, and alcohol according to the magnitude of mixing heat. Because of electron negativity of phosphorus contained in the [EMIM][DMP], hydrogen bonds are formed between [EMIM][DMP] and water, alcohol, and methanol, respectively. This property is especially important for the working pair of the absorption heat pump or refrigeration. They are ranked in the order of water, methanol, and alcohol according to the magnitude of mixing heat [1]. The three binary solutions, [MMIM][DMP] + water/ethanol/methanol systems, have exhibited some important characteristics which are needed for an absorption heat pump working pair. Their basic thermodynamic properties including vapor pressure, heat capacity, excess enthalpy, viscosity, and density were measured [18]. The viscosity of pure [MMIM][DMP] was very high, but it would decrease sharply when either [MMIM][DMP] was heated or the water/ethanol/methanol was added. As the absorption and generation temperatures of an absorption heat pump or absorption heat transformer are usually high, the viscosity of the [MMIM][DMP] + water/ethanol/methanol pairs is expected to be low. Consequently, the high viscosity of the ionic liquid would not limit its use as a heat pump absorbent. Zhao et al. [29] investigated the vapor pressure data for nine binary systems at varying temperature, and IL contents were measured using a quasi-static method. The effect of ILs on the vapor pressure lowering of solvent follows the order [MMIM][DMP] > [EMIM][DEP] > [BMIM][DBP] for water and [BMIM][DBP] > [EMIM][DEP] > [MMIM][DMP] for organic solvent methanol and ethanol. This suggests that the “ionic” is specifically dominant in water and “molecule” is prevailing in organic solvents.
Research on the solubility of ammonia in four ILs which contain [EMIM][Ac], [EMIM][SCN], [EMIM]-[EtOSO3], and [DMEA][Ac] has been studied by Yokozeki and Shiflett [30]. The very high solubility behavior has been clearly demonstrated in terms of the thermodynamic excess functions based on the present EOS. Discussion shows that there is opportunity for the absorption cycle application using ammonia-IL systems replacing the traditional ammonia–water system. Also, the COPs and flow ratios of series of ammonia-IL and NH3 + water have been reported with the temperatures of 100, 40, 30, and 10°C corresponding to the generator, condenser, absorber, and the evaporator temperatures, respectively. These COPs and flow ratios obtained from studies [30–32] and compared in [30] are displayed in graphical form in Figures 2 and 3.


Among ammonia-ILs pairs studied, [DMEA][Ac] has shown the best result. The performance (COP) of the present ammonia-ILs systems is somewhat lower than that of the ammonia-water system. The functional capability of replacing LiBr with ILs as an absorbent for H2O in absorption heat pumps and chillers has been demonstrated using theoretical absorption cycle analysis in some experimental works [10, 28, 33–35]. Yokozeki and Shiflett [28] used twelve ILs with H2O in a simple cycle configuration analysis, using coefficient of performance and flow ratio as comparing parameters, which explains the efficiency and compactness of the system. In the analysis, [MMIM][(CH3)2PO4] and [EMIM][(CH3)2PO4] indicate the best results, with the highest COP and the lowest flow rate values. The COP result was about 85–88% of that of the H2O + LiBr system. The results of cycle simulations demonstrate that IL + H2O systems could be competitive with H2O + LiBr system particularly for optimized ILs.
The possible application of water and room-temperature ionic liquids (RTIL) mixtures for the absorption cooling cycle has been investigated using the present EOS and a simple absorption cycle model. It was found that an H2O + RTIL system, when used with an optimized RTIL, could compete with an existing H2O + LiBr system [28]. Also the COPs and flow ratios of series of H2O + IL and NH3 + water have been reported with temperatures of 100, 40, 30, and 10°C corresponding to the generator, condenser, absorber, and evaporator temperatures, respectively. The data used for the graphical representations in Figures 4 and 5 are taken from Yokozeki and Shiflett [28].

COP of series of H2O + IL and H2O + LiBr previously presented in [28].

Flow ratio of series of H2O + IL and H2O + LiBr previously presented in [28].
3. Discussion
As illustrated in Table 1, a comparison was made among different ionic liquids in order to choose suitable working pairs. Among most of the ionic liquids, water was chosen as refrigerant due to its superior properties as a heat transfer fluid, such as large latent heat of evaporation followed by extremely small flow ratio that resulted in its high performance. Selecting working fluids for absorption cycles should be suitable for initial operating conditions and limitations, such as crystallization, corrosion problems, and environmental limits, and also material requirement should be considered.
Characteristic comparison of different working fluids.
4. Conclusions
In this paper a number of researches about working fluids of absorption cycles, which contain absorption heat pumps, absorption chillers, and absorption heat transformers including ionic liquids, were reviewed. Environmental issues and saving energy concerns have always been a major global problem. Absorption cycles can play an important role due to their capability of reducing CO2 discharge and to reuse large amount of industrial waste heat. Since the performance of absorption cycles mainly depends on the working fluids, the research aspect of improving working fluids is quite essential and in progress. Using ionic liquids as the working fluids of absorption cycles can lead us to benefit from factors such as less crystallization, less corrosion, low toxicity, and nonflammability in comparison with conventional working fluids including (NH3 + water and LiBr + water).
Footnotes
Acknowledgment
The authors gratefully acknowledge Canay Ataoz librarian of the Eastern Mediterranean University for her endless support and kindness.
