Abstract

We previously reported the case of a young woman with a syndrome of recurrent, burning, aching head and neck discomfort with superimposed episodes of severe jabbing pain in discrete areas of the temporal, parietal and occipital scalp (1). The jabbing pain was followed within hours by the beginning of hair loss from areas of scalp affected by the pain. Initially, the episodes were sufficiently infrequent to allow hair re-growth between attacks. After several months, the jabbing pain became daily and large areas of the scalp exhibited alopecia (Figure 1). Biopsy of the affected scalp revealed lymphocytic peribulbar inflammation similar to that seen in alopecia areata (Figure 2). The biopsy showed no features of psychogenic hair pulling (trichotillomania). Although she was unresponsive to multiple medication trials, treatment with local injections of botulinum toxin A (BoNT/A) induced remission of the head pain lasting about 6 weeks, with regrowth of hair. Subsequent courses of BoNT/A treatment induced remissions lasting 4–12 weeks.
Hair follicles are innervated by neuropeptide-containing, unmyelinated neural plexuses (2) with varicosities located in close proximity to the likely site of hair follicle stem cells (3). Perifollicular nerve fibres contain neuropeptides including substance P (SP) and calcitonin gene-related peptide (CGRP), which are associated with the neurogenic inflammatory response (4) including vasodilation. SP release from C and A δ fibres is associated with mast cell degranulation both in skin (5) and dural vessels (6). In the skin, perifollicular SP is also involved in the induction and regulation of hair growth (5) and CGRP inhibits antigen presentation by Langerhans cells and other dendritic cells (7) and regulates T-cell proliferation (8) when released. Disruption of the physiological, tonic release of neuropeptides from perivascular nerve fibres may, therefore, result not only in derangement of trophic vasomotor regulation but also in altered peribulbar antigen presentation, premature loss of hair and inhibition of further hair growth.
We therefore hypothesized that chronic activation of the c-fibres, in addition to mediating inflammatory pain and follicular injury, might reduce SP and CGRP concentrations in affected cutaneous terminals causing disruption of the physiological, tonic release of trophic neuropeptides from chronically activated neurons resulting in altered peribulbar antigen presentation and inhibition of further hair growth. We further hypothesized that the hair regrowth observed after successful BoNT/A injections was due to restoration of tonic cutaneous SP and CGRP release. To test our hypotheses, we obtained scalp biopsies from the patient under two conditions: (i) when she was experiencing both pain and alopecia and (ii) during a period of improved pain control and hair regrowth following BoNT/A injections. We then examined the samples for the presence of cutaneous SP- and CGRP-containing nerve fibres.
Subject soon after presentation with severe pain and alopecia. H&E staining of hair follicle with inflammatory cells (arrows indicate perifollicular inflammatory cells).

Methods
After obtaining consent under a protocol approved by the Human Studies Institutional Review Board, the patient underwent scalp biopsies:
At three sites when she was very symptomatic (> 4 months after previous botulinum toxin treatment) with both severe scalp pain and areas of alopecia. We obtained two 3-mm punch biopsy specimens at sites in which she had jabbing neuralgiform pain and hair loss (samples A and B) (Figure 3) and one punch biopsy specimen at a site which at the time of the biopsy was not painful and had sustained no hair loss (sample C)
At one site 34 days after treatment with BoNT/A (sample D) (Figure 4). At this site, which prior to treatment had been very painful and had exhibited hair loss, she reported that she had been pain free for almost 4 weeks. She had experienced hair regrowth at this site. During the 12 h just prior to her return visit, she had begun to re-experience scalp pain but had no loss of the regrown hair.

Subject at first biopsy with focal alopecia before treatment.

Subject with decreased alopecia after treatment.
Immunohistochemical methods
Zamboni's fixed biopsy specimens were cryoprotected and sectioned with a freezing sliding microtome (Leica, Nussloch, Germany). Diluent and washing solutions were 1% normal donkey serum (Jackson ImmunoResearch, West Grove, PA, USA) in phosphate-buffered saline (PBS) with 0.3% Triton X-100 (Sigma, St Louis, MO, USA). Floating sections were blocked with 5% normal donkey serum in the diluent solution. Nerve and tissue antigens were localized using primary antibodies to protein gene product (PGP) 9.5 (1:800; AbD; Serotec, Kingston, NH, USA), SP (1:1000; Diasorin, Stillwater, MN, USA), CGRP (1:1000; Diasorin), tryptase (1:4000; Chemicon, Temecula, CA, USA) and type IV collagen (1:800; Chemicon), with each diluted in PBs–Triton X-100–normal goat serum. Biotinylated Ulex europaeus agglutinin type I (1:200; Vector, Burlingame, CA, USA) was visualized with Cy5-labelled Biotin-SP (Jackson ImmunoResearch). Non-immune serum was used with negative controls. Secondary antibodies labelled with cyanine dye fluorophores 2, 3 or 5 (Jackson ImmunoResearch) were used to locate two or three antigens in each section. After immunofluorescent processing, sections were adhered to coverslips with agar, dehydrated via an alcohol series, cleared with methyl salicylate, and mounted in DPX (Fluka BioChemika, Ronkonkoma, NY, USA).
