Abstract
The COVID‐19 pandemic presented the world to a novel class of problems highlighting distinctive features that rendered standard academic research and participatory processes less effective in properly informing public health interventions in a timely way. The urgency and rapidity of the emergency required tight integration of novel and high‐quality simulation modeling with public health policy implementation. By introducing flexibility and agility into standard participatory processes, we aligned the modeling effort with the imposed reality of the emergency to rapidly develop a regional system dynamics (SD) model integrating diverse streams of data that could reliably inform both health system restructuring and public health policy. Using Lombardy data, our SD model was able to generate early projections for the diffusion of the pandemic in neighbor Ticino. Later, it projected the timing and size of peak patient demand. Our work also supported the need for reorganization of the healthcare system and volume flexibility strategies increasing hospital capacity (e.g., intensive care unit [ICU] and ward beds, medical and nursing staff, and oxygen supply) in Ticino. Counterfactual analyses quantify the impact of the decisions supported by our interventions. Our research contributes to our understanding of volume flexibility strategies used by healthcare organizations during emergencies, highlighting the critical role played by available response time in the deployment of strategies that either prioritize critical services or leverage available resources. It also contributes to the literature on participatory systems modeling by describing a flexible and agile participatory process that was successfully deployed in a rapidly evolving high‐stakes emergency.
Keywords
INTRODUCTION
A novel class of problems
Covid19 presented the world to a novel class of problems highlighting distinctive features that rendered more standard academic research and traditional implementation pathways less effective. Characterized by a rapidly evolving situation with high stakes impacting a significant number of lives, attempts to address such problems required very rapid response times. In addition, the presence of strong reinforcing loops driving the exponential growth in the number of severe cases meant that policy officials could not rely solely on qualitative analysis to guide public policy. Furthermore, involvement of multiple stakeholders, availability of different streams of data requiring vetting and integration, and diverse policy levers with unclear and uncertain effectiveness exacerbated intrinsic challenges. These problems require tight integration of novel and high‐quality simulation modeling with policy implementation. However, due to their novelty, rapidity, and high impact, no existing off‐the‐shelf models were available to properly and promptly quantify the likely magnitude of outcomes. This research seeks to inform key aspects of decision‐making during emergencies, to ensure practitioners (e.g., policymakers and health officials) and academics (e.g., operations manager researchers) can better respond and prepare to future emergencies. It focuses on the rapid response of our research team to support public healthcare decisions during the first wave of the COVID‐19 pandemic in Canton Ticino, south of Switzerland.
The emerging problem in Ticino (Feb–Mar 2020)
In mid‐February 2020, health authorities in Ticino watched with alarm the rapid spread of the COVID‐19 disease in Lombardy, the first region in Europe to have been severely affected by COVID‐19. The dramatic evolution of the COVID‐19 pandemic in Lombardy, with rapidly growing number of confirmed cases and severe patients completely overwhelmed Lombardy's hospital capacity. A taskforce in Lombardy quickly identified the need to adjust regional hospital capacity to care for the expected high inflow of patients infected with SARS‐CoV‐2 virus (Grasselli et al., 2020a). Given that a large fraction of COVID‐19 patients develop respiratory failure, requiring mechanical ventilation, it further recognized the urgent need of increasing ICU bed capacity in Lombardy (Grasselli et al., 2020b). Due to its geographic proximity (only 100 km south) and intense socioeconomic relationship with Lombardy, Ticino experiences a daily inflow of over 60,000 Italian workers. Fearing that they soon could experience a similar fate, health officials in Canton Ticino also established a taskforce of key stakeholders (e.g., with representation from the government, police, army, ambulance services, public and private hospitals, and family doctors) to design an emergency plan to meet possible needs of COVID‐19 patients.
Key challenges and concerns
The COVID‐19 taskforce immediately implemented basic emergency actions to support their response, including (i) declaring a state of emergency, (ii) mandating coordinated collaboration between public and private hospitals, (iii) centralizing the supply of essential drugs through the cantonal pharmacy, and (iv) early engagement of the armed forces. Collaborating with the cantonal health authorities and the taskforce, the Ente Ospedaliero Cantonale (EOC), responsible for managing and operating the public hospital network, identified several challenges requiring urgent attention to prepare for the likely spread of the COVID‐19 disease in Ticino. First and foremost, the explicit concern was the ability of the hospital system in Canton Ticino to meet the surge in COVID‐19 patient demand. Second, the dispersed nature of the cantonal hospital network—composed of four public hospitals (all equipped with an intensive care unit [ICU] and four private clinics [only one with an ICU])—posed challenges with respect to the coordination and allocation of medical and nursing staff, and oxygen and personal protective equipment (PPE) supplies. Finally, due to the need of mechanical ventilation, the EOC also recognized the urgent need to increase ICU beds with full ventilation capacity as well as the need to ensure the availability of oxygen supply. In parallel, the cantonal health authorities were concerned about the need to implement a lockdown and other social distancing policies. Confronted with these concerns, health authorities were uncertain about which hospital capacity and social distancing policies to put in place. They also feared that those policies could be insufficient to address the surge in COVID‐19 patient demand and curb the spread of the disease. Moreover, given the devastating effect observed in Lombardy, they knew they could not rely solely on qualitative analysis and simple heuristics to guide regional public health policy. Table 1 summarizes the stakeholders involved and their main concerns and decisions.
Key stakeholders, concerns, and decisions associated with Ticino's COVID‐19 pandemic
Note: Main concerns and key decisions in italics were explored by our research team.
Key stakeholders, decisions, and model prediction tasks
Our research team worked closely with the EOC and the cantonal health authorities to inform decisions requiring immediate answers. With the EOC, we emphasized informing decisions associated with (i) the need to consolidate hospital capacity and the need to increase ward and fully ventilated ICU bed capacity and (ii) hospital operations strategy, estimating the need for additional ward and fully ventilated ICU bed capacity, the associated need for medical and nursing staff, as well as oxygen. We used established insights from healthcare operations management to investigate the soundness of the dedicated COVID‐19 hospital decision. From our projections of patient demand, we estimated the requirements for ICU and ward bed capacity, medical and nursing staff, and oxygen, and evaluated whether the proposed capacity changes would suffice to meet the surge in patient demand. With the cantonal health authorities and the COVID‐19 taskforce, we focused on the need to inform the decision to introduce social distancing measures, developing a system dynamics (SD) model to project the contagion trajectory with and without social distancing measures in place.
As the COVID‐19 pandemic progressed and fatigue of medical and nursing staff increased, health authorities prioritized estimating the timing and size of the peak in patient demand. Insufficient capacity (e.g., ICU and ward bed capacity, medical and nursing staff, and oxygen requirements) to meet patient demand at the peak could translate into limited hospital admissions, inability to treat the sick, and unnecessary deaths. At that time, our modeling shifted to the ability of the hospital system to meet the surge in patient demand. To inform that decision, we projected growth in patient demand and estimated the timing and size of peak patient demand in the first wave of the pandemic. Our model was able to predict within 1 week the timing and size for the peak patient demand in Ticino. After months of lockdown, falling numbers of cases and hospitalizations, and facing increased pressure to reopen the economy, health authorities wondered they could attempt to return to normal by lifting social distancing measures. At the same time, they worried about a possible resurgence. To inform that decision, we projected the contagion trajectory with and without removal of ongoing social distancing measures. We predicted that a second wave of infections was likely to occur if social distancing policies were lifted. Table 2 provides an overview of main concerns, decisions, inherent needs, and the model prediction tasks used to inform those COVID‐19 decisions.
Concerns, decisions, need, and model prediction tasks associated with the COVID‐19 pandemic in Ticino
Note: Nonshaded cells relate to concerns, decisions, need, and model predictions tasks early on; shaded cells relate to exponential growth phase.
Contributions and broader insights
This research contributes to two streams of literature: volume flexibility in healthcare operations and participatory systems modeling. From a healthcare operations perspective, the research contributes to the literature on volume flexibility strategies during emergencies, exploring the effective options available to healthcare providers when (i) demand uncertainty is high, (ii) required level of flexibility is high, and (iii) time to respond is scarce. According to Jack and Raturi (2002), when health organizations face high levels of uncertainty and require high volume flexibility, they should adopt mitigating strategies. Typically, those strategies would include restructuring, risk pooling, outsourcing, and developing strategic alliances. However, the limited time available to respond during emergencies prevents the successful implementation of typical mitigating strategies. Instead, the short response time to achieve high volume flexibility to meet high demand uncertainty during emergencies forces healthcare organizations to deploy strategies that either prioritize critical services or leverage internal resources. For instance, a draconian government mandate to stop all nonessential surgeries was implemented as demand management strategy. While demand management is typically deployed as a shielding strategy to achieve low level of flexibility, it worked as a mitigating strategy yielding high volume flexibility during an emergency.
In addition, a combination of different workforce flexibility strategies (e.g., creation of nurse pools, increasing shift length, and internal sourcing of medical and nursing staff) was used as a mitigating strategy to accommodate the high uncertainty in demand. In contrast, the literature prescribes that individual workforce flexibility strategies can be deployed as a containing strategy to meet low demand uncertainty, when the demand for healthcare services exceeds organizational capacity. This research contributes to Jack and Powers’ (2004) call for future research and further understanding on “linkages associated with the combination of strategies, and their impact on organizational performance” as well as “specific tactics chosen by management in each quadrant of the volume flexible strategies framework.”
From a participatory system modeling perspective, the research contributes to the literature by describing a flexible and agile participatory SD modeling process that was successfully deployed in a rapidly evolving high‐stakes emergency. While participatory systems modeling processes have a well‐documented track record of public policy impact (Andersen et al., 2017; Rouwette et al., 2011; Scott et al., 2016), the urgency and rapidity of the emergency, requiring tight integration of novel and high‐quality simulation modeling with policy implementation, render traditional participatory approaches inadequate to inform public health policy decisions properly and promptly. By introducing flexibility and agility into traditional participatory systems modeling processes, we aligned the modeling effort with the imposed reality of the emergency to rapidly develop a regional SD model integrating diverse streams of data that could reliably inform both health system restructuring and public health policy.
This research also contributes to a long tradition of SD simulation modeling applied to health, healthcare policy, and epidemiological modeling (e.g., Hirsch & Homer, 2017), and more recently to COVID‐19 modeling estimating the disease diffusion and informing health policy decisions (Ghaffarzadegan & Rahmandad, 2020; Rahmandad et al., 2021a, 2021b; Struben, 2020). It combines dynamic modeling and empirical analysis to inform policies in the domain of healthcare operations management (Dai & Tayur, 2019; Denton, 2013; Jha et al., 2016; Keskinocak & Savva, 2020; McLaughlin & Hays, 2008). The SD model captures the complex structure of the epidemiological and socioeconomic system (e.g., stocks and flows, feedback loops, nonlinearities, and delays) (Forrester, 1961; Meadows, 1989; Sterman, 1994). By combining SD modeling with data analytics to inform decision making, we were able to effectively inform the reconfiguration of the public hospital system in Canton Ticino, Switzerland, dramatically improving service efficiency and patient care.
