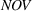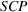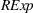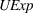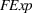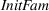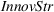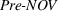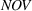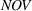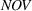Abstract
The accumulation of experience that occurs with production is likely to impact an organization's ability to develop manufacturing process innovations. However, how different types of manufacturing experience relate to the characteristics of an organization's process innovation output is an open question. In this study, we investigate how a firm's accumulated related and unrelated manufacturing experiences are associated with this firm's ability to innovate its production methods. To characterize firms' process innovation output, we observe their portfolios of patented manufacturing inventions, which we qualitatively evaluate over time, through a unique collaboration with expert patent attorneys, along two critical dimensions: novelty and scope. We argue that related manufacturing experience leads to a better understanding of parts of the focal product's technological landscape that will allow the development of inventions of broader scope. However, it may also contribute to inertia in that it might restrict the firm's innovative activity to more familiar regions of the landscape, thereby limiting inventions' novelty. Conversely, manufacturing experience with products that are unrelated to the focal product is expected to stimulate and support a broader search that includes more distant regions of the focal product's technological landscape, which would lead to more novel manufacturing inventions. Yet, the application of this unrelated experience to the production of the focal product is likely to require additional exploratory effort in a not‐well‐understood region of the focal product's landscape, likely resulting in inventions of limited scope. In line with our hypotheses, we find that related (unrelated) manufacturing experience is positively (negatively) associated with inventions' scope, and negatively (positively) associated with inventions' novelty. In addition to supporting the relevance of a multidimensional evaluation of innovations, our findings provide practical guidance regarding the strategic implications of a firm's knowledge management.
INTRODUCTION
Process innovation, defined as “new elements introduced into an organization's production or service operations (e.g., input materials, task specifications, work and information flow mechanisms, and equipment) to produce a product or render a service” (Damanpour, 2008, p. 698), plays a strategic role in the long‐term survival of manufacturing organizations (Pisano, 1997). It can enable improvements in production yields (Tushman & Nadler, 1986), reductions in production costs (OECD, 2005), and other productivity enhancements (Ettlie & Reza, 1992). For mature products, it represents the dominant form of innovation (Utterback & Abernathy, 1975). Yet, while considerable research effort has been expended to understand the drivers of product innovation, process innovation remains largely understudied (Crossan & Apaydin, 2010). While past literature (e.g., Leiblein, 2009) suggests that an organization's manufacturing experience is crucial for generating new process‐improvement ideas, that is, process innovations, a more granular understanding of this relationship is missing (Argote & Miron‐Spektor, 2011). In particular, how different types of manufacturing experience relate to an organization's process innovation output remains an open question (Cohen, 2010), which motivates our study.
Experience is “what transpires in the organization as it performs its task” (Argote & Miron‐Spektor, 2011, p. 1024). Through manufacturing experience, organizations develop valuable skills, an improved understanding of the conditions required by the technology and/or equipment (Leiblein, 2009), and gain knowledge, which becomes embedded in various repositories such as an organization's members, tasks, and tools (Argote & Hora, 2017). As a result, the type of manufacturing experience that an organization accumulates plays an important role in shaping its ability to devise innovative solutions to its process development needs (Chari et al., 2007), allowing it to remain competitive. Specifically, when looking at the output produced through a focal process, a distinction has often been made between related and unrelated manufacturing experience (Cohen & Levinthal, 1990; Fong Boh & Slaughter, 2007).
In manufacturing environments, technical knowledge is likely to be transferable across the production processes of related products, thus benefiting innovation (Garcia‐Vega, 2006). Indeed, experience with manufacturing related products, that is, related experience, suggests an exposure to a variety of tasks and technical challenges, which are likely to be relevant when attempting to solve problems associated with the focal manufacturing process (Lawrence, 2018). However, the accumulation of related manufacturing experience can also, overtime, create inertia and act as a deterrent to process innovation as the firm becomes trapped in its current competencies (Lawrence, 2018; Levitt & March, 1988). This is because process innovation often entails significant changes such as the introduction of new manufacturing equipment or the adoption of new production routines, all of which can drastically impact existing production practices (Leiblein, 2009).
In contrast, experience with operating manufacturing processes that create unrelated products, that is, unrelated experience, broadens the knowledge base of a firm (Leten et al., 2007) and enables proficiency across various technologies (Argote & Miron‐Spektor, 2011). As such, it can give rise to potentially greater innovation than if relying on related experience alone (Nelson, 1959). Indeed, when firms diversify into unrelated fields, more learning opportunities are available through higher cross‐fertilization between different technologies (Garcia‐Vega, 2006) and through learning from diverse errors (Egelman et al., 2017). Yet, even though unrelated experience may protect organizations from becoming locked into a particular set of technologies, it can also put a strain on a firm's limited financial and cognitive resources (Hitt et al., 1997), thereby increasing the opportunity cost of not pursuing innovation in related fields.
The above discussion suggests that, even when conceptually differentiating between related and unrelated manufacturing experience, their respective relationship with process innovation remains ambiguous, which calls for greater empirical investigation. In particular, it appears worthwhile to distinguish between the scope and the novelty of a firm's innovative output in order to reconcile the contradicting conceptual arguments presented above. Using a firm's portfolio of manufacturing process patents as the main indicator of its innovative output (Jain, 2013), we define novelty as the technological distance between the patented innovations and prior art 1 (Reitzig, 2003) and scope as the number of substitute process configurations that are simultaneously covered by the patented innovations (Gilbert & Shapiro, 1990; Lerner, 1994). On the one hand, our previous depiction of the relationships between experience and innovative output would indicate that related knowledge, because of its transferability, has the potential to enhance one aspect of innovation—its scope—but also carries the risk of lodging the firm into a competency trap, which would restrict its novelty. On the other hand, because it broadens an organization's knowledge base, unrelated experience could enhance a firm's ability to identify highly novel solutions for the focal process. However, these inventions would also push the firm outside of its typical operating practices. This unfamiliarity implies that the identification of substitute process configurations would not only be relatively harder to envision but would also involve significant exploratory effort. Hence, cognitive and financial constraints could limit the scope of these inventions.
To study these relationships, we choose as our context the manufacturing of active pharmaceutical ingredients (APIs) for drugs that have lost product‐patent protection. Product‐patent expiration implies that the originating pharmaceutical firm faces competition from bio‐equivalent versions, that is, “generics,” which contain the same API. In these relatively saturated markets, process innovation becomes the main way by which firms can gain a competitive advantage and secure a greater market share (Morton, 1999). We examine how a pharmaceutical firm's accumulated related and unrelated experiences in API production are associated with this firm's ability to innovate its manufacturing methods, as reflected in the characteristics of its portfolio of manufacturing process patents.
