Abstract
Activation of metabotropic glutamate receptors (mGluRs) often produces long-lasting effects on the excitability of cortical neurons. For example, mGluR stimulation induces long-term potentiation or depression of excitatory synaptic transmission in the hippocampus. Similarly, the effects of mGluRs on cortical epileptiform activities also are enduring. A transient application of group I mGluR agonists to hippocampal slices produces ictal-like discharges that persist for hours after the removal of the applied agonist. This action of group I mGluRs—transforming “normal” hippocampal slice into an “epileptic-like” one—may represent a form of epileptogenesis. The advent of such a model, in which epileptogenesis can be reliably induced in an in vitro preparation and the process is complete within hours, may facilitate the exploration of cellular mechanisms underlying epileptogenesis.
Introduction
Eight subtypes of mGluRs (mGluR1–8) have been cloned and classified into three different groups based on biochemical and pharmacologic properties (6,7). Activation of each group of mGluRs elicits different effects on epileptiform activities. Specifically, stimulation of group II (mGluR 2 and 3) and group III (mGluR 4, 6, 7, and 8) mGluRs (Gi protein–coupled receptors) suppresses seizures (8,9). In contrast, agonists of group I (mGluR 1 and 5) mGluRs (G protein–coupled receptors) elicit convulsive activities (10,11). Group I mGluRs have widespread distribution in the brain and are especially prominent in the CA3 region of the hippocampus (12–14).
An interesting feature of mGluR-mediated effects is that they are often long lasting. For example, activation of mGluRs sustains long-term potentiation (LTP) (15–17) and induces long-term depression (LTD) (18–20) of the CA3–CA1 synaptic connection. Similarly, mGluR stimulation produces long-lasting effects on cortical epileptiform activities. Transient activation of group I mGluRs by agonist application elicits epileptiform discharges in hippocampal slices that linger for hours after the washout of the agonist (21). The anticonvulsive action of group III mGluR agonist on the hippocampal neuronal network is also long lasting, in that the anticonvulsant effects persist long after agonist removal (22).
In this review, we describe the group I mGluR–induced epileptiform discharges and their long-lasting properties. In addition, we discuss plausible biochemical and cellular mechanisms underlying the plasticity process that induces and maintains the epileptiform discharges.
Group I mGluR-mediated Epileptiform Discharges
Group I mGluR agonists [e.g. (S)-3,5-dihydroxyphenylglycine; DHPG] elicit synchronized episodes of oscillatory synaptic depolarizations and overriding action potentials (episodes of oscillations) in the CA3 neuronal population of hippocampal slices. The episodes of oscillations have duration between 2 and 15 s, and they occur rhythmically at intervals of 10–20 s (Fig. 1A) (21,23). Within an episode, synaptic depolarizations appear rhythmically at 12–27 Hz (Fig. 1B; β-oscillations). The synaptic depolarizations reach firing threshold, and each depolarization may then elicit one to three action potentials. The hypersynchronized nature of an episode of oscillations, the frequency of oscillations within an episode, and the duration of an episode are comparable to those occurring in an ictal discharge. As such, information on the generation and plasticity mechanisms associated with the episodes of oscillations (ictal-like discharges) may shed light on the generation of ictal discharges and on epileptogenesis, respectively.
As mentioned earlier, ictal-like discharges such as those shown in Fig. 1, once induced by an agonist, persist for hours after agonist washout.
