Abstract
Background:
Moxibustion treatment has been found to ameliorate clinical symptoms including abdominal pain, diarrhoea and constipation in patients with irritable bowel syndrome (IBS). Herein we investigated the mechanisms underlying the use of moxibustion in a rat model of IBS.
Methods:
In our study, an IBS model was established in rats by colorectal distension (CRD) stimulus and mustard oil enema. The rats were randomly divided into a normal group, model group, mild moxibustion group, electroacupuncture group, probiotic group and dicetel group. Abdominal withdrawal reflex (AWR) scores were determined within 90 min of the last treatment. The expression of GDNF/GFRα3 protein and mRNA in the colon and spinal cord were detected by immunohistochemistry and quantitative real-time-PCR, respectively.
Results:
The IBS model rats had significantly higher AWR scores than the normal group (P<0.01). After mild moxibustion treatment, the AWR score was significantly reduced (20 mm Hg, P<0.05; 40 mm Hg, 60 mm Hg and 80 mm Hg, P<0.01). The model group showed significantly more colonic glial cell line-derived neurotrophic factor (GDNF/GFRα3 (GDNF family receptor α3) protein and mRNA expression in the colon and spinal cord than the normal group (P<0.01). Compared with the model group, the expression of GDNF/GFRα3 protein and mRNA in the colon and spinal cord of the rats were significantly decreased in the mild moxibustion group (colon: GDNF and GFRα3 protein, P<0.01; GDNF and GFRα3 mRNA, P<0.01; spinal cord: GDNF and GFRα3 protein, P<0.01; GDNF mRNA, P<0.05, GFRα3 mRNA, P<0.01).
Conclusions:
Our data suggest that moxibustion therapy may mitigate CRD-induced increases in the expression of GDNF and its receptor GFRα3 in the colon and spinal cord in a rat model of IBS.
Keywords
Introduction
Irritable bowel syndrome (IBS) is a common chronic functional bowel disease. Its main features include abdominal pain, changes in defecation, abnormal stool and a lack of significant organic changes. An epidemiological study has shown the global prevalence of IBS to be 11.2%. 1 Current therapeutic methods mainly include medicinal, psychological and dietary therapy. 2 However, medicinal therapy is limited to symptomatic treatment and is associated with certain side effects. Furthermore, the long-term effects are unsatisfactory. Acupuncture has been used as a safe and effective therapy to treat IBS and related functional bowel disorders.3,4 It not only mitigates clinical symptoms, including abdominal pain, diarrhoea and constipation, but also regulates intestinal substance P, vasoactive intestinal peptides and expression of the P2x7 receptor.5,6
The pathogenesis of IBS has not been fully characterised. Recent studies indicate that it might be associated with brain–gut interactions, visceral hypersensitivity, colonic dysmotility, and genetic and psychological factors.7,8 In recent years, studies have indicated that visceral hypersensitivity is the main cause of abdominal pain and diarrhoea in patients with IBS. 9 Reducing visceral hypersensitivity and relieving clinical symptoms in IBS patients are important therapeutic strategies. Glial cell line-derived neurotrophic factor (GDNF) is a neurotrophic factor with several biological activities. Studies have shown that the visceral hypersensitivity rat model has a high expression of GDNF. 10 Tanaka et al. found a high expression of GDNF in the dorsal root ganglia of the visceral hypersensitivity rat model. 11 Moxibustion is an effective method for treating IBS.12,13 In this study, we focused on visceral hypersensitivity and GDNF/GFRα3 (GDNF family receptor α3) to investigate the mechanism of action underlying the effects of moxibustion on IBS.
Methods
Animals
Sixty male Sprague-Dawley rat pups were obtained 8 days after birth from the Experimental Animal Center, Shanghai University of Traditional Chinese Medicine. The laboratory temperature was 22±2°C and humidity was 60±5%. All experimental procedures were performed according to the protocols approved by the University Animal Care and Use Committee of Shanghai University of Traditional Chinese Medicine (IACUC protocol no. SYXK (Shanghai) 2009–0082) and in accordance with the National Institutes for Health (NIH) ‘Guide for the Care and Use of Laboratory Animals’ to reduce pain and to avoid harm.