To qualitatively characterize firms' portfolios of patented manufacturing inventions, we collaborate with expert patent attorneys. This unique characteristic of our study allows us to simultaneously evaluate the novelty and scope of these portfolios. By focusing our study on firms that all belong to a single industrial sector, that is, the pharmaceutical industry, where the patent mechanism is widely considered as the preferred mechanism for protecting a firm's intellectual property (Mansfield, 1986), we also reduce the need to control across firms “for fundamental differences in technological opportunity and propensity to patent” (Lieberman, 1987, p. 258).
Among pharmaceutical products, we focus on APIs for antineoplastic drugs—the broad family of drugs, which are used in the treatment of cancer—because these drugs are significant from both an economic and a political point of view. Antineoplastics “rank first in terms of global spending by therapeutic class” and “figure prominently in discussions over health reform, alternatively symbolizing wasteful spending and biomedical progress” (Howard et al., 2015, p. 140). Moreover, compared to other families of drugs, anticancer drugs are complex, challenging‐to‐produce, and chemically diverse drugs that affect a variety of organs, tissues, and tumors (Chabner & Longo, 2011). As such, to remain innovative, a firm must be capable of developing processes that successfully integrate a complex technological skill set that relies on engineering, chemistry, and microbiology (Bennett & Cole, 2003). Manufacturing experience is expected to play a crucial role in shaping this organizational skillset.
We find that related and unrelated experiences have contrasting relationships with the novelty and scope of a firm's innovative output. Specifically, results suggest that related experience allows inventions of broader scope for the focal API's manufacturing process but limits their novelty while unrelated experience allows for inventions of higher novelty but narrower scope.
Overall, we make the following significant contributions. First, we respond to persistent calls in the innovation literature (e.g., Crossan & Apaydin, 2010) for more research specifically focused on process innovation, and its determinants. While process innovation differs from product innovation in terms of its nature, determinants, and potential impact (Becheikh & Amara, 2006), researchers frequently fail to distinguish between the two. This overlooks the fact that the knowledge that is necessary to enable process innovations is of a more complex and tacit nature. Indeed, process innovations result from hands‐on experience and a more intimate understanding of how a process' components interact with each other and with the organizational setting in which the process is executed (Gopalakrishnan & Bierly, 1999). Moreover, because process innovations can have a cross‐functional impact, their development requires input from the different functions of an organization whose operation will be affected by the new technology (Boer & During, 2001). Our work is motivated by these important distinctions between product and process innovation. By focusing on the manufacturing of APIs for drugs open to competition from generics, we obtain an ideal setting for a deeper investigation of manufacturing experience as a key determinant of process innovation. Through our analysis, we show that manufacturing experience has the potential to benefit certain attributes of process innovation while hurting others.
Second, while related and unrelated experiences have frequently been considered as sources of learning in the operations management (OM) literature, existing studies have mostly focused on individuals and teams (e.g., Fong Boh & Slaughter, 2007; Gino et al., 2010) and frequently in single‐firm service settings. In manufacturing settings, where again a large number of studies are single‐firm or even single‐site studies (e.g., Egelman et al., 2017; Levitt et al., 2013), production experience has mostly been considered in relation to the learning curve (e.g., Sinclair et al., 2000) and has thus been examined in relation to measures of productivity and not of process innovativeness. As such the association between manufacturing experience and manufacturing process innovation is not well understood and existing results from OM studies that focus on productivity are unlikely to directly carry over to process innovation, which tends to occur via nonroutine and nonrepetitive activities (Jain, 2013). The few studies that have examined the association between manufacturing experience accumulation and process innovation have found a positive association between learning by doing and the rate of patenting (Lieberman, 1987), that engineering changes are driven by the accumulation of process experience (Adler & Clark, 1991), and that introduction of new processes into a production facility is disruptive for learning relative to existing processes (Hatch & Mowery, 1998). Thus, these studies remain at a high level and do not distinguish between different types of experience or different qualitative characteristics of process innovativeness. Our multifirm and multiproduct data set allows us to compare the innovativeness of manufacturing methods used by firms to produce essentially the same output, that is, a given API. Thus, by considering a variety of firms, with each having unique related and unrelated product portfolios relative to a focal product, we are able to build and expand on prior literature on manufacturing process innovativeness.
Third, we also contribute more broadly to the literature on organizational learning, where it has been observed that “a more fine‐grained analysis of the experience‐creativity link will help reveal underlying mechanisms and boundary conditions that explain how, when, and why prior experience affects knowledge creation in organizations” (Argote & Miron‐Spektor, 2011). This analysis is especially needed in multiproduct environments where different products share common resources and therefore less is known about how different types of experience affect learning, knowledge transfer, and the generation of new ideas (Egelman et al., 2017). With this research, we address this need while also considering multiple qualitative dimensions of generated (and patented) ideas. To the best of our knowledge, such an in‐depth examination of the association between manufacturing experience and process innovation is unique in the literature.
Our work also provides important insights for practitioners. When a firm's product portfolio is concentrated on a set of related products, new manufacturing methods that the firm invents for these products over time are likely to be characterized by broad scope (i.e., cover a large number of possible alternative process configurations) but to lack novelty. Conversely, a diverse product portfolio might enable the firm to invent more novel manufacturing methods (i.e., methods that differ more fundamentally from existing practices) but these are likely to be of a narrower scope. This suggests that firms' product portfolio and specific managerial practices targeted toward experience and knowledge management could play a key role in shaping their competitive strategy.
LITERATURE REVIEW AND HYPOTHESIS DEVELOPMENT
Past literature has extensively discussed some of the characteristics that affect a firm's ability to innovate. For instance, a firm's level of R&D spending and the extent of competition in the markets in which a firm operates have both been found to be important factors (Cohen, 2010; Matraves, 1999). Less well understood is the role of the firm's manufacturing experience. We begin our review by focusing on available evidence on the link between a firm's manufacturing experience and its innovative capabilities. We then introduce the concept of a “technological landscape” as the organizing framework for our arguments and use it to offer a more nuanced discussion of this link by distinguishing between related and unrelated manufacturing experience and narrowing our focus on process innovation.