The rest of the article is organized as follows. In Section 2, we provide a brief history and structure of the project, describe general aspects of the participatory modeling process adopted, detail the structure of the SD simulation model, and present the set of structural and behavioral validity tests used to build confidence in our model. In Section 3, we describe modeling outcomes, including (i) early projections (based on data from Lombardy) and (ii) projections for the timing and size of peak patient demand. Section 4 discusses changes to healthcare practice, including requirements for public hospital operations and deployed strategies to reach a high level of volume flexibility and meet the high demand uncertainty. In Section 5, we provide further analyses and counterfactual simulations. We conclude with a discussion of process findings, broader lessons from the research, and implications for the management of future emergencies.
METHODS
This section offers a brief background of the project, describes general aspects of the participatory modeling process, and provides an overview of the model structure.
Project background
The global impact of COVID‐19 galvanized our team to work collaboratively in support of public health efforts against the pandemic. Our collaboration was rapidly and fortuitously forged in the early days of March after exchanges with the research development office at the Università della Svizzera italiana (USI), Lugano, where we are faculty members. All of us are experts in our fields: epidemiology, health economics, healthcare management, and mathematical modeling. In addition to an amazing combination of expertise, our team had substantial discretion policy implementation at the EOC, as well as sizable regional influence. One of us is the Chief Medical Officer (CMO) of the EOC. One of us is an economist specialized in public health policy, member of the board of directors of EOC, and member of the COVID‐19 taskforce. One of us is actively involved in applied epidemiological and public mental health research. One of us is actively involved with public policy modeling and advisory. Together the team possessed complementary skills, research‐oriented fact‐driven perspectives, responsibility for decision making, experience in epidemiology and policy advisory, and pragmatism. As we discussed the possibilities of our collaboration, the need to build a simple SD model to complement ongoing efforts at the EOC to forecast the regional impact of the pandemic became clear. With little time to spare, the forecasting effort emphasized developing a model with just a sufficient level of aggregation to inform ongoing needs. The small size of our group allowed frequent interactions throughout the week. In the first 5 weeks of the pandemic, we met (via Microsoft Teams) regularly (twice a week) to share updated data, model developments and insights, and discuss challenges and policy requirements. Any new government dispositions, research discoveries, complications, or modeling insights would lead to an ad hoc request for a meeting.
Our team members also consulted and exchanged ideas frequently with different support groups invested in the success of the project. Two of the team members had frequent meetings with an advisory group composed of public health experts that were part of the COVID‐19 taskforce. Through those contacts, our research team engaged in “collaborative sessions” to discuss our findings, policy recommendations, and implications, with cantonal health authorities and other key stakeholders. 1 The expert modeler met regularly with a support group of colleagues also modeling COVID‐19 in other countries (e.g., the United Kingdom and the United States), sharing specific insights, research articles, and feedback on the modeling approach. Finally, all our members met informally, but regularly with experts to share the state‐of‐the‐art knowledge, up‐to‐date information on new COVID‐19 research insights. This ongoing exchange informed and enhanced our overall understanding of different aspects of the problem. Table 3 summarizes the organization and roles of our research team and different support groups.
Organization and roles of our research team and different support groups
Because our team was small and included key stakeholders and expert modelers, our frequent interactions facilitated building mutual trust and respect. From this solid foundation, we could openly discuss specific modeling results, their implications for policy decisions, and possible ways to implement them. While our collaboration benefitted from serendipity, it was also informed by participatory systems modeling.
Participatory modeling process
Computational models (e.g., SD, discrete event simulation, and agent based) can support decision‐makers by (i) synthesizing extant knowledge in a systematic and structured way (Addison et al., 2013) and (ii) allowing them to explore the long‐term impact of policy decisions (Sterman, 2006). While nowadays it is more common for decision‐makers to resort to such models to help them develop, implement, and assess public policy (Freebairn et al., 2022; Gilbert et al., 2018; Mabry et al., 2010; Sterman, 2006), their use is still not widespread. Among the barriers to broader use of computational models, Addison et al. (2013) mention that the models (i) can be prohibitively expensive or too time consuming, (ii) do not properly capture the decision makers’ knowledge, (iii) lack the proper data, or (iv) are too complicated to be understood; and that decision makers’ acceptance of model outputs can be compromised due to inadequate communication by the modeler during the model building process. Within SD modeling the barriers above can be addressed with the use of participatory systems modeling (e.g., participatory modeling, group model building, and participatory simulation) (Freebairn et al., 2022).
Participatory systems modeling is frequently used to support decision‐making in dynamic complex settings involving multiple stakeholders (Freebairn et al., 2018; Kopainsky et al., 2017; Osgood, 2017; Rouwette et al., 2016; Vennix, 1999). While participatory systems modeling can use different methods and tools, they typically share common attributes, such as (i) active engagement of decision‐makers in model development and (ii) coproduction of knowledge and collaborative learning (Kopainsky et al., 2017; Rieder et al., 2021). As a result, models resulting from participatory processes tend to be more robust and credible to decision‐makers, who engage with modelers to include realistic concepts and assumptions (Freebairn et al., 2022), and more useful and fit for purpose, since they do a better job capturing the available policy landscape (Gilbert et al., 2018). While the benefits of participatory systems modeling are well documented (Andersen et al., 2017; Rouwette et al., 2011; Scott et al., 2016), adoption suffers due to the need of an expert SD modeler and facilitator (Andersen et al., 1997) and the intense time requirement from stakeholders (Stave et al., 2019).
As the COVID‐19 pandemic took Ticino by surprise, decision‐makers had to make high‐stakes policy decisions that would affect hundreds of thousands of lives. As two of the members of our research team were among those decision‐makers, our focus was on ensuring that the impact from our intervention led to successful decisions, placing more emphasis on the positive impact than on the protocol of the research design. That is, we adopted an action research orientation (Schein, 1987). In SD, group model building (Vennix, 1996) has traditionally adopted an action research orientation, focusing first on the success of the intervention. Anand and Gray (2017) discuss how the action research in operations management (OM) can contribute to strategy and organization literature, and Srai et al. (2021) provide an example applying action research to agricultural supply chains in India.
To ensure our ability to deliver results, our research team adapted the traditional participatory modeling process to mitigate the intense time requirements and the need for a facilitator. To reduce the prohibitive time requirements during an emergency, we kept the compositions of the advisory, modeling, and consultation groups to the absolute minimum. Instead of forming large comprehensive groups spanning all constituencies, we took the opposite approach and formed small groups that could deliver on their specified roles. Because all our meetings were small and informal, we did not find it necessary or appropriate to appoint a formal facilitator. Ad hoc interactions between members of the research team and the advisory, modeling, and consultation groups were distributed across members, who later reported back to our frequent research meetings. That flexible format allowed us to quickly learn and assess knowledge and expertise from different groups. It also avoided long delays until all members of different groups could find common slots of available time. Focusing on flexibility and access, our process design avoided “typical” participatory workshops with all stakeholders. Instead, we engaged in focused and middle sized “collaborative sessions” involving only critical stakeholders. Unlike typical participatory systems modeling workshops where all stakeholders are involved in coproducing all aspects of knowledge, our “collaborative sessions” had much more focused goals. For instance, the “collaborative sessions” focused on (i) eliciting participants’ extensive expertise that could be incorporated into model development, (ii) identifying and prioritizing gaps in the model, (iii) incorporating regionally relevant conditions and policy opportunities, and (iv) discussing ethical issues relevant for public policy implementation. We specifically left out of the “collaborative sessions” the development of a joint problem framing and the appropriate level of aggregation, because we felt the need to progress rapidly and help address ongoing needs during the emergency. We relied on the input and expertise of members of our diverse research team to ensure that we developed a model with an appropriate level of aggregation that properly addressed the problem with an adequate understanding of the policy domain. Still, our process enabled us to achieve a broad agreement on the problem definition and overall public health policy. It ensured active engagement of decision‐makers in the model development and led to better understanding and alignment among the stakeholders involved.
Beyond action research, Oliva (2019) suggests leveraging interventions (that improve a problem situation), using them as a mechanism to develop or test existing theories. By adapting the participatory systems process to the imposed reality of the emergency, we managed to mitigate typical barriers to adoption of participatory processes, leveraging our intervention, and further developing the theory supporting participatory processes.
Model structure
Figure 1 captures the basic components of our SD model. It borrows from the traditional SEIR epidemiological model, with stocks capturing the Susceptible, Exposed (asymptomatic), Infected (symptomatic), and Recovered populations, and flows capturing the advancement of the disease. Reinforcing loops (R1 & R2) capture the contagion effect (from exposed and infected individuals) driving the spread of the disease. Balancing loop (B1) captures the depletion of the susceptible population. At the early stages of the disease, when the susceptible population is still large, the impact of B1 is quite limited. As the stock of susceptible people decreases, the strength of loop B1 increases effectively limiting the spread of the disease.
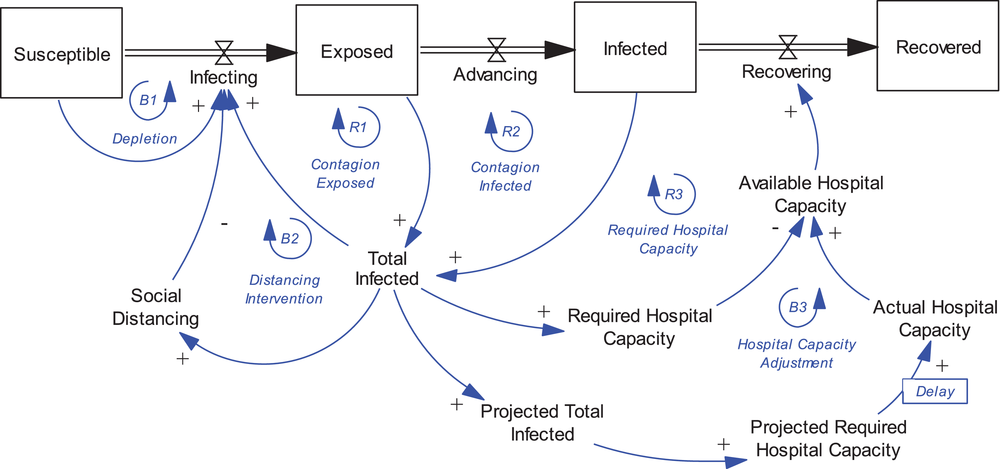
Stock and flow structure of the SEIR model with social distancing and hospital feedback processes
A more effective loop to counter the spread of the disease early on is balancing loop (B2), capturing the impact that social distancing has on limiting the number of contacts leading to new effective infections. As the total number of confirmed cases and deaths rose, the Cantonal authorities introduced very strict social distancing regulations (activating B2). One of the initial concerns in Ticino was to understand whether the available capacity of the system would be sufficient to meet the need. Projecting the pattern of diffusion from the COVID‐19 pandemic data from Italy, the task force projected the likely impact in Ticino, helping them inform the decision to set up a dedicated COVID‐19 hospital and rapidly and significantly increase ICU bed capacity. These decisions are captured in balancing loop (B3) hospital capacity adjustment. Naturally, as the infected developed the symptoms, they were admitted to the hospital and consumed part of the available capacity, closing the reinforcing loop (R3).