Sections were evaluated visually with a Nikon Microphot-SA fluorescent microscope (Lake Success, NY, USA) for qualitative assessment of nerve morphology and neuropeptide density. CGRP- and SP-positive fibres in the sub-epidermal neural complex, superior to the papillary capillary complex, were counted as they approached, in a vertical orientation, the epidermal basement membrane. Neuropeptide-positive nerve fibres within or underlying the capillary complex were not counted. Thick sections were confocally imaged (CARV non-laser confocal microscope imaging system; Atto Bioscience Inc., Rockville, MD, USA) and then epidermal nerve fibres (ENFs) (immunostained for PGP 9.5 and type IV collagen) were traced with Neurolucida software (MicroBrightField, Colchester, VT, USA) according to established counting criteria and reported as number of ENFs per mm2 of skin surface (9). Mast cells were visualized with anti-tryptase and evaluated based on the extent and location of degranulation: 0, no degranulation; −1, degranulation in deep dermis; −2, degranulation in mid and deep dermis; −3, degranulation throughout dermis; −4, extensive degranulation throughout dermis.
Results
Untreated samples (A, B, C) had a weak non-robust staining pattern with the pan-neuronal marker, PGP 9.5 (Figures 5 and 6, top and middle right). The subepidermal neural plexus (SNP) was thin with appreciably fewer nerve fibres. ENFs were abundant, but were faintly stained, often appearing fragmented. Perifollicular innervation in untreated samples was also less dense than in treated samples. Sweat glands were well innervated, although nerves in untreated areas had thinner calibre nerve fibres than those seen in the treated sample.
Quantification of nerve fibres
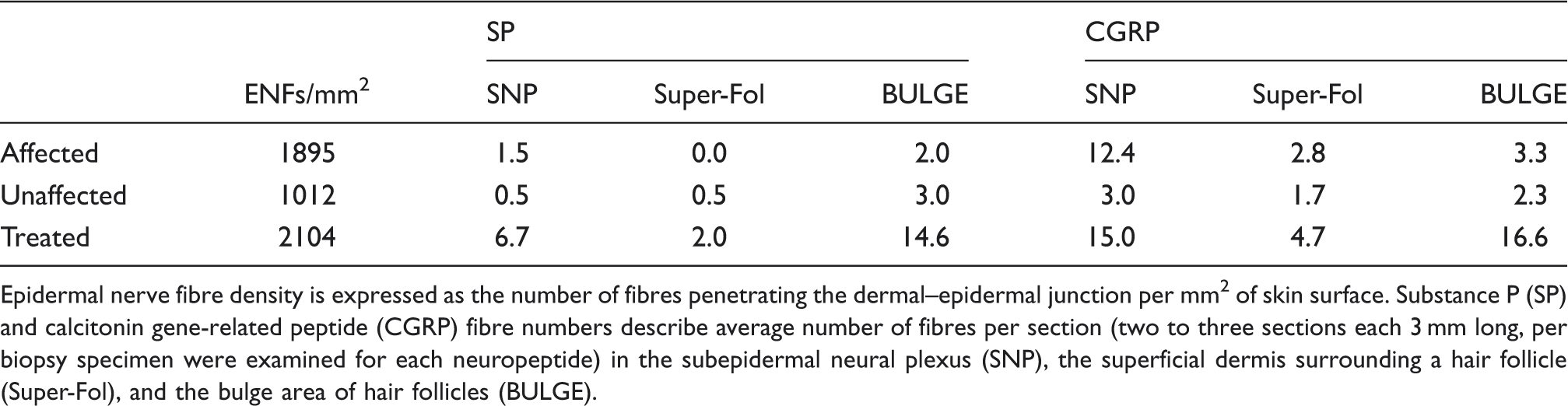
Epidermal nerve fibre density is expressed as the number of fibres penetrating the dermal–epidermal junction per mm2 of skin surface. Substance P (SP) and calcitonin gene-related peptide (CGRP) fibre numbers describe average number of fibres per section (two to three sections each 3 mm long, per biopsy specimen were examined for each neuropeptide) in the subepidermal neural plexus (SNP), the superficial dermis surrounding a hair follicle (Super-Fol), and the bulge area of hair follicles (BULGE).
