Abstract
Background:
Acupuncture has been recommended as an alternative therapy for migraine. Emerging evidence suggests that the 5-HT7 receptor (5-HT7R) plays a significant facilitatory role in descending modulation in migraine pathophysiology, and that activation of 5-HT7R in the descending pathway is involved in migraine central sensitisation.
Objective:
To investigate the ability of electroacupuncture (EA) to ameliorate central sensitisation via modulation of 5-HT7R in the descending pain pathways using a rat model of migraine induced by repetitive dural electrical stimulation (DES).
Design:
64 male Sprague-Dawley rats were randomly divided into four groups: Normal group; DES group (receiving dural electrical stimulation only); DES+GB20 group (DES model group treated with EA at GB20); and DES+Sham group (DES model group treated with EA at a non-traditional (sham) acupuncture point). The presence of cutaneous allodynia was determined by measuring facial and hind-paw withdrawal latencies to electronic von-Frey. The expression of 5-HT7R in the descending pathways (periaqueductal grey, raphe magnus nucleus, and trigeminal nucleus caudalis) was assessed using immunofluorescence and Western blotting.
Results:
Facial and hind-paw withdrawal thresholds were significantly increased in the DES+GB20 group compared with the untreated DES group. The expression of 5-HT7R was significantly decreased in the DES+GB20 group compared with the DES group (one-way analysis of variance (ANOVA), P<0.05). No significant differences in behaviour or expression were found between the rats in the DES+Sham group and the untreated DES group (one-way ANOVA, P>0.05).
Conclusion:
EA at GB20 may ameliorate central sensitisation in migraine by inhibiting the activation of 5-HT7 receptors in the descending pain pathway in a rat model of migraine.
Keywords
Introduction
Migraine is a perplexing, recurrent, and often disabling neurological disorder affecting approximately 15% of people worldwide 1 . It is typically characterised by unilateral throbbing head pain and/or an aura preceding the episode. 2 Another key feature of migraine is cutaneous allodynia (CA), a sensation of pain or discomfort in response to a normal, non-noxious stimulus that develops in 50–70% of migraineurs. CA is believed to result from central sensitisation of the trigeminovascular pain pathway,3,4 and the presence of this external symptom is considered to be a risk factor for the progression from episodic to chronic migraine. 5 Additionally, patients who experience CA tend to respond poorly to conventional therapy. 6 Furthermore, no specific medications have been developed to inhibit or delay central sensitisation, prompting patients to search for alternative therapies, including acupuncture.
The serotonin (5-hydroxytryptamine, 5-HT) system, originating from the raphe nuclei in the brainstem, can exert differential effects (facilitatory versus inhibitory) on the processing of nociceptive information depending on the subtype of the activated receptor. 7 The 5-HT7 receptor (5-HT7R), the most recently identified 5-HT receptor subtype, has been observed to play a pro-nociceptive role in the modulation of signals along the descending pain pathway; activation of 5-HT7R distributed along the descending pathway results in facilitatory modulation of signals along this pathway, mediating the type of central sensitisation observed in migraine.8,9 Thus 5HT7R is a potential target in the treatment of migraine.
The effectiveness of acupuncture in the treatment of migraine has been validated by a recent systematic review, 10 but its mechanism of action is still poorly understood. Preclinical experiments have shown that the effects of EA may be mediated by type 1 cannabinoid (CB1) receptors and 5-HT, focused mostly on the level of peripheral vascular/body fluid metabolism and less on the central nervous system, especially the descending pathway.11–13
Our previous report showed that electroacupuncture (EA) applied at GB20 in rat models of migraine can reduce grooming behaviour (reflective of CA) and inhibit c-fos expression in the three main brain nuclei comprising the descending pathway: the periaqueductal grey (PAG); the raphe magnus nucleus (RMg); and the trigeminal nucleus caudalis (TNC). 14 These data support the notion that acupuncture intervention may ameliorate central sensitisation via modulation of the descending pathways. The present study aimed to explore the deep mechanisms underlying the efficacy of EA for migraine.
Based on the aforementioned evidence, we hypothesised that the therapeutic effects of EA at GB20 on CA in migraine involves inhibition of the facilitatory effects of 5HT7R in the descending pathway. Accordingly, the aim of the present study was to examine the effect of EA on CA (both cephalic and extra-cephalic cutaneous mechanical sensitivities) and the expression of 5-HT7R in the PAG, RMg and TNC in a rat model of migraine induced by repetitive dural electrical stimulation (DES).
Methods
Ethical statement
All experiments adhered to the national guideline for the care and use of laboratory animals, amendment 2 (State Council of China, 2013). These experiments were reviewed and approved by the Beijing Institutional Review Board for Animal Experiments and Use Committee of Capital Medical University, Beijing (permit no. AEEI 2015–075). All surgeries were performed under anaesthesia, and all efforts were made to reduce suffering.
Animals
Male Sprague-Dawley rats weighing 210±10 g at the time of purchase (Vital River Laboratories (VRL), Beijing, China) were individually housed under a 12/12 hour light/dark cycle (lights on at 8 am), at room temperature 23±2°C and humidity 50±10%. Food and water were provided ad libitum. After a 1-week acclimation period, formal experimentation was initiated.