Our SD model expands on the traditional SEIR epidemiological model, adapting it as the understanding of the disease improved. It captures major stocks of individuals advancing through the system and the broader aspects of testing and social distancing policies resulting with the spread of COVID‐19. The final SD model distinguishes between Exposed Pre‐symptomatic Infectious (E), Asymptomatic Infectious (A), and Symptomatic Infectious (I). The SD model also explicitly captures testing, which is required to identify positive COVID‐19 individuals. Early in the pandemic, when the number of tests were limited, mainly high risk or severely symptomatic individuals tested for COVID‐19. Of those, only individuals that were both COVID‐19 positive and severely symptomatic could be hospitalized. Infected individuals testing positive but without severe symptoms were sent back home. Infected symptomatic individuals, whose conditions never become severe, eventually recover on their own. Infected asymptomatic individuals also recover spontaneously. Infected individuals (e.g., symptomatic, and asymptomatic) who are never hospitalized continue to have contact with susceptible and continue to contribute to the spread of the virus. As the pandemic evolved and more tests became available, testing became more ubiquitous across all cohorts. Model formulations reflect such conditions.
Given the availability of data regarding ward (W) and ICU (U) hospitalizations, our model could explicitly capture hospitalizations into those components. Typically, individuals admitted to the hospital would first enter the ward. However, some critical patients would be admitted directly into ICU. Patients in the ward with severe symptoms either recover, eventually being discharged, or deteriorate and turn critical. Critically ill individuals would either get admitted to ICU (if space were available), or would remain hospitalized in the ward, eventually recovering or potentially dying. After hospitalizations, patients can either be discharged, after recovering, or die. Those outflows from the hospital system are, respectively, captured in the stock of Cumulative Recovered (R) and Cumulative Deaths (D). Importantly, deaths also occur out‐of‐hospital, most commonly in nursing homes to the elderly.
Figure 2 presents a high‐level view of the expanded SD model, summarized by the acronym Susceptible, Exposed Pre‐symptomatic, Infected Symptomatic, Asymptomatic, Hospitalized Ward, Hospitalized ICU, Recovered, Deaths (SEI(AWU)RD) (full model documentation is available in the Supporting Information). Several articles estimating COVID‐19 diffusion rates adopt expanded versions of the SEIR model accounting for population stocks that include asymptomatic (Arcede et al., 2020; Chen et al., 2020; Rahmandad et al., 2021a), quarantined (Lu et al., 2021), isolated (Nadim et al., 2021), detected/active cases (Rahmandad et al., 2021a; Warne et al., 2020), hospitalized (Betcheva et al., 2020; Favero, 2020; Rahmandad et al., 2021a). We found only one other article that accounts for ward and ICU hospitalizations (Betcheva et al., 2020). 2 Our model also contributes to the Operations and Healthcare Management literature by offering an expanded SEIR model that explicitly tracks patients in hospital's acute ward (W) and ICU.
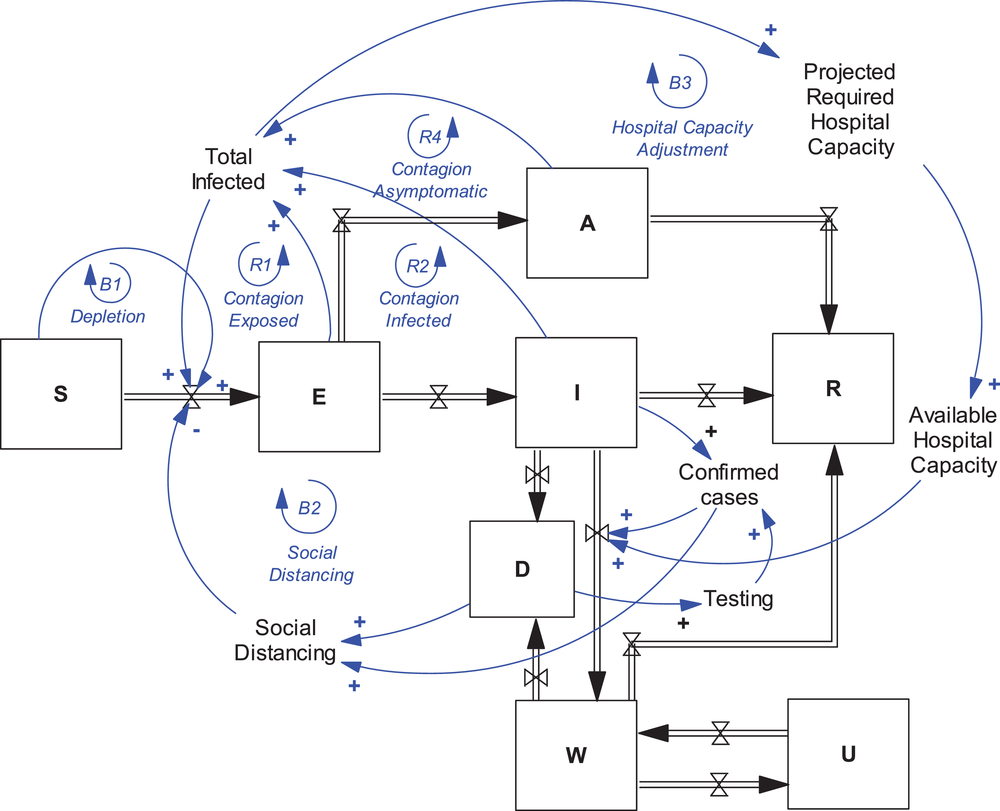
Schematic of the expanded system dynamics SEI(AWU)RD model capturing social distancing, testing, and hospital feedback processes
Finally, we include an endogenous formulation for the implementation of social distancing and lockdown policies deployed to reduce the diffusion of the disease. Infection rate is given by the product of infective contacts and a fraction. The fraction is determined by a logistic function (Ft
), with values ranging from 1 to a minimum value (0 < F
min < Ft
< 1). When no social distancing policies are in place, the logistic function multiplying contacts equals one, leaving contacts unaffected. The logistic function captures the effect of dread on social distancing. Dread associated with the disease increases with cumulative pandemic‐related deaths. As dread increases, the value of the logistic function decreases toward its minimum. Dread changes over time with an asymmetric time constant: It increases quickly with cumulative deaths and decreases slowly with time. The formulation for the logistic function (Equation (1)) follows closely Kapmeier and Gonçalves (2018) and Betcheva et al. (2020):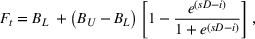
Structural and behavioral validity
To build confidence in our model, we subjected it to a series of structural and behavioral validity tests, following a process suggested by Barlas (1996) and Sterman (2000). All tests (structural and behavioral) were performed incrementally. Starting with a simple SEIR model and iterating with a subset of stakeholders, we verified the structural relationships captured in our formulations. As a deeper understanding of the disease became available, we adjusted the model to account for such insights. For instance, as knowledge about the role of asymptomatic individuals became available (Bai et al., 2020; Hu et al., 2020) or information about the infectivity of presymptomatic (Qian et al., 2020) became available, we quickly incorporated them into our model. The first version of the model considered only the epidemiological diffusion. A revised version included hospital structure and its internal dynamics. A later version incorporated testing to the model structure.
With respect to behavioral tests, we used model calibration to test the ability of the model to replicate observed behavior, comparing the simulated behavior of the model against available time series data from real counterparts (Forrester, 1979; Homer, 2012). Model calibration drives a real‐time series input data through model structure and compares the simulated output behavior with the associated time series data for the output. Both partial and full model calibration requires time series data on several variables. The COVID‐19 taskforce maintained a database of the stock of available ward and ICU beds, and bed occupancy and the flow of new daily admissions, discharges, and deaths, including out‐of‐hospital deaths. Additional data from patient flow within the hospital system were available from the EOC electronic medical record system and were used to source additional information and patients’ disposition. EOC made the data available to our team, allowing us to analyze the dynamics of the COVID‐19 epidemic in Ticino as well as its future developments. To calibrate the model to the available data, we set up an optimization problem that minimizes the sum of squared differences between a set of model variables (
MODEL RESULTS
As the number of COVID‐19 cases, hospitalizations, and deaths increased during the surge phase, our team worked with the EOC to present model outcomes that could inform their critical decisions. In this section, we present the key results of our modeling effort. In particular, the section focuses on (i) early projections of pandemic diffusion (based on Lombardy data) and (ii) projections of the timing and size of peak patient demand.
Early projections of pandemic diffusion
The first COVID‐19 cases were detected in Lombardy on February 21, 2020. Nine days later, on March 1, the first confirmed case and hospitalization took place in Ticino. By then, Lombardy faced a calamity with its hospital system overwhelmed by the number of severe COVID‐19 patients. Without much data available early on, our team used Lombardy data (e.g., confirmed cases, hospital admissions, ward and ICU hospitalizations, discharged, and deaths) to forecast the pandemic progression in Ticino. Starting with a preliminary SD model (Figure 2), we constantly updated the model to track the likely progression of the pandemic in Ticino. The progression of confirmed cases, hospitalizations, and deaths in Ticino and Lombardy suggested a 1‐week delay between the two outbreaks. That window of opportunity allowed Ticino just enough time to learn from its neighbor's experience and anticipate the likely impact of the COVID‐19 pandemic.
Early simulations with Lombardy data (Figure 3) showed a rapid increase in the number of confirmed cases, hospitalizations, and deaths. In the first 3 weeks of the outbreak, Lombardy faced almost 6000 hospitalizations, with a 23% daily growth rate and a doubling time of 3 days.
