Abstract
Although significant progresses have been made in sensor technology, it is still a challenging task to develop ultrasensitive sensors to monitor very weak and low frequency vibration signal for early warning of natural disasters and efficient structural health monitoring of infrastructures. It has been reported from previous experiments that some fishes have acute sensitivities to extremely low frequency linear acceleration due to the otolith organs of the inner ear. In this paper, based on the experimental results and qualitative mechanism of the infrasound sensitivity of some fishes conducted by other researchers, a bioinspired gating spring model with negative stiffness is established to simulate the mechanical-electricity transduction of the hair cell in fish ear. Then, numerical analyses of the mechanical model subject to static and dynamic loading are conducted, respectively. It is shown that the gating model has adaptive amplification capability to weak and low frequency excitation compared with the corresponding linear model. This mechanism can be used for the design of bioinspired ultrasensitive sensors for monitoring weak and low frequency vibration signal.
1. Introduction
It is essential to develop high sensitive sensors for early warning of natural disasters and efficient health monitoring of infrastructures [1, 2]. Nowadays, infrastructures are large in size. Ambient vibration signals of these large scale infrastructures including tall buildings and long span bridges are weak, low-frequency dominated with frequencies less than 1 Hz and sometimes around 0.1 Hz, and flooded in the ambient noise as the spectra of typical ambient noise grow toward the lower frequencies [3]. Also, structural damage is essentially a local phenomenon; thus only high sensitive sensors near the site of damage can sense the minor changes of vibration signal caused by damage. Moreover, weak and low frequency vibration signals can also be used to monitor natural hazards other than earthquakes, such as rock avalanches, debris flow, and flooding [4]. Amitrano et al. [5] indicated that the frequency range of 0.1–1 Hz is the most sensitive to mudslide velocity and that the frequency range of 0.01–10 Hz is associated with landslide deformation. Kao et al. [6] found two distinct seismic signatures of landslides with tremor-like waveforms and a predominant frequency band of 0.5–5 Hz. Feng [7] indicated that the frequency range of 0.5 to 1.5 Hz is the main frequency of the seismic signal of the Shiaolin landslide occurring in Taiwan in 2009. Dammeier et al. [8] showed that it is possible not only to locate the landslides to within a few kilometers but also to extract information about the characteristics of the landslide from the seismic signals. Thus, seismic signals may be abnormal and in the range of infrasound during geological disasters, such as landslides, and spread to the surrounding air medium. Velocity of infrasound is much larger than that of landslide, so imminent landslides can be forecasted through infrasound monitoring by advancements in ultrasensitive low frequency sensors.
Although a lot of advanced sensors have been developed for monitoring weak and low frequency vibration signal, these sensors are too bulky, large energy consuming, and expensive; for example, a Streckeisen STS-2 seismometer weights 9 kg and costs more than 10 k US dollars, which can hardly meet the demands on a large number of sensitive sensors for monitoring large size structures and natural disasters. Also, current sensors do not have adaptive sensitivity; that is, sensors with high sensitivity to weak signals, intended for ambient monitoring, will saturate during strong excitation.
On the other hand, former experiments have shown that some fishes, such as teleost species, can detect very weak and low frequencies motion. At 0.1 Hz, the particle acceleration threshold of Atlantic cod was about 10−5 m/s2 [10, 11]. All plaice readily responded to infrasound down to 0.1 Hz with threshold of approximately
The otolith organs contain a dense calcified matrix (the otolith) overlying a sensory epithelium (or macula) containing hair cells as shown in Figure 1 redrawn from Popper [9]. The basic structure of the otolith organs of fishes closely resembles that of an accelerometer—a mass that moves in a relative manner to some kind of receptor. The otolith serves as the mass, whereas the sensory hair cells serve as the receptors [16]. Sand and Karlsen [17] indicated an otolith organ as a simple harmonic oscillator. The unaided otolith organs of the inner ear are not sensitive to sound pressure but to linear accelerations, which is the more relevant stimulus parameter at very low frequencies [11]. Actually, the hair cells display adaptive signal amplification to mechanical stimuli, leading to high sensitivity to weak input and low sensitivity to high input, thus expanding the dynamic range through adaptive amplification [18].