Dura mater electrode implantation
Rats were anaesthetised with intraperitoneal sodium pentobarbital (Sigma-Aldrich, 60 mg/kg) as previously described. 14 The dura was exposed by drilling two burr holes (1 mm) adjacent to the superior sagittal sinus (SSS), 4 mm anterior and 6 mm posterior to the bregma on the midline suture, respectively. A pair of electrode fixtures (JiAnDeEr Ltd, Beijing, China), insulated at all points except the tips, were carefully inserted into the burr holes and implanted on the dural surface (extending 1.8–1.9 mm from the skull surface). Dental acrylic (New Century Dental Materials Co, Ltd, Shanghai, China) and stainless steel screws were then used to secure the electrode fixtures. Penicillin (0.04 million IU/100 g, Harbin Pharmaceutical Group Co, Ltd, China) was administered following the surgical procedure, and the rats were allowed to recover for 1 week.
Experimental design
After 7 days of post-surgical recovery, 64 animals were randomised into equally-sized groups (n=16 rats each) according to a randomisation list: a DES+GB20 group that received both dural electrical stimulation and electroacupuncture at GB20 (Fengchi); a DES+Sham group that received dural stimulation and EA at a sham location not corresponding to any traditional acupuncture point; a DES group that received only dural stimulation; and a Normal group that underwent electrode implantation but received neither DES nor EA. Investigators were blind to treatment allocation during data analysis.
Establishment of the recurrent rat migraine model
Following baseline behavioural testing, rats in the DES+GB20, DES+Sham, and the untreated DES groups underwent DES. They were electrically stimulated with monophasic square-wave pulses (0.5 ms pulse duration, 1.8~2.0 mA current intensity, 20 Hz stimulus frequency) by a programmed electrical stimulator (YC-2 stimulator, Chengdu) for 15 min every other day (day 1, day 3, day 5 and day 7). 14 Rats in the Normal group were connected to the stimulator but received no stimulation.
Electroacupuncture intervention
Following the DES procedure, rats in the DES+GB20 and DES+Sham groups were restrained in tailored rat fixtures, with their heads and necks sufficiently exposed, for successive EA treatment on days 1 through 7, for a total of seven treatments. In the DES+GB20 group, stainless filiform needles (Suzhou Medical Appliance Factory, Suzhou, China; diameter 0.25 mm; length 25 mm) were inserted bilaterally at GB20 (located 3 mm away from the centre of an imaginery line joining the two ears) to a depth of 2–3 mm directed toward the opposite eye, as described previously. 14 The needles were then attached to a Hans’s acupuncture point nerve stimulator (HANS-200, Nanjing, China), and the stimulus parameters were as follows: 2/15 Hz amplitude-modulated wave (pulse width 0.6 ms); 0.5–1.0 mA intensity (depending on the reaction of the rat); and 15 min duration. In the DES+Sham group, an equivalent EA procedure was performed, except that needles were placed at bilateral, neuroanatomically remote sites not corresponding to traditional acupuncture points (about 10 mm above the iliac crest).
Induction of mechanical hyperalgesia
To evaluate the effect of EA on mechanical hyperalgesia (reflective of CA), an electronic von-Frey anesthesiometer (Model 2390; IITC Life Science, California, USA) fitted with a rigid tip (1.6 mm tip diameter) was used to measure facial/hind-paw withdrawal thresholds with respect to mechanical stimuli. Behavioural tests were conducted on all animals in the same feeding room from 8 am to 10 am on day 0 (baseline), day 2, day 4, and day 6. Withdrawal data were collected for the right and left periorbital and hind-paw regions for each rat by a blinded observer.
Facial allodynia
Rats were placed in a tailored plastic tube restraint (25 cm in length, 8 cm inner diameter) with wire mesh inlay in the front, leaving the periorbital region accessible. After a 30 min adaptation period, the von-Frey tip was applied to the periorbital region with steady vertical pressure until a positive withdrawal response was elicited, namely, vigorous stroking of the face with the ipsilateral fore-paw or quick recoil of the head from stimulation or vocalisation. 15
Hind-paw allodynia
Animals were individually placed on a dedicated elevated testing frame for 30 min before testing to acclimatise them to the arena. The elevated testing frame consisted of a row of 12 transparent perspex enclosures separated by opaque dividers with wire mesh at the bottom. Punctuated mechanical stimuli were applied until the paw withdrew from the tip or was lifted off the mesh floor. All procedures were repeated three times on each side with 30 s intervals between stimulations. The mean value of the three (facial)/six (hind-paw) measurements was considered the withdrawal threshold, with a cut-off value of 60 g, as described by Boegel et al. 16 All testing was performed blind to group assignment.
Immunofluorescence
Following the final DES and EA procedures (day 7), animals were anaesthetised with intraperitoneal 10% chloral hydrate (15 mL/kg) and transcardially perfused through the ascending aorta with 100 mL of 0.1 M phosphate-buffered saline (PBS), followed by 500 mL of 4% paraformaldehyde (PFA) in PBS. The brains were removed and postfixed overnight at 4°C in 4% PFA/PBS, then transferred to 30% sucrose/PBS for 72 hours for cryopreservation. Coronal sections (40 µm thick) through the PAG, RMg and TNC were cut using a cryostat (Leica CM3050S, Leica Inc, Wetzlar, Germany), blocked with goat serum, and incubated with primary antibody against 5-HT7R (1:200, Santa Cruz Biotechnology Inc, USA) overnight at 4°C and then treated with Alexa-488-conjugated secondary antibodies (1:500, Jackson ImmunoResearch Laboratories Inc, Pennsylvania, USA) for 1 hour at room temperature. The sections were then mounted, dehydrated and cover-slipped in antiquenching medium (Fluoromount G, Southern Biotechnology Associates).