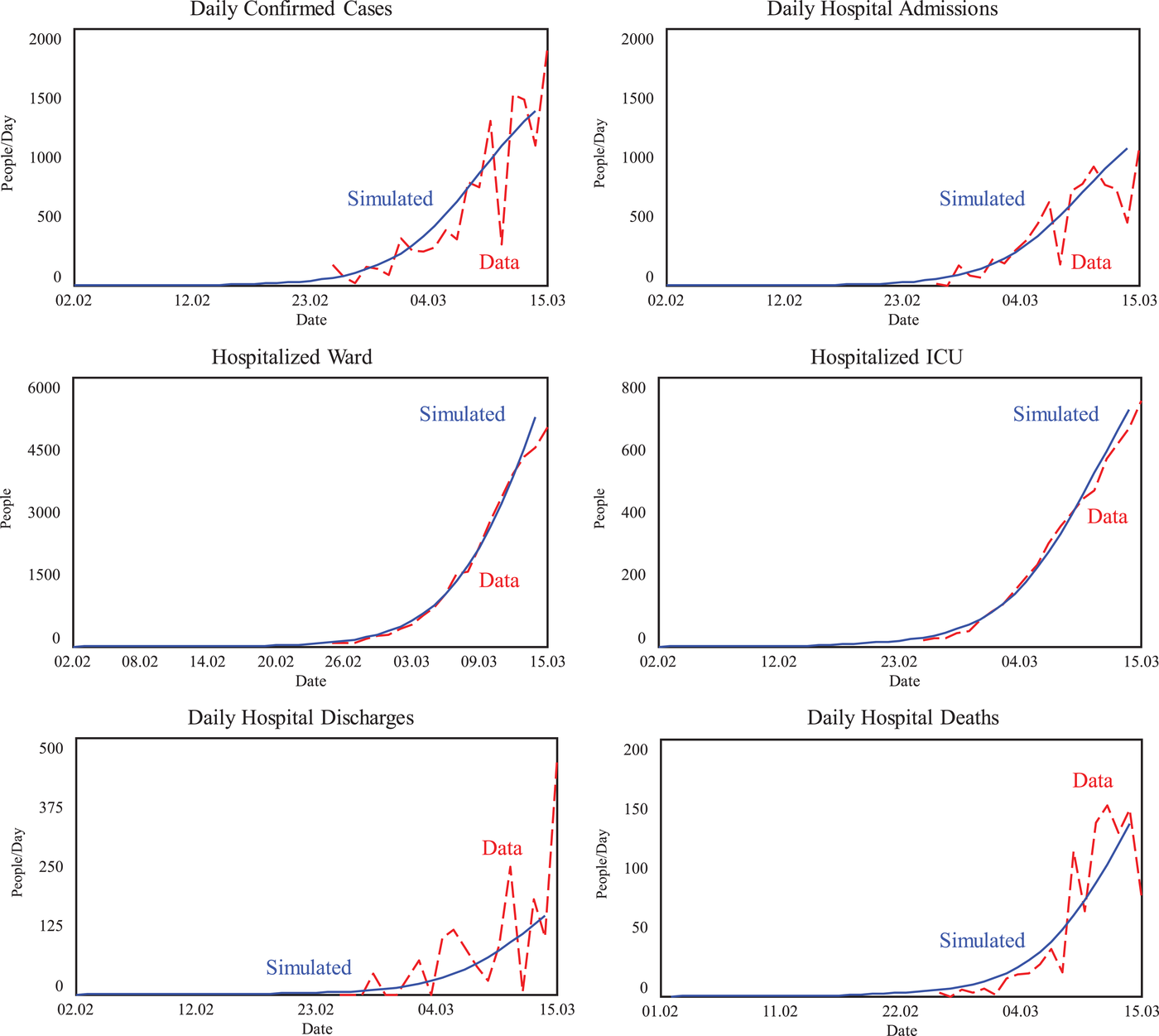
Simulations estimating COVID‐19 progression in Lombardy (Feb 01–Mar 14, 2020)
To generate 1‐week‐ahead range forecasts for the pandemic in Lombardy, we allowed simultaneous changes in multiple key parameters (e.g., incubation time, illness duration, fraction progressing to asymptomatic, hospitalization time, ICU time). Then, we simulated the model 20,000 times with independent randomly selected parameter values from uniform distributions within specified ranges, to generate multivariate Monte Carlo sensitivity projections. 4 To adjust the range forecasts to Ticino, we used a 3.5% ratio (1/28.5), since Lombardy's population (e.g., 10.1 million people) is 28.5 times higher than that of Canton Ticino. The projections for Lombardy for patients hospitalized in the ward (from 8000 to 12,000) and hospitalized in ICU (from 1500 to 3000), implied possible ranges for Ticino from 280 to 420 hospital beds including anywhere from 52 to 105 ICU beds (see Table 4). 5 Informed by the delay between the two outbreaks and extrapolating the data from Lombardy, we were able to quickly extrapolate (and anticipate) the hospital capacity and personnel needs, emphasized the high‐end projections to avoid possible bottlenecks.
Projected patient demand in Ticino during the surge phase (based on Lombardy data)
Note: It is assumed that the fraction of hospitalizations from confirmed cases remains stable at 70%, a fraction of ward hospitalizations from total remains stable at 85% (in the first three weeks). Ticino peak estimates extrapolate the likely impact using a 3.5% population ratio.
Projections of the timing and size of peak patient demand
In the weeks following the changes in hospital operations, there was continued concern about the ability of the hospital system in Canton Ticino to meet peak COVID‐19 patient demand. As the pandemic spread and fatigue among medical and nursing staff increased, the timing and size of the peak began to matter. We used the SD model to generate frequent forecasts to predict the likely timing and size for the peak patient demand. Figure 4 shows the comparison of the data and simulated model for Ticino data.
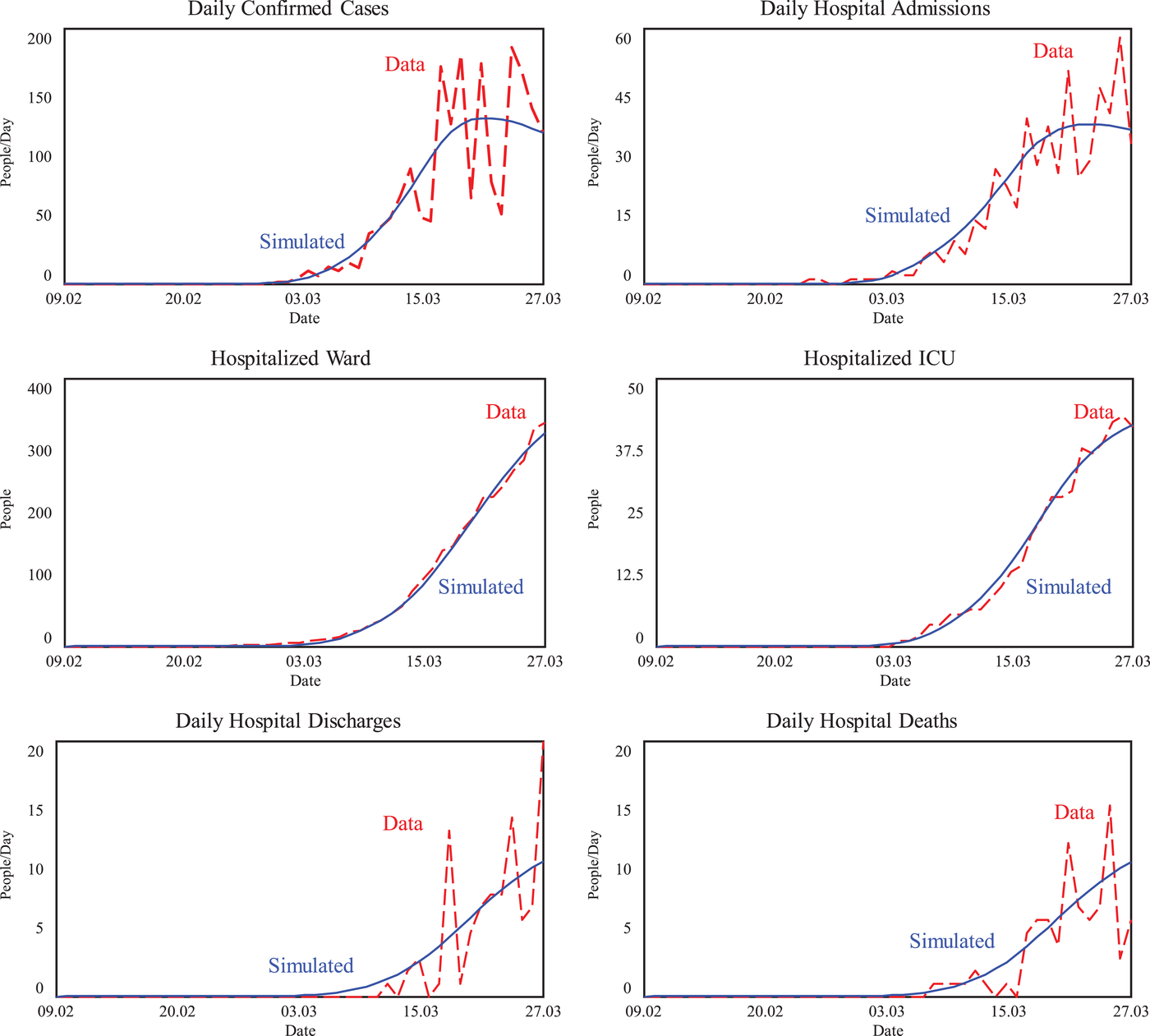
Simulations estimating COVID‐19 progression in Ticino (Feb 09–Mar 27, 2020)
Sensitivity analysis allowed us to create range forecasts for the progression of the disease in Ticino. While it was not possible to predict exactly the timing or the size of the peak, the range forecasts gave us an understanding of what was likely to come within the next week. 6 In addition, as the days passed the cone of uncertainty decreased providing more accuracy in the forecasts for the coming days. Our range forecasts provided confidence that the capacity changes implemented were sufficient for the hospital system in Ticino to meet peak COVID‐19 patient demand.
CHANGES TO PRACTICE
In this section, we present the impact on hospital practice and design resulting from our analysis. First, we describe the hospital network and resources available before the onset of the COVID‐19 pandemic in Ticino (pre‐March 2020). During this prepandemic phase, we characterize the baseline hospital capacity of the system. Next, during the surge in patient demand, Ticino experienced a large increase in the number of COVID‐19 cases, hospitalizations, and deaths, with mounting pressure to increase baseline hospital capacity. During this surge phase, we characterized the required changes and managed hospital practices and network design to address pandemic challenges that were informed by our analysis. In particular, the section focuses on (i) the need to consolidate hospital network and (ii) the need to increase hospital capacity to sustain demand surge, including fully ventilated ICU bed capacity, medical and nursing staff capacity, and oxygen supply.
Prepandemic baseline hospital capacity
Prepandemic, the cantonal hospital network composed of four public hospitals (all equipped with ICUs and a private clinic with an ICU, had a total of 52 ICU beds (45 and 7, respectively) of which 45 had full ventilation capacity. If each of the four public hospitals operated their ICUs, maintaining an 8‐h shift for medical staff, each facility would require three senior consultant doctors to ensure 24/7 service care. That is, 12 senior consultant doctors would be required if doctors would work only in a single hospital. At the same time, prepandemic the EOC ICU medical staff included only two senior consultant doctors 7 and because COVID‐19 affected Italy and nearby Cantons, it was not possible to source other senior doctors from nearby locations. Moreover, there was a limited number of appropriately skilled (i.e., anesthetists with at least one term of ICU experience) registrars and residents that could be added to EOC. There were also insufficient numbers of specialist nursing (SN) staff capable of managing ventilators, oxygen pumps, and monitors, required to treat COVID‐19 patients developing respiratory failure. While registered nursing (RN) staff were more available, they were not skilled to work in ICU, and could not be promptly trained since that would require 2 years.