Model establishment
The number of rats was based on the power calculation of a previous study. 14 According to a random number table generated by SPSS version 21.0, 10 rats were selected as a normal group. According to the protocol described by Al-Chaer et al., 15 the remaining 50 rats formed the model group and received colorectal distension (CRD) via balloon stimulation, which is one of the most widely used methods to induce visceral hypersensitivity, 16 on a daily basis between the age of 8 and 21 days. A 3 cm balloon stimulator was connected to a 10 cm long latex tube. At the end of the tube, a Y connector was attached to a syringe and sphygmomanometer, which provided constant dilation pressure and stimulation. The balloon stimulator was first lubricated with petrolatum and inserted into the rat colon in line with its physiological curvature. The balloon was inserted 2 cm deep to the anus and then inflated with 0.5 mL air for 1 min; this was repeated after 1 hour and again once a day for 14 consecutive days. After the daily procedure was finished, the rats were put back into the cages with their mothers. After 4 weeks rest, the model rats then received colonic injections of 0.2 mL 4% mustard oil once a day for 14 consecutive days. The abdominal withdrawal reflex (AWR) was tested by different observers. The model was considered successfully established when the AWR score in model rats was significantly greater than that of the normal rats.
Grouping and processing
The rats were randomly divided into the following groups:
Normal group (NG) and Model (CRD) groups: no treatment.
Mild moxibustion group (CRD+MM): Mild moxibustion (MM) was applied at bilateral ST25 (Tianshu) and ST37 (Shangjuxu). Special fine moxa sticks used for this experiment were 4 mm in diameter and 12 cm in length, and were provided by the Nanyang Hanyi Moxa factory in China. The moxibustion was performed 2–3 cm above the traditional acupuncture points for 10 min, once a day for 7 days.
Electroacupuncture group (CRD+EA): Sterilised stainless steel acupuncture needles (Yunlong, Wujiang Yunlong Medical Instruments Factory, Wujiang, China) 25 mm in length and 0.3 mm in diameter were inserted to a depth of about 2–3 mm at ST25 and ST37 bilaterally. An electrical acupuncture point stimulator (HAN-200A, Jisheng Ltd, Beijing, China) was used to stimulate the needles using dilatational waves (2–100 Hz, intensity 1mA). Each treatment was performed for 20 min once per day for 7 days in total.
Probiotic group (CRD+PB): Bifidobacteria Triple Viable (SFDA approval no. S19980004) was administrated via oral gavage. Daily dosage was calculated according to the ratio between adult humans (70 kg body weight) and rats (200 g body weight), which was 1:0.018 (based on the Methods for Pharmaceutical Experiment). The treatment was performed once a day for 7 days in total.
Dicetel group (CRD+DG): Dicetel (SFDA approval no. H20120127) was administrated via oral gavage. The same dosing and timing as the Probiotic group were applied.
One rat died during the preparation of the model, two rats in the CRD+PB group and two rats in the CRD+DG group died after gavage, and three rats died for unknown reasons. The final number of rats in each group were: NG, n=8; CRD, n=8; CRD+MM, n=8; CRD+EA, n=8; CRD+PB, n=6; and CRD+DG, n=7.
Sample collection and processing
After treatment, all rats were weighed and anaesthetised with 2% pentobarbital sodium (2 ml/kg), and then euthanased by cervical dislocation. Spinal cords (L6–S2) and colonic samples (2–3 cm) from the distal colon were collected immediately. The samples were stored at −80°C for immunohistochemistry and real-time PCR analysis.
AWR scoring
Within 90 min of the last treatment, the AWR score was calculated according to Al-Chaer et al. 15 Balloon dilation pressures had four pressure grades: 20, 40, 60 and 80 mm Hg. Each distension stimulation lasted about 20 s and was repeated three times. The average of the scores for each rat was used for statistical analysis. The AWR scoring standards applied were as follows: no reaction to the colorectal balloon dilation stimulation, score 0; a motion stop and a brief head movement, score 1; abdominal muscle contraction but without abdominal lifting, score 2; abdominal lifting and lifting off the platform, score 3; arching of body and lifting of pelvis and scrotum, score 4.
Immunohistochemical analysis of GDNF and its receptor GFRα3
Formalin-fixed paraffin-embedded sections (4 μm thick) were immunostained using anti-GDNF (ab18956, 1:100, Abcam) and anti-GFRα3 (ab8028, 1:100, Abcam) antibodies according to the manufacturer’s instructions. The primary antibodies were added and incubated at 4°C overnight, and the corresponding secondary antibodies (BA1003, 1:150, Wuhan Boster Biological Technology Co Ltd) were added and incubated at room temperature for 1 hour. After DAB reaction for 3–5 min, the sections underwent 2 min of haematoxylin staining and were then viewed under an optical microscope.
Using Image-Plus Pro image analysis software, three randomly selected sections were counted from each rat per group and analysed for the average optical density of the positive target.
