Abstract
We determined the cerebrospinal fluid (CSF) and serum neuron-specific enolase (NSE) concentrations in 19 patients with acute benign headache. All patients had normal neurological examination, CSF and head computed tomography scan. The final diagnoses were: primary thunderclap headache (n = 7), primary exertional headache (n = 3), primary cough headache (n = 1), migraine without aura (n = 4), headache unspecified (n = 2), probable infrequent episodic tension-type headache (n = 1), headache attributed to hypertensive crisis without hypertensive encephalopathy (n = 1). A group of 108 healthy subjects served as controls. CSF NSE concentration was 14.16 ng/ml [95% confidence interval (CI) 11.86, 16.47)] in the headache sample (controls 17.19 ng/ml, 95% CI 16.23, 18.15). Serum NSE concentration was 7.50 ng/ml (95% CI 5.20, 9.80) in the headache sample (controls 8.45 ng/ml, 95% CI 7.67, 9.23). CSF/serum ratio was 2.81 (95% CI 2.21, 3.40) in the headache sample (controls 2.23, 95% CI 2.03, 2.42). Acute benign headache is not associated with neuronal damage as estimated by means of CSF and serum NSE concentration.
Introduction
Neuron-specific enolase (NSE) is the γγ-isoform of the glycolytic enzyme 2-phospho-D-glycerate hydrolase. It is contained in relatively large amounts in neurons, peripheral nervous system tissue and neuroendocrine cells and it is therefore one of the most investigated biochemical markers of nervous tissue damage. Increased NSE levels in serum, cerebrospinal fluid (CSF) or both have been reported in several neurological diseases, including hypoxic brain injury (1–4), Creutzfeldt–Jakob disease (5), acute ischaemic stroke (6–8), head injury (9–11) and status epilepticus (12, 13).
Few papers (14–16) have reported data about CSF NSE in headache patients, with conflicting results: according to Royds et al. (15) and Vermuyten et al. (16), CSF NSE was elevated in a minority of patients with headache, whereas it was normal according to Jacobi et al. (14). However, these reports included very small numbers of patients and clinical details about headache were not provided.
We determined CSF and serum NSE concentrations, as indicators of neuronal damage, in a group of patients with acute benign headache.
Materials and methods
Patients
Patients referred from the Emergency Department for neurological evaluation of acute headache were eligible, provided that neurological examination, brain computed tomography (CT) scan and lumbar puncture were normal. Additional investigations (magnetic resonance imaging of the brain, cerebral angiography and others) were performed in single cases, as indicated on the basis of clinician judgement.
As regards the diagnosis of headache, the clinical data were independently reviewed by two neurologists (M.C. and L.V.), who formulated a diagnosis for each patient applying the International Classification of Headache Disorders (2nd edn). The patients' follow-up was based on the clinical records of Ravenna Local Health Unit.
Controls
The control group consisted of 108 healthy subjects (40 women, mean age 68.7 ± 16.7 years, range 19–92) in which CSF and serum samples for NSE determination were obtained during spinal anaesthesia for various diseases; details about this group have been reported elsewhere (17).
Neuron-specific enolase
NSE was determined by means of an immunofluorescent assay (monoclonal antibody against NSE) with time-resolved amplified cryptate emission (TRACE) technology (Kryptor-NSE; BRAHMS, Hennigsdorf, Germany). Details about NSE determination have been previously published elsewhere (17). CSF and serum samples were collected simultaneously; blood-contaminated samples were discarded.
Statistics
Univariate
Results
A summary of demographic features of the patients is presented in the Table 1: 19 patients (16 women, mean age 47.8 ± 16.3 years, range 23–78) were included. After discussing disagreements the final diagnoses were as follows: primary thunderclap headache (n = 7), migraine without aura (n = 4), primary exertional headache (n = 3), primary cough headache (n = 1), headache unspecified (n = 2), probable infrequent episodic tension-type headache (n = 1), and headache attributed to hypertensive crisis without hypertensive encephalopathy (n = 1).
Demographic, clinical data, cerebrospinal fluid and serum neuron-specific enolase concentrations in 19 patients with acute benign headache

Deceased at follow-up.
NA, Not available.
The mean time interval between headache onset and brain CT scan was 39.4 ± 41.1 h (range 4–168), whereas the interval between headache onset and CSF examination was 64.1 ± 55.8 h (range 2–216). Two patients (cases 9 and 12) also underwent magnetic resonance study of the brain; cerebral angiography was performed in one patient (case 2); additional blood tests to rule out vasculitis were performed in three other patients (cases 9, 14 and 19). All such additional tests gave normal results.