Furthermore, due to PPE precautions—requiring residents and nurses to dress‐up/‐down and disinfect between patients—typical prepandemic resident‐to‐patient ratio (1:12) and nursing‐to‐patient ratio (1:6) would need to be reduced. The EOC realized that on a COVID‐19 ward it was only possible to assign 10 patients per resident (i.e., a 1:10 resident‐to‐patient ratio) and four patients per nurse (i.e., a 1:4 nursing‐to‐patient ratio). This decision implied a significant increase in required resident (∼20%) and nursing (∼30%) staff capacity.
Finally, to support patients developing respiratory failure it was critical to ensure the availability of oxygen for mechanical ventilation. Prepandemic, we established average daily oxygen requirements of about 250 kg/day (i.e., 219 L of compressed oxygen/day, or 187,000 L of high flow oxygen/day). On March 8, 1 week into the pandemic, with 7 acute wards and 15 in ICU patients, the daily oxygen requirements had almost tripled to 578 L of compressed oxygen/day (almost 500,000 L of high flow oxygen/day).
Need to consolidate hospital capacity
As the number of COVID‐19 cases, hospitalizations, and deaths increased during the surge phase, public health authorities focused on the need to consolidate and better manage hospital capacity. Considering only the senior consultant doctor requirements, it was clear that the EOC would not have sufficient capacity to run four independent ICUs. Consolidating ICU services in a single hospital would allow the EOC the ability to better share scarce medical and nursing staff resources across different patients, achieving significant economies of scale. For instance, operation of a single ICU would require only three senior consultant doctors (instead of the 12 for 4 independent ICUs) to ensure 24/7 service care. Changing the medical staff shift to 12 h (instead of the 8‐h shift prepandemic) would reduce further the requirements to only two senior consultant doctors. Based on simple back‐of‐the‐envelope calculations, the EOC designated its hospital in Locarno as a COVID‐19 dedicated facility on March 11, 2020. 8
The EOC decision to setup a dedicated COVID‐19 facility followed a focus as emphasis perspective (McDermott & Stock, 2011), emphasizing two “focus dimensions” (Dabhilkar & Svarts, 2019), such as Knowledge areas (i.e., referring to physicians’ training, skills, or experience) and Medical conditions (i.e., referring to the disease or injury causing a patient to seek healthcare), in its effort to treat COVID‐19 patients. The consolidation decision addressed the critical needs of public authorities, exemplifying a healthcare strategy that improved allocation of available medical and nursing staff, improving operational performance (Hyer et al., 2009; McDermott & Stock, 2011) and patient care (Goldstein et al., 2002).
Need to manage hospital capacity
ICU and ward bed requirements
Volume flexibility in healthcare operations refers to the ability of an organization to adapt its volume of service flexibly to meet patient demand (Jack & Powers, 2004; Koste & Malhotra, 1999). The projected surge in demand required the EOC to respond strategically to use any and every available resource. Within 4 weeks, the EOC managed to create 55 new ICU beds at the COVID‐19 dedicated hospital, including 22 intermediate care beds for tracheotomy patients after weaning from mechanical ventilation, while maintaining 18 ICU beds for non–COVID‐19 patients. In parallel, the private clinic also increased its ICU bed capacity. While these increases were gradually introduced over 4 weeks, they allowed Ticino to increase ICU bed capacity by over 150%, ensuring that it could meet the sharp expected increase in patient demand. In addition to increasing the bed capacity of its acute ward and ICU, the EOC implemented several other measures, building on internal resources around workforce flexibility, reallocation of available capacity, and effective schedule management, to achieve further volume flexibility. 9
Medical and nursing staff requirements
The surge in patient demand and the increased ICU bed capacity had implications for medical and nursing staff that required the allocation of staff with the necessary skills in a coordinated way. While the decision to set up a dedicated COVID‐19 facility allowed the EOC to operate its ICU without an increase in the number of senior consultant doctors, it still had a limited and insufficient number of medical and nursing staff. Volume flexibility in medical staff was achieved through the implementation of four policies:
Stopping all nonessential elective surgeries and procedures. In the second half of March 2020, the government issued a measure ordering hospitals in Ticino to put on hold all nonessential elective surgical activities. This allowed the EOC to free up not only operating rooms (ORs), but also medical and nursing staff with appropriate knowledge (i.e., conduct tracheotomies, operate the ventilators, change flows/pressure, regulate the pumps, and read the monitors) to be redeployed to COVID‐19 units. While highly unusual, this strategy can be classified as a demand management strategy.
Increasing shift length from 8 to 12 h. The major impact of the 50% increase in shift length was to reduce patient handovers in half, allowing a single handover instead of two per day. This change also simplified the handover process reducing the time required and associated errors, while improving patient care. The change increased workforce flexibility.
Sourcing medical and nursing staff from other departments. An additional workforce flexibility strategy leading to additional volume flexibility was achieved by sourcing adequately qualified junior medical staff, that is, anesthetists with at least one term of ICU experience, and physicians from internal medicine and operating theater. Implementation of such volume flexibility policies allowed the EOC to increase its ICU medical staff during COVID‐19 to 45 FTE (a 40% increase from the original level), with most of the adjustment (96%) taking place among registrars (38%, 14.8 FTE) and residents (58%, 24.5 FTE). The 40% increase of ICU medical staff had to be coordinated with an equivalent (+44%) increase in nursing staff. Due to insufficient numbers of SN staff, the EOC also had to source nurses from different departments, reallocating the SNs with appropriate skills from anesthesia departments and the operating theater.
Creating nursing pools. As a final workforce flexibility strategy, the EOC supplemented the scarce stock of SN staff with RN staff, by establishing a nurse buddy system that paired one registered nurse (one RN) with two specialist ones (two SNs). While RNs were not skilled to work in an ICU, which would require two additional years of training, and were not able to treat a patient on their own, they could assist the SNs in standard patient care. In the EOC's “buddy system,” nurses could tackle different tasks and together they could satisfy the surge in patient demand. RNs would perform usual nursing work (e.g., treat patients, wash and turn them, inject medication) and could also support SNs work (e.g., managing ventilators, pumps, and monitors).
Oxygen requirements
Baseline assessment for daily oxygen consumption requirements prepandemic and early surge established that patients with hypoxemic respiratory failure could use up to 20 L/min of oxygen, and patients in the ward with nasal prongs could consume up to 6 L/min of oxygen. Our early projections, emphasizing the high end to avoid possible bottlenecks, suggested up to 420 acute ward patients and up to 105 ICU patients. From those values, it was straightforward (see Table 5 for details) to extrapolate the oxygen needs of about 6.7 million liters of high flow oxygen/day (or about 7800 L of compressed oxygen/day, or 8900 kg of oxygen/day) at the peak. To ensure continuity of operations the EOC considered the reserve oxygen tank capacity at each of its public hospitals. It established a requirement of 72‐h service autonomy of oxygen during the forecasted surge in patient demand, which translated into a consumption of 26,700 kg of oxygen/day.
Projected assessment for daily oxygen consumption requirements at the peak of demand
None of its hospitals had such capacity. A location (in Faido) being considered for the dedicated facility had its oxygen capacity set up in bottles, which would provide at most a 16‐h service autonomy (about 5900 kg of oxygen/day) during the forecasted surge in patient demand. Given the limited oxygen capacity at Faido, that facility was discarded. Moreover, because oxygen is manufactured outside Canton Ticino, the EOC focused on establishing strategic alliances with oxygen suppliers early on, setting up service‐level agreements (SLAs) that ensured the delivery of higher quantities (e.g., filling up a tank of 20,000 kg of compressed oxygen) with higher frequency (e.g., every other day), instead of once per week prepandemic. 10
Summary volume flexibility strategies
Jack and Powers (2004) provide a framework to explore different volume flexibility strategies (see Figure 5) considering the level of demand uncertainty faced by the health organization (x‐axis) and the range of flexibility provided by a given strategy (y‐axis). When demand uncertainty is high and an organization's ability to implement flexibility is low, it adopts a Shielding Strategy to try to mitigate the negative effects of demand uncertainty. When demand uncertainty and desired flexibility are low, it can adopt an Absorbing Strategy using its internal buffers to mitigate uncertainty. In contrast, when demand uncertainty is low but desired flexibility is high, the organization can adopt a Containing Strategy leveraging its internal resources. Finally, when demand uncertainty and desired flexibility are high, it can adopt a Mitigating Strategy leveraging internal and external resources to handle the high uncertainty.
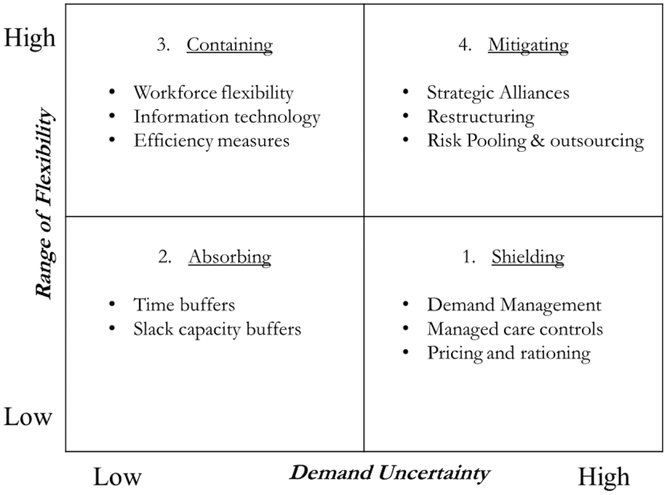
Volume flexibility strategy framework in normal healthcare operations (adapted from Jack & Powers, 2004)
During the pandemic surge, health organizations faced high levels of uncertainty and required high volume flexibility from their strategies. According to Jack and Raturi (2002), when health organizations face high levels of uncertainty and require high volume flexibility, they should adopt a mitigating strategy, including specific strategies such as restructuring, risk pooling, outsourcing, and developing strategic alliances. However, during emergencies there is limited time available to deploy such strategies. The need to respond quickly means that only strategies that can be deployed quickly can be considered. It also means that implementable strategies (i) leverage already available internal resources or already existing collaborations or (ii) prioritize critical services. As an example of the former, the strategic alliance that the EOC implemented for oxygen delivery (e.g., noncritical support services) was the only mitigating strategy adopted that was prescribed in the literature. Such alliance was only possible because it built upon an already existing collaboration (see Figure 6). As an example of the latter, the draconian government mandate to stop all nonessential surgeries, prioritizing all resources to critical COVID‐19 services, can be interpreted as a demand management strategy. While traditional demand management strategies (e.g., health promotion programs, forecasting systems) implemented by healthcare organizations yield low volume flexibility and are deployed as a shielding strategy, the one mandated by the government (e.g., stop all nonessential surgeries) yielded high volume flexibility and worked as a mitigating strategy (see Figure 6).