Manufacturing experience and innovative output
Through manufacturing experience, firms develop operational (i.e., know‐how) and conceptual (i.e., know‐why) learning (Malerba, 1992), both of which are crucial drivers of process innovation (Cornelius et al., 2020). This link between manufacturing experience and process innovation has been demonstrated in past literature (e.g., Lieberman, 1987), which has found that learning‐by‐doing influences the innovativeness of a firm's manufacturing processes and enhances their productivity. However, this literature has focused on the volume of patenting activity without examining its qualitative attributes. It also does not distinguish between the different types of accumulated manufacturing experience, which, in pharmaceuticals, are likely to play an even more pronounced role in a firm's ability to innovate its processes because an intimate understanding of how complex scientific principles translate into manufacturing practice is essential for process innovation to occur (Jain, 2013). For instance, the development over time of knowledge crucial for manufacturing process innovation (e.g., knowing which pharmaceutical excipients would perform best, and worst, under a particular set of process conditions or under a particular manufacturing setup) can only be achieved through manufacturing experience.
More generally, Xie and O'Neill (2013) explain that as firms accumulate experience manufacturing their products, they gain technological knowledge and skills, that is, know‐how, which can then be continuously applied to follow‐on research and problem solving, and contribute to the development of strong internal R&D capabilities, that is, know‐why (Yeoh & Roth, 1999). These R&D capabilities, in turn, are crucial for solving future manufacturing problems, uncovering opportunities for long‐lasting process improvements, and so forth, and eventually enabling the development of manufacturing process innovations. This strong interdependence between R&D and manufacturing in driving process innovation explains why pharmaceutical firms strive to streamline information flows between the lab and the shop floor (Cardinal et al., 2001).
To better illustrate how an organization's accumulated manufacturing experience might inform its R&D efforts and influence its innovative output in our setting, consider the following statement from the production and R&D head of a leading manufacturer of APIs
2
:
The information flows described in the above statement are not necessarily limited to a single manufacturing process. Instead, solutions are often derived from the combination of knowledge that has been obtained across the firm's different manufacturing processes. For example, as explained by Chaudhuri (2013),
The technological landscape of pharmaceutical manufacturing and the novelty and scope of innovation
The design of a manufacturing process consists in several configuration decisions along a set of technological attributes (Mihm et al., 2015). These technological attributes can be thought of as the different dimensions of a “technological landscape,” which can be defined as “the space that inventors must search when attempting to discover useful new inventions” (Fleming & Sorenson, 2004, p. 912). As such, new inventions can be considered as recombinations of existing “technological components,” which themselves can be thought of as “fundamental bits of knowledge or matter that inventors might use to build inventions” (Fleming & Sorenson, 2004, p. 910).
The concept of a technological landscape has been used extensively in the operations management and strategy literatures (e.g., Aharonson & Schilling, 2016; Mihm et al., 2015) as the organizing framework for examining a firm's innovation activities. Any given set of decisions along the landscape's different technological attributes corresponds to a specific point on the landscape and can be referred to as a “technological configuration.” Manufacturing inventions of the type included in our sample of patents can be considered as choices along attributes such as input materials, solvents, catalysts, production steps and their sequencing, equipment and their configuration, and/or other operating conditions such as temperature, light, humidity, and pressure. Because these inventions typically capture entire decision ranges along certain attributes, each corresponding patent is going to protect “not just one point but instead an entire area in the technology landscape” (Mihm et al., 2015, p. 2669).
Seen from this angle, each API in our study, can be thought of as having its own technological landscape. A firm's position on the landscape for a focal API is then determined by the firm's set of manufacturing choices across the entire API production process (Kauffman et al., 2000). According to this conceptualization, manufacturing process innovation, as reflected in a firm's portfolio of process patent(s) for a focal API, becomes a means to occupy a unique, and potentially more valuable, position on that API's technological landscape. Consistent with our formal definitions of novelty and scope provided in Section 1, portfolios of higher novelty would correspond to technological configurations that are located in a more distant region of the technological landscape, relative to the state of the art, while portfolios of broader scope allow the firm to maintain exclusive access to a broader set of related technological configurations or equivalently, a wider region of the technological landscape (Lugovoi et al., 2021).
While scope has not received the same amount of attention as novelty in the literature, it represents an important dimension of innovation because it captures the degree of difficulty for competitors to operate similar technological configurations without infringing on any of the patent‐holding firm's patented manufacturing inventions. In markets that are highly saturated, such as the pharmaceutical markets for generics, having inventions of broad scope can be particularly critical to maintain differentiation from competition, thereby achieving long‐term profitability and survival (Schmidt & Porteus, 2000; Yiannaka & Fulton, 2001).
Because novelty and scope reflect distinct characteristics of a firm's innovative output, patent portfolios can be high in novelty but low in scope or vice versa. To illustrate the former, consider Mesna, a drug in our sample that is used in the treatment of chemotherapy‐related cystitis (Vidal, 2013). UCB Pharma developed a new manufacturing process for the Mesna API, which involves a hydrolysis step followed by an isolation step and results in an active ingredient of higher purity profile relative to prior art (Leveque et al., 2011). While the novelty of the process for the preparation of this API was high, the resulting patent was of narrow scope, as, according to our patent experts, competitors bypassed this invention in a relatively effortless manner. For example, Fresenius AG, Sagent Pharmaceuticals Inc., and Altan Pharma Ltd all implemented minor changes to the invention disclosed by UCB Pharma and achieved production of the API with similar purity without violating UCB Pharma's patent (IMSHealth, 2015). Conversely, an example of a low‐novelty but broad‐scope patent can be found with Bevacizumab, a drug mainly used to treat colorectal and lung cancers (Vidal, 2013). F. Hoffmann–La Roche developed a new method for increasing the yield of antibodies in cell culture, which consists in substituting specific residues during the manufacturing process. The low novelty of the invention is explained by the existence, in prior art, of many techniques for producing antibodies using a variety of host cells. The high scope is justified because the invention encompasses a wide range of potential residues' substitutions and describes comprehensively both the residues that could be substituted and the substitutes that could be used.
Related and unrelated manufacturing experience
As described in Section 2.1, a firm's past manufacturing experience is a key determinant of its ability to innovate. However, as noted at the end of that section, different types of experience could have different effects. In particular, the extent to which past experience readily applies to the process at stake appears as an important differentiator.
Related manufacturing experience
As firms accumulate manufacturing experience, they build core skills that are more likely to be applicable to innovation activities that take place in technological landscapes of related—rather than unrelated—products (Cohen & Levinthal, 1990). This is especially the case in the pharmaceutical industry given the complexity and specificity of the underlying science. In this study, we treat as “related” the APIs that belong to the same subclass of anticancer APIs. Related APIs have similar molecular structures and, consequently, can be produced through similar production steps using similar manufacturing technologies (Anderson, 2012). For example, Doxorubicin and Epirubicin—two APIs present in our sample—both belong to the same subclass and can be produced through similar production steps (i.e., halogenation, hydrolysis, chromatographic purification, and crystallization) and may even share production equipment. However, their production will differ in the starting materials, the execution of each production step (e.g., volume and concentration, type of solvents), and the operating conditions.