Schematic of the hair cells in relation to the otolith, redrawn from Popper [9].
Recently, some researchers have studied ultrasensitive low frequency sensors using the high sensitivity of fish's hearing ability to infrasound. Lim and Park [21] investigated the mechanical model of gating spring mechanism of stereocilia. Zhang et al. [22] simulated the octopus' hearing system as dynamic oscillating system. The modeling results agree reasonably well with the experimental audiograms (acceleration threshold, velocity threshold, and sound pressure level) of the three cephalopod species. Zheng et al. [23] developed capacitive accelerometer incorporating positive and negative feedbacks into the system, inspired by the operation mechanisms of the inner ear hair cells. Lee and Park [24] developed a mechanical model consisting of an inverted pendulum and a fixed T-bar that mimic the interaction of two adjacent stereocilia, and a magnet pair was attached to the top of the inverted pendulum and the fixed T-bar to emulate the negative resistance of the tip link due to the transient stiffness softening by the gating ion channel. The authors have also studied the mechanism of gating model of hair cell subject to static and dynamic loading at the tip of the tallest stereocilia [25].
In this paper, based on observed experimental results from other researchers [10–13] on the mechanism of the sensitivity of fish's inner ear to extremely weak low frequency motion, a bioinspired mechanical model with gating spring for simulating the mechanical-electricity transduction of fish's hair cell is established. Then, numerical responses of the mechanical model subject to static loading and ground motion are analyzed by the Open System for Earthquake Engineering Simulation (OpenSees), a popular software for simulating the seismic response of structural systems. Finally, mechanical functions of the proposed mechanical model on the design of new high sensitive bioinspired low frequency sensors for effective monitoring the vibration signals of civil infrastructures and early warning of natural disasters are addressed.
2. Mechanical Behaviors of Fish's Hair Bundle
A fish's hair cell bundle may contain from 20 to 300 stereocilia aligned in the order of height, which are interconnected by long protein strings called tip links. Stereocilia are relatively stiff and pivot at a tapered base. In hair cells of the inner ear, mechanical stimulation supplies the work necessary to open transduction channels. The dense otolith provides a stimulus for the hair cells, which results in the shear deformations of the hair cell [11]. In Figure 2(a), Stereocilia bundles are in resting state and ion channels are closed. When a hair bundle is deflected by the positive phase of a sinusoidal stimulus (Figure 2(b)), increased tension of tip link opens the ion channels at the tip of the stereocilia, and Ca2+ enters through the transduction channel (Figure 2(c)). Then the associated tip link shortens, and the tension in the link falls. Relaxation of the tip link acts like an external force in the positive direction, causing the bundle to move still further. The reduced tension in the tip link then permits the channel to reclose (Figure 2(d)). As the channel shuts, the increased tension in the tip link exerts a force that moves the bundle in the negative direction. When the ion is extruded by a membrane Ca2+ pump, the hair bundle is primed to repeat the cycle (Figure 2(e)) [14, 15].

3. A Bioinspired Mechanical Model
3.1. Modeling
Based on the mechanical behaviors of fish's hair bundle and the bioinspired mechanism, an electromechanical model that extends the gating-spring model described by Lim and Park [21] is established to simulate the adaptive signal amplification functions of the auditory hair cells of fish. This model consists of an inverted pendulum array of 21 stereocilia bundles supported by pivotal springs at the base and interconnected by tip link springs. Each stereocilium is modeled as a rigid bar with a hinge joint at its base that approximates the nonbending, rotational motion of a stereocilia's tapered base, as shown in Figure 3(a). Based on the biomechanics characteristics of tip link and ion channel described in Figure 2, the gating spring is modeled by the nonlinear spring characteristics to simulate the tip-link and ion gating channel and connects the tip of the adjacent stereocilium, as shown in Figure 3(b).

Simulation model of stereocilia bundle. (a) Stereocilia bundle; (b) gating spring (tip link) and ion channel; (c) sketch of stiffness of the gating spring.
The gating spring is defined as a nonlinear elastic spring with variational stiffness shown in Figure 3(c). The gating force
Mechanical model of the above stereocilia bundles is established by OpenSees with the parameters of mechanical model shown in Table 1. Detailed explanations of the parameters in the table are shown after Table 1.
Parameters of simulation model.

Details of some parameters are listed below:
(1)
(2)
(3) α—angle of tiplink with respect to the horizontal direction, estimated from TEM and SEM by Cotton and Grant [19].
(4) k—pivotal stiffness of a single stereocilia; the table value shown was determined to make the stereocilia bundles have sufficient stiffness.
(5)
(6) ζ
3.2. Analyses of the Model Subject to Static Loading
A static external force 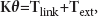

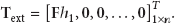

The magnitudes of the static external load range from 0 to 100 pN with an interval of 0.02 pN. The displacements at the tip of the tallest stereocilium in the two models with gating and without gating are calculated by OpenSees, respectively.
Figure 4(a) shows the comparisons of displacements at the tip of the tallest Stereocilia in the two models subject to variation of static loading. The threshold force required to open the first gate spring is 5.76 pN, which is a very small value. It is clear that twenty gates are opened sequentially one by one. The amplification of gating model is defined as the ratio of the amplitude of displacement of gating model and that of the model of without gating. As shown in Figure 4(b), the amplification is in rising tendency with the increasing of external load for small values of loading and trends to decrease for larger values of external loading. Therefore, the model is highly sensitive to weak external loading and lowly sensitive to large external loading, due to the negative stiffness of gating spring.