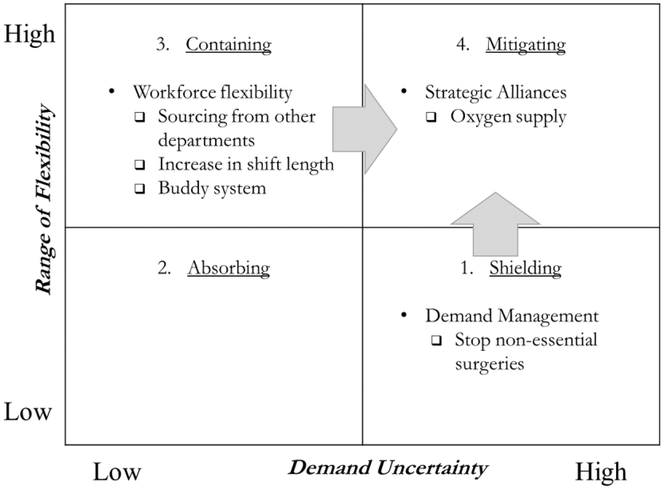
Summary of volume flexibility strategies adopted by the EOC during the pandemic surge
Interestingly, the EOC also implemented a combination of workforce flexibility strategies that yielded high volume flexibility (Bates, 2013; Bloom, 1997; Fagefors et al., 2020; Lebanik & Britt, 2015). Deployment of those strategies was only possible because it leveraged already available internal resources. The workforce flexibility strategies included increasing shift length of medical and nursing staff from 8 to 12 h, sourcing medical and nursing staff from different departments, and creating nursing staff pools, through a nurse buddy system. The literature typically prescribes workforce flexibility strategies as a containing strategy when faced with low levels of demand uncertainty. Jack and Powers (2004) recognize that when the demand for healthcare services exceeds organizational capacity, a containing strategy can be used to ensure additional operational flexibility. During the pandemic surge, however, the combination of workforce flexibility strategies was used as a mitigating strategy to address the high uncertainty in demand (see Figure 6).
In summary, the EOC hospital operations strategy ranged from consolidation of services through the establishment of a dedicated COVID‐19 facility, and a suite of mitigating (e.g., strategic alliances, demand management, and workforce flexibility) strategies that could address the high demand uncertainty and yield the high‐volume flexibility required. These coordinated strategies translated into increased operational capability (Goldstein, 2003) and improved overall performance (Goldstein & Iossifova, 2012). Finally, the fact that a physician, with vast international experienced, was at the helm of the EOC, ensured that their service operations strategy benefitted from pragmatic policies that were more likely to perform well in practice (Goldstein & Ward, 2004).
COUNTERFACTUAL ANALYSES
In this section, we present three counterfactual analyses considering the impact of options different than those supported by the SD model. First, we analyze the impact of a 1‐week delay in the consolidation and expansion of hospital capacity. Second, we consider on March 16 the impact of not implementing social distancing policies at Canton Ticino. Finally, we consider the impact of removal of social distancing measures on May 31. 11
One‐week postponement of hospital capacity expansion
The 1‐week delay between the Lombardy and Ticino outbreaks provided a window of opportunity that allowed Ticino and the EOC not only to generate early projections of likely cases, hospitalizations, and deaths in Ticino, but also informed the urgent need to expand bed capacity. We estimate that such intervention anticipated in at least 1 week the decision to initiate the increase in ICU and ward bed capacity. In the counterfactual analysis, we estimate the impact of a 1‐week delay in the actual ICU and ward bed capacity trajectory. Figure 7 shows the counterfactual analysis displaying the impact of delaying the process of increasing ICU and ward bed capacity in Ticino. Because we have the actual bed capacity data and patients hospitalized (i.e., the actual number of beds required) in both the ICU and ward, we can easily assess the impact of 1‐week delay in the growth in bed capacity. Figure 7 also shows bed shortages and occupancy percentages in the ICU and the ward. Shortages are computed by subtracting the data on patients hospitalized (actual demand) from the 1‐week delayed bed capacity. Occupancy percentages are computed as the ratio between occupied and total available beds.
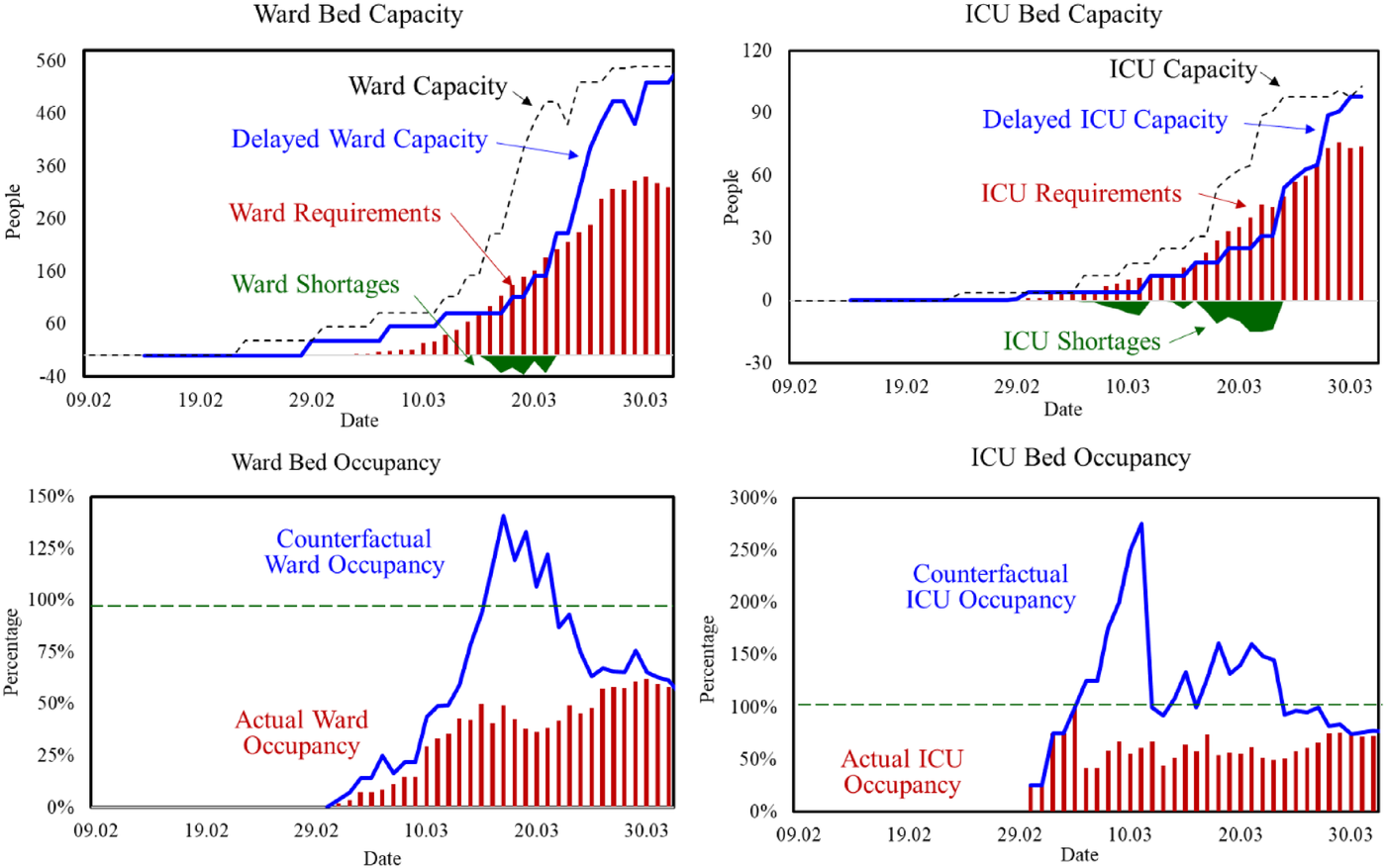
Counterfactual analysis displaying the impact of delay in adding ICU and ward bed capacity in Ticino (Feb 09–Mar 30, 2020)
The counterfactual analysis shows that in both the ICU and the ward a 1‐week delay would shift the mode of operation dramatically. With one week delay in initiating the capacity expansion, both the ward and ICU departments would have faced shortages. In the ward, the shortages would have started on March 16, affecting 13 patients, and would have lasted for 6 days. In its maximum, the hospital system would be short 37 ward beds (i.e., 33% of the projected available capacity on March 19). In the ICU, the shortages would have taken place already in the first week of March, lasting for almost 3 weeks. In its maximum, the hospital system would be short 15 ICU beds (i.e., 60% of the projected available capacity on March 17). Considering occupancy percentages, ICU would have to turn down patients throughout the first 3 weeks of the pandemic, until March 23; the ward would do the same from March 15 to 21. In the ward, the high occupancy rates would be somewhat contained to 6 days, reaching “only” a maximum of 133%. In contrast, in ICU high occupancy rates would last for 18 days (three times longer) and reach at its peak an occupancy of 275%. Our results shed light on the value and impact of our early intervention in collaboration with the EOC.
Delayed implementation of social distancing measures (March 16)
On March 16, 2020, Canton Ticino introduced strict regulations to enforce social distancing, with a complete lockdown of all nonessential services, including schools, attendance to church services, pubs, and restaurants. At the time that such policies were introduced, health officials were uncertain about the effectiveness of social distancing policies in curbing the spread of the disease, especially since reports alerted them about the role of presymptomatic and asymptomatic individuals contributing to the diffusion of the pandemic. Because our model captured such stocks explicitly, we hoped to be able to estimate their likely contribution to the diffusion of the pandemic and later compare their results with specific serological surveys.
Figure 8 shows the simulated behavior of the model from February 09, 2020, to April 01, 2020, for daily values of confirmed cases and hospital admissions. The figure also shows the actual data on confirmed cases and hospital admissions, and the simulated behavior of two polar social distancing policies (e.g., one with full lockdown and another without any distancing measures). For each simulated policy, the SD model uses only data available until March 16 to calibrate its behavior. (Later data are included as a baseline behavior for comparison purposes only.) Social distancing policies limit the number of contacts. The lockdown simulation (with social distancing policies in place) shows that confirmed cases and hospital admissions follow more closely the actual data. Still, the lockdown simulation overshoots “future” data (beyond March 16). That is, with the limited amount of data available in the first 2 weeks, the endogenous social distancing formulation does not manage to fully account for the actual effectiveness of the social distancing policies. Despite the overshoot beyond March 16, the lockdown simulation projects much lower values than the simulation without social distancing policies in place. In fact, they project values for confirmed cases and hospital admissions by April 1 that are half of those simulated without any distancing. Distancing policies yield slower diffusion with significantly smaller numbers of cases, hospital admissions, and deaths.