SP-positive nerve fibres in the SNP were more plentiful in the treated sample than affected samples and unaffected untreated samples (Table 1, Figure 5). A cluster of these SP-positive fibres was observed adjacent to the follicle infundibulum in the treated sample and a single SP fibre was observed near the infundibulum in the unaffected untreated sample, while superficial SP fibres were not seen near the follicle in affected untreated samples. At the bulge level of the follicle, a network of SP-positive fibres was seen, in contrast to the paucity of individual fibres seen in untreated samples.
Confocal images of epidermis (EPID) and superficial dermis from scalp biopsies. Top row: Scalp 
CGRP-positive nerve fibres were almost continuous along the SNP in the affected samples (A and B) and treated sample (D), but were reduced in the unaffected sample and seen in only a portion of the SNP (C) (Figure 6). CGRP-positive fibre density along the follicle was similar to that observed for SP, with increased density of fibres seen at the surface and bulge area of the treated sample as opposed to the untreated sample.
Confocal images of epidermis (EPID) and superficial dermis from scalp biopsies. Top row: Scalp 
Mast cell degranulation was observed in untreated scalp areas but not in the treated area (Figure 7). In affected samples (A and B) degranulation was evident surrounding the bulge area of the follicle. In the unaffected biopsy specimen (C) degranulation was minimal and seen in the deep dermis with no apparent predilection for perifollicular areas.
Mast cells, 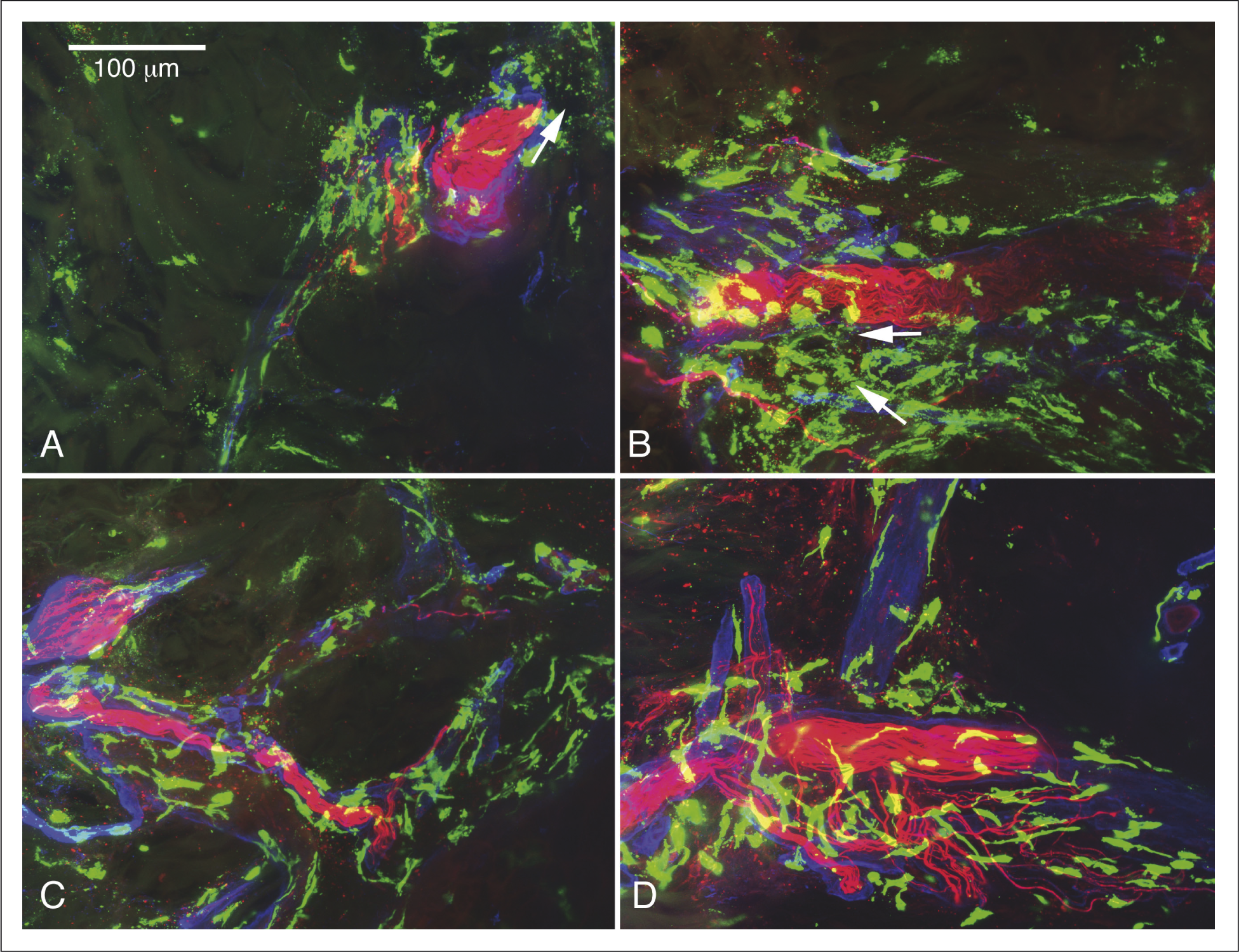
Discussion
In this case of cephalalgia alopecia, biopsy specimens from untreated scalp areas (both symptomatic and not yet symptomatic) showed altered immunohistochemical staining patterns. The fibres were abnormally fine and often fragmented when compared with the more normal staining pattern of the BoNT/A-treated sample. In the SNP, SP was markedly reduced in the untreated samples compared with the treated sample. Interestingly, the CGRP staining of neurons in the SNP was reduced, clustering inconsistently in the untreated sample unaffected by hair loss, but was less reduced in the untreated sample obtained from an area exhibiting alopecia, perhaps reflecting partial recovery in the sample from the symptomatic site. This would be consistent with the early reappearance of pain but not alopecia just prior to sampling. However, in contrast to the SNP, perifollicular SP and CGRP concentrations both appeared reduced in untreated samples when compared with the treated sample. These findings provide preliminary support for the hypothesis that the alopecia that follows neuralgiform pain in this patient may be due to loss of tonic SP and CGRP release from depleted C fibre terminals and subsequent disinhibition of perifollicular antigen presentation and loss of SP-mediated promotion of hair growth. The study is limited by lack of ENF density reference ranges for scalp and our findings need confirmation in subsequent cases.