Protein extraction and Western blotting
After decapitation, the brains were rapidly removed and processed on a frozen microtome. Bilateral punches of the PAG, RMg and TNC regions were selected using a magnifying glass with stainless steel cannulae of 1000 µm inner diameter and subsequently pooled. Then the tissues were homogenised in 400 µl of ice-cold lysis buffer, supplemented with proteinase inhibitors, and centrifuged for 10 min at 4°C. Protein concentration in the supernatants was determined using a Micro BCA protein assay (Thermo Scientific). For Western blotting, the samples (40 µg protein) were loaded onto SDS-PAGE gels and transferred to PVDF (polyvinylidene fluoride) membranes. The membrane was blocked for 1 hour at room temperature in Tris-buffered saline (TBS) containing 0.1% Tween-20 (TBST), 5% skim milk and 0.2% bovine serum albumin (BSA), and washed four times for 5 min each time in TBST. The membrane was incubated for 2 hours at room temperature with rabbit anti-5-HT7 (1:600; Millipore Corporation, USA) mouse monoclonal anti-β-actin (1:2000; Millipore Corporation, USA) primary antibodies in TBST. After incubation with a horseradish peroxidase-conjugated secondary antibody (anti-rabbit; 1:1000) for 1 hour, the primary antibodies were visualised through enhanced chemiluminescence (Amersham Biosciences, Buckinghamshire, UK). Kodak film was used to detect chemiluminescent signals. The band intensities of the detected proteins were analysed using Quantity One software (Bio-Rad). The control gel used for gel loading and protein transfer was β–actin.
Image analysis
Square cross-sectional images of immunofluorescence were captured using a Leica semiautomatic light microscope (Leica DM5500 B, Germany) at 20× magnification. Cells positive for 5-HT7R and exhibiting protein grey values in the PAG, RMg and TNC were assessed by a blinded observer using Leica-accompanying software and Image J software, respectively. Anatomical boundaries were determined according to a rat brain atlas. 17
Statistical analysis
All data are presented as mean±SD. Data were analysed using one-way analysis of variance (ANOVA) followed by Bonferroni (homogeneity of variance) or Tamhane (heterogeneity of variance) post hoc analysis. Statistical evaluation was performed with the Statistical Package for the Social Sciences Software (SPSS) version 12.0 (SPSS Inc, Chicago, IL, USA). Values of P<0.05 were considered statistically significant.
Results
Facial and hind-paw allodynia
The nociceptive threshold results for each group at all time points are shown in figure 1. Before the first/baseline DES procedure, no significant differences in facial or hind-paw nociceptive thresholds were observed between groups (P>0.05).
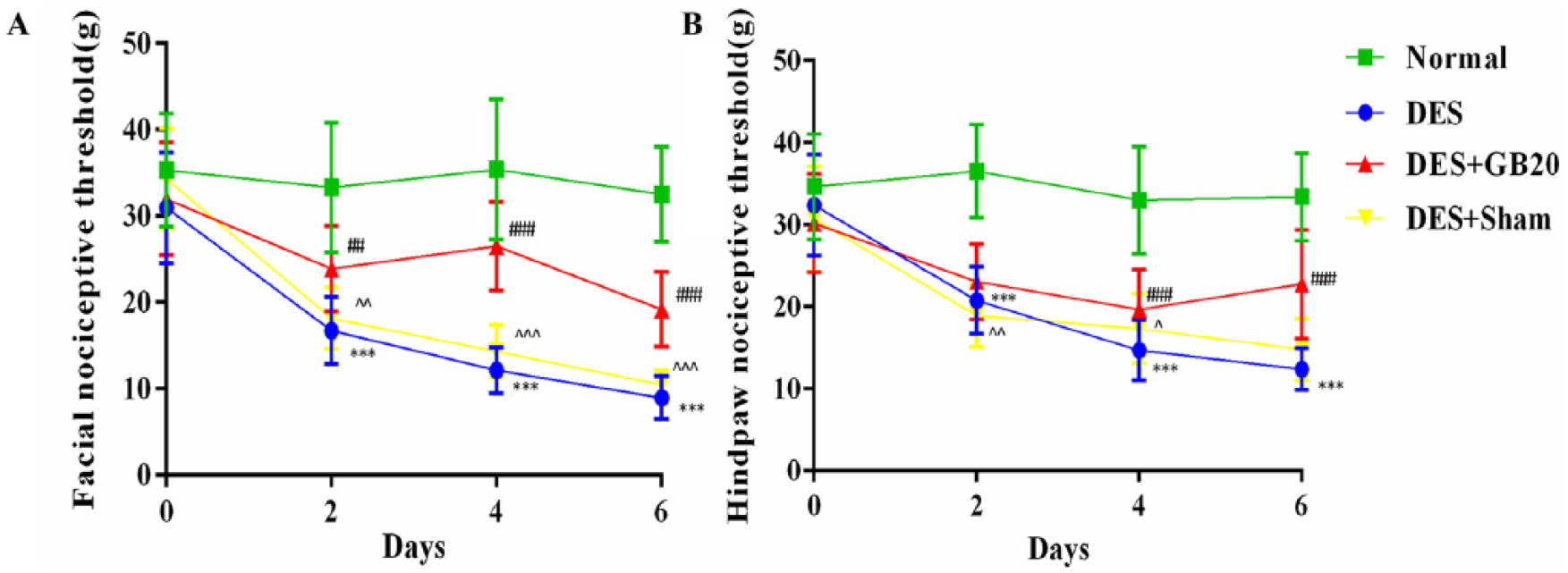
Nociceptive threshold results over the course of the experiment. Quantification of (A) facial allodynia and (B) hind-paw allodynia. Data are presented as mean±SD (n=16 per time point per group). * P<0.05, ** P<0.01, *** P<0.001, DES versus Normal. # P<0.05, ## P<0.01, ### P<0.001, DES+GB20 versus DES. ^ P<0.05, ^^ P<0.01, ^^^ P<0.001, DES+Sham versus DES+GB20. DES, dural electrical stimulation.