Real-time quantitative PCR analysis of GDNF and its receptor GFRα3
Total RNA was extracted using TRIzol LS (Invitrogen Corp) according to the manufacturer’s instructions. The PCR reaction mixture (16 µL) comprised 5 µl of H2O, 8 µl of 2×SYBR Green PCR mix, 1 µl of upstream primer (10 pm/μl), 1 µl of downstream primer (10 pm/μl) and 1 µl of reverse transcription product (cDNA). Reaction conditions were as follows: 95°C for 2 min, 94°C for 10 s, 60°C for 10 s and 72°C for 40 s, 40 cycles. The sequences of the target genes were set according to the Gene Bank database. Primers were designed using ABI Primer Express Software v 2.0 and synthesised by BGI Inc. The primer sequences were as follows:
GAPDH: upstream 5′-GGCAAGTTCAACGGCACAGT-3′; downstream 5′-ATGACATACTCAGCACCGGC-3′.
GDNF: upstream 5′-TCGAAGAGAGAGGAACCGGC-3′; downstream 5′-TTAAGACGCACCCCCGATTT-3′.
GFRα3: upstream 5′-AAAATGCGAGGCTAATCCCG -3′ downstream 5′-CTTCAAGGCACGCTGCAGAT-3′.
Relative mRNA expression of the target genes was calculated using the 2-ΔΔCt method: target gene ΔCt=target gene Ct − reference (GAPDH) Ct, ΔΔCt=target gene Ct − average of blank ΔCt.