This suggests that the landscapes of related APIs are populated by configurations that arise from decisions along closely related—or even identical—attributes that interact in similar ways across landscapes. Because specific operating decisions will still differ across related APIs, accumulation of experience manufacturing them, that is, related experience, will contribute to a firm's deeper understanding of how the associated technological attributes interact over wider decision ranges (Malerba, 1992) and such understanding will apply and transfer across landscapes. Consequently, related experience can help the firm better understand the corresponding regions of the focal API's technological landscape and enable it not only to identify new technological configurations that are valuable for the focal API's production process (Nerkar & Roberts, 2004) but also to patent them in ways that are relatively general, that is, of relatively broad scope. These will then be the elements of a patent portfolio that protects a wider area of the focal API's technological landscape from competition (Leonard‐Barton, 1988).
Nevertheless, these new technological configurations, identified because of the availability of related know‐how and know‐why, will typically be of an incrementally, rather than of a radically, different nature (Malerba, 1992; Solheim et al., 2018) and will mostly correspond to regions of the focal API's technological landscape that are adjacent to the ones the firm already operates in. This is a consequence of the fact that production of related products tends to happen via similar process configurations within a given firm (Aharonson & Schilling, 2016), such that transfers of know‐how and know‐why across related products are unlikely to result in radically different process configurations for the focal API.
Moreover, as firms become more experienced in the manufacturing of related products, and therefore more capable at activities similar to the ones in which they already engage, accumulation of related experience does not contribute to the knowledge diversity that is critical to creating something substantially novel. Indeed, accumulation of related manufacturing experience can permit the development of incremental solutions that improve efficiency and productivity but can also contribute to a firm's rigidity in dealing with novel situations and become a source of inertia or “competency trap” (Levitt & March, 1988; Yeoh & Roth, 1999). Expertise and routines combined with organizational biases tend to endorse local rather than global search and the firm may have difficulties interpreting alternatives, their consequences, and their potential influence on problem solving when performing more distant searches (Afuah & Tucci, 2012). For example, Knudsen and Levinthal (2007) point out that screening ability may depend on experience when stating that “actors may become quite skillful and accurate in evaluating one class of alternatives, but rather inaccurate in evaluating a different and for them novel set of alternatives” (p. 52). Moreover, related experience may induce an organization to become inflexible such that, over time, technological strategies that once ascertained firm's superiority can even turn into impediments—for instance, to preserve the existing state of affairs, management may try to conceal difficulties related to the traditional approaches (Katila & Ahuja, 2002).
It is therefore unlikely that accumulation of related experience will push a firm's inventive activities toward more distant regions of the focal API's landscape. Instead, such activities will remain concentrated to the regions of the landscape that the firm understands well. This is also reflected in the organizational learning literature (e.g., Gupta et al., 2007), which explains that prior experience can constrain creative thinking as it can lead to drawing on familiar processes and heuristics when solving a problem. Exploring more distant regions of the technological landscape would require the consideration of more numerous alternatives and, hence, more numerous experiments with uncertain results given the large number of unfamiliar technological configurations (Afuah & Tucci, 2012). Such exploration, when combined with the need for an eventual integration to production, requires time, resources, and effort that the firm may be unwilling or unable to invest (Katila & Ahuja, 2002). Relatedly, the fear of failure, together with mental and social fatigue, may function as additional impediments as inventors become “locked into established thought patterns” (Fleming, 2001, p. 1025).
This is echoed by the senior vice president of R&D for an API manufacturer who we interviewed for this study. He explained to us that the R&D team at his firm is populated with scientists (typically chemists), each of whom specializes in a set of related APIs among the firm's product portfolio. Such specialization is necessary in order to achieve the needed expertise in understanding the chemistry and other specificities of associated molecular structures and production processes. Inventive activities around a focal API are guided by the corresponding R&D specialists and the latter are typically inclined to first search for solutions within the more easily accessible related knowledge base. Moreover, such activities are typically carried out under tight time and budget constraints that arise from the intense competition that characterizes the generics industry. Thus, accumulation of related experience will limit the likelihood of searching for solutions beyond the related know‐how and know‐why.
The above discussion implies that, as a firm accumulates related manufacturing experience, it will develop a deeper understanding of parts of the focal API's technological landscape that will permit the development of inventions of broader scope. However, it will also contribute to inertia that will constrain the firm's inventive activity to more familiar regions of the landscape, thereby hurting the inventions' associated novelty. We thus state the following two hypotheses:
The magnitude of a firm's related manufacturing experience is negatively associated with the novelty of process inventions for a focal product. The magnitude of a firm's related manufacturing experience is positively associated with the scope of process inventions for a focal product.
Unrelated manufacturing experience
Unrelated products are produced through manufacturing processes characterized by different production technologies. In our setting, we define APIs to be unrelated when they belong to different subclasses of anticancer APIs. As such, unrelated APIs have different molecular structures, which necessitate different production approaches with very limited “functional overlap” (Cornelius et al., 2020). Typically production processes for unrelated APIs consist of different steps that are also of highly different nature in terms of equipment and operating conditions owing to the differences in the underlying chemistry. As an example, consider two unrelated APIs from our sample: Everolimus and Doxorubicin. While production of Everolimus—and of other APIs in the same subclass—is characterized by the use of an enzymatic process (Guengerich et al., 2004), production of Doxorubicin—and of other APIs in the same subclass—relies on semisynthetic molecules, which require radically different manufacturing technologies (Madduri et al., 1998). Despite these differences, experience with producing Doxorubicin through a one‐pot process was key in inspiring and informing the development of a new, much shortened, process for Everolimus that bypassed separate preparation of an intermediate product. 3
This example suggests that the presence of manufacturing experience with unrelated (to the focal) APIs, which are produced through highly distinct manufacturing processes, can lead the firm to conduct a broader search that includes more distant regions of the focal API's technological landscape and identify production methods that might have otherwise remained unexploited. This is possible because the presence of unrelated know‐how and know‐why can inspire firms to consider combining technological components in the focal API's landscape in radically different ways (Fleming & Sorenson, 2004). This is further supported by Sting et al. (2020), who argue that simultaneous input from multiple parties with different expertise or knowledge levels in joint problem solving leads to broader searches and enables a higher degree of creativity. Moreover, it may “unlock new component combinations” (Albert & Siggelkow, 2021, p.15). Thus, the experience that has been accumulated from producing unrelated APIs (e.g., developing and using special equipment designs, executing various production techniques) can inspire the firm to approach the manufacturing process of the focal API more creatively and, “by systematically broadening the search,” lead to the development and patenting of more novel manufacturing inventions that would not have been possible by only relying on available related manufacturing experience (Baumann et al., 2019, p. 289).