Across all subsequent time points (day 2, day 4 and day 6), repetitive DES produced tardive and significant CA (DES vs. Normal: P<0.001 for facial/hind-paw across the three time points). For the facial nociceptive threshold, these effects were all significantly reversed by EA at GB20 in the DES+GB20 group at the previously indicated time points (DES vs. DES+GB20: day 2, P=0.002; day 4/6, P<0.001). The lowered hind-paw nociceptive threshold was significantly attenuated by EA treatment at days 4 and 6 (but not day 2) (DES vs. DES+GB20: day 2, P=0.254; day 4/6, P<0.001). Nevertheless, EA at the non-acupuncture point did not significantly alter any sensory thresholds (DES vs. DES+Sham: facial: day 2, P=0.723; day 4, P=0.078; day 6, P=0.085; hind-paw: day 2, P=0.96; day 4, P=0.137; day 6, P=0.077).
Immunofluorescence analysis of 5-HT7R
Representative fluorescent images and results depicting 5-HT7R immunopositive cells in the PAG, RMg and TNC are respectively shown in figures 2-4. As shown in these figures, the brain sections of animals in the DES group contained more 5-HT7R-positive cells than those of animals in the Normal group (DES vs. Normal: P<0.001 for all). However, this effect were significantly reversed/attenuated by repeated EA at GB20, indicated by the reduced number of 5HT7R-positive cells (DES vs. DES+GB20: PAG/TNC P<0.001; RMg P=0.001), while they were not reversed/attenuated by EA at the non-acupuncture point (DES vs. DES+Sham: PAG P=0.295; RMg P=1; TNC P=0.094).
