Abstract
Objective:
Cannabinoid CB2 receptors (CB2Rs) are mainly present on immune cells including mast cells, which participate in 2,4-dinitrofluorobenzene (DNFB)-induced allergic contact dermatitis (ACD). In this study, we aimed to investigate whether inhibition of mast cell degranulation was involved in the anti-ACD effect of electroacupuncture (EA) at ST36 via CB2R.
Methods:
Sprague-Dawley rats were sensitised and challenged with DNFB following EA stimulation for 1 week. Ear swelling, serum IgE levels, local cytokine production and mast cell infiltration were evaluated. Additionally, rat peritoneal mast cells (RPMCs) were isolated and cultured for detection of CB2R expression, mitogen-activated protein kinase (MAPK) signalling activation and mast cell degranulation (including β-hexosaminidase and histamine release) in the presence or absence of CB2R antagonists.
Results:
EA treatment inhibited ear swelling, suppressed IgE and cytokine production, decreased the number of mast cells and curbed mast cell degranulation, which was associated with the inhibition of p38 phosphorylation in DNFB-induced ACD. Importantly, EA enhanced the expression of CB2R mRNA and protein in the RPMCs. CB2R antagonist AM630 but not CB1R antagonist AM251 effectively reversed the suppressive effect of EA on p38 activation, mast cell infiltration and degranulation.
Conclusion:
These findings provide more evidence to support the hypothesis that EA promotes CB2R expression in mast cells, which is followed by inhibition of the p38 MAPK pathway, potentially resulting in the anti-ACD effect of EA. This suggests that EA at ST36 may be an effective candidate therapy for treating inflammatory skin diseases such as ACD.
Keywords
Introduction
Allergic contact dermatitis (ACD) is a well-known T-cell-mediated inflammatory skin disease, which is characterised by typical allergic symptoms of the skin including redness, oedema and pruritus, accompanied by scaling and dryness. 1 Besides T cells, Langerhan cells, keratinocytes, dermal dendritic cells (DCs), natural killer (NK) cells and mast cells (MCs) are also involved in the immunomodulatory effects of ACD.2,3 On activation, MCs release bioactive substances such as histamine, tumour necrosis factor α (TNFα) and interleukin 8 (IL-8), which are major contributors to the late-phase reactions and inflammation accompanying the recruitment and activation of other immune cells. 4 In addition, MCs are helpful in activating cutaneous DCs once they have accepted the stimulation of haptens in the sensitisation phase of ACD. 5
Increasing evidence indicates that MCs can be influenced differently by the cannabinoid system. 6 The biological effects of cannabinoids are determined via their binding to two cell membrane G0-protein coupled receptors, named cannabinoid CB1 receptor (CB1R) and cannabinoid CB2 receptor (CB2R). CB1R is also referred to as the ‘central’ cannabinoid receptor because it is mainly expressed in the central nervous system. Conversely, CB2R is referred to as the ‘peripheral’ cannabinoid receptor because it is mostly found on immune cells. 7 A previous study showed that CB1R and CB2R are co-expressed in MC line rat basophile leukaemia cells but function through different pathways. 8 A recent publication reported that inhibition of MC degranulation, at least partly, was attributed to activation of CB2R. 9 Moreover, mice lacking CBRs developed an exacerbated ear swelling in response to 2,4-dinitrofluorobenzene (DNFB), which indicated that the endocannabinoid system possessed the potential to attenuate ACD. 10
In recent decades, some studies have indicated that there is a greater concentration of MCs at the location of traditional acupuncture points in the skin of rats and humans compared to the neighbouring tissues. 11 However, few studies have focused on the effect of electroacupuncture (EA) stimulation on MCs and the potential role of the cannabinoid receptors. The results of the present study demonstrate that the inhibition of MC degranulation was involved in the suppressive effect of EA on DNFB-induced ACD, which was associated with enhanced CB2R expression and limited p38 MAPK activation. These findings provide new evidence to help clarify the potential mechanisms underlying the anti-ACD effect of EA treatment, and suggest EA stimulation at ST36 may be effective at treating skin inflammatory disease.
Methods
Animals
Male Sprague-Dawley rats (7 weeks old) were obtained from Hubei Research Centre of Laboratory Animals (Wuhan, China). The rats were maintained under specific pathogen-free conditions at the Animal Care Facility of Hubei University of Chinese Medicine (Wuhan, China). Animal care and use were in compliance with guidelines of the Animal Care and Use Committee of Hubei University of Chinese Medicine.
DNFB-induced ACD
To model ACD, rats were sensitised with 25 µL 0.5% DNFB (Sigma-Aldrich, St Louis, MO, USA) in acetone/olive oil (4:1) applied topically to the shaved hind flank on days 1 and 2. After 5 days, the left ears were challenged with 10 µL 0.2% DNFB. The ear swelling change was used to measure the allergic responses.