Statistical analysis
Statistical analysis was performed using SPSS 21.0 (IBM Co, Armonk, NY, USA). Data were expressed as mean±SD (
Results
AWR score
The average stimulation score of the CRD group was significantly higher than that of the normal group (P<0.01). The various treated groups showed lower AWR scores than the CRD group in response to different stimulation intensities. Among them, the AWR scores of the CRD+MM group were significantly reduced (20 mm Hg, P<0.05; 40, 60 and 80 mm Hg, P<0.01); AWR scores of the CRD+MM group under 60 and 80 mm Hg were significantly lower than in the CRD+PB and CRD+DG groups (CRD+PB group: 60 mm Hg, P<0.05, 80 mm Hg, P<0.01; CRD+DG group: 60 mm Hg, P<0.01, 80 mm Hg, P<0.05) (Figure 1).
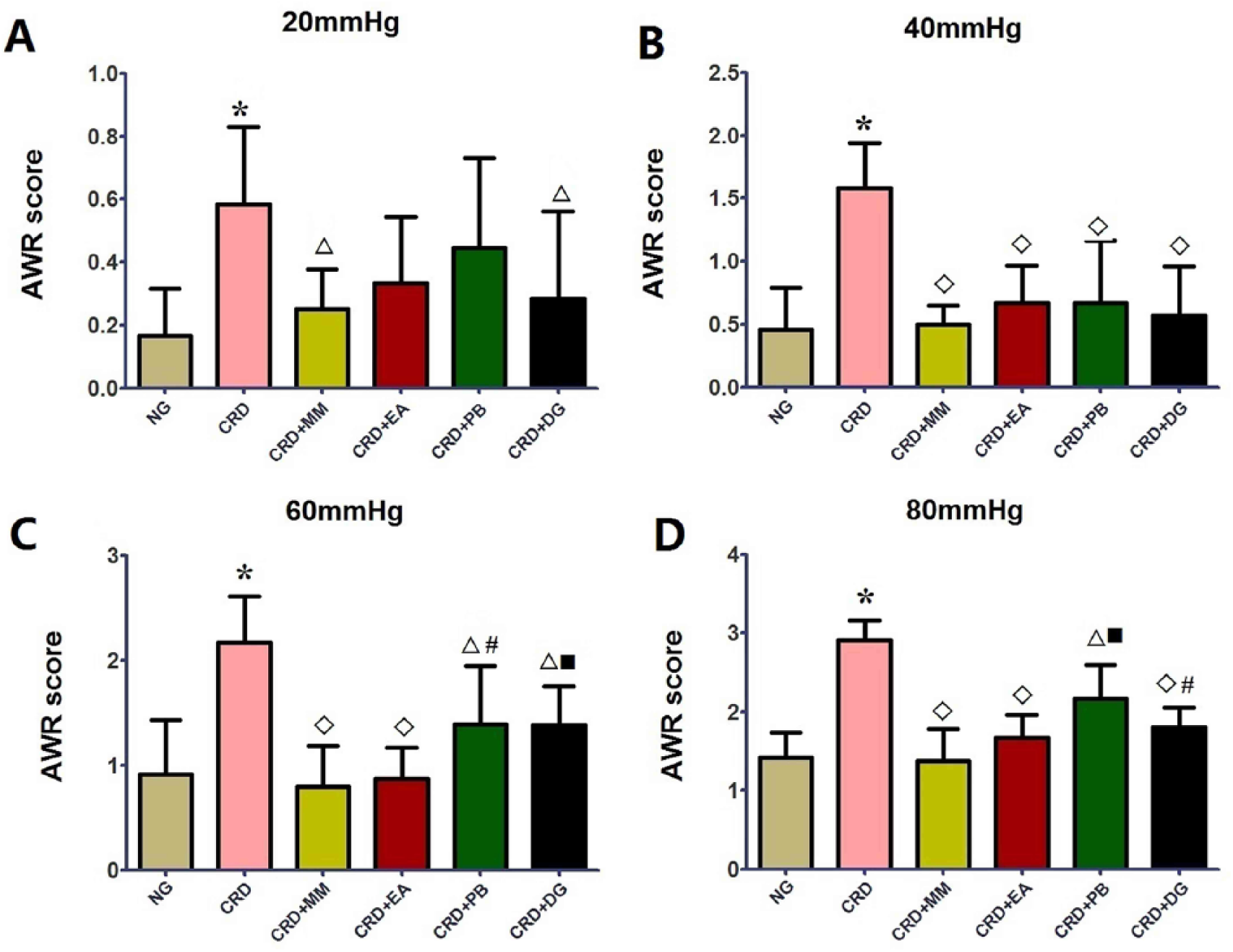
Abdominal withdrawal reflex (AWR) scores under different distension pressures (A: 20 mm Hg, B: 40 mm Hg, C: 60 mm Hg, D: 80 mm Hg) in different groups (Normal group: NG, n=8; Model group: CRD, n=8; Mild moxibustion group: CRD+MM, n=8; Electroacupuncture group: CRD+EA, n=8; Probiotic group: CRD+PB, n=6; Dicetel group: CRD+DG, n=7). *P<0.01 versus NG; P<0.05, P<0.01 versus CRD; #P<0.05, ■ P<0.01 versus CRD+MM.