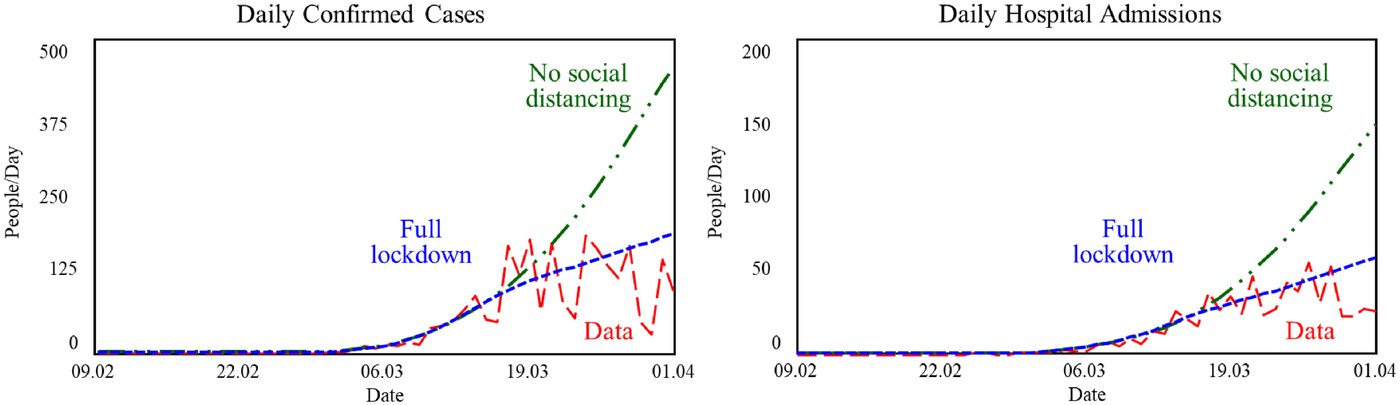
Counterfactual simulations estimating the impact of social distancing policies in Ticino (Feb 09–April 01, 2020)
The counterfactual simulations of the SD model with and without social distancing policies shed light on the likely impact of the lockdown policies. Our results reiterated public health officials’ expectations on the critical importance of imposing strict social distancing policies, also providing a reference for the likely impact if no social distancing policies were put in place.
Early removal of social distancing measures (May 31)
Figure 9 compares simulated behavior with time series data for Ticino (from February 09, 2020, to July 01, 2020) for daily values of confirmed cases and hospital admissions. Inspection shows that the simulation has a good fit for the data across both variables. Figure 9 also shows the projected behavior of the model for different levels of “effectiveness” of social distancing policies. Such effectiveness levels, implemented from May 31, capture different levels of compliance with ongoing lockdown policies. When “effectiveness” equals 1 (100%), there is widespread compliance with full lockdown policies; when “effectiveness” equals 0 (0%), there is absolutely no compliance with social distance policies, and people operate as if they were in normal conditions. The figure also shows the behavior of the model for values of effectiveness in between.
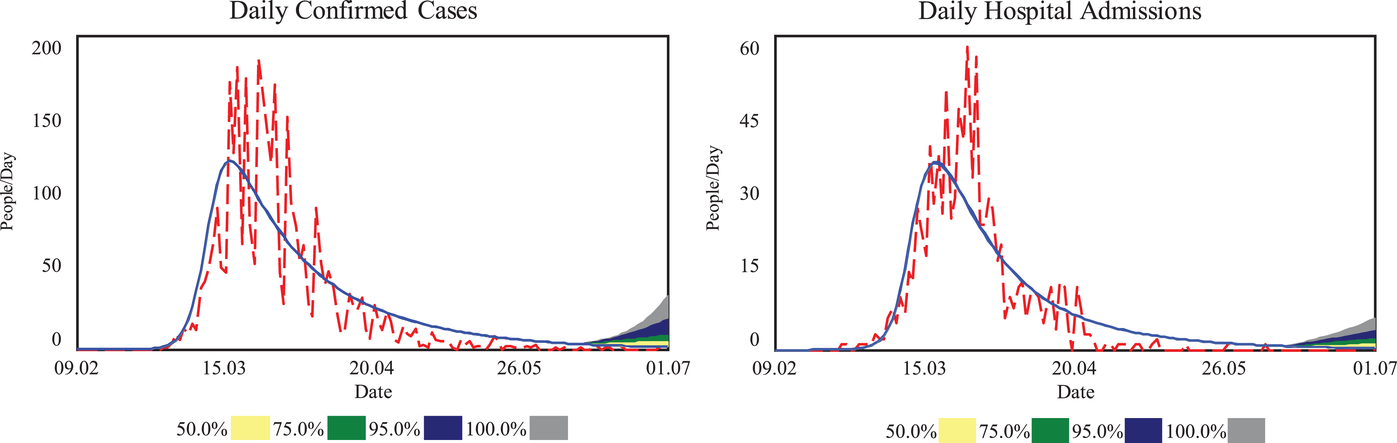
Counterfactual simulations estimating the impact of relaxing social distancing policies in Ticino (Feb 09–July 01, 2020)
The counterfactual simulations performed with the SD model confirm that a second wave of infections was likely to occur if social distancing policies were lifted to all individuals (even at the end of May 31). The information shared with health policy officials helped maintain a policy of caution and a range of social distancing policies in place.
KEY PROCESS LESSONS
Below, we reflect on aspects of our research effort that were well‐aligned with the challenging conditions imposed by the COVID‐19 emergency. We also distill key process lessons regarding the development of collaborative systems models to inform public health policies during emergencies.
Process lessons
Due to the rapid growth of severe COVID‐19 cases, a multitude of affected stakeholders, various streams of data, no existing off‐the‐shelf quantitative model, and a diverse set of policy levers, our research team had to quickly develop a regional model capturing the different stakeholders’ perspectives, reliably quantifying the progression of the pandemic vis‐à‐vis the data, and capable of identifying and testing different policies, before selecting the high‐leverage ones. As is common in participatory systems modeling processes, which have a well‐documented track record of public policy impact (Andersen et al., 2017; Rouwette et al., 2011; Scott et al., 2016), the novel and high‐quality SD model had to be tightly integrated with policy implementation. However, traditional participatory processes were ill aligned with the urgency imposed by the emergency. To timely support public health authorities, it was critical to introduce more agility in our participatory systems modeling process. By adapting the participatory systems process to the imposed reality of the emergency, we managed to mitigate typical barriers to adoption of participatory processes.
To design the key attributes of the agile participatory systems process, we considered specific contextual challenges imposed by the emergency, identified the specific modeling needs, and the key process characteristics required to support public health policy decisions (see Table 6). For instance, our research team was composed of experts with knowledge in the areas that were specific to the challenges imposed by the emergency. The research team met frequently. We structured small informal advisory, consultation, and modeling groups. Members of the research team held independent and ad hoc meetings with those groups. Lessons learned in those interactions were shared frequently with the research team. We replaced workshops with “collaborative sessions.” Such sessions had focused goals and a limited number of critical stakeholders, avoiding the need for a facilitator.
Challenges, impact from mistakes, identified needs, and key process characteristics for participatory systems modeling during emergencies
While our agile participatory process was more suitable for the reality imposed by the emergency, the changes also carry downsides. For instance, small “collaborative sessions,” with few stakeholders, focusing on specific goals, are not as inclusive or open. Frequent iterations, rapid model development, emphasis on constant progress hampered model transparency and a shared understanding. Many stakeholders were not involved, and their concerns were not included in the modeling process. And, due to the sense of urgency, there was no time to develop a thorough stakeholder engagement plan. Given the emergency and the need to engage critical stakeholders in a participatory process, we deemed these shortcomings acceptable. However, such shortcomings and their benefits must be considered carefully before an actual engagement. Still, we believe that agile participatory processes can inform key aspects of decision‐making during emergencies, ensuring that researchers are better prepared to support practitioners’ (e.g., policymakers and health officials) needs during future emergencies.
Discussion
COVID‐19 presented the world to a novel class of rapidly evolving high‐stakes problems, requiring very rapid response to develop novel, high‐quality, data‐intensive dynamic simulation models tightly integrated with policy implementation. In such rapidly evolving contexts, traditional time‐intensive participatory systems modeling processes are less effective, despite their range of other benefits. This research describes the rapid development and application of an agile participatory SD modeling process to help reorganize the public health system and inform public health policies during the COVID‐19 pandemic in Ticino, Switzerland. The agile participatory process has some advantages over the traditional participatory process with respect to (i) the speed of putting together a research team (and other support groups), (ii) the pragmatism associated with determining the level of model aggregation, (iii) the engagement and collaboration with decision‐makers, and (iv) its ability to deliver rapid and informative public policy analysis. The process characteristics of agile participatory modeling can support public health policy decisions during emergencies.
Our work informed and supported the need for the reorganization of the healthcare system in Ticino. Early on, the SD model was used with data from Lombardy to project the likely needs of ICU and ventilated bed capacity (as well as medical and nursing staff capacity) in Canton Ticino. Comparative analysis of the outbreaks in the two different regions allowed an educated guess regarding the needs in Ticino. As the COVID‐19 pandemic progressed, the SD model could predict the timing and size for the peak patient demand in Ticino and inform whether the additional ICU and ventilated bed capacity (as well as medical and nursing staff capacity) would suffice to meet the peak patient demand. The SD model was able to inform the decisions on required bed capacity, and that the added capacity was sufficient to cater for the surge in patients.
Counterfactual analyses consider the impact of (i) a 1‐week delay in the consolidation and expansion of hospital capacity, (ii) not implementing social distancing policies on March 16, and (iii) removing social distancing measures on May 31. Most dramatically, counterfactual analysis of the consolidation and expansion of hospital capacity shows that a single‐week delay would lead to bed shortages both at the ICU and the ward, such shortages would last for almost 3 weeks in ICU and would represent 60% of the available capacity at the time. Estimated ICU occupancy rates would rise sharply reaching a peak occupancy of 275%. Counterfactual analysis of the removal of social distancing measures on May 31, at the end of the first wave of the pandemic, confirmed that the second wave of infections were likely to occur if social distancing policies were lifted to all individuals.
Using established insights from healthcare operations management, we were able to inform the EOC decision to designate one of its hospitals as a COVID‐19 focused facility. The decision exemplifies a hospital focused on the treatment of COVID‐19 patients (i.e., medical conditions) and respiratory and pulmonary training (i.e., knowledge areas). The estimated surge in demand provided a clear signal for the need to increase hospital capacity (i.e., volume flexibility). The EOC's early efforts emphasized, first and foremost, a sufficient increase of ICU bed capacity. At the peak of the pandemic, the proportion of hospitalized patients admitted to ICU was 19.1%. ICU bed occupancy reached 80% and general ward bed occupancy reached 62%. In Ticino, the availability of critical care beds for COVID‐19 patients did not create a bottleneck for the access of patients to ICU.