Groups
The rats were randomly divided into six groups (n=8 per group): group 1 (Control); group 2 (DNFB-induced ACD); group 3 (EA+ACD)—receiving EA treatment at bilateral ST36 (Zusanli) located 5 mm below the fibular head and 2 mm lateral to the anterior tubercle of the tibia continuously for 1 week, then sensitised and challenged with DNFB; group 4 (Sham+ACD)—receiving EA treatment at a location not corresponding to any traditional acupuncture point (sham point, localised 5 mm below ST36) followed by repeated DNFB application as in group 3; group 5 (AM630+EA+ACD); group 6 (AM251+EA+ACD).
AM630 and AM251 (specific CB2R and CB1R antagonists, respectively) (Tocris Bioscience, Bristol, UK) were dissolved in 5% dimethyl sulfoxide (DSMO) before use. Based on another report, 12 AM630 or AM251 (1 mg/kg) was administered intraperitoneally 1 hour before EA treatment continuously for 7 days in rats of group 5 and 6, respectively.
EA treatment
A Hans acupuncture point nerve stimulator (HANS, Beijing, China) was used to administer electrical stimulation at ST36 or the sham point bilaterally via two needles. The needles (length 3.0 cm, diameter 0.20 mm, Suzhou Medical Appliance Factory, Suzhou, China) were inserted perpendicular to the skin. The following parameters were used: continuous wave at 2 Hz and 1 mA for 5 min; 2 Hz and 1.5 mA for 5 min; and 2 Hz and 2 mA for 20 min.
Ear thickness measurement
Ear thickness was measured 24 hours after the challenge with a thickness gauge (Digimatic calliper, Mitutoyo, Japan) and the swelling assessed as follows: ear swelling = thickness of DNFB-challenged ear (left) − thickness of unchallenged ear (right).
Determination of IgE levels in serum
Twenty-four hours after elicitation, blood samples were collected for detection of IgE using an ELISA kit (Kamiya Biomedical, Seattle, WA, USA) according to the manufacturer’s instruction.
Isolation of rat peritoneal mast cells
The rats were terminally anaesthetised with 7% chloral hydrate intraperitoneally (0.5 mL/100 g). Twenty millilitres of Dulbecco’s Modified Eagle Medium (DMEM) with 10% fetal bovine serum were injected into the peritoneal cavity. After the lavage was completed, the rats were euthanased by cervical dislocation. Rat peritoneal mast cells (RPMCs) were collected and centrifuged at 400 g for 15 min. Cell pellets were resuspended in 1 mL serum-free DMEM, and the macrophages were separated from MCs by differential centrifugation using a Percoll solution as previously described. 13
Toluidine blue staining
The left ear of each rat was removed and fixed in 4% paraformaldehyde (Sigma-Aldrich). Fixed tissues were embedded in paraffin for 24 hours and serially sectioned to a thickness of 5 µm for histological analysis. Tissue sections were stained with toluidine blue and observed under a Nikon Eclipse Ti-S microscope (Nikon, Tokyo, Japan). Ten different fields of each section were counted manually and evaluated in a blinded fashion. The number of metachromatic cells were counted in five sections per rat, and the results from eight rats were averaged.
Analysis of β-hexosaminidase release
RPMCs were cultured in 12-well plates at a density of 5×105 cells/well and incubated overnight with anti-dinitrophenyl immunoglobulin E (Anti-DNP IgE, 1 µg/mL), followed by stimulation with dinitrophenyl-human serum albumin (DNP-HSA, 20 ng/mL) for 4 hours. Then, the cells were centrifuged at 150 g for 10 min at 4°C. The supernatant (50 µL) was transferred to 96-well plates and incubated with an equal volume of substrate solution (1 mM 4-nitrophenyl N-acetyl-β-D-glucosaminide in 0.1 M citrate buffer, pH 4.5) for 1 hour at 37°C. The cells were lysed with 0.5% Triton X-100 (50 µL) and lysates were collected to measure total β-hexosaminidase activity. The reaction was stopped by adding 150 µL stop solution (0.1 M Na2CO3-NaHCO3, pH 10). The absorbance was measured at 405 nm. β-hexosaminidase release (%) = (ODsupernatant – ODcontrol)/(ODtotal – ODcontrol) × 100%
Histamine release assay
Histamine concentrations in the culture supernatant of RPMCs were detected using an ELISA kit (Kamiya Biomedical) according to the manufacturer’s instructions.