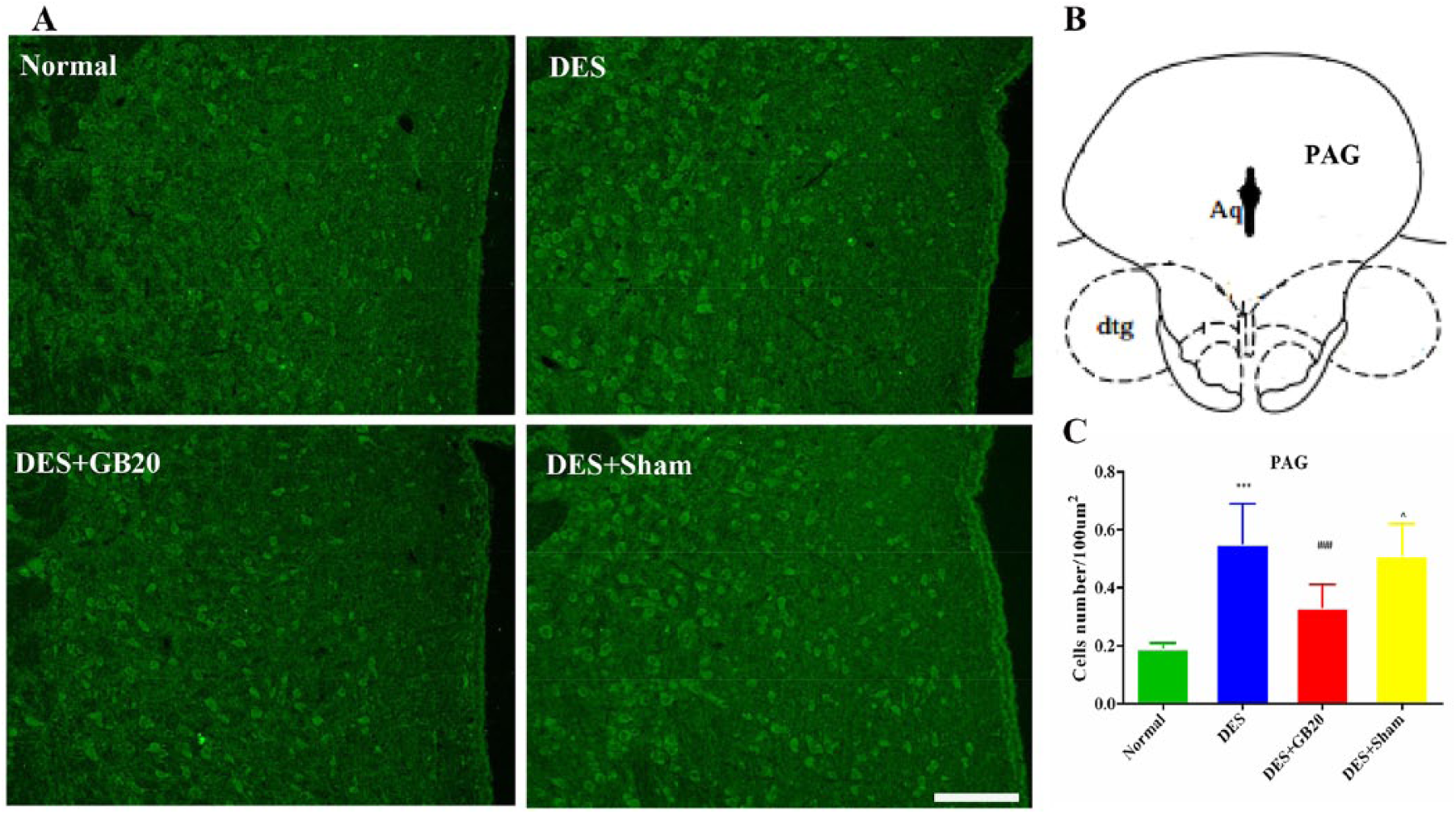
Representative fluorescent images and results depicting 5-HT7R immunopositive cells in the periaqueductal grey (PAG). Panel A: immunofluorescence expression of 5-HT7R-positive cells in the PAG (200×, bar=50 µm). Panel B: anatomical localisation of PAG. Panel C: quantification of the number of 5-HT7R positive cells in the PAG (/100 µm2). Data are presented as mean±SD (n=8 per group). *** P<0.001, DES versus Normal; ### P<0.001, DES+GB20 versus DES; ^ P<0.05, DES+Sham versus DES+GB20. Aq, aqueduct (of Sylvius); dtg, dorsal tegmental bundle.
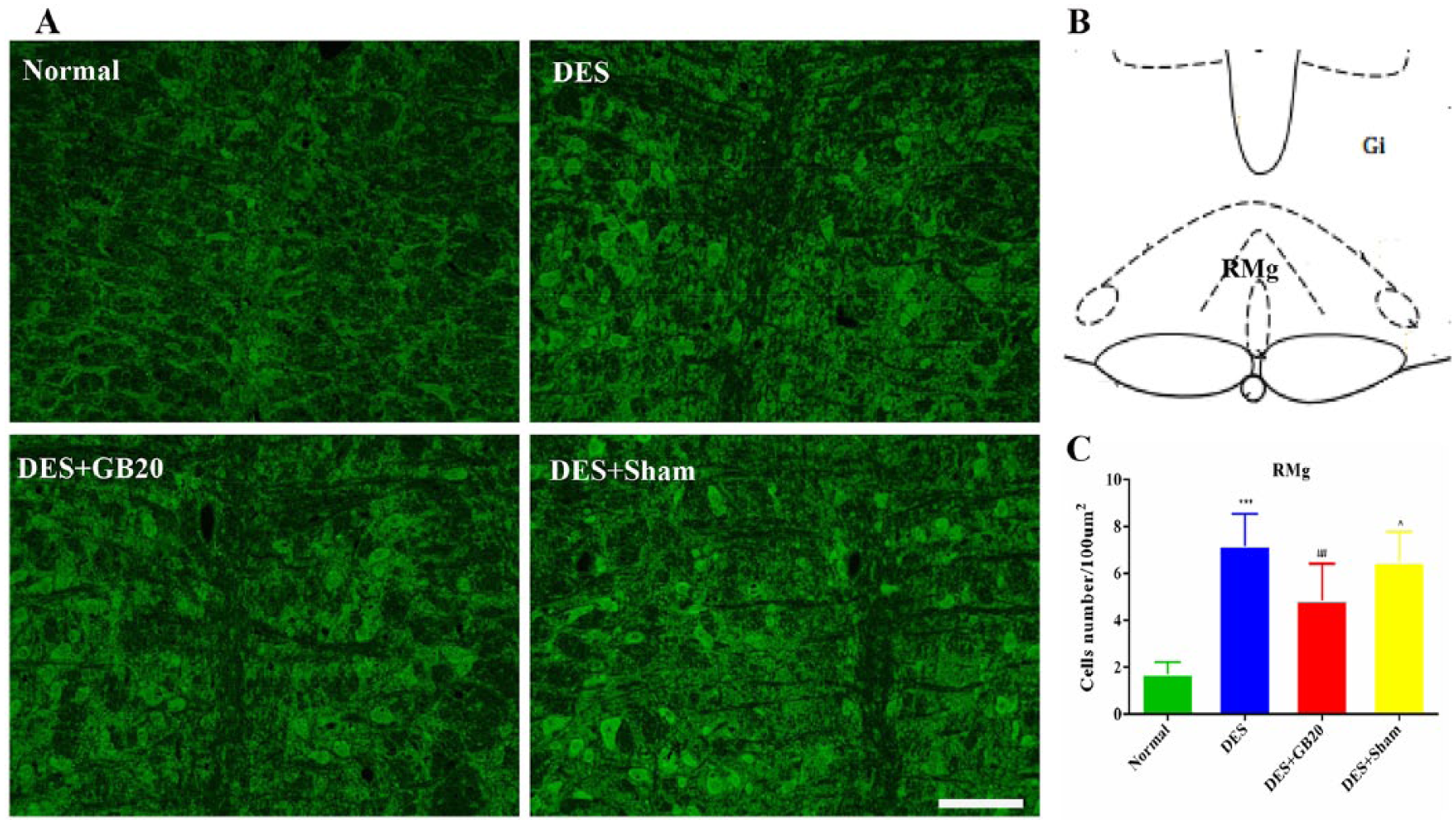
Representative fluorescent images and results depicting 5-HT7R immunopositive cells in the raphe magnus nucleus (RMg). Panel A: immunofluorescence expression of 5-HT7R-positive cells in the RMg (200×, bar=50 µm). Panel B: anatomical localisation of RMg. Panel C: quantification of the number of 5-HT7R positive cells in the RMg (/100 µm2). Data are presented as mean±SD (n=8 per group). *** P<0.001, DES versus Normal; ## P<0.01, DES+GB20 versus DES; ^ P<0.05, DES+Sham versus DES+GB20. DES, dural electrical stimulation; EA, electroacupuncture; Gi, gigantocellular reticular nucleus.
