Abstract
A male, 34 years of age, suffers from headaches, red and watery eyes. The headaches began in childhood; the frequency of headaches has increased over the years and in the last decade headaches have occurred on a daily basis. If he wakes up before sunrise he feels much better and free of a headache; however, once he continues to sleep during and after sunrise, he suffers from tiredness, headache and nervousness. On magnetic resonance imaging (MRI), benign neuroepithelial cysts or a chronic infarct area was reported at the junction of the left medio-lateral zone of hypothalamus. After repeated MRI examinations, it was decided that the lesion on the left medio-lateral zone of hypothalamus may have disrupted the pineal gland and changed melatonin secretion. It was decided to treat him with 3 mg melatonin daily before going to bed. After a week of treatment, the patient reported that he felt very fresh and was virtually free of headaches.
In mammals, information on environmental lighting conditions that is neurally encoded by the retina is finally converted into nocturnally elevated synthesis of the pineal hormone, melatonin. In the photo-sensitive mammalian pineal gland, the message of darkness seems to depend on the master circadian pacemaker in the suprachiasmatic nucleus (SCN) (1,2). Although the mechanism underlying the circadian variation of migraine is not known, there is increasing evidence that melatonin secretion and pineal function are related to migraine with and without aura (3), menstrual migraine (4) and chronic migraine (5). It has been shown that light exposure suppresses melatonin more markedly in migraineurs compared with controls in symptom-free intervals (6,7).
A number of abnormalities in visual function have been described in migraine, some of which indicate hyperexcitability of the visual pathways (8,9). Clinical observations suggest that hyperexcitability occurs not only during attacks, typically in the form of photophobia, but also between episodes. Furthermore, compared with headache-free subjects, migraineurs are more sensitive to light (10). Since melatonin secretion is inhibited during the day through the retino-hypothalamic pathway (of which a main transmitter is glutamate) and the SCN is also involved in rapid changes of light-induced pineal metabolism, we suggest that hypersensitivity of melatonin suppression to light in migraine is at least in part the result of an increase of glutamate transmission of the pathway (11,12).
It is known that circadian photoreception depends on the ganglion cell layer of the retina. In these cells, photopigments different from opsins have been described and are involved in this non-visual pathway (the so-called ‘non-image-forming system’) that carries photoperiodic information to the SCN of the hypothalamus. These photopigments are melanopsins and chryptochromes (13) and they respond to a narrow band of blue wavelengths. Furthermore, this system appears to send photic information not only to the endogenous clock in the SCN, but also to other brain areas involved in irradiance detection, such as the light-activated pupil response. An abnormality at the level of this irradiance detection system could thus be suspected as a contributing factor in migraine.
Given that migraine attacks are triggered by light and changes in lifestyle, evidence suggests a pivotal role of the SCN and pineal gland, a structure involved both in long-term phase adjustments and rapid changes of light-induced pineal metabolism. Melatonin rhythmicity is now accepted as a core regulator in the pathogenesis of headache related to the sleep–wakefulness cycle or light–dark cycle. In the current case, a patient with sunrise-related headache was evaluated. Because the pineal gland plays an important role in maintaining homeostatic equilibrium in close relationship with changing environmental conditions, the possibility that the pineal hormone melatonin is deficient in migraine is proposed.
Case report
A man, 34 years of age, suffered from headaches, red and watery eyes. The headaches had begun in childhood; the frequency of headaches had increased over the years and over the last decade headaches had occurred on a daily basis. He noted that the headaches began in the temporal region, dispersed towards the apical region of the head and eventually became throbbing in intensity. Sleep deprivation and travel also triggered the headaches.
He also noted increased sensitivity to watching TV, using computers, bright light and hot water shower in the last 4 years. His headaches had become worse along with eye problems (red and watery eyes) after watching TV for a couple of hours or working on a computer. Apart from this, if he awoke before sunrise he felt much better and free of headache; however, once he continued to sleep during and after sunrise, he suffered from tiredness, headache and nervousness. He said that he tested this by sleeping after sunrise and he had the same painful results every time.
He had had a car accident at the age of 7 years, but no complications had been observed in the emergency room. He had had allergic rhinitis starting from age 9 years and allergies to pollen and feathers had been diagnosed. He suffered from frequent upper respiratory tract infections and mouth ulcerations two to three times a year. He was examined neurologically and was found to have a visual defect in the right eye. Visual capacity of left eye was 100%, but was only 60% for the right eye. Examination of the fundus revealed scar tissue on the optic disc and epirential gliosis on the macula. Ophthalmological consultation documented that the eye problems were related to a toxoplasma infection.