Yet, because application of this unrelated experience to the production of the focal API is not as straightforward as the application of related experience, the resulting invention is likely to be more limited in scope. Indeed, knowledge gained from unrelated manufacturing experience is likely to push the firm outside its typical operating practices along one or more attributes of the focal API's production. The closely interconnected nature of the different attributes of pharmaceutical production suggests that such deviation would place the firm in an unfamiliar area of the technological landscape (Novelli, 2015). Hence, the development of a solution that is general enough to merit a patent of broad scope requires additional exploratory effort (e.g., through experimentation) in a not‐well‐understood region of the focal API's landscape. This is reflected in Sting et al. (2020), who explain that joint problem solving based on different types of expertise or knowledge is more time consuming because of the broader nature of the associated search. Increased experimentation is needed to achieve more extensive familiarity with feasible alternatives (Leonard‐Barton, 1988), and a deeper engagement with a wider range of associated production technologies and technological configurations (Mihm et al., 2015). Similarly, Albert and Siggelkow (2021) argue that finding large sets of interaction changes that improve performance does become increasingly difficult. Hence, the resources necessary to carry out such exploratory activities are less likely to be invested due to financial and time constraints. Moreover, cognitive constraints to consider “all the components and their potential relationships simultaneously” (e.g., within R&D) can limit the extent to which the firm can even recognize broader parallels between unrelated knowledge and the manufacturing of the focal API (Fleming, 2001, p.1025; Fong Boh & Slaughter, 2007).
The chief scientific officer and head of R&D at a vertically integrated pharmaceutical company that we interviewed explains that:
The above discussion suggests the following two hypotheses:
The magnitude of a firm's unrelated manufacturing experience is positively associated with the novelty of process inventions for a focal product. The magnitude of a firm's unrelated manufacturing experience is negatively associated with the scope of process inventions for a focal product.
DATA SET AND VARIABLES
Data set
To test our hypotheses we construct a unique data set using both primary and secondary data sources. We focus on antineoplastic (or anticancer) APIs, that is, APIs identified as belonging to the class L1 of the European Pharmaceutical Marketing Research Association's (EphMRA) drug classification system (EphMRA, 2015). Our unit of analysis is the API–firm–quarter observation. Our initial sample consists of all 208 antineoplastic APIs that existed between January 2005 and December 2015. Because of a 1‐year lag between the independent experience variables and the dependent innovation variables in our main analyses, the measurement window for our independent variables extends from January 2005 to December 2014 while our dependent variables are evaluated between January 2006 and December 2015.
To measure our dependent variables of novelty and scope of process innovation, we focus on U.S. manufacturing process patents to control for differences among country‐level regulations that govern competition and innovation. The U.S. market represents 33.7% of global generics' sales (Source: Marketline Industry Profile). Of the 208 APIs initially considered, 148 APIs lost product‐patent protection either before or during our study's timeframe. Among these 148 APIs, 80 APIs were sold in the United States by at least one firm, out of which 30 had at least one new process patent issued and sales by the patent‐holder during our timeframe. Thus, the sample for our dependent variables' measures (herein referred to as the “focal” sample) includes 30 APIs (our “focal” APIs) produced by 24 firms (our “focal” firms). This results in a data set, which contains 223 API–firm–quarter observations.
To measure our independent variables of related and unrelated experience, which are API–firm‐level characteristics, we expand our sample to account for those 24 focal firms' global production across all APIs included in our initial sample (i.e., 208 APIs). In particular, we record quarterly API sales across the United States and three major European countries, 4 namely, France (9.8% of the European generics market), Spain (6.5% of the European generics market), and Italy (2.5% of the European generics market). We choose these specific markets because while the United States is the largest pharmaceutical market worldwide, France, Italy, and Spain are all within the top five European pharmaceutical markets and among the most important markets globally for the sale of anticancer drugs specifically (Aitken et al., 2018). 5 Among the 208 APIs contained in our initial sample, 73 APIs—including our focal 30 APIs—are sold in at least one country by at least one of our 24 focal firms within our timeframe. We provide a list of these 73 APIs with the 30 focal APIs in bold in Table A.1 of Supporting Information A.
Finally, the measurement of three of the control variables, that is, market concentration, market growth, and innovation intensity, also requires us to expand our focal sample in order to assess these API‐level characteristics. In particular, for market concentration and market growth, we consider all firms that sold any of our focal APIs in the United States during our study's timeframe. The resulting sample includes 58 firms, that is, 34 firms in addition to the 24 focal firms. For innovation intensity, we narrow this 58‐firm sample to the 28 firms that owned any patent related to the sold API. We note that these patents do not necessarily qualify as manufacturing process patents, in which case, they were not considered in building our focal sample.
Variable definitions and sources
Table 1 provides summary statistics for all variables presented in this section. Pairwise correlations are reported in Table 2.
Summary statistics
Pairwise correlations
Performance: Firm's innovative output
We measure pharmaceutical firms' innovative output for the APIs in our sample by observing their respective U.S. patent portfolios. We rely on the Thomson Reuters Newport database, which is recognized in the pharmaceutical industry as the reference database for patents and other intellectual‐property information (Grimaldi et al., 2015). Patents are frequently used in the operations management (e.g., Chan et al., 2018) and organizational learning (e.g., Jain, 2013) literatures as a valid indicator of a firm's innovative output (Griliches, 1990). For any given API, since the end product is essentially identical across firms (Babar, 2019), all patents contained in our sample introduce innovative manufacturing methods, and are thus referred to as “process patents” (Lieberman, 1987). As an illustrative example, consider Finasteride—a prostate cancer prevention drug. In Q1 of 2007, Aurbindo Pharma Ltd. developed and patented a manufacturing method that avoided the use of expensive and toxic reagents that required production under extreme reaction conditions. Additionally, the introduction of novel intermediates, which could be easily purified to permit obtaining a purer API, resulted in an overall higher yield of production compared to the prior art. This process innovation enabled Aurbindo to achieve a significant reduction in its manufacturing costs for Finasteride and observe an increase of its U.S. market share from 3% in 2008 to 26% in 2014, thereby becoming the market leader.