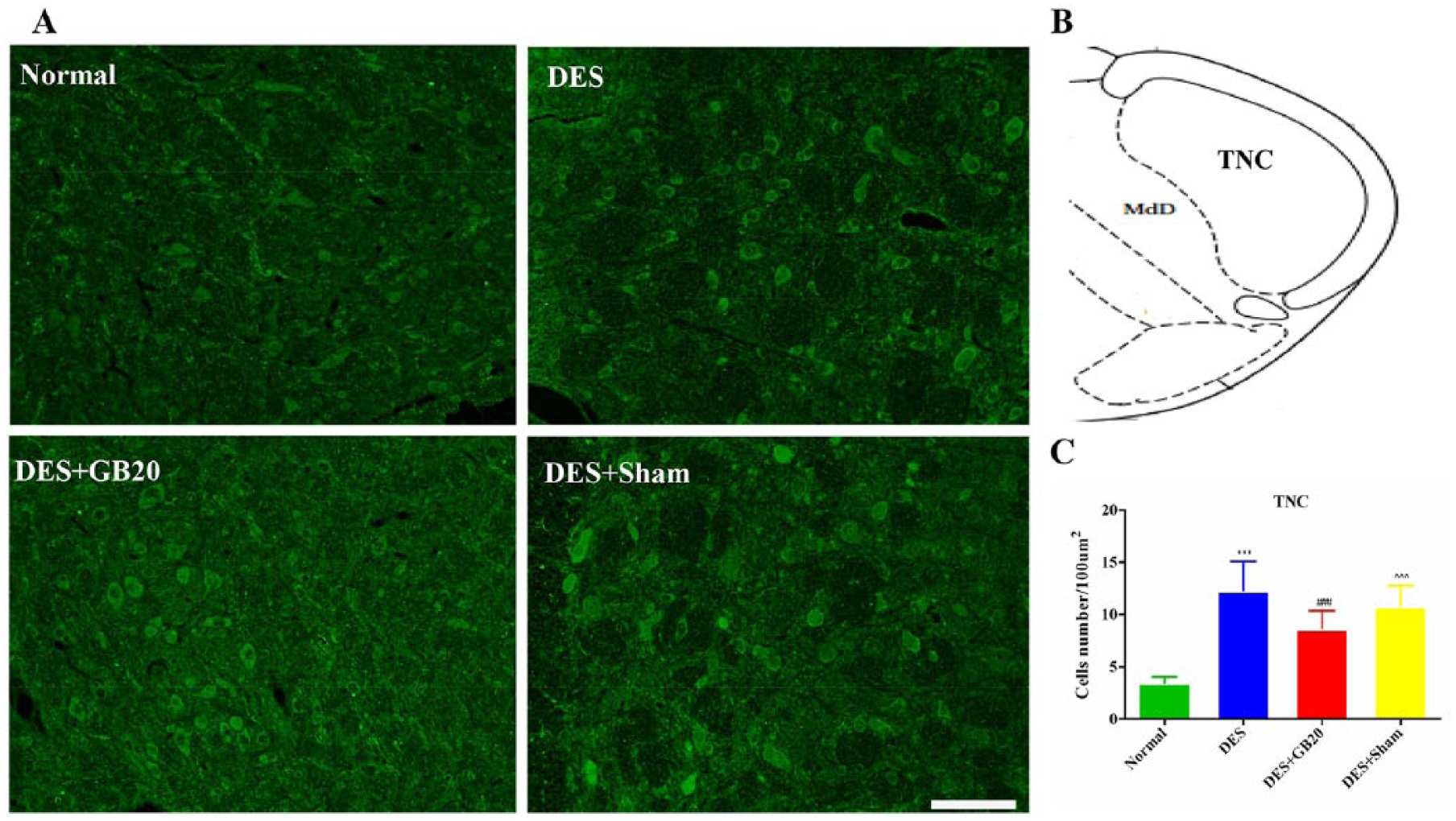
Representative fluorescent images and results depicting 5-HT7R immunopositive cells in the trigeminal nucleus caudalis (TNC). Panel A: immunofluorescence expression of 5-HT7R-positive cells in the TNC (200×, bar=50 µm). Panel B: anatomical localisation of TNC. Panel C: quantification of the number of 5-HT7R positive cells in the TNC (/100 µm2). Data are presented as mean±SD (n=8 per group). * P<0.05, ** P<0.01, *** P<0.001, DES versus Normal. # P<0.05, ## P<0.01, ### P<0.001, DES+GB20 versus DES. ^ P<0.05, ^^ P<0.01, ^^^ P<0.001, DES+Sham versus DES+GB20. DES, dural electrical stimulation; MdD, medullary reticular nucleus, dorsal part.