Protein expression of GDNF and GFRα3 in the colon
The CRD group showed significantly more colonic GDNF and GFRα3 protein expression than the normal group (P<0.01). The CRD+MM, CRD+EA, CRD+PB and CRD+DG groups all showed significantly less GDNF protein expression than the CRD group (CRD+MM, CRD+EA and CRD+DG groups, P<0.01; CRD+PB group, P<0.05). The CRD+MM, CRD+EA, CRD+PB and CRD+DG groups also showed significantly less GFRα3 protein expression than the CRD group (P<0.01). Compared with the CRD+MM group, protein expression of GFRα3 was higher in the CRD+PB and CRD+DG groups (P<0.01) (Figure 2).
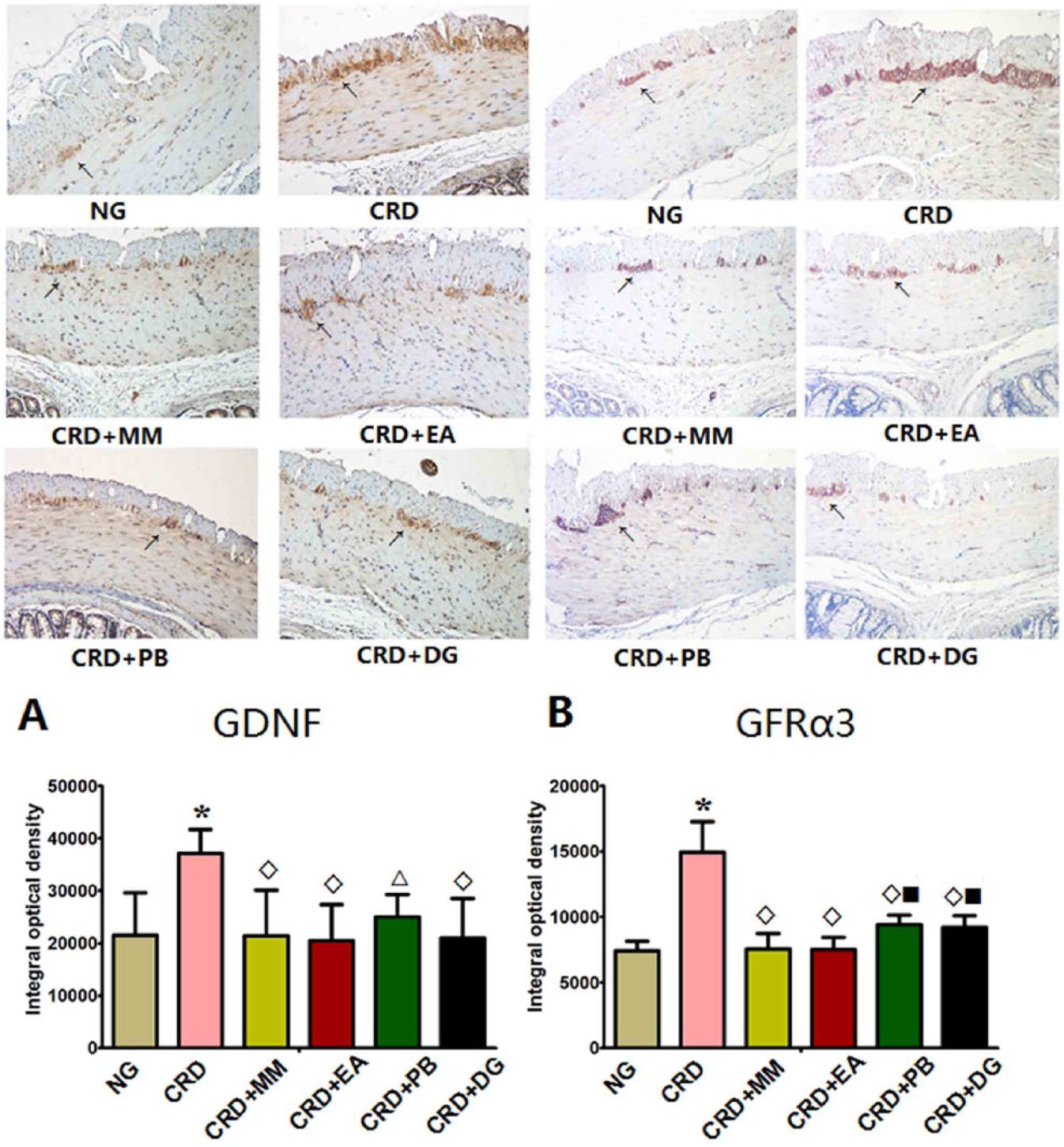
Representative immunohistochemistry images taken at
Protein expression of GDNF and GFRα3 in the spinal cord
The CRD group showed significantly more GDNF and GFRα3 protein expression than the normal group (P<0.01). The CRD+MM, CRD+EA, CRD+PB and CRD+DG groups all showed significantly less GDNF and GFRα3 protein expression than the CRD group (P<0.01) (Figure 3).