As we describe at the beginning of Section 4, at any given quarter, we associate the accumulated levels of related and unrelated experience with innovation output (i.e., newly issued patents) during the following year (i.e., four quarters). We do so to avoid spurious correlations between our dependent and independent variables and buttress causality interpretations. For each portfolio of patents issued during the four postexperience quarters, we evaluate two distinct dimensions, namely, novelty and scope, at the API–firm–quarter level. The variable 
Specifically, we followed a two‐step process to obtain portfolio‐level 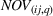
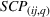
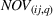
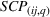
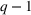
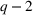
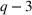
We note that the literature also includes other measures of novelty, such as classes to which a patent has been assigned (Fleming, 2001; Strumsky et al., 2012) and backward citations (Schoenmakers & Duysters, 2010; Verhoeven et al., 2016). Researchers have also measured scope using the number of technological classes in which a patent is classified (e.g., Fischer & Leidinger, 2014; Lerner, 1994) and number of claims included in a patent (e.g., Funk & Owen‐Smith, 2016; Lanjouw & Schankerman, 1997). However, such approaches have significant drawbacks. For example, a key problem with using citations to measure patent novelty is how to account for the timing of different patent grants and how these relate with a study's timeline. One of the most popular approaches, which suggests truncating using a 5‐year time span (e.g. Fischer & Leidinger, 2014; Huenteler et al., 2016), is hardly applicable to our 10‐year data set. Moreover, the references associated with a patent are subject to strategic decisions, including possible manipulation (Gerken & Moehrle, 2012), and the citation inflation problem (Hall et al., 2001). More generally, the problem of bibliographic information is that it ignores the description section of patents (Reitzig, 2004). During our interviews, pharmaceutical industry experts acknowledged that the number of claims in a patent and the number of classes are far less important than the wording of the claims that describe physical limitations (Haeussler et al., 2014). Finally, we note that these measures refer to individual patents. To the best of our knowledge, there is no published work that establishes how such individual patent measures could be combined at the multipatent portfolio level (Fischer & Leidinger, 2014).
Predictors: Firms' manufacturing experience
Our experience measures are based on quarterly sales data for the APIs in our sample from the Intercontinental Medical Statistics (IMS) Health's MIDAS pharmaceutical sales proprietary database. This database tracks quarterly sales for almost every pharmaceutical product sold by every pharmaceutical firm worldwide and is considered to be the most reliable source of sales information in the pharmaceutical industry (Kanavos, 2014). Given that several final dosage forms may exist for the same API, we measure sales for a focal API as the total number of kilograms of the focal API sold by a firm in a quarter (Caves et al., 1991). In our 10‐year study horizon, we observe sales from 24 distinct corporate groups 7 —hereafter referred to as “firms.” In Table A.2 of Supporting Information A we provide a list of the 24 firms appearing in our sample.
In line with the organizational learning literature (e.g., King & Tucci, 2002), we measure a firm's accumulated manufacturing experience for a focal API in a given quarter using adjusted cumulative sales. Specifically, let 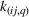
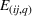
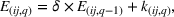
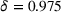
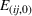
To distinguish between related and unrelated experience, we rely on the EphMRA classification. Within the broad L1 class of anticancer drugs, nine subclasses exist of which eight are present in our sample (specifically, the L1AB, L1C, L1D, L1F, L1G, L1H, L1X1, and L1X9 subclasses). In Table A.3 of Supporting Information A, we present how the 73 APIs that we use to construct our experience measures are grouped across the nine subclasses of L1 APIs.
Let 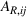
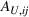
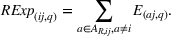
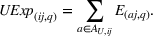
Control variables
Market concentration
Past research has found that market concentration, a measure of a market's competitiveness, influences firms' innovation efforts. For example, Schumpeter (1942) argues that higher market concentration may increase firms' innovation efforts as higher market power might translate into higher profits that can be invested in R&D, whereas Arrow (1972) asserts that higher market concentration eliminates or diminishes competition and may reduce the urgency to innovate. In the pharmaceutical industry, Matraves (1999) finds a positive relationship between market concentration and innovation. In line with previous research (Rego et al., 2013), we measure market concentration for API 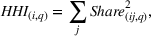
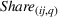
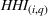
Market growth
Past literature (e.g., Cohen et al., 1987) suggests that a higher market growth rate implies higher product demand, which supports firms' innovative efforts. We measure the growth of the market for API 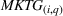
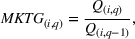
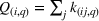
Innovation intensity
To control for heterogeneous opportunities for innovation among APIs, we consider the overall level of innovation intensity for API 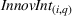
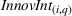
R&D expenses
The magnitude of a firm's R&D expenses has been found to be associated with the quality and quantity of a firm's innovation output (Bhaskaran & Ramachandran, 2011). Thus, we log‐transform R&D expenses for firm 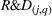
Focal experience
We control for firm's direct experience with manufacturing the focal product. Similar to our measures of related and unrelated experience, we define the experience “focal” to API 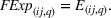
Initial familiarity
To control for the familiarity that firm 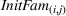
Innovation strategy
Corporate strategy could potentially influence both production volumes and innovation outcomes (Custódio et al., 2017). For instance, firms that make a given L1 subclass a strategic priority, would see increased related production volumes as well as additional R&D efforts into developing patents with wide scope in that subclass. Thus, we introduce 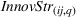
Pre‐experience novelty and scope
We control for novelty and scope of portfolios of process patents that were previously granted to the focal firm for the focal API and remain active during a given quarter. We do so because past levels of a firm's inventions' novelty and scope may influence their future levels, that is, our dependent variables 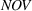
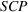
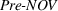
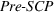
These two control variables, similar to our dependent variables, also rely on expert patent evaluations and are constructed using a two‐step process similar to the one we described in Section 3.2.1 after the evaluation of 173 additional patents. Given the long timeframe considered, we allow here for the possibility that patented inventions may progressively lose some of their relative novelty or scope due to increased competition (Mansfield, 1985). For example, the scope of a patent relative to the state of the art may drop over time because imitation may lead competitors to research and adopt manufacturing practices that are “similar” to the focal firm's, that is, closer to the focal firm's position on the technological landscape (Aharonson & Schilling, 2016). Similarly, the novelty of a patent relative to the state of the art may drop over time as new ideas partially or wholly supersede existing practices (de Rassenfosse & Jaffe, 2018), understanding of the technological landscape's structure improves, and/or the technological landscape itself is reshaped by technological progress. For these reasons, we depreciate the patent experts' initial patent evaluations over time.