Real-time RT-PCR
Total RNA from RPMCs was extracted using Trizol reagent (Invitrogen, Carlsbad, CA, USA). Quantitative PCR was performed using SYBR-Green Master PCR Mix (Applied Biosystems, Foster City, CA, USA) on a TP800 qPCR System (Takara, Japan). The following forward (F) and reverse (R) primer sequences were used: CB1R: F, 5′-CTACGTGGGCTCGAATGACA-3′; R, 5′-GACCAACGGGGAGTTGTCTC-3′; CB2R: F, 5′-GCCTGGTCATGGCTGTTCTG-3′; R, 5′-CAGCAGAGCGGATCTCTCCA-3′; GAPDH: F, 5′-CCCCCAATGTATCCGTTGTG-3′; R, 5′-TAGCCCAGGATGCCCTTTAGT-3′. The relative CBR mRNA levels were normalised to those of GAPDH. For relative quantification, 2-(Ct-Cc) (where Ct and Cc are the mean threshold cycle differences after normalising to control) was calculated and used as an indicator of relative expression. 14
Western blot analysis
RPMCs were obtained and lysed with RIPA Lysis Buffer (Cell Signalling Technology Inc, Beverly, Massachusetts, USA). A total of 20 µg protein per lane was separated by 10% SDS-PAGE. Subsequently, blots were transferred onto polyvinylidene difluoride membranes and blocked for 2 hours with 5% non-fat dry milk at room temperature. Membranes were incubated with primary anti-rat antibodies against ERK, p38, JNK, p-ERK, p-p38, p-JNK, GAPDH, CB1R or CB2R (Santa Cruz Biotechnology Inc, Santa Cruz, CA, USA) overnight at 4°C, followed by incubation with the appropriate horseradish peroxidase (HRP)-conjugated secondary antibody (Santa Cruz). The bands were visualised by enhanced chemiluminescence detection (HP Scanjet 7400C; Hewlett-Packard Co, Palo Alto, CA, USA). Optical density for each band was assessed using ImageJ analysis software (National Institutes of Health, Bethesda, MD, USA). Sample loading was normalised to quantities of GAPDH detected in parallel.
Statistical analysis