Assessment of 5-HT7R protein level
As shown in figure 5, the levels of 5-HT7R protein expression in the PAG, RMg and TNC were significantly increased by repeated DES in the DES group (DES vs. Normal: P<0.001 for all). After EA at GB20, the DES-induced increases in protein expression were significantly attenuated in the same brain areas (DES vs. DES+GB20: PAG P=0.02; RMg/TNC P=0.001). However, EA at the non-acupuncture point did not significantly influence the level of 5-HT7R protein expression in these brain areas (DES vs. DES+Sham: PAG P=0.996; RMg P=1; TNC P=0.783).
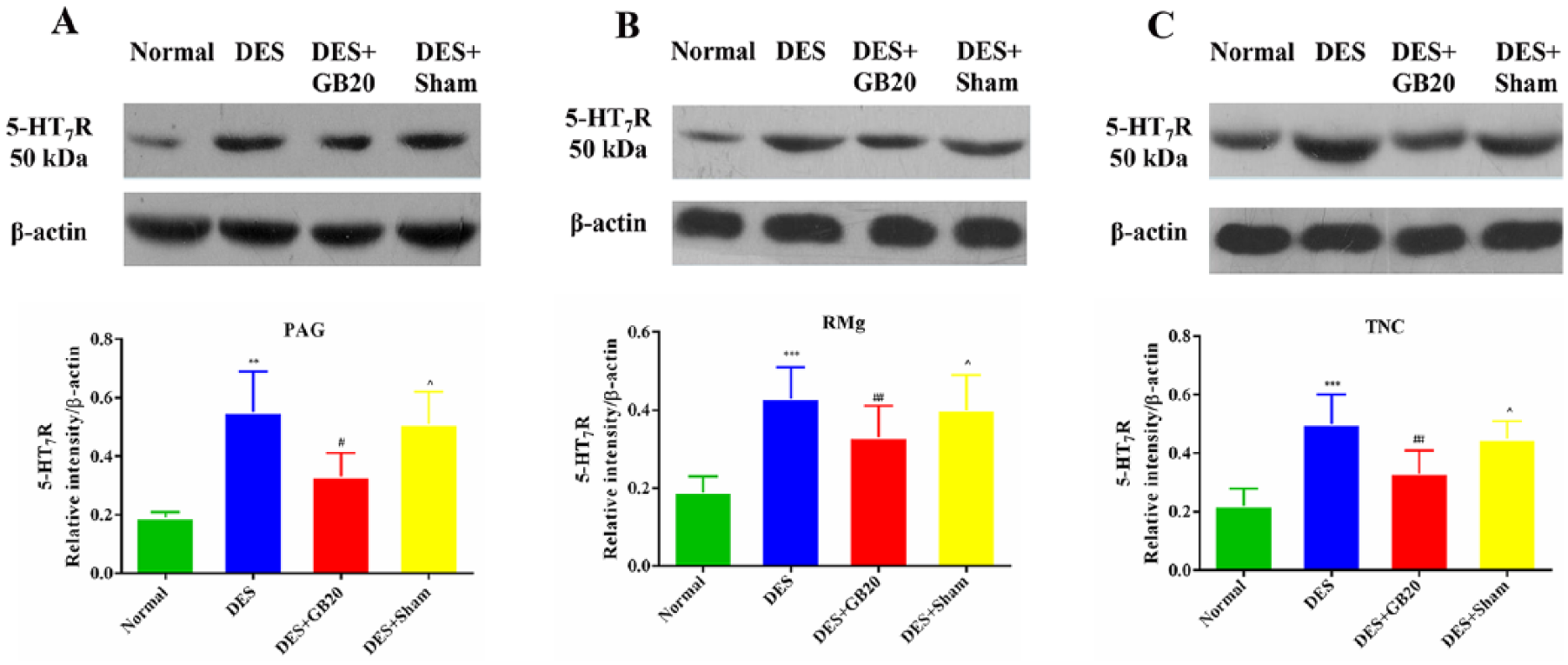
Levels of 5-HT7R protein expression in the periaqueductal grey (PAG), raphe magnus nucleus (RMg) and trigeminal nucleus caudalis (TNC). Representative immunoblots and densitometry analysis of 5-HT7R in PAG (A), RMg (B) and TNC (C). Data are presented as mean±SD (n=8 per group). *** P<0.001, DES versus Normal; ### P<0.001, DES+GB20 versus DES; ^^^ P<0.001, DES+Sham versus DES+GB20. DES, dural electrical stimulation.
