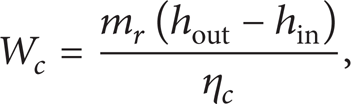Abstract
Energy consumption of heating and domestic hot water is very high and will keep increasing. Air source absorption heat pump (ASAHP) was proposed to overcome the problems of low energy efficiency and high air pollution existing in boiler systems, as well as the problem of bad performance under low ambient temperatures for electrical heat pumps. In order to investigate the crystallization possibility of ammonia-salt ASAHP, crystallization margin (evaluated by solution mass concentration) at generating temperature ranging from 100 to 150°C, evaporating temperature from −30 to 10°C, and condensing temperature from 30 to 65°C are analyzed. To prevent the NH3–NaSCN solution from crystallizing, ASAHP integrated with pressure booster located between the evaporator and absorber is simulated. Analysis and comparisons show that NH3–NaSCN is easy to crystallize at relatively high generating temperature, low evaporating temperature, and low condensing temperature. But crystallization margin of NH3–LiNO3 can always stay above 5% for most conditions, keeping away from crystallization. Pressure booster can effectively avoid the crystallization problem that will take place in the NH3–NaSCN ASAHP system.
1. Introduction
Energy consumption of heating and domestic hot water is very high and is predicted to keep increasing rapidly [1]. For the existing heat supply systems, boilers are of low energy efficiency and high air pollution [2], electrical heat pumps show bad performance under low ambient temperatures [3, 4]. In order to solve these problems, heat supply systems based on air source absorption heat pump (ASAHP) were put forward in the previous work [5, 6]. Analysis on energy saving potential indicated that the ASAHP systems are promising for heating and domestic hot water. Regarding the selection of working fluids, ammonia-based pairs, such as NH3–H2O, NH3–NaSCN, and NH3–LiNO3 [7, 8], are suitable for ASAHP systems in cold regions, where the evaporating temperature is always below 0°C, and water-based working pairs cannot be used due to the freezing point limitation of refrigerant water [9, 10]. Among those ammonia-based working fluids, NH3–NaSCN and NH3–LiNO3 obtained wide attentions since the rectifier is not required anymore compared with the traditional NH3–H2O [11, 12].
Crystallization is a big problem for actual applications of H2O–LiBr chillers, which has been studied in numerous works [13, 14]. As for ammonia-based working fluids, there is little research on the crystallization possibility except several statements that NH3–NaSCN chiller cannot work when the evaporating temperature is below −10°C for the concern of crystallization [15–18], without any further analysis. And there is no crystallization analysis on another ammonia-salt absorption system, NH3–LiNO3 ASAHP. As a result, the detailed investigation on crystallization problem of ammonia-salt absorption systems should be carried out for better design and operation of the absorption systems. For the ASAHP heating systems used in cold regions, the evaporating pressure will be very low and the decreased pressure difference will weaken the absorption between solution and refrigerant. Besides, the solution temperature in the absorber of ASAHP is much higher than that of the cooling applications, which is also adverse to the absorption ability. Consequently, the mass fraction of salt in the ammonia-salt solution needs to be much higher, or the cooling water temperature needs to be lower to maintain a smooth absorption process, which may be unfavorable for staying away from crystallization. This paper is to investigate whether the crystallization crisis will happen and in which situation it happens for both NH3–NaSCN and NH3–LiNO3 ASAHP used in cold regions. As one of the crystallization control methods [19] in H2O–LiBr refrigeration systems, pressure boosting between the evaporator and absorber is introduced to the ASAHP heating systems to avoid the potential crystallization of ammonia-salt solutions.