The results were presented as mean±SD. Data were analysed with SPSS 16.0 software (SPSS Inc, Chicago, IL, USA). Significance was assessed by one-way analysis of variance (ANOVA) followed by Dunnett’s test. A value of p<0.05 was considered to indicate a statistically significant difference.
Results
EA treatment at ST36 suppresses DNFB-induced ACD in rats
We first investigated the effect of EA stimulation at ST36 on DNFB-induced ACD in rats. The Sprague-Dawley rats were sensitised and challenged with DNFB to induce contact hypersensitivity (Figure 1A). The DNFB challenge induced a significant increase in ear swelling (Figure 1B). However, EA treatment at ST36 led to an incremental decline in ear thickness. EA also significantly inhibited IgE levels in serum (Figure 1C), suggesting an anti-allergic effect of EA on DNFB-induced ACD. Interestingly, repeated DNFB application increased MC infiltration in the ear skin (Figure 1D). Conversely, EA treatment at ST36 evidently decreased the number of MCs in the ear dermis (Figure 1E).
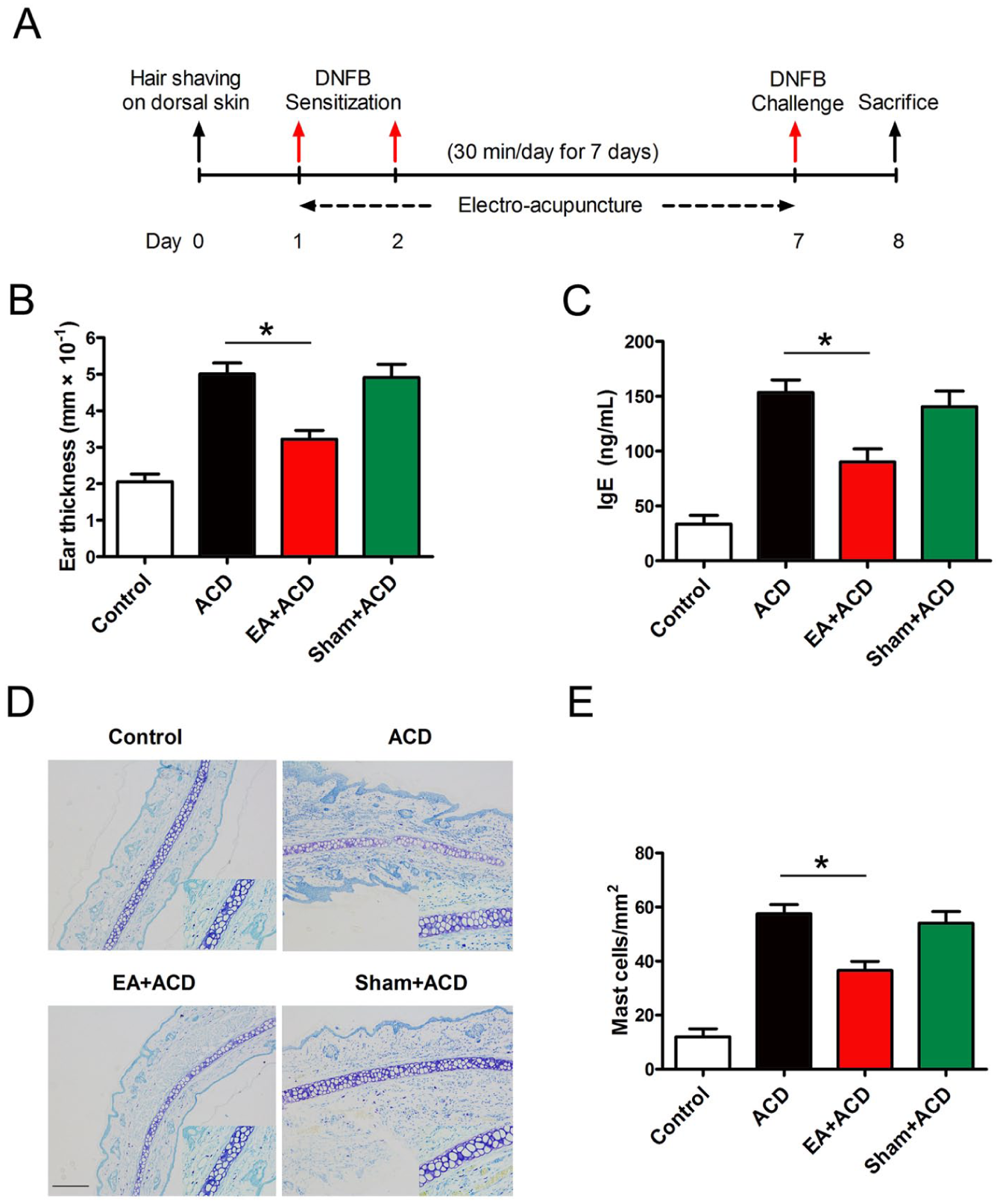
Electroacupuncture (EA) treatment at ST36 relieved 2,4-dinitrofluorobenzene (DNFB)-induced allergic contact dermatitis (ACD) in rats. (A) Experimental schedule for DNFB-induced ACD and EA treatment. (B) Thickness of ear swelling and (C) serum IgE levels were recorded. (D) Ear tissues were sectioned and stained using toluidine blue to observe mast cells. High magnification images are displayed in the small windows. Scale bar=20 µm. (E) The number of mast cells in ear sections was statistically analysed (n=8, *P<0.05).
EA treatment inhibits MC degranulation associated with p38 signalling
Next, we observed the effect of EA treatment on MC degranulation. The results showed that EA significantly lowered the levels of β-hexosaminidase and histamine (Figure 2A and B) and suggested that inhibition of MC degranulation was involved in the suppressive effect of EA on DNFB-induced ACD.
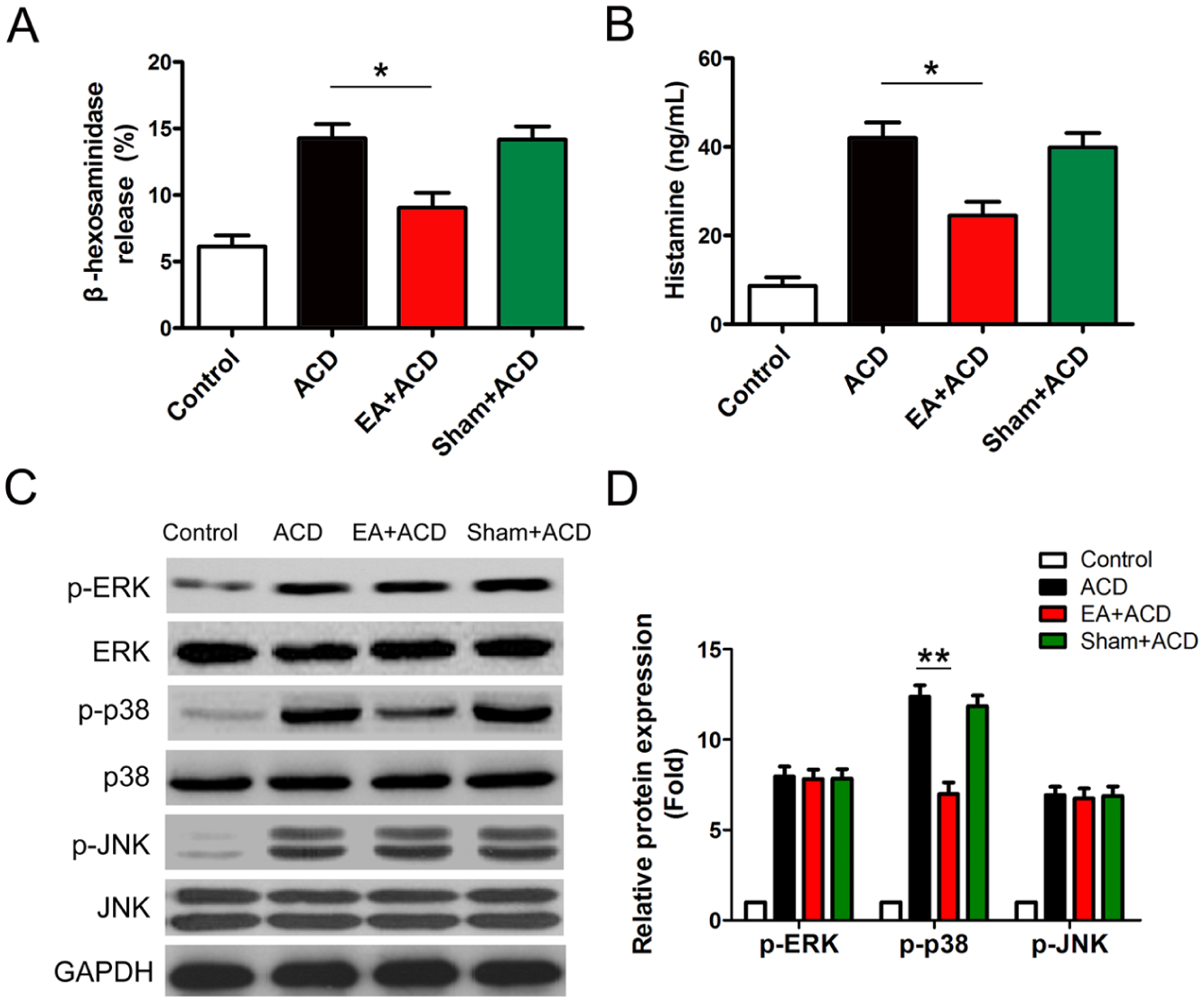