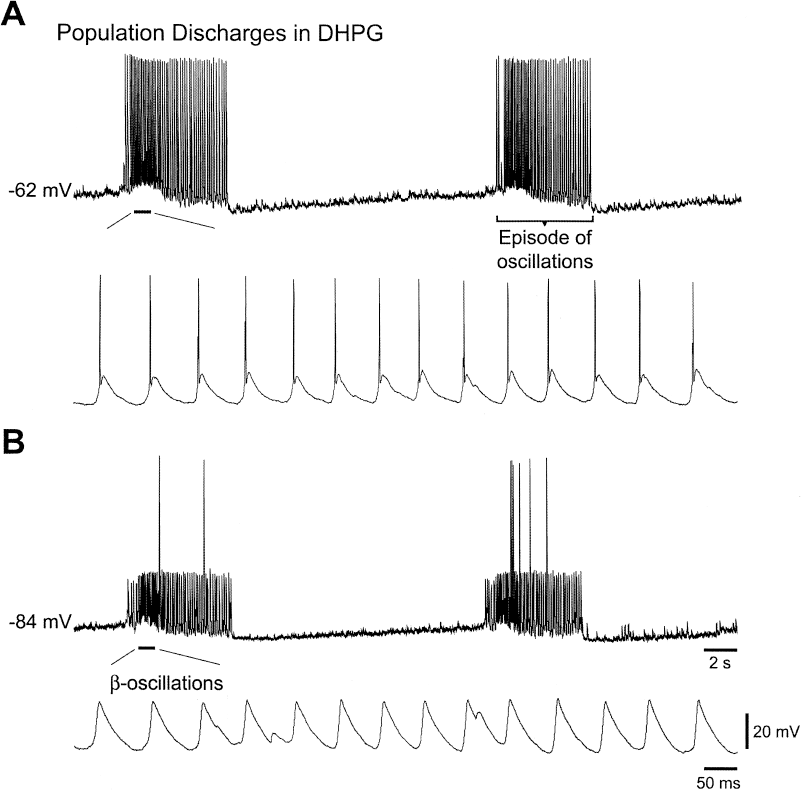
Group I mglur activation induces ictal-like discharges in the CA3 region of the hippocampus. Intracellular recordings from a CA3 pyramidal neuron of a mouse hippocampal slice 70 min after addition of the Group I mGluR agonist (S)-3,5-dihydroxphenylglycine (DHPG; 50 μM) to the perfusing solution. Top traces in A and B: two episodes of oscillations, each of 6–7 s in duration, recorded at resting membrane potential (A) and at a potential hyperpolarized with −1 nA DC current injection (B). The indicated membrane potential values refer to the beginning of the traces. Bottom traces in A and B: portions of the top traces indicated by bars shown on a faster time scale. Calibrations in B also apply to A. The hyperpolarization in B reveals that excitatory postsynaptic potentials (EPSPS) occurring at about 14 Hz (β-oscillations) underlie the rhythmic discharges during an episode of oscillations. The episodes of oscillations are population discharges because their frequency was not altered by membrane potential changes, and they were recorded as extracellular field potentials.
Plasticity Mechanisms Underlying Group I mGluR-Induced Long-Lasting Ictal-Like Discharges
Plastic changes in neuronal excitability, including those leading to the generation of long-lasting ictal-like discharges, generally proceed in two temporal stages: induction and maintenance. During induction, biochemical and/or molecular activities modify the properties of synaptic and/or ionic channel (cellular) processes. The plastic phenomenon is then expressed by the activation of the modified cellular processes. Induction is followed by maintenance, during which the cellular modifications are stabilized for the long-term expression of the plastic phenomenon.
We present information on the induction, maintenance, and expression of long-lasting ictal-like discharges.
Induction
Group I mGluR-induced plasticity generally involves translational modifications of protein function. For example, mGluR-dependent LTP (17) and LTD (20,24) are both blocked by pretreatment with protein-synthesis inhibitor. Biochemical studies in synaptoneurosomes prepared from cortical neurons (25,26) confirm that group I mGluR activation does lead to protein synthesis, including the production of fragile X mental retardation protein.
Consistent with this general scheme, induction of group I mGluR-dependent ictal-like discharges is also protein synthesis-dependent. Pretreatment of slices with a protein-synthesis inhibitor (anisomycin or cycloheximide) prevents the appearance of ictal-like discharges after agonist application (27).