Upon routine blood examination, total cholesterol, low-density lipoprotein,
very-low-density lipoprotein and triglyceride were found to be elevated. A variety
of hormone levels, including FT3-4, thyroid-stimulating hormone, cortisol,
follicle-stimulating hormone, luteinizing hormone, prolactin, free testosterone and
total testosterone, were found to be in normal range, although adrenocorticotropic
hormone levels were high. Benign neuroepithelial cysts or a chronic infarct area was
reported at the junction of the left medio-lateral zone of hypothalamus upon
magnetic resonance imaging (MRI) (Fig. 1). Benign neuroepithelial cysts or a chronic infarct area were reported at
the junction of the left medio-lateral zone of hypothalamus upon
magnetic resonance imaging. T1, hypointense; T2, hyperintense
(arrow).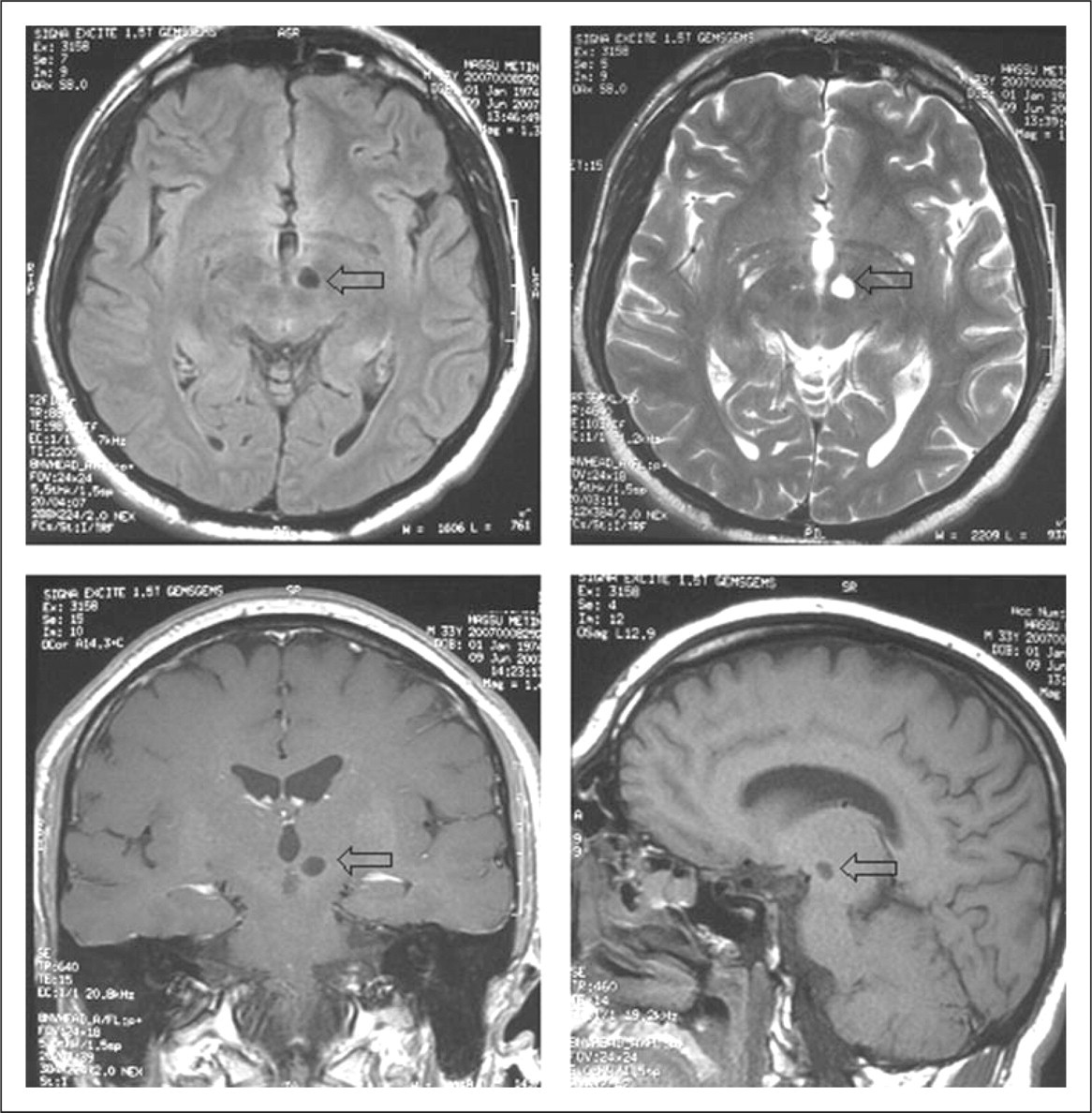
His EEG was normal. The patient had been given selective serotonin reuptake inhibitors, non-steroidal anti-inflammatory drugs, sodium valproate, triptans, etc. at several hospitals over the years, but his complaints remained unchanged.