Electroacupuncture (EA) treatment inhibited mast cell (MC) degranulation associated with p38 signalling. Rat peritoneal MCs (RPMCs) were isolated and cultured. MCs (5×105/well) were incubated with anti-dinitrophenyl (anti-DNP) IgE (1 µg/mL) for 24 hours, followed by stimulation with DNP-human serum albumin (DNP-HSA) (20 ng/mL) for 4 hours. (A) β-hexosaminidase and (B) histamine release were measured and statistically analysed. All experiments were done in triplicate. (C) Phosphorylation of ERK, p38 and JNK was measured by Western blotting, and representative results are shown. (D) Statistical analysis of relative protein expression is shown (n=8, *P<0.05 and **P<0.01). ACD, allergic contact dermatitis.
MAPK signalling has been reported to be associated with the regulation of mast cell degranulation. We proceeded to investigate the effect of EA on phosphorylation of ERK, p38 and JNK (Figure 2C). As shown in Figure 2D, EA treatment effectively prevented p38 MAPK activation but not phosphorylation of ERK and JNK. The results indicated that inhibition of p38 activation was associated with an anti-ACD effect of EA.
EA treatment enhances CB2R expression
As CB2R is mainly present on immune cells such as MCs, we further investigated the effect of EA stimulation on CBRs. Compared with the untreated ACD group, EA treatment obviously induced the expression of CB2R (but not CB1R) mRNA and protein in RPMCs (Figure 3A and B). This suggested that CB2R was involved in the anti-ACD effect of EA.
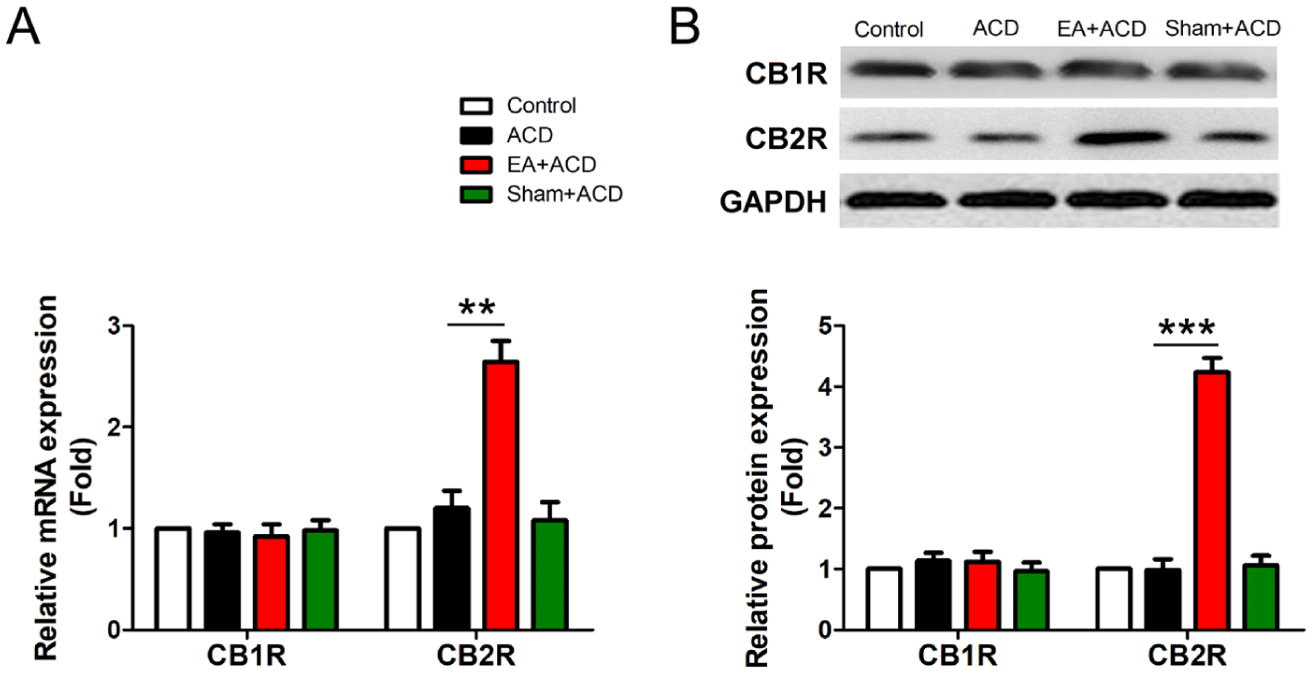
Electroacupuncture (EA) treatment attenuated CB2R expression. Rat peritoneal mast cells (RPMCs) were isolated and cultured for the detection of cannabinoid receptors (CBRs). (A) mRNA expression of CBR was measured by quantitative real-time PCR and (B) protein expression of CBR was measured by Western blotting, and the statistical analysis is shown. A representative Western blot is also shown (n=4, **P<0.01 and ***P<0.001).