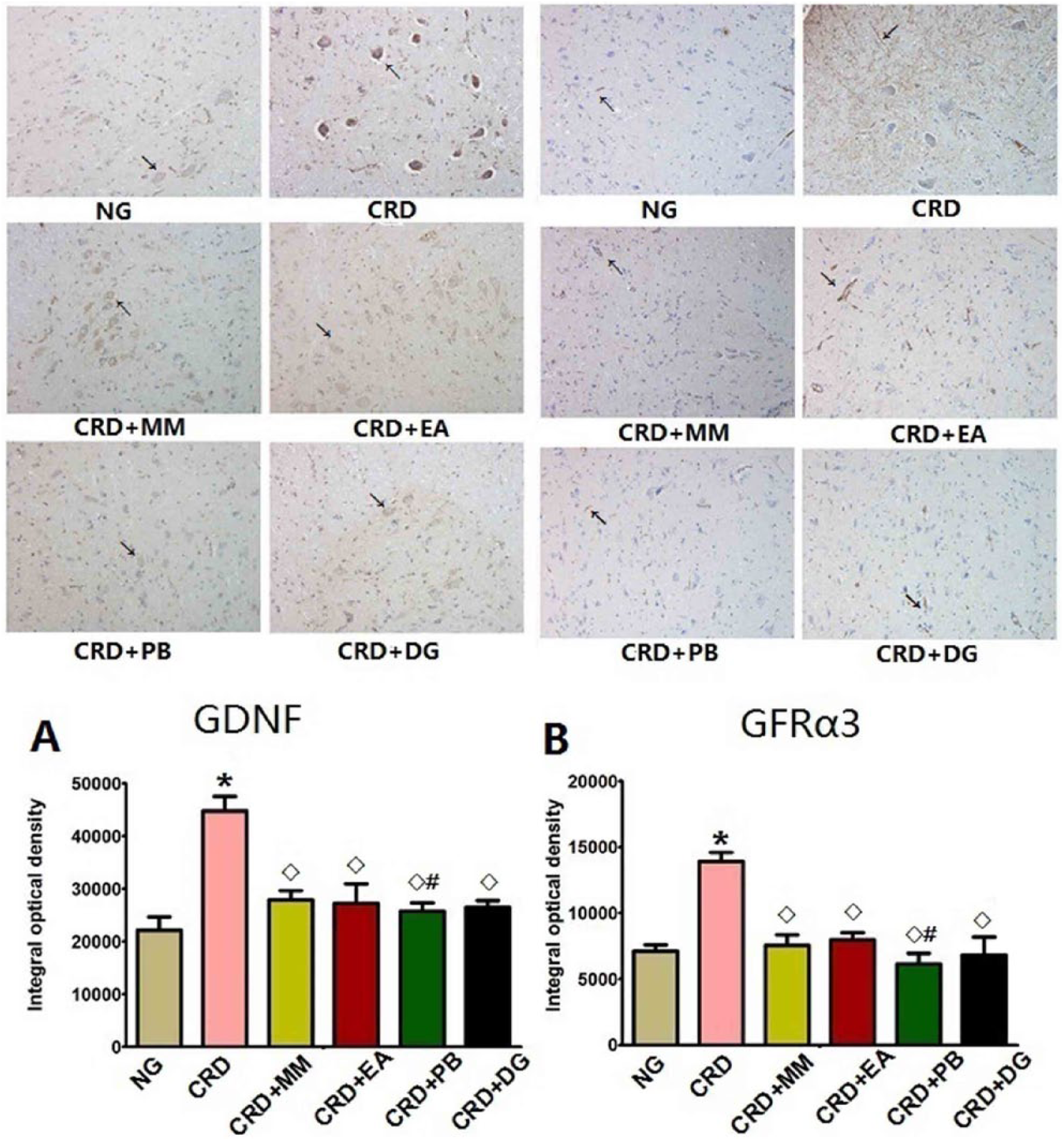
Representative immunohistochemistry images taken at
mRNA expression of GDNF and GFRα3 in the colon
Compared with the normal group, the CRD group showed a significant increase in colonic GDNF and GFRα3 mRNA expression (P<0.01). Compared with the CRD group, the CRD+MM, CRD+EA, CRD+PB and CRD+DG groups all showed a significant decrease in colonic GDNF mRNA expression (P<0.01). The CRD+MM, CRD+EA, CRD+PB and CRD+DG groups also showed a significant decrease in colonic GFRα3 mRNA expression (CRD+MM, CRD+EA and CRD+DG groups, P<0.01; CRD+PB group, P<0.05) compared with the CRD group (Figure 4).
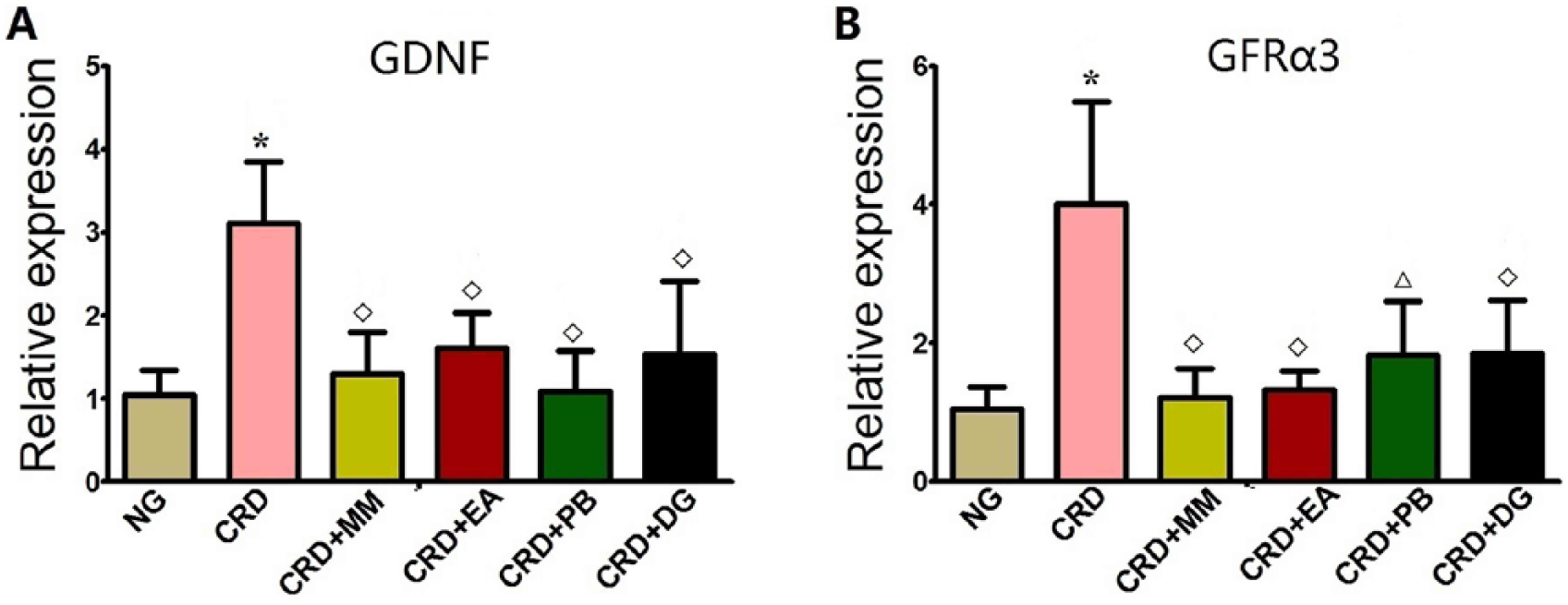
Expression of GDNF (A) and GFRα3 (B) mRNA in colon measured by real-time-PCR. Normal group: NG, n=8; Model group: CRD, n=8; Mild moxibustion group: CRD+MM, n=8; Electroacupuncture group: CRD+EA, n=8; Probiotic group: CRD+PB, n=6; Dicetel group: CRD+DG, n=7. Data are presented as mean±SD *P<0.01 versus NG; P<0.05, P<0.01 versus CRD; #P<0.05, ■ P<0.01 versus CRD+MM.