Since (to the best of our knowledge) no existing literature considers how individual patent novelty and scope may depreciate over time, we rely on the literature on depreciation of R&D expenditures and intellectual property assets to obtain an appropriate formula—patent novelty and scope are key determinants of patent value (Novelli, 2015; Reitzig, 2003). The central idea in that stream of literature is that the value of intangible intellectual property assets (such as patents) depreciates over time due to the decay of appropriable revenues that these assets generate because of imitation from other firms and technological progress (Khorasani, 2019; Pakes & Schankerman, 1984). Specifically, we follow standard practice in the literature (e.g., Bessen, 2008; Nordhaus, 1967) and apply the following exponential decay formula: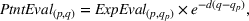
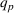
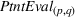
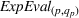
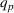
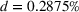
We obtain the portfolio‐level measures of 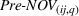
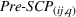
ANALYSES AND RESULTS
Model specification
To test the association between a firm's related and unrelated experience and the qualitative characteristics of its manufacturing inventions, we employ an ordinary least squares (OLS) approach. In addition to the controls discussed earlier, we control for unobserved invariant characteristics through subclass (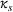
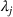
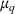
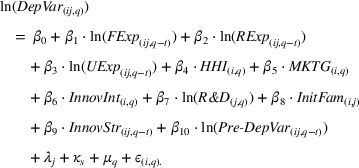
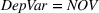
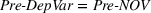
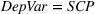
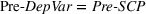
To account for potential simultaneity bias and buttress causality interpretations, we lag our experience measures by 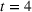
In Models 1, 2, and 3 of Table 3 (Models 4, 5, and 6) we present our results for H1a and H2a (H1b and H2b). Specifically, we build toward our main models, that is, Models 3 and 6, as follows: First, we present Models 1 and 4, which only include 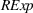
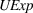
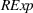
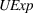
H1a, H1b, H2a, and H2b estimation results


Related manufacturing experience
H1a posits that the magnitude of a firm's related experience is negatively associated with the novelty of its portfolio of patented manufacturing inventions for a focal product. The coefficient for 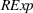
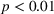
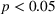
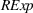
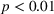
Unrelated manufacturing experience
H2a states that the magnitude of a firm's unrelated experience is positively associated with the novelty of its portfolio of patented manufacturing inventions for a focal product. The coefficient for 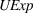
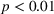
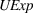
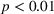
Robustness checks
We now assess the robustness of the results for Models 3 and 6 (i.e., full models for H1 and H2 hypotheses sets) through several checks.
Endogeneity
Unobserved time‐varying factors
To more precisely account for unobserved time‐varying factors, we introduce half‐year fixed effects in Models 3 and 6 (Reagans et al., 2005)—in our baseline analysis, we included full‐year fixed effects. Overall, our results remain consistent (cf. Table C.1 in Supporting Information C).
In‐house versus outsourced production
Some of the production that we attribute to the firms in our sample might be outsourced to third‐party manufacturers (Gray et al., 2015). As such, the experience is not generated in‐house, which suggests that, with respect to a firm's learning, one unit of outsourced production may not be as valuable as one unit of in‐house production (Sting & Loch, 2016). However, we also note that given the stringent regulations that govern pharmaceutical manufacturing, third‐party manufacturers are in very close collaboration with the outsourcing firm regarding process design and execution, how to handle production issues, and so forth, and typically have limited freedom in terms of executing changes on their own (Bruccoleri et al., 2019). As such, any learning opportunity that arises in the environment of a third‐party manufacturer is likely to also benefit the outsourcing firm. Nevertheless, to account for the potentially different learning benefits from in‐house versus outsourced production, we discount outsourced production quantities. Thus, we change Equation (1) as follows: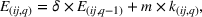
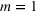
Firm size
The literature suggests that firm size is a key determinant of innovation output. This is explained by the fact that larger firms tend to have more R&D spending (Cohen, 2010), which is supported by the high correlation between firm size and R&D expenses in our data set (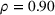
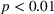
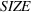
Alternative model specifications
Nonlinear effects of experience
Related and unrelated manufacturing experience could have some nonmonotonic effects on the novelty and scope of manufacturing process inventions. For example, while too much related experience might result in a competency trap, some related experience could be beneficial for novelty. Similarly, there could be an optimal level of unrelated experience, below or above which the benefits of having a diverse knowledge base would either be insufficient or be outweighed by the costs of diverting an organization's cognitive and financial resources. Thus, we include quadratic terms for 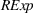
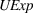
Specifically, the squared term for 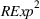
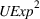
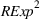
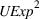
Sensitivity to choice of model parameters
Quarterly experience retention rate
We consider alternative values for the quarterly experience retention rate δ (cf. Equation (1)). Specifically, we set δ to 1.000, 0.987, 0.960, and 0.946, which (respectively) correspond to annual experience depreciation rates of 0%, 5%, 15%, and 20% (our baseline value is 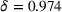
Lag of experience measures
We re‐estimate Models 3 and 6 by setting 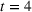
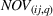
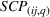
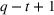
Duration of experience measures' calibration period
While in our main analysis we rely on the four initial quarters of our time window to calibrate our experience measures, we also tested Models 3 and 6 by initializing our experience measures using two, six, and eight quarters and found that results continue to hold (cf. Table C.8 in Supporting Information C).
DISCUSSION AND CONCLUDING REMARKS
Our research suggests that related and unrelated manufacturing experience have contrasting relationships with the novelty and scope of a firm's process innovative output. Specifically, we find that, on the one hand, experience with production of related products is associated with a decrease in the novelty and an increase in the scope of the manufacturing methods that a pharmaceutical firm develops and patents for a focal product. Hence, while the decrease in novelty points toward a risk of falling into a competency trap, the increase in scope suggests that the accumulation of related experience enables firms to better understand the structure of a focal product's technological landscape and develop a broad set of applicable innovative production methods. On the other hand, experience with manufacturing unrelated products is associated with an increase in a focal product's patents' novelty and a decrease in its patents' scope. Thus, our study suggests that engagement with a diverse set of products and, as a result, various manufacturing technologies, permits firms to identify and patent more novel methods that apply to the production of a focal product. However, it also highlights that the strain on firms' cognitive and financial resources that comes with engaging in the manufacturing of a diverse set of products, could prevent firms from having a well‐secured position on the focal product's technological landscape.
Our study contributes to the operations, innovation, and organizational learning literatures in the following ways. First, by focusing on products whose patent protection has expired, we are able to study exclusively manufacturing process innovation—an important competitive dimension that has been understudied relative to product innovation. Second, by considering multiple firms and multiple products produced by these firms, and by distinguishing between process innovation novelty and scope—a feature made possible by our collaboration with expert patent attorneys—we are able to observe how different types of experience affect different qualitative characteristics of process innovation. As such, we are able to contribute to the OM literature that has mostly examined how manufacturing experience accumulation relates to patenting activity. Third, because of our multiproduct setting, we are also able to answer calls in the organizational learning literature for a deeper exploration of the experience–creativity link in multiproduct environments. Collectively, our research allows us to reconcile several seemingly contradicting conceptual arguments by showing that manufacturing experience may benefit one dimension of process innovation while having a negative impact on another.