CB2R activation participates in the anti-ACD effect of EA
Lastly, we detected the anti-ACD effect of EA utilising AM251 and AM630, which are specific CB1R and CB2R antagonists, respectively. In contrast to AM251, AM630 clearly abrogated the inhibitive effect of EA on MC infiltration (Figure 4A and B). Additionally, AM630 distinctly dampened the suppressive effect of EA on MC degranulation, resulting in an increased release of β-hexosaminidase and histamine (Figure 5A and B). It is worth noting that p38 phosphorylation was also reversed in response to AM630 (Figure 5C and D). This suggests that CB2R activation participated in the inhibition of MC degranulation associated with the anti-ACD effect of EA.
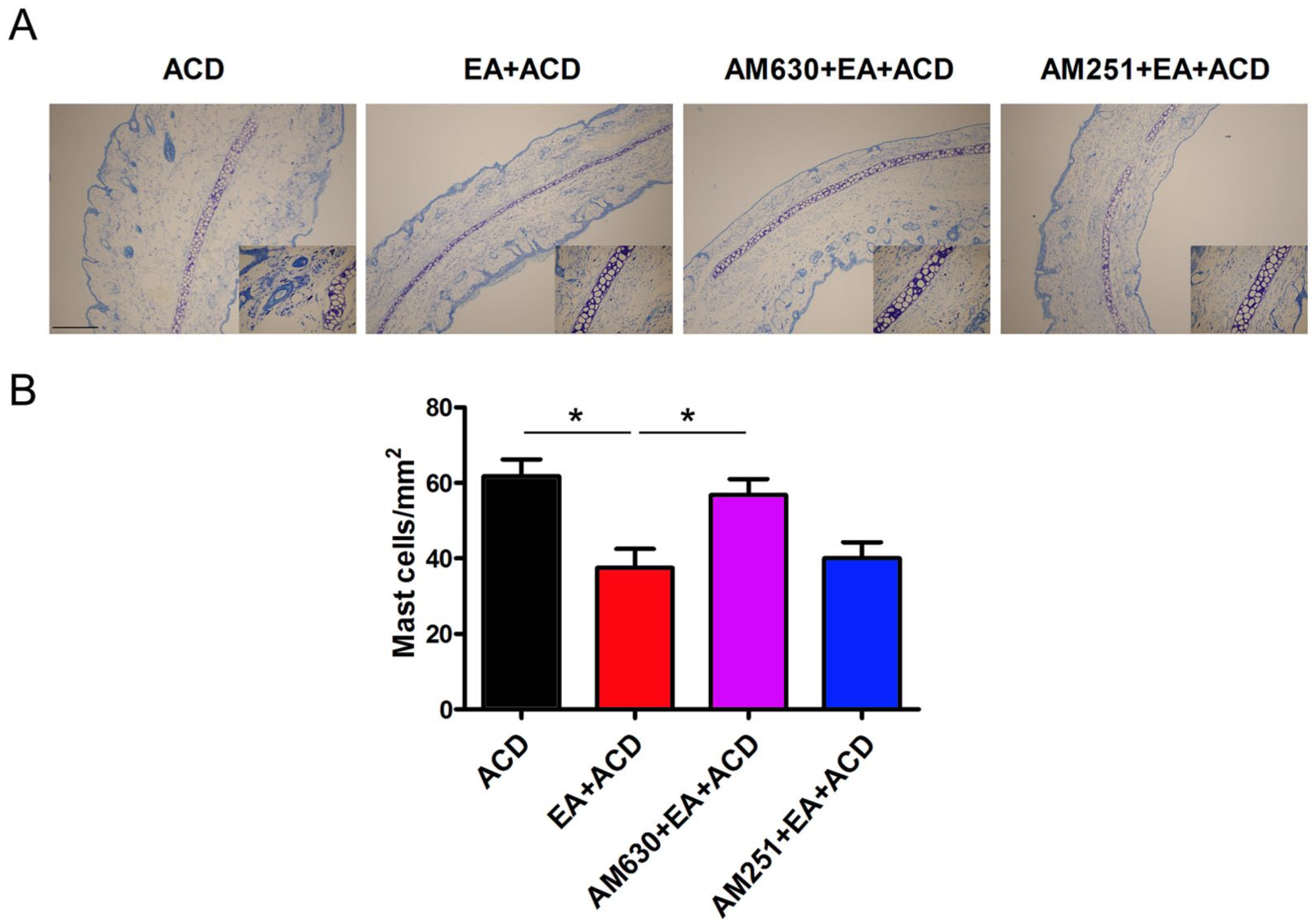
CB2R antagonist AM630 abrogated the suppressive effect of electroacupuncture (EA) on mast cell infiltration. (A) Sections of ear tissues were stained with toluidine blue and observed for mast cells. High magnification images are shown in the small windows. Scale bar=20 µm. (B) The numbers of mast cells in the ear sections were statistically analysed (n=8, *P<0.05). ACD, allergic contact dermatitis.