mRNA expression of GDNF and GFRα3 in the spinal cord
The CRD group showed significantly more GDNF and GFRα3 mRNA expression in the spinal cord than the normal group (P<0.01). The CRD+MM, CRD+EA, CRD+PB and CRD+DG groups all showed a significant decrease in GDNF mRNA expression in spinal cord (CRD+MM group, P<0.05; CRD+PB, CRD+EA and CRD+DG groups, P<0.01). The CRD+MM, CRD+EA, CRD+PB and CRD+DG groups also showed significantly less GFRα3 mRNA expression than the CRD group (P<0.01) (Figure 5).
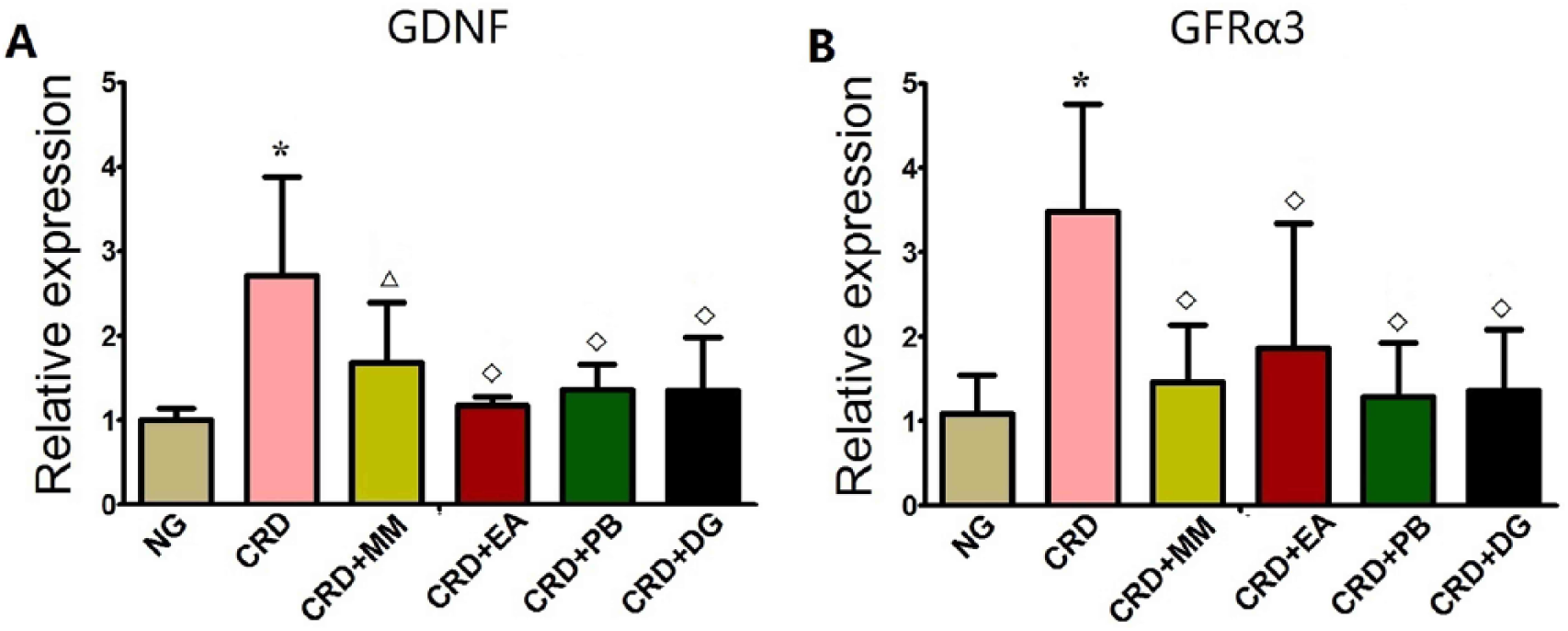
Expression of GDNF (A) and GFRα3 (B) mRNA in spinal cord measured by RT-PCR. Normal group: NG, n=8; Model group: CRD, n=8; Mild moxibustion group: CRD+MM, n=8; Electroacupuncture group: CRD+EA, n=8; Probiotic group: CRD+PB, n=6; Dicetel group: CRD+DG, n=7. Data are presented as mean±SD. *P<0.01 versus NG; P<0.05, P<0.01 versus CRD; #P<0.05, ■ P<0.01 versus CRD+MM.