One possible scenario that explains our results consists of the following. (i) In normal human scalp, trophic release of perifollicular SP stimulates hair growth in resting hair follicles (5) and baseline CGRP release mediates inhibition of antigen presentation (7), preserving immune privilege. (ii) An initial vigorous c-fibre activation, from whatever cause, results in massive SP release from c-fibres, which meditates an inflammatory response (10) including mast cell degranulation (11), which in turn prolongs the activation of inflamed perifollicular nociceptive c-fibres (12) and perhaps recruits neighbouring fibres into the response. (iii) Released SP causes the short-term loss of immune privilege and regression in the hair follicle (11) resulting in hair loss. (iv) Ongoing perifollicular c-fibre activation, whether mediated entirely within the central nervous system or in combination with local mast cell degranulation (12) results in further hair follicle regression, eventual depletion of SP and CGRP within perifollicular terminals and a prolonged delay of hair regrowth. (v) Treatment with BoNT/A inhibits further depolarization-induced release of SP and CGRP from nerve fibre terminals, allowing repletion and ending mast cell degranulation. (vi) There is evidence that BoNT/A does not block low-level trophic release of neuropeptides such as CGRP (13), thus allowing resumption of SP and CGRP baseline regulation of the hair follicle and hair regrowth. (vii) Finally, when the effect of BoNT/A in this patient wanes, the c-fibres are again strongly activated and depolarization-induced release and terminal depletion of SP and CGRP are resumed. As a result, the process of painful inflammatory activation, hair follicle regression and hair loss is repeated.
Since the identification of this patient, we have seen at least three other similar cases of patients with severe medication-refractory neuralgiform head pain and hair loss. In one of these cases, botulinum toxin was administered with hair regrowth and reduction of head pain. In addition, we have observed several other cases of more generalized hair loss in patients with severe primary headache disorders.
Pareja and colleagues (14) recently reported a series of five patients with scalp pain that met criteria for nummular headaches and in whom the painful areas also exhibited skin changes. Three of the five patients developed alopecia and redness at site of pain. As in our patient, a larger area of pain encompassed the area of hair loss or other trophic change. Furthermore, in all cases hair loss followed the onset of pain and the pain had a neuropathic quality. The pain intensity was very severe at times in three of the five reported cases, as it was in our patient. In two of the three patients biopsied, there was a slight cellular infiltration: in one subject a minimal lymphocytic infiltration, in the other a slight monocytic infiltrate. This differed from the rather obvious peribulbar lymphocytic infiltration observed in our case.
What these cases of neuralgiform pain and hair loss represent is unclear. They may constitute a rare clinical syndrome with a single underlying cause; however, we think that it is more likely that the co-occurrence of hair loss and pain reflects physiological interaction between two ectodermally derived structures, skin and nerve. They suggest the importance of neural regulation of dermal structures and the consequences of the loss of that regulation. The cases also remind us that in addition to their afferent sensory function, neural structures play important roles in modulation of the local immune response, both excitatory as in neurogenic inflammation, and inhibitory as we see in the CGRP-mediated inhibition of antigen presentation by Langerhans cells.
Footnotes
Acknowledgement
This study was funded in part by an unrestricted research grant from Allergan Inc.