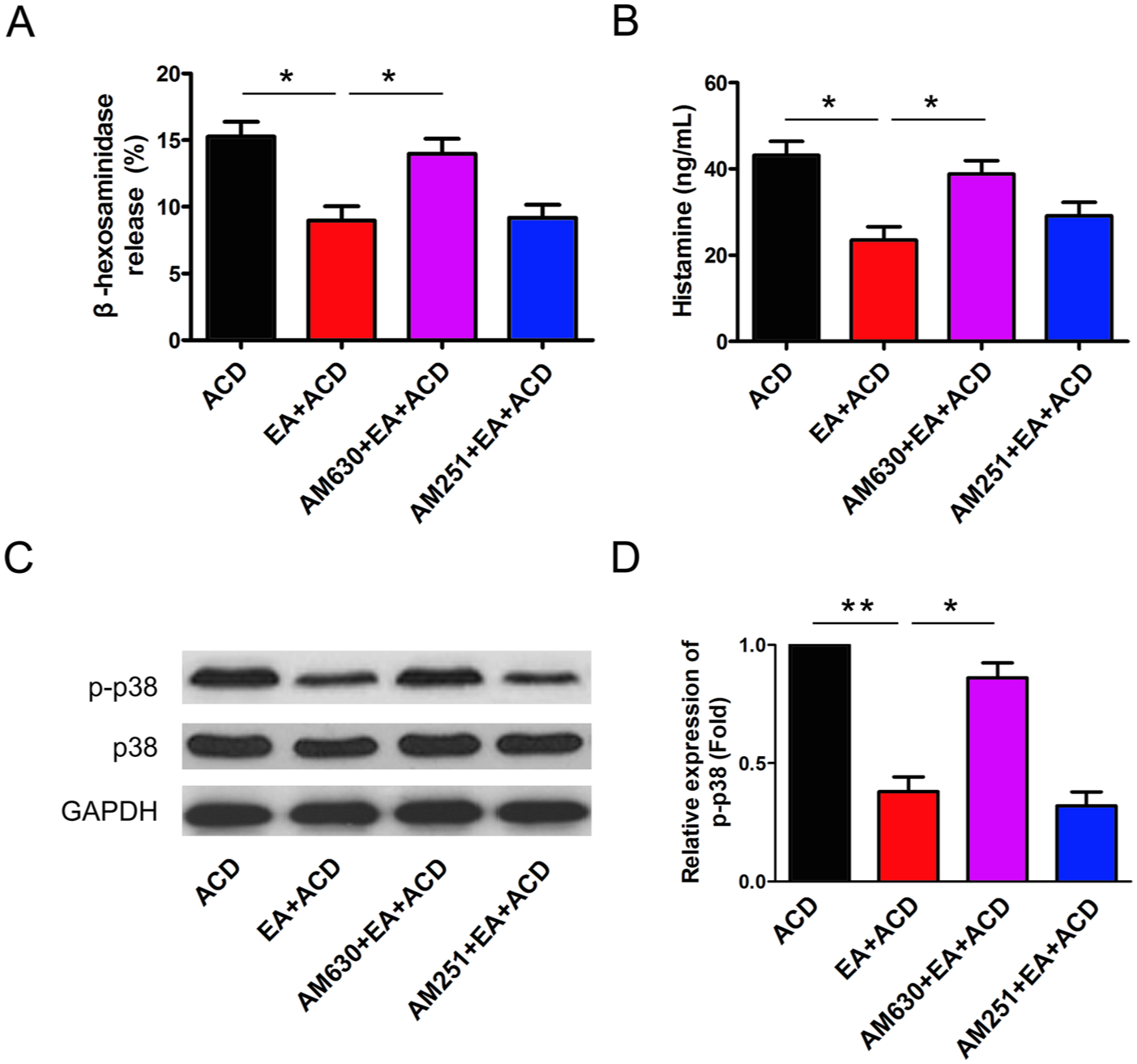
Inhibition of mast cell (MC) degranulation in the effect of electroacupuncture (EA) on allergic contact dermatitis (ACD) via CB2R. Rat peritoneal MCs (RPMCs) were isolated and cultured as described. (A) β-hexosaminidase and (B) histamine release were measured and statistically analysed. All experiments were done in triplicate. (C) Phosphorylation of p38 was measured by Western blotting, and representative results are shown. (D) Statistical analysis of relative protein expression of p-p38 is shown (n=8, *P<0.05 and **P<0.01).