From a practical standpoint, our findings underline how a firm's past product decisions—that is, emphasis on a more diverse versus more concentrated product lineup—might influence its current innovation trajectory—that is, the novelty and scope of manufacturing inventions for a focal product. However, and perhaps more importantly, by highlighting this influence, our study also provides insights into how manufacturing knowledge accumulated through production could be proactively managed in alignment with an organization's strategic goals.
For the senior vice president of R&D of a generics‐focused manufacturer with whom we communicated in the context of this project, a key priority is developing and patenting inventions that are as broad in scope as possible, whereas novelty is secondary in importance. This choice was justified by this manufacturer's competitive strategy, which emphasizes growing its existing position in order to limit competition rather than venturing into more novel, but also more risky, endeavors. In the context of API manufacturing, this then translates into promoting the identification and patenting of a broad number of different routes of synthesis through which a given API might be produced, and this even if all of these routes may not necessarily be implemented in practice. This would be important from a competitive standpoint because it would prevent competitors from taking advantage of production methods that are proximal to these of the innovating firm and, presumably, of comparable efficiency. Our analysis suggests that for firms with such a strategic objective, a key priority should be to emphasize related‐knowledge retention practices. This could be achieved through formal organizational mechanisms (Anand et al., 2012) such as a heavy reliance on standard operating procedures and documentation of production and R&D activities (Argote & Miron‐Spektor, 2011), purposely organizing the workforce to promote team continuity (Sting et al., 2020) and facilitate transfer of knowledge across related product lines (Cornelius et al., 2020), and using dedicated production equipment for related products to minimize forgetting due to changeovers (Egelman et al., 2017).
Conversely, firms might favor novelty over scope. Innovations of higher novelty typically have a higher long‐term economic value and the potential to more profoundly impact the competitive environment by disrupting existing competencies or even eliminating existing players from the market (Bessen, 2009). Teva pharmaceuticals, a large generics manufacturer with a diverse product portfolio, provides a relevant example. Teva developed and patented a very novel process for producing Letrozole, a drug used in the treatment of breast cancer, that substantially increased yield relative to the state of the art at the time (Vogel, 1995). While implementation required Teva to build a new manufacturing facility, the new process allowed Teva to achieve a level of productivity that permitted it to double its market share and eliminate major competitors such as Bristol‐Myers Squibb and Hikma Pharmaceuticals from the U.S. market (IMSHealth, 2015). Our results imply that when a firm's strategic priorities involve developing a portfolio of novel manufacturing inventions, investing on specific training to deal with product variety (Ramdas et al., 2017) and emphasizing the development of transactive memory systems, where members possess meta‐knowledge of who knows and does what (Argote & Miron‐Spektor, 2011) is likely to be invaluable. In line with this emphasis, informal networks should be encouraged, as they have been shown to support collaborative problem solving through the exchange of tacit knowledge (Soda & Zaheer, 2012).
These findings are likely to carry implications for other science‐based, capital‐intensive, and high‐technology industries (e.g., semiconductors or chemicals). Indeed, the development of innovative manufacturing methods requires not only pharmaceutical firms, but also other firms in science‐based, high‐technology industries, to “couple the worlds of leading‐edge science with the realities of plant operations” (Pisano, 1997, p. 21). As such, innovating manufacturing processes in these settings is both technically and organizationally complex as technical uncertainties associated with process choice must be dealt simultaneously with scaling challenges associated with product commercialization. Yet, for mature product markets such as the ones studied in this paper, process innovation appears as the main remaining opportunity to increase profits (Utterback & Abernathy, 1975).
As is inevitable with studies of this kind, our work has some limitations. First, not all patentable inventions are patented (Griliches, 1990). Nevertheless, the pharmaceutical industry is characterized by a very high propensity to patent. Pharmaceutical firms generally prefer patenting relative to other forms of intellectual property protection (e.g., trade secrecy) and this results in more than 80% of patentable inventions being patented (Mansfield, 1986). This is particularly true for the case of new manufacturing methods, which is the form of inventions that we study. Infringement of a corresponding patent may be identified from the “chemical fingerprint” that a manufacturing process leaves on the final product (Deconinck et al., 2008) or through on‐site inspections of suspected infringing competitors (Blakeney, 2005). Moreover, as a pharmaceutical firm must disclose in detail the characteristics of its manufacturing process when applying to public health authorities for a legal license to sell a product (Cartwright, 2016), such details could potentially be obtained by competitors through future court orders. Also, protection via trade secrecy is inherently risky because competition may succeed in independently discovering a firm's invention, reverse engineer it, or hire away from the inventing firm key personnel with the necessary technical knowledge (Arundel & Kabla, 1998). Second, our study is focused on the pharmaceutical industry and more specifically on the production of anticancer APIs that are no longer patent‐protected. Future studies that test our findings in other settings would be especially valuable. Third, additional insight into firms' learning could be obtained from the analysis of data that provide visibility into which facilities manufacture which products (Egelman et al., 2017) or from the analysis of even larger data sets that could permit considering alternative outcome measures (e.g., individual patent issuances). Finally, future research may compare our measures of novelty and scope to existing measures of these constructs after first adapting them to the portfolio level.
Footnotes
ACKNOWLEDGMENTS
The authors would like to thank Prof. Glen Schmidt, the senior editor, and three referees for their very helpful and insightful comments from which the paper greatly benefitted. The first two authors gratefully acknowledge the financial support from the HEC Paris Foundation and Labex Ecodec: Investissements d'Avenir (ANR‐11‐IDEX‐0003/Labex Ecodec/ANR‐11‐LABX‐0047).
1
Prior art includes all publicly available knowledge at the date of the patent filing (35 U.S. Code, §102).
2
Additional details on this and other private communications with practitioners from the pharmaceutical industry that we have initiated in the context of this study can be provided upon request.
3
The API manufacturer that communicated to us this example asked us not to publicly disclose its name.
4
The European generics market accounts for 20.1% of the global generics market (Source: Marketline Industry Profile).
5
We could not use data from the other two top European countries (i.e., Germany and the United Kingdom) because pharmaceutical firms' names may not legally be disclosed for sales in the United Kingdom, and Germany has a very fragmented market for pharmaceuticals, owing to its federal structure and complex procurement systems.
6
Details about all three patent experts are available from the authors upon request.
7
In the Newport database, a corporate group is the parent company to which the marketer (i.e., the company responsible for sales of a drug product in a specific market) and the patent holder (i.e., the company that holds the rights granted by a patent) belong.
8
A quarterly retention rate of 0.975 implies an annual retention rate of 0.9754 = 0.9 or, equivalently, an annual depreciation rate of 0.1.
9
While
10
All results, which are discussed but not presented in the paper, are available in Supporting Information.