2. Methodology
2.1. System Description
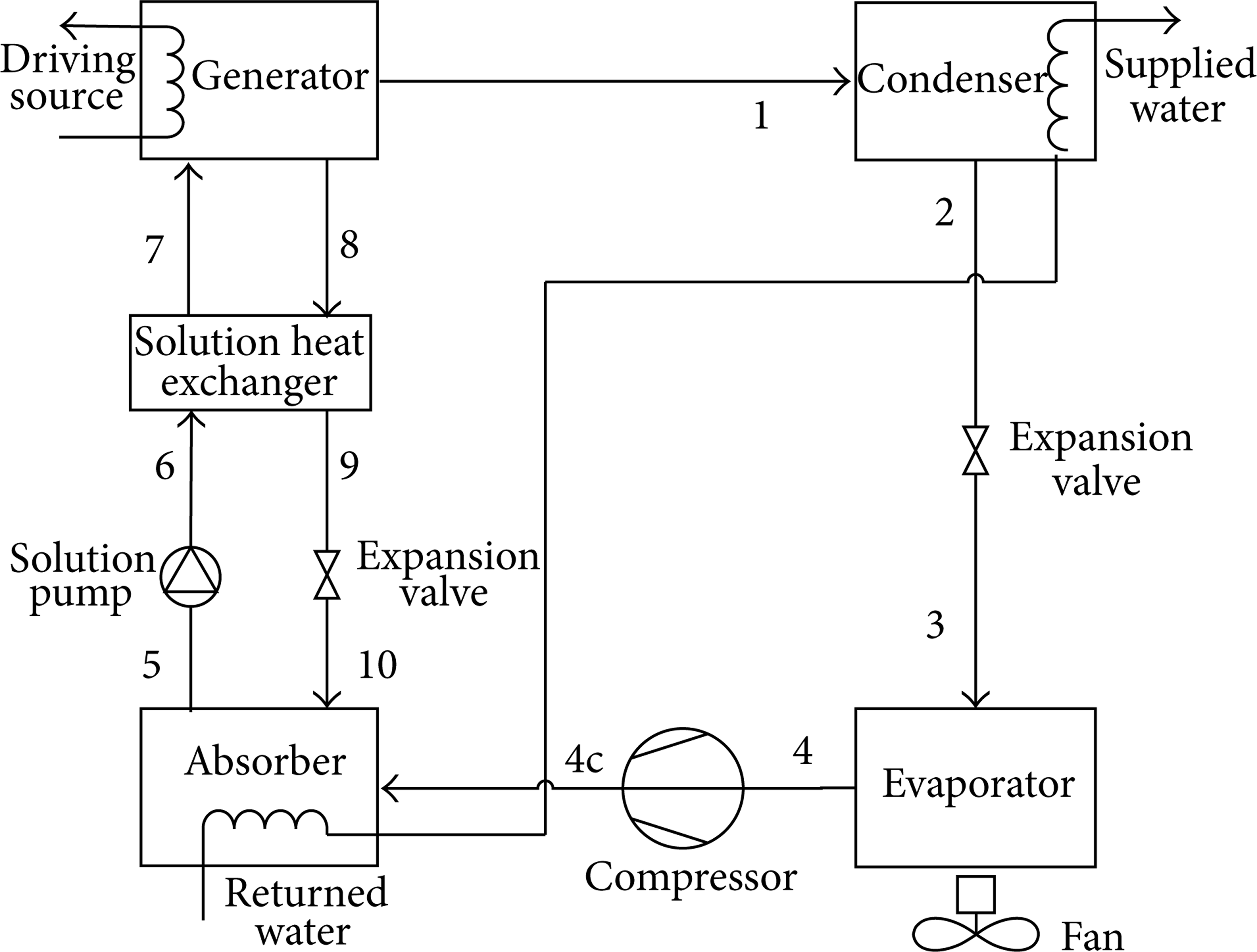
The schematic diagram of ASAHP is shown in Figure 1. Low-grade heat in the ambient air is extracted by the air-source evaporator, and the returned water is heated to the required temperature in the absorber and condenser. The produced hot water is arranged to pass through the absorber first to obtain good absorption ability under low evaporating temperatures [6]. The detailed operation principle and mathematical model of ASAHP can be found in [5, 6].