Discussion
A recent publication has shown that MCs might have an influence on the course of chronic ACD. Utilising MC-deficient KitW-sh/W-sh (Sash) mice, local engraftment with MCs protected against exacerbated ear swelling and pro-inflammatory cytokine production in chronic ACD. 15 Murine studies of DNFB-induced ACD in vivo and RBL-2H3 cells demonstrated that MC degranulation, including β-hexosaminidase and histamine release, participated in its pathogenesis.16–18 To some extent, inhibiting MC activation and degranulation is a viable therapeutic strategy for ACD. In this study, EA at ST36 repressed DNFB-induced ACD, resulting in the relief of ear swelling and a decline in IgE production. Interestingly, inhibited MC degranulation was also associated with the suppressive processes of EA.
As one of the main acupuncture-related therapeutic techniques for treating systemic inflammatory disorders, EA treatment is becoming more and more widely used with few side effects. 19 Our previous study investigated the immunomodulatory effect of EA, which resulted in inhibition of inflammation associated with ACD via suppression of p38 activation. 20 An earlier study also suggested that MC degranulation was regulated by the p38 signalling pathway. 21 Consistent with the above findings, our results showed that EA significantly restricted p38 but not ERK or JNK phosphorylation, suggesting that the p38 MAPK pathway possibly contributes to the anti-ACD effect of EA.
In view of the increasing interest in the relationship between EA stimulation and cannabinoid receptors, many studies have explored their cross-effects in different pathological models.22,23 EA pretreatment at GV20 (Baihui) induced neuroprotection against cerebral ischaemia and reperfusion, and this protection was mediated by the activation of STAT-3 via CB1R. 24 In addition, EA at GB30 (Huantiao) and GB34 (Yanglingquan) reduced inflammatory pain through CB2R activation, which was significantly attenuated by the application of the CB2R antagonist AM630. Conversely, the CB2R agonist AM1242 produced analgesia similar to EA stimulation, and the effect was also blocked by AM630. 25 Our results showed that EA at ST36 enhanced CB2R expression at both mRNA and protein levels. Importantly, the CB2R antagonist AM630 reversed the suppressive effect of EA, which strongly suggests that CB2R activation was beneficial in EA treatment of ACD.
Some specific studies have implied that CB2R might execute a biological function as an immunomodulator and play a different role in MCs. 26 Endogenous cannabinoids acting on CB2R represented an autacoid mechanism in modulating the immunological response of guinea-pig MCs and human basophils. 27 CB2R activation and decreasing MC degranulation partly contributed to the inhibitive effect of the cannabinoid quinol VCE-004.8 on scleroderma. 28 According to these reports and our own findings, we speculate that EA at ST36 could enhance CB2R expression on MCs at the site of acupuncture stimulation and facilitate p38 phosphorylation, resulting in decreased MC degranulation induced by DNFB in rats.
To our knowledge, this is the first study to explore the immunomodulatory effect of EA at ST36 based on the intrinsic relationship between MCs and CB2R. Our results have provided new evidence on the possible molecular mechanism for the anti-ACD effect of EA, and also suggested EA at ST36 could be used as a potential therapy in the clinical treatment of allergic inflammation of the skin, although further clinical studies are needed to confirm its efficacy in humans. There are some limitations of the present study that should be mentioned. CB2R-/- animal or bone marrow-derived MCs could not be used because of the rat model, and further studies are required to clarify if nuclear factor κ–light–chain–enhancer (NF-κB) signalling or another pathway are involved in the anti-ACD effect of EA.
In conclusion, our results strongly suggest that EA-induced CB2R activation followed by p38 phosphorylation may contribute to the anti-ACD effect of EA at ST36. Although further investigations are needed to illuminate the detailed signal cascades underlying the CB2R-p38 pathway and the downstream events, the present study provides a new perspective on the use of EA stimulation at ST36 and may help to develop therapeutic strategies for the treatment of inflammatory skin diseases such as ACD.
Footnotes
Contributors
ZGW, ML and ZBC designed the study. ZGW and ML finished the experiment and wrote the manuscript. JR, ML and LYC performed partial research. XXW helped to analyse the data. ZBC revised the manuscript. All authors read and approved the final version of the manuscript accepted for publication.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Specialized Fund of Hubei Provincial Department of Education in Hubei Provincial Collaborative Innovation Center of Preventive Treatment by Acupuncture and Moxibustion (no. HBPCIC-2016-020).
Patient consent
Not required.
Provenance and peer review
Not commissioned; externally peer reviewed.