Follow-up
After repeated MRI examinations, it was decided that the lesion on the left at the junction of the medio-lateral zone of hypothalamus may have disrupted the pineal gland and changed melatonin secretion. The decision was made to treat him with 3 mg melatonin daily before going to bed. After a week of treatment, the patient reported that he felt very fresh, had almost no headache, and a reduced sensitivity to hot water. The eye problems also diminished significantly when he spent time watching TV or using the computer. The patient has been under control for 1 year and has had no problems except for rare light headaches.
Discussion
Day, night and seasons stem from the fact that our planet rotates 360° every 24 h and moves around the sun on an annual basis. This is the case in the circadian rhythms, which are synchronized with the external day and night as zeitgebers. The circadian clock (meaning about a day, from the Latin term circa diem) measures time on a 24-h basis, perhaps in all living creatures. The core oscillator is assumed to be located in the SCN and produces a rhythmic oscillation of about 24 h. The input component allows the clock to be entrained by external cues, synchronizing circadian time with the environment. Finally, the output component is required to use time information for controlling circadian gene expression, physiology and behaviour (14). The overt circadian rhythms of temperature, cortisol, wake–sleep, blood pressure, heart rate and plasma glucose levels to which we are common can be viewed as the hands of this clock system. Further characterization of these molecular cogwheels has revealed the existence of numerous peripheral clocks working in harmony with the central clock (15). Consequently, at least 10% of all cellular transcripts oscillate in a circadian manner, underscoring the global regulatory capacity of the circadian transcriptional machinery (16).
The photoneuroendocrine system (PNES) must be intact for the light–dark cycle for central melatonin production due to the inherent rhythmic activity of the SCN. In the absence of retinal input, cyclic production of melatonin is maintained but it ‘free runs’. With a totally intact pathway, light detected by the retina inhibits the SCN and prevents melatonin synthesis. Plasma glucose concentrations increase just before the onset of the active period known as ‘down phenomenon’ for humans (17,18). Apart from increasing plasma glucose, the PNES renders tissues more tolerant to glucose and increases insulin sensitivity (19) during the active period. At this time, blood pressure, plasma glucose and heart rate are higher but remain in the normal range. There is evidence that this mechanism is driven by neural collections from the SCN, paraventricular nucleus, area postrema and rostral ventrolateral medulla (20–23). Moreover, the PNES, via its direct neuronal connection, shares the light information with the intermediolateral cell (IMLC) column of the spinal cord and, thereby, apprises the pineal gland of the photoperiodic environment; this adds another level of rhythmic metabolic regulation. In reference to man, absence of light signals the end of the active period of day and down-regulation of sympathetic tone, resulting in decreased blood pressure, heart rate and blood glucose (dusk phenomenon). Therefore, the IMLC column of the spinal cord has a dual regulatory effect. In addition to influencing pineal melatonin production, it informs other organs about the autonomic shift. Before any other upcoming central preparation or commands (e.g. changes of plasma hormones or neurotransmitter levels), the IMLC via sympathetic chain keeps peripheral organs updated about pending events.
The exciting point is that the nocturnal third ventricular cerebrospinal fluid melatonin levels in sheep are several orders of magnitude higher than simultaneous concentrations measured in the peripheral blood (24,25). Similar results have been obtained in healthy individuals (26) and patients with meningitis (27). Therefore, melatonin has a variety of physiological regulations in the central nervous system and peripheral tissues. In the current case, the hypothalamic lesion suggests that the disrupted intactness of PNES may prevent melatonin peaking during the dark period. Three milligrams of melatonin may readily restore peak plasma levels during night and may mimic intact pineal melatonin production. The reason for the sunrise-related headaches may be partly explained by the failure of the down phenomenon and sympathetic takeover.
Since melatonin is not per se patentable, the pharmaceutical industry has pursued the development of melatonin analogues which can be patented (e.g. agomelatine and ramelteon). The affinity of agomelatine for cloned human melatonin receptors is similar to the binding affinities of melatonin to the same receptor subtypes (28). Studies confirm that agomelatine is effective in the treatment of depression, including the most severely depressed patients, and has a good safety and tolerability profile (29–31). Among susceptible persons, decreased seasonal exposure to light may mediate seasonal affective disorder (SAD) through phase shifts in circadian rhythms, with resulting alterations in several aspects of serotonin metabolism. However, agomelatine has also been found effective in treatment of SAD (32).
Lack of central effects of melatonin may also be involved in the pathogenesis of several types of headache. For example, melatonin facilitates GABAergic transmission by modulating the GABA receptor (33–35) and enhances the release of β-endorphin, a potent opioid in humans, from the pituitary gland (36). The synthesis of β-endorphin fluctuates rhythmically (37) in accordance with higher pineal melatonin secretion during the night. Future studies are warranted to reveal the exact mechanism of beneficial effects of melatonin in the treatment of several headaches.