Schematic diagram of ASAHP heating system.
2.2. Fluid Properties
The properties of pure ammonia involved in this work are called from NIST [20]. Infante Ferreira [16] provided the thermodynamic and physical properties correlations of NH3–LiNO3 and NH3–NaSCN. The solubility properties of the salt solutions are especially essential for crystallization analysis. And the crystallization concentration of NH3–LiNO3 and NH3–NaSCN was fitted as functions of solution temperature in different concentration ranges, as shown respectively in
where x c is the crystallization concentration, %; T is solution temperature, °C.
2.3. Crystallization Margin
In order to evaluate how close the concentrated solution is near crystallization, crystallization margin xmargin is defined as the difference between the concentration of strong solution (with strong absorption ability) and the crystallization concentration corresponding to the solution temperature at the absorber inlet, the lowest temperature in the strong solution circulation. xmargin is expressed as
where xa inlet is the concentration of strong solution (the mass fraction of ammonia in the ammonia-salt solution) at the absorber inlet, %; x c (Ta inlet) represents the crystallization concentration corresponding to the temperature of strong solution at the absorber inlet (Ta inlet), %.
A negative valve of xmargin means that ammonia mass fraction of strong solution is smaller than the crystallization concentration. Consequently, crystallization will appear in this situation. The more negative the valve of xmargin is, the more seriously the crystallization occurs. On the contrary, the more positive the valve of xmargin is, the farther the solution is away from crystallization.
2.4. System Performance
To analyze the effect of crystallization avoiding and system performance provided by the integrated ASAHP + compressor system, the model of compressor should be built, apart from the basic model of AHP in the previous studies [5, 6]. The electricity consumption of compressor can be calculated as [21]
where W c is the electricity consumption, kW; m r is the mass flow rate of refrigerant ammonia, kg/s; hout and hin are the refrigerant enthalpy at compressor outlet and inlet, kJ/kg; η c is the efficiency of compressor, which is taken as 0.65 [21, 22] in this work.
The coefficient of performance (COP) of the integrated cycle is
where Q a and Q c is the heat capacity of absorber and condenser, kW; Q g is the heat consumption of generator, kW; ηpower is the efficiency of coal-fired electricity generation, which is taken as 0.33 in this work [23].
Based on the previously equations and the mathematical models in the previous studies [5, 6], both single ASAHP and hybrid ASAHP + compressor cycles are simulated to investigate the crystallization crisis and the effect of crystallization avoiding. In addition, the performances of these two systems are calculated and compared.
3. Crystallization Analysis and Comparisons
3.1. Crystallization Analysis on PTX Diagram
Two typical working conditions are chosen to illustrate the ASAHP cycle in pressure-temperature-concentration (PTX) diagram of NH3–NaSCN and NH3–LiNO3, which can directly show the possibility of crystallization. The two working conditions are as follows: condition 1 (T g 140°C, T c = 45°C, T a = 40°C, T e = – 20°C) and condition 2 (T g = 115°C, T c = 40°C, T a = 35°C, T e = – 12°C). The corresponding supplied hot water temperature is respective 40°C and 35°C, which is typical for low-temperature heating, such as floor radiation. The crystallization analysis of NH3–NaSCN and NH3–LiNO3 is shown in Figures 2 and 3, respectively. The state point numbers are in accordance with Figure 1.

Crystallization analysis of NH3–NaSCN under typical working conditions.

Crystallization analysis of NH3–LiNO3 under typical working conditions.
It can be found from Figure 2(a) that crystallization occurs in working condition 1. The crystallization is very serious, since not only the process line 8→10 (strong solution flowing from generator to absorber) crosses the crystallization line, but also the process line 5→7 (weak solution flowing from absorber to generator) approaches crystallization. In working condition 2, the NH3–NaSCN ASAHP has no danger of crystallization at an evaporating temperature of −12°C, as shown in Figure 2(b).