Discussion
In our experiments, the AWR score was adopted to evaluate intestinal sensitivity. We found that the AWR score in the CRD group was significantly higher than in the normal group, indicating that visceral hypersensitivity in IBS rats plays an important role in the pathogenesis of the condition, which is consistent with previous findings. 17 Visceral hyperalgesia may be the result of a combination of peripheral and central sensitisation mechanisms. The enteric nervous system (ENS), spinal cord and central nervous system (CNS) are involved in the occurrence of visceral sensation. Studies have demonstrated that the ENS contributes to visceral hypersensitivity. 18 Therefore, it is likely that GDNF in the ENS is also involved in the formation of visceral hypersensitivity. In view of this, we observed the expression of GDNF and its receptor GFRα3 in the colon and spinal cord.
Studies have shown that acupuncture and moxibustion can regulate gastrointestinal function and relieve visceral hyperalgesia, and exert their therapeutic effect mainly by regulating, abnormally expressed 5-HT, TRPV1 and HSP70.19,20 In this study, AWR score was significantly decreased in the CRD+MM, CRD+EA, CRD+PB and CRD+DG groups compared with the CRD group, indicating that moxibustion can effectively alleviate visceral hypersensitivity in IBS rats as well as probiotics and dicetel. Under 60 and 80 mm Hg, the AWR score in the CRD+MM group was significantly lower than the CRD+PB and CRD+DG groups, indicating that moxibustion may be more beneficial at reducing IBS rat visceral pain. Recent studies have shown that electroacupuncture, 21 probiotic 22 and dicetel 23 have good effects in the treatment of IBS. Our results suggest that moxibustion may potentially be used as an alternative treatment to probiotics and dicetel.
GDNF can be used to treat diseases of the CNS such as Alzheimer’s disease and Parkinson’s disease.24,25 In this way, GDNF and its receptor GFRα3 are critical to maintaining the CNS and ENS. Therefore, GDNF and GFRα3 are likely to play a role in intestinal inflammation. Our study has shown that the expression of GDNF and GFRα3 in the colon of the IBS model rats were higher than in the normal group. It is implicated that the need of neurons and glial cells for GDNF and GFRα3 was increased, suggesting that GDNF and GFRα3 play a protective and promotional role in the regeneration of enteric neurons, are involved in the regulation of intestinal inflammatory processes, and may have a role in promoting recovery of the intestinal mucosa in IBS.
In this study, we also found that, compared with the rats in the normal group, the expression of GDNF and GFRα3 in the spinal cord of rats in the CRD group was increased. Similar to the results reported here, previous studies have also indicated that GDNF is upregulated in IBS patients. 26 Compared with the CRD group, the CRD+MM group showed a reduced expression of GDNF and GFRα3 in the colon and spinal cord. This suggests that moxibustion therapy may regulate the increased expression of GDNF and GFRα3 in the colon and spinal cord of IBS rats. Studies have shown that moxibustion can regulate intestinal inflammation to treat inflammatory bowel disease. 27 Based on the results of our study, it is possible that moxibustion causes the enteric glial cells to secrete GDNF by regulating intestinal inflammation. Consequently, it may alleviate visceral hypersensitivity, reduce the pain threshold and relieve visceral pain. However, the exact mechanism requires further study.
Footnotes
Acknowledgements
The authors would like to thank the Experimental Animal Center, Shanghai University of Traditional Chinese Medicine for providing the animals.
Contributors
XW conceived and designed the study. YW and DJ performed the animal experiments and immunohistochemical analysis. CW and YL performed the PCR experiments and statistical analyses. HW and XJ prepared the figures. XW and QQ drafted the main manuscript text. All authors revised the manuscript and approved the final version accepted for publication.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Natural Science Foundation of Shanghai (grant no. 14ZR1438700) and National Basic Research Programme of China 973 programme (grant no. 2015CB554501).
Patient consent
Not required.
Provenance and peer review
Not commissioned; externally peer reviewed.