From a healthcare operations perspective, our research contributes to the literature on volume flexibility strategies during emergencies, exploring the effective options available to healthcare providers when (i) demand uncertainty is high, (ii) required level of flexibility is high, and (iii) time to respond is scarce. Previously, Jack and Raturi (2002) prescribe that when faced with high levels of uncertainty and requiring high volume flexibility, health organizations should adopt a mitigating strategy. However, during emergencies, when the required response time is short, health organizations cannot deploy those strategies. Our research suggests that health organizations deploy strategies that either (i) leverage already available internal resources or already existing collaborations or (ii) prioritize critical services. A combination of different workforce flexibility strategies (e.g., creation of nurse pools, increasing shift length, and internal sourcing of medical and nursing staff) captures EOC's choice to leverage available resources. As the EOC recognized the need for high volume flexibility, they drew on internal resources available in the Cantonal hospital system—reallocating medical and nursing staff across different units, increasing the length of doctors and nurse shifts, and creating nursing staff pools. Principles of healthcare operations informed the need for coordinated actions across groups of staff (e.g., medical and nursing staff) as well as within groups of staff (e.g., SNs and RNs, and residents, registrars, consultant doctors). The adoption of nursing pools proved particularly useful in a resource‐constrained setting with limited time or ability to train the additional resources required. An important aspect of the successful COVID‐19 response in Ticino was the coordination of measures addressing different possible issues or areas (e.g., ward and ICU beds, ventilation, oxygen, medical and nursing staff, hospital focus). The comprehensiveness of the measures ensured that no single item became a bottleneck that could cripple the response. The combination of healthcare management policies implemented in Ticino provide an exemplar of a successful healthcare management response to an emergency.
Post hoc insights and reflections
To collect additional lessons learned and insights related to the implemented work, we held a roundtable conversation with the EOC's CMO, representing the public hospital system, and the member of the EOC board of directors and member of the Ticino COVID19 taskforce. 12
From a public hospital system perspective, the EOC realized its lack of preparedness to deal with pandemics. In the first weeks, it took a long time to populate the model with validated data. The necessary data were simply not available. Data had to be collected manually. Data quality was low, containing several inconsistencies. Such problems highlighted the need for the EOC to improve its data collection system to ensure reliability, availability, and quality. During the project, the EOC continuously improved its IT system to enable automatic extraction of the required data. Today, the EOC has a data dashboard that it uses on a regular basis, providing a much‐improved understanding of EOC's overall operations and effectiveness. Lack of adequate data was also an insight from the cantonal and federal public health perspective. While the federation collected data on a regular basis on sick people, they realized that since pandemics infect healthy people, it is critical to frequently collect data (e.g., social determinants, nutrition, biomarkers) on a representative sample (∼100,000) of the entire population to better understand the progression of pandemics. Because no such data (and no serological survey data) were available during the first wave, there were no data on the number of presymptomatic, asymptomatic, and infected to better estimate the disease progression.
The public health system's CMO reflected that our work transformed the EOC's mindset toward resource effectiveness. Due to the constraints imposed by the pandemic, the EOC had to learn how to better use the overall resources available (e.g., medical and nursing staff, ICU and ward beds, ORs). After the first wave of COVID‐19, there were about 1000 postponed elective interventions on stand‐by that the EOC needed to schedule and execute promptly. Due to the learning from improved planning, the EOC changed its OR allocation scheme, basing it on need instead of by specialty, which allowed it to quickly process all pending interventions. The resource effectiveness mindset also led management to stress test a previous capacity expenditure decision. Before the start of the COVID‐19 pandemic, the EOC's board of directors had approved an investment of CHF 250 Million to expand EOC's ICU capacity. Due to the changes implemented during the pandemic and improved OR scheduling, the EOC deemed the capacity expansion unnecessary.
In our conversation, we also investigated what the public hospital system and health officials could do differently to better prepare for future pandemics. The Cantonal and Federal authorities have explored two regulatory changes that would lead to improved flexibility and faster services: intervention authorizations and financial responsibility for COVID tests. Regarding the former, authorizations for different medical interventions are currently given to specific hospital sites (e.g., to a location), not to doctors, whose skills and training enable such interventions. The regulation for intervention authorization is based on an outdated concept that did not foresee doctors’ moving across hospitals. During the crisis, this regulation was temporarily suspended to authorize the COVID‐19 dedicated hospital to execute the required medical interventions. A change to this regulation is currently under discussion in Ticino. Regarding the latter, lack of clarity regarding financial responsibility for COVID‐19 tests resulted in limited test capacity and focused testing of severely symptomatic. Because Switzerland has a federalist health system, Cantons are the main actors planning and providing public health services. These services are complemented by those offered by multiple health insurance companies. In the first months of the pandemic, it was unclear whether the financial burden would be borne by Cantonal authorities, insurance companies, or the Federation. Influenced by a policy paper, written by members of the Ticino COVID‐19 taskforce, the Federation took over financial responsibility for COVID‐19 tests. Such change ensured broader availability of tests and a better understanding of the progression of the disease. Finally, at the EOC, a possible change under consideration deals with training of nursing staff. The public health system struggled with enhancing nursing ICU capacity during the pandemic. Due to a siloed training approach, nurses train only in a specific area and tend to remain there. The public health system realized the need to change the training system, making it more integrated and allowing nurses to learn different specializations through rotation across departments. This approach would make the job more interesting for nurses, while also ensuring that the system would have a sufficiently large (and occupied) well‐trained staff to provide flexibility in future emergencies.
Future research
Naturally, there are ample opportunities for further research both on agile participatory systems modeling processes and on volume flexibility strategies during emergencies. In terms of the former, our design was driven by an urgent need to address a pressing challenge, not by meticulous consideration of the best possible choices. Hence, we conjecture that there would be further opportunities to improve on the proposed design. Regarding “collaboration sessions,” future research could focus on specific designs for these sessions, including ideal number of sessions, frequency, critical composition, content, focus, objectives, and communication protocol. Regarding the adequate composition of advisory, consultation, research, and modeling groups, future research could consider specific criteria for the inclusion (or exclusion) of different stakeholders in each of the groups. Future research could also consider: What would be an ideal communication protocol across groups? Should the frequency of meetings be dictated by the speed that the problem evolves?
There were also unique factors contributing to the formation of our research team and the success of our intervention. The sense of urgency and the high‐stakes situation created a favorable environment to form a new collaboration that was both pragmatic and results oriented. The research team included a policymaker, a decision‐maker, an SD modeler, and a topic expert, which gave us significant discretion regarding policy implementation in the public hospital system and the ability to influence Cantonal policy decisions. At the same time, our team was embedded in a much larger and influential network (i.e., through our informal meetings with our advisory, consultation, and modeling groups), which provided us with state‐of‐the‐art knowledge and up‐to‐date information on the new COVID‐19 research insights globally. Given the uniqueness of the context and our team, future research efforts could consider specific ways in which the lack of diversity (e.g., policymaker, decision‐maker, SD modeler, topic expert) in the composition could impose limitations on the ability of the team to achieve success.
Our research on volume flexibility strategies during emergencies suggests that the need to respond quickly, limits the meaningful strategies that can be deployed to those that (i) leverage already available internal resources or already existing collaborations or (ii) prioritize critical services. Future research could explore other general conditions (and provide examples) that would allow other strategies to be deployed. Future research could also consider other types of deviations from traditional strategies deployed during normal times that take place during emergencies. In addition, our research highlighted that to address the high level of uncertainty and achieve high volume flexibility, healthcare organizations were tweaking their strategies to meet their needs. For instance, independent workforce flexibility strategies typically deployed as a shielding strategy were instead implemented in combination as a mitigating strategy to yield high flexibility. Future research could explore further examples of similar strategic deviations due to contextual conditions. Finally, it would be interesting to develop future research and further understanding of what, Jack and Powers’ (2004) call, “linkages associated with the combination of strategies” and “specific tactics” deployed by managers during emergencies.
In summary, our dynamic modeling research clarified existing interconnections, established hierarchies and interdependencies, identified, and informed policies, and quantified their long‐term impact. Our research on volume flexibility strategies during emergencies showed new insights when compared to the deployment of such strategies in normal times. Our agile participatory systems modeling seemed to address typical shortcomings of traditional participatory processes. We believe that the contributions described in this research can not only be applied to improve healthcare operations in future emergencies but also motivate further research.
Footnotes
ACKNOWLEDGMENTS
We are grateful to Edward Anderson, Sushil Gupta, Nitin Joglekar, and Martin Starr, editors of this Special Issue, and two anonymous referees for guiding this research with extremely helpful comments and suggestions. Their advice was instrumental in improving the paper. We also thank Ottavio Beretta, Feryal Erhun, Antoine Feylessoufi, Martine Bouvier Gallacchi, Houyuan Jiang, Paul Kattuman, Tse Yang Lim, Anees Pari, Hazhir Rahmandad, Stefan Scholtes, John Sterman, and Jeroen Struben for their constructive and insightful comments on earlier versions of the paper. We thank Maria Andreeva for excellent data support and Raquel Buzogany Froese and Alireza Akhavan for research support.
Open access funding provided by Universita della Svizzera italiana.
1
Key participants in these “collaborative sessions” were, among others, public officials working for the Department for Health and Society (Dipartimento della sanità e della socialità—DSS), a department responsible for cantonal health policy. “Collaborative sessions” typically involved no more than 10 people and were relatively short lasting typically about 90 min. The relative success of these sessions led later on to a DSS initiative to fund and setup a separate project to track the second wave of the epidemic and formally support public health policy decisions.
2
A research effort in which our mathematical modeler expert is also a coauthor.
3
4
The use of uniform distribution for parameter ranges leads to wider confidence intervals for the sensitivity analysis. Our rationale for using ad hoc parameter ranges and a high variance distribution is to capture qualitative insights about parameter ranges that could potentially move beyond values reflected in historical data.
5
6
7
In total, the EOC had about 32 full time equivalent (FTE) staff, organized in a structured hierarchy with senior consultants (two FTE), junior consultants (3.4 FTE), registrars (9.8 FTE), and residents (17 FTE).
8
Supporting Information Appendix A4 provides further details on the hospital infrastructure available prepandemic and the changes implemented in response to the expected surge in patient demand.
9
Supporting Information Appendix A5 provides further details on the allocation of ICU beds to the COVID‐19 dedicated facility in Locarno.
10
In addition to oxygen capacity, it was important to ensure a robust oxygen delivery system (i.e., that oxygen from the tanks would arrive at patients’ beds). Because high oxygen flows can cause a dramatic drop in temperature around the pipes, there was a heightened risk of failure during the surge in demand. To avoid failures and increase system reliability, hospital operations increased insulation of pipe systems, reviewed the battery systems and implemented a backup in case of failure.
11
Supporting Information Appendix 6 presents a counterfactual for an early forecast (on March 7) of required hospital capacity in Ticino, anticipating the timing of the forecast in one week.
12
Both of them are members of our research team.