Besides, Figures 3(a) and 3(b) indicate that there is no possibility of crystallization for NH3–LiNO3 ASAHP in both two working conditions. And the cycle is far from crystallization in working condition 2.
The parameters including pressure, concentration, temperature, and COP of NH3–NaSCN and NH3–LiNO3 are compared in Table 1. It can be more specifically seen that the crystallization margin of NH3–LiNO3 is much larger than that of NH3–NaSCN. Consequently, NH3–LiNO3 has less possibility of crystallization. Moreover, COP of NH3–LiNO3 is also higher, which is 1.418 in working condition 1 and 1.474 in working condition 2.
Crystallization comparisons under typical working conditions.

3.2. Crystallization Analysis in Whole Temperature Ranges
The previous analysis in two typical working conditions implies that NH3–NaSCN is more probable to crystallize than NH3–LiNO3. However, the ASAHP cycle has a much wider operation range of temperature actually. It is necessary to investigate the crystallization possibility in the whole temperature ranges for these two absorption pairs. To conduct this analysis, generating temperature is set from 100°C to 150°C, evaporating temperature is set from −30°C to 10°C, and condensing temperature is set from 30°C to 65°C. Crystallization margin xmargin and COP of ASAHP using NH3–NaSCN and NH3–LiNO3 as working fluids are simulated, and the results are compared in Figures 4∼6.

Crystallization margin and COP versus generating temperature.
In Figure 4, condensing temperature and absorbing temperature is respectively kept constant at 50°C and 45°C, while evaporating temperature is set as −5°C and −20°C in two cases. When the evaporating temperature is −5°C, xmargin of NH3–NaSCN is below 0% and the solution is in danger of crystallization in the generating temperature range of 140∼150°C. In the generating temperature of 115∼138°C, xmargin of NH3–NaSCN is 0%∼6%, and crystallization is absent, but the COP is much lower than that of NH3–LiNO3. For NH3–LiNO3 ASAHP, xmargin stays above 5%, keeping far away from crystallization.
When the evaporating temperature is as low as −20°C, xmargin of NH3–NaSCN is below −1% in the generating temperature range of 140–150°C. And in the range of 100–140°C, the generating temperature is not high enough to drive the NH3–NaSCN ASAHP at such a low evaporating temperature. As for NH3–LiNO3, xmargin can stay above 4% in the generating temperature range of 125–150°C without any crystallization concern.
In Figure 5, generating temperature is kept constant at 130°C, while condensing temperature is set as 50°C and 40°C in two cases. When the condensing temperature is 50°C, xmargin of NH3–NaSCN is above 1%, without the danger of crystallization in the evaporating temperature range of −14∼10°C. But in the range of −30∼−14°C, the NH3–NaSCN ASAHP cannot work due to the negative concentration difference caused by low evaporating temperature. For NH3–LiNO3 ASAHP, xmargin stays above 8% in the evaporating temperature range −24∼10°C, being far away from crystallization.

Crystallization margin and COP versus evaporating temperature.

Crystallization margin and COP versus condensing temperature.
When the condensing temperature is 40°C, the evaporating temperature suitable for NH3–NaSCN can be much lower and the COP can also be higher. However, the xmargin keeps negative and crystallization always exists in this case. When it comes to NH3–LiNO3, the evaporating temperature can be very low and the COP can reach 1.38 when it is as low as −30°C. In addition, xmargin can keep higher than 5% in the whole range of −30–10°C, so crystallization is always impossible.
In Figure 6, generating temperature is kept constant at 130°C, while evaporating temperature is set as −5°C and −20°C in two cases. When the evaporating temperature is −5°C, NH3–NaSCN has the danger of crystallization in the condensing temperature range of 30–43°C. Though no crystallization occurs when condensing temperature is higher than 45°C, the COP of NH3–NaSCN ASAHP will decrease greatly. For NH3–LiNO3 ASAHP, crystallization is still impossible.