The induction of long-lasting ictal-like discharges is accompanied by the development of spontaneous episodes of synchronized discharges of increasing duration and frequency (21). During each episode of synchronized discharge, CA3 pyramidal cells are excited by their recurrent synaptic connections through activations of postsynaptic N-methyl-D-aspartate (NMDA) and non-NMDA receptors. The discharges also produce prolonged neuronal depolarizations and activation of voltage-gated Ca2+ channels. Because NMDA-receptor activation and Ca2+ entry are cellular events known to induce plastic changes in neuronal excitability, the involvement of these events in the generation of long-lasting ictal-like discharges must be considered. Data indicate that induction of long-lasting epileptiform discharges can occur independent of a contribution by NMDA receptors, as these discharges are induced in the presence of NMDA-receptor blockers (28). Furthermore, the induction process is not affected by blocking both the NMDA and non-NMDA receptors (29). Under this condition, application of group I mGluR agonist no longer elicits synchronized discharges. Interestingly, after a period of “silent” agonist treatment, long-lasting ictal-like discharges emerge on the washout of the agonist followed by the removal of iGluR blockers in the perfusing solution.
The data indicate that induction of long-lasting ictal-like discharges depends primarily on an activation of group I mGluRs. Coactivation of NMDA receptors or endogenous Ca2+ channels associated with synchronized discharges is not required for the induction of long-lasting ictal-like discharges. Studies are now beginning to assess the relative importance of subtypes of mGluRs (mGluR1 and mGluR5) in the induction and maintenance of long-lasting ictal-like discharges. The results suggest a more important contribution of mGluR5 in the induction process, whereas mGluR1 appears to play a more dominant role in maintenance (30).
Maintenance
Ictal-like discharges, once induced, are no longer affected by protein-synthesis inhibitors. The data suggest that new proteins, once synthesized, can function for hours to sustain ongoing activities (27). Ictal-like discharges during the maintenance phase are blocked by group I mGluR and mGluR1 antagonists (21,30). Because endogenous glutamate is the only agonist available during the maintenance phase, the data suggest that ictal-like discharges are maintained by synaptically released glutamate.
Expression
The cellular processes activated synaptically during maintenance for the expression of ictal-like discharges have not been identified. ImGluR(V), a voltage-dependent depolarizing current, elicited by group I mGluR stimulation (31), is a candidate (32).
ImGluR(V), a Candidate Cellular Response Sustaining Long-lasting Ictal-like Discharges
ImGluR(V) appears in the presence of a group I mGluR agonist (31). The current increases the excitability of CA3 pyramidal cells and can elicit rhythmic firing in individual cells (32). Data suggest that population ictal-like discharges are generated by the synchronization of the single-cell rhythmic firing through the recurrent excitatory synapses (23,32).
Recent studies showed that pharmacologic blockade of phospholipase C (PLC) or knockout of the gene for PLCβ1 prevents the induction of ImGluR(V). As a result, single-cell rhythmic firing is suppressed, and the mGluR agonist no longer activates ictal-like discharges. These results are consistent with the hypothesis that the group I mGluR/PLC-dependent Im>GluR(V) is a cellular response necessary to sustain the population ictal-like discharges.
At present, it is unclear whether ImGluR(V) is active during the maintenance phase. For ImGluR(V) to be active in the maintenance phase, plastic changes must have occurred during the initial exposure to group I mGluR agonist, so that the responsiveness of ImGluR(V) to endogenous glutamate is increased. This increased responsiveness would then enable the current to be recruited synaptically and to participate in the production of ictal-like discharges during maintenance.
Summary
Studies in hippocampal slices show that a transient application of a group I mGluR agonist produces long-term changes in the cellular responses of the CA3 neuronal network, enabling the network to generate recurring ictal-like discharges. The cellular mechanisms underlying the mGluR-dependent epileptogenesis are largely unexplored. Group I mGluRs turn on a voltage-gated ionic current (ImGluR(V)) in CA3 pyramidal cells. Potentiation of the ImGluR(V) response may constitute one of the plastic changes underlying group I mGluR-mediated epileptogenesis. The group I mGluR model of epileptogenesis may have particular relevance to epilepsy because the induction mechanism is dependent on neuronal responses activated by glutamate, an endogenous neurotransmitter.
Footnotes
Acknowledgments
We thank Dr. Lisa R. Merlin for helpful comments. Work supported by NS 35481.