Analysis result of the two models subject to static loading. (a) Comparisons of displacement at the tip of the tallest stereocilium under static loading. (b) The sensitivity of the gating model subject to static loading.
3.3. Analyses of the Model Subject to Ground Motion
The whole stereocilia bundles and the otolith are subject to the ground acceleration motion as shown in Figure 5. The mass of each stereocilia bundle is ignored because it is negligible when compared with that of the otolith.

Model subject to ground acceleration motion.
The governing equation of the stereocilia array and the otolith can be obtained as follows:

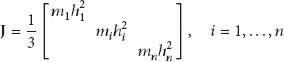
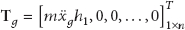
To examine the adaptive amplification of the gating model to the amplitude of external excitation, it is assumed that the gating model and model without gating are subject to the ground acceleration motion in sinusoidal wave; that is,
The displacement responses at the tip of the tallest stereocilium in the two models (with gating spring and without gating spring) are calculated by OpenSees. Figure 6 shows the time history of the relative displacement responses of the tip of the tallest stereocilium of the two models subjected to sinusoidal ground motion with a different amplitude

The time history chats on displacement at the tip of the tallest stereocilium of two models under ground motion at 0.1 Hz.

Analysis result of the two models subject to ground acceleration motion. (a) Comparisons of the amplitudes of relative displacements at the tip of the tallest stereocilium in the two models subject to ground acceleration motion. (b) The sensitivity of the gating model subject to ground acceleration motion.
To examine the adaptive amplification of the gating model to the frequency of external excitation, the gating model and model without gating are subject to sinusoidal ground acceleration motion with variational frequency ω. The amplitude of ground acceleration

The time history chats on relative displacements of two models to frequency ratio.

The comparisons of relative displacements to frequency ratio of two models.
3.4. Low Frequency Thresholds for Gating Hair Cell Model
Low frequency thresholds for gating hair cell model are defined as the amplitude of ground motion required to open the first gate spring; that is, the first ion channels is opened, because opening the transduction channel in the cilia and allowing the entry of calcium ions into the cell result in a cascade of events that leads to the release of chemical neurotransmitters from the base of the cell. The neurotransmitter crosses a small gap between cells and excites the endings of the nerve that innervates the cell. This, in turn, results in an electrical potential (the action potential) in the nerve that is carried to the brain.
Acceleration threshold of ground motion for gating hair cell model is shown in Figure 10(a). The thresholds at 0.01 Hz and 0.1 Hz are all approximately 0.58 μm/s2 (or −11 dB re: 1 μm/s2). It is shown clearly that acceleration threshold is in sharp increase with the rise of frequency when the exciting frequency exceed the nature frequency (0.927 Hz). Meanwhile, below the upper frequency cutoff, the acceleration thresholds audiogram is virtually flat, as predicted by Sand and Karlsen [17]. The acceleration thresholds audiogram of gating hair cell model is also similar with that of perch tested by Karlsen shown in Figure 10(b) [13]. These two figures both depict that when thresholds are related to particle acceleration, which is the relevant stimulus parameter at very low frequencies, the apparent drop in sensitivity towards low frequencies disappears.

Acceleration threshold of fish ear. (a) Acceleration threshold for gating hair cell model; (b) acceleration thresholds for perch stimulated at different frequencies from Karlsen [13].
4. Conclusions
In this paper, based on the experimental results of the infrasound sensitivity of some fishes from other researchers, a bioinspired gating spring model with negative stiffness is established to simulate the mechanical-electricity transduction of the hair cell in fish inner ear. Through numerical analyses of the model by OpenSees, it is shown that the gating model is more sensitive to weak excitation and low frequency ground acceleration motion due to the negative stiffness of whole stereocilia bundles caused by the opening of gating spring.
The gating model simulating the mechanical-electricity transduction of hair cell and its mechanical function can be used as the theoretical basis for the design of bioinspired sensors with adaptive amplification capability and ultrasensitivity to weak and low vibration signals for efficient monitoring of infrastructures and early warning of natural disasters.
This paper only conducts numerical analyses of the mechanism of a hair cell bioinspired sensor. Further experiment validation work of presented mechanism is necessary.
Footnotes
Acknowledgments
This work is sponsored by the Natural Science Foundation of China (no. 51308482) and by the Key Project of Fujian Province of China (no. 2013Y0079). The authors are also grateful to Professor Y. F. Zhang of University of Maryland for his great contributions to the research project.