When the evaporating temperature is as low as −20°C, NH3–NaSCN crystallizes in the condensing temperature range of 30–46°C and refuses to work when it is above 46°C. When it comes to NH3–LiNO3, crystallization will not happen as long as the ASAHP can work normally.
Besides, it is obvious that COP of NH3–LiNO3 ASAHP is higher than that of NH3–NaSCN ASAHP in most conditions. The advantage is especially great at lower generating temperatures, lower evaporating temperatures, and higher condensing temperatures, as illustrated in Figures 4∼6.
In summary of the above analysis, it can be concluded that NH3–NaSCN is easy to crystallize at relatively high generating temperature, low evaporating temperature, and low condensing temperature when other parameters are fixed. However, when the working conditions lead no crystallization to NH3–NaSCN, the COP will be much worse. Comparatively speaking, NH3–LiNO3 can always keep away from crystallization problem in all the operation conditions with higher operation performance.
4. Crystallization Avoiding by Pressure Boosting
In order to prevent the NH3–NaSCN solution from crystallizing, a pressure booster can be located between the evaporator and absorber to enhance the pressure of refrigerant at the absorber inlet. In this way, the solution in absorber does not need to be so strong (and the cooling water temperature does not need to be low enough) to obtain good absorption ability when the evaporating temperature is too low. In this paper, a compressor is used to enhance the pressure of refrigerant leaving evaporator and to supply the compressed refrigerant to absorber, as shown in Figure 7.
Schematic diagram of ASAHP with compressor.
Figure 8 illustrates the cycle comparison in PTX diagram between NH3–NaSCN ASAHP and ASAHP + compressor. The working condition designed for fan coil heating (supplied hot water temperature 45°C) is set as T g = 140°C, T c = 50°C, T a = 45°C, and T e = – 15°C. The compression ratio is CR = 2.5. Process 7 → 8 → 10 → 5 represents the solution cycle of ASAHP without compressor, and 7c → 8 → 10c → 5c represents the solution cycle of ASAHP with compressor. It is obvious that the compressor can effectively avoid the crystallization problem that will take place in the NH3–NaSCN ASAHP system.
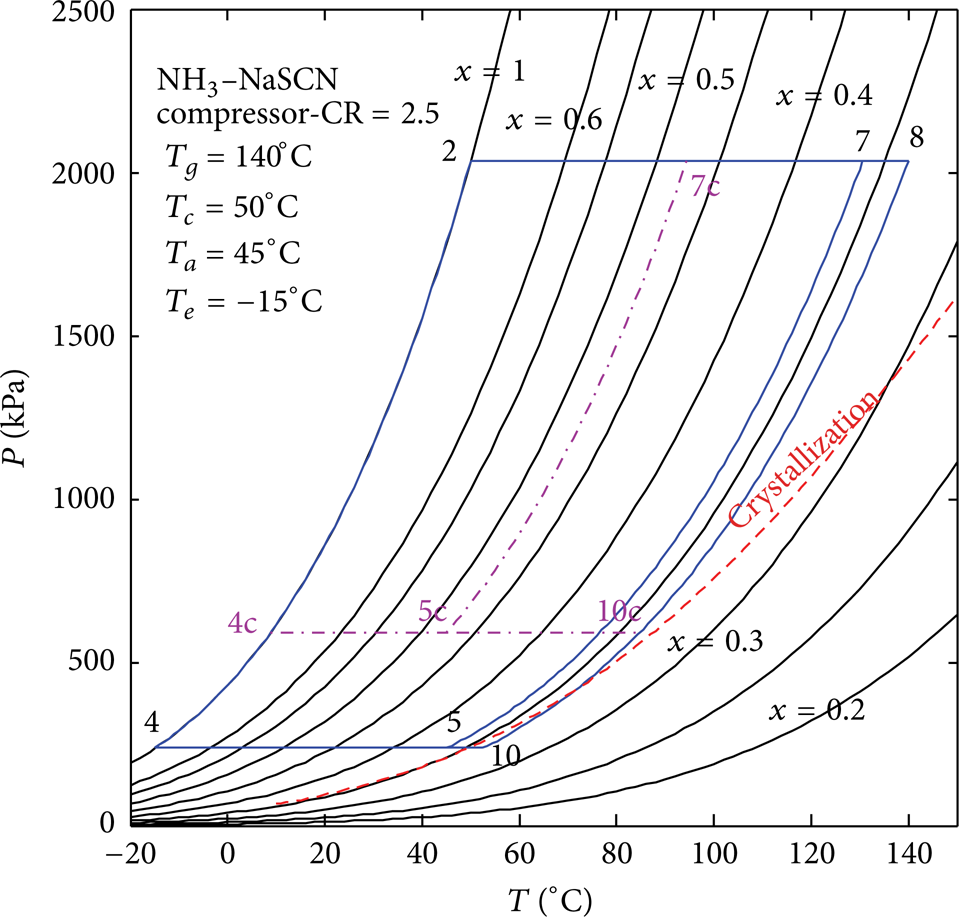
Crystallization analysis of NH3–NaSCN ASAHP with compressor.