Discussion
The ability of EA at GB20 to ameliorate migraine-like symptoms via alteration of the descending pathway in a rat model, indicated by decreased c-fos expression in the PAG–RMg–TNC, has been previously elucidated. 14 In the present study we found that EA at GB20 can increase facial and hind-paw withdrawal thresholds, decrease the expression of 5-HT7 receptors in the aforementioned nuclei, and provide evidence that amelioration of central sensitisation and CA occurs at least in part due to the inhibitory effect of EA at GB20 on 5-HT7R along the descending pathways.
EA at GB20 can ameliorate migraine central sensitisation status
CA is an abnormal skin sensitivity invoked by innocuous stimuli and commonly occurs in conjunction with migraine episodes.18,19 As the frequency of migraine increases, this hypersensitivity of the skin can develop in the ipsilateral craniofacial region and then beyond the territory of trigeminal nerve innervation, spreading to the limbs and, in some cases, the whole body. 5 CA has been recognised as a clinical marker of central sensitisation in the trigeminovascular pain pathways induced by repetitive firing of dural nociceptors, and its presence renders the patient more susceptible to the development of chronic migraine. 5 In animal studies, repetitive DES is capable of inducing migraines that successfully imitate recurrent migraine conditions, and facial and hind-paw withdrawal latencies to electronic stimulation with von-Frey filaments reflect cephalic and extra-cephalic allodynia, respectively.
Evidence from functional imaging and electrophysiological studies supports the notion that the development of cephalic/cutaneous allodynia suggests a sensitisation of second-order trigeminovascular neurons in the TNC, while extra-cephalic allodynia is attributed to sensitisation of third-order thalamic nociceptive neurons and/or the dysfunction of the descending pathway.20,21 In the present study, EA at GB20 was observed to significantly reverse the DES-induced reduction in facial (days 2, 4 and 6) and hind-paw (days 4 and 6) nociceptive threshold, providing behavioural evidence in support of the notion that EA at GB20 may ameliorate CA and central sensitisation.
Potential mechanism of anti-hyperalgesia in recurrent migraine
Growing evidence supports the concept that the imbalance of descending pathways in migraine (impaired descending inhibition and/or enhanced descending facilitation) is implicated in the development and maintenance of central sensitisation, indicated by the presence of extra-cephalic allodynia.5,20
Sufficient evidence exists to support the notion that descending serotonergic projections arising from the RMg play an important role in the bidirectional modulation of pain in the descending pathways, which is dependent on the activation of different 5-HT receptor subtypes.22–24 The 5-HT7R, the most recently identified 5-HT receptor subtype, is positively involved in generalised nociceptive processing. In migraine, 5-HT7R expression has been widely observed at different levels of the trigeminovascular pain pathway, including the descending pathways. This 5-HT receptor subtype has also been associated with meningeal vasodilatation, neuropeptide release and central sensitisation.8,23 Several preclinical findings have additionally revealed that selective inhibition of 5-HT7R results in anti-migraine effects, while agonists of 5-HT7R can reverse the inhibitory effects of these antagonists.9,25 Evidence from the present study, taken together with the aforementioned observations, would suggest a facilitatory role of 5-HT7R in the modulation of nociception. 8 Increased descending facilitation mediates central sensitisation and extra-cephalic allodynia, and activation of 5-HT7R results in the facilitation of pain signals. It can therefore be inferred that the enhanced descending facilitation stems partly from the activation of 5-HT7R along the descending pathway, and that 5-HT7 receptors mediate central sensitisation by affecting the transmission of nociceptive information. 26
We previously demonstrated increased c-fos expression in the descending pathway of DES-treated rats, and that this increase in expression can be reversed by EA at GB20. 14 Consistent with the results of previous studies, 9 the results of the present study indicate significantly increased 5-HT7R expression within the PAG, RMg and TNC in repetitively DES-exposed rats, suggesting that the descending pathway participates in the development of central sensitisation. However, repeated EA at GB20 results in significant decreases in 5HT7R expression in these same regions. The altered nociceptive thresholds and levels of 5-HT7R expression in the PAG, RMg and TNC for rats in the verum EA-treated group may be regarded as the result of diminished 5HT7R facilitation of the descending pathways.
Conclusion
EA at GB20 may decrease facial and hind-paw withdrawal thresholds and inhibit 5-HT7R expression within the PAG, RMg and TNC in a rat model of migraine. Overall, EA at GB20 may ameliorate CA and central sensitisation in migraine via inhibition of 5-HT7R in the descending pain modulatory system. Ongoing studies are aimed at exploring the roles of the remaining 5-HT receptor subtypes in the inhibition or facilitation of pain via the descending pathways.
Footnotes
Contributors
PP, LL, LPZ, ZYQ and CYT performed the experiments. PP and LL drafted the manuscript. LPW and WMY supervised the research programme, contributed to the integration of the research team and revised the manuscript. All authors read and approved the final version of the manuscript accepted for publication. PP and LL contributed to this research equally.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Basic Research Program of China (grant no. 2014CB543203) and National Natural Science Foundation of China (grant no. 81803903, 81603683).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Provenance and peer review
Not commissioned; externally peer reviewed.