More detailed analysis with different compression ratio is listed in Table 2. It can be found that xmargin can be improved by enhancing the refrigerant pressure entering the absorber (p a ). After combining a compressor, COP of the hybrid ASAHP will become 1.214 at a CR of 2.5 and will become 1.171 at a CR of 3.0. For comparison, another working condition designed for floor radiant heating (supplied hot water temperature: 35°C; T g = 130°C, T c = 40°C, T a = 35°C, and T e = – 5°C) is also considered, which is shown in Table 3. It is indicated that crystallization can be avoided by lower compression ratio under lower hot water temperature. In this situation, the COP will become 1.312 at a CR of 2.0 and will become 1.251 at a CR of 2.5.
Crystallization control effect of NH3-NaSCN. ASAHP + compressor for fan coil heating.

Crystallization control effect of NH3-NaSCN. ASAHP + compressor for floor radiant heating.

In summary, crystallization of NH3–NaSCN ASAHP in cold regions can be effectively avoided by low pressure boosting at the cost of lower performance. For lower temperature heating, such as floor radiant heating, the performance will be better. And NH3–LiNO3 is the better choice for heating in cold climates in the point view of higher COP as well as no crystallization crisis in the operation ranges.
5. Conclusions
Heating and domestic hot water systems based on ASAHP were put forward to overcome the problems of the commonly used boiler and the electrical heat pump systems. To investigate the crystallization possibility of NH3–NaSCN and NH3–LiNO3 ASAHP, crystallization margin xmargin at generating temperature from 100°C to 150°C, evaporating temperature from −30°C to 10°C, and condensing temperature from 30°C to 65°C are analyzed. To prevent the NH3–NaSCN solution from crystallizing, a compressor is located between the evaporator and absorber, and the integrated ASAHP + compressor cycle is simulated. From the analysis and comparisons, conclusions can be draw as follows.
When other parameters are fixed, NH3–NaSCN is easy to crystallize at relatively high generating temperature, low evaporating temperature, and low condensing temperature.
xmargin of NH3–LiNO3 stays above 5% for most conditions, keeping away from crystallization problem.
Crystallization of NH3–NaSCN ASAHP can be effectively avoided by low pressure boosting at the cost of lower performance. For lower temperature heating, the performance will be better.
NH3–LiNO3 is the better choice for ASAHP in cold climates in the point view of higher COP as well as no crystallization crisis in the operation ranges.
Footnotes
Nomenclature
Acknowledgment
The authors gratefully acknowledge the support from the Natural Science Foundation for Distinguished Young Scholars of China (Grant no. 51125030) and the National Basic Research Program of China (Grant no. 2010CB227305).