CSF and serum NSE values of each patient are also reported in the Table 1 and plotted in Fig. 1; in Fig. 1 the CSF/serum NSE ratio is also plotted. CSF NSE was available for 18 patients and serum NSE for 10 patients; CSF/serum ratio could be calculated for nine patients.
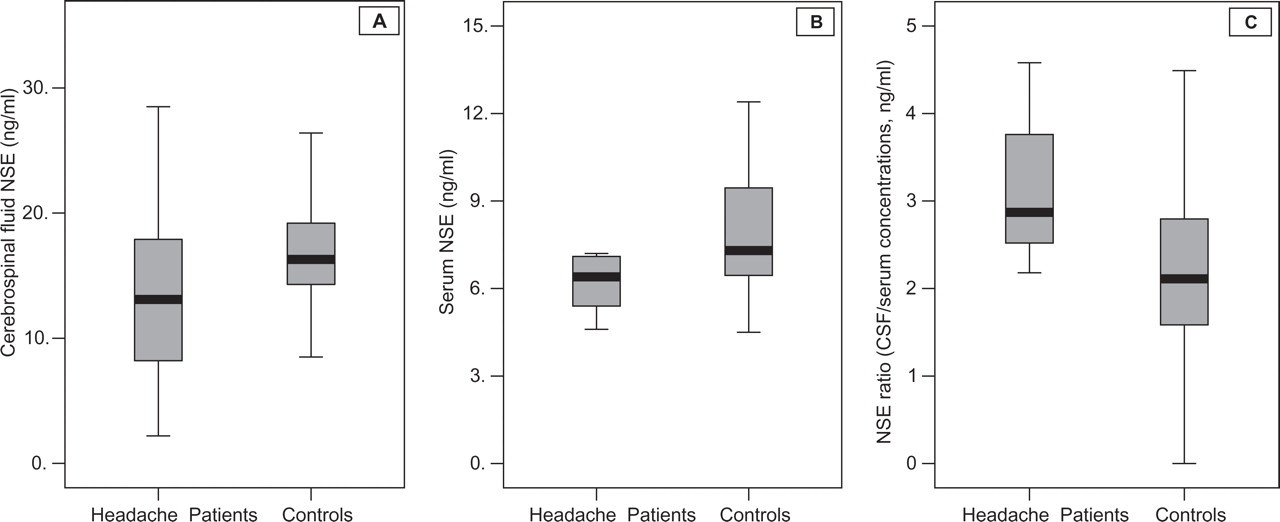
Boxplot showing distribution of cerebrospinal fluid (CSF) neuron-specific enolase (NSE) (A), serum NSE (B) and NSE ratio (CSF/serum concentrations) (C) in headache patients and healthy controls.
After correction for age as covariate, the mean CSF NSE concentration was 14.16 ng/ml [95% confidence interval (CI) 11.86, 16.47] in the headache sample vs. 17.19 ng/ml (95% CI 16.23, 18.15) in the normative group; mean serum NSE concentration was 7.50 ng/ml (95% CI 5.20, 9.80) in the headache sample vs. 8.45 ng/ml (95% CI 7.67, 9.23) in the healthy control group. CSF/serum ratio was 2.81 (95% CI 2.21, 3.40) in the headache sample (in the normative group 2.23, 95% CI 2.03, 2.42).
A follow-up was available for 18/19 patients (mean follow-up duration 74.9 ± 31.7 months; range 17–123). At follow-up two patients had deceased: in both cases death was unrelated to neurological causes (case 5, death caused by rectal carcinoma; case 10, death caused by non-Hodgkin's lymphoma; the time interval between neurological observation for headache and death was, respectively, 17 and 23 months). The other 16 patients were alive at follow-up; none had been subsequently admitted because of subarachnoid haemorrhage or any other neurological disease.
Discussion
We examined a group of patients whose common clinical features were the following: acute headache which prompted evaluation in the Emergency Unit, normal neurological examination, normal findings at cerebral CT scan and CSF examination, absence of brain haemorrhage or other cerebral events at follow-up. In these patients we determined the serum and CSF NSE values while the headache was in the acute phase.
Some articles have reported, with conflicting results, about NSE values in headache patients, but in these studies headache patients were included within heterogeneous groups of neurological diseases (14–16). CSF γ-enolase was increased in approximately half of the cases of migraine (three out of seven patients) of Royds et al. (15) and in two out of nine cases of Vermuyten et al. (16). In contrast, CSF NSE was normal in a group of 24 patients with ‘functional disorders’, including headache, reported by Jacobi et al. (14). Remarkably, all these reports concern very small numbers of headache patients and clinical details about headache are not provided.
In our group of patients, acute headache proved not to be related to brain damage, as determined by means of extensive clinical investigations and follow-up. The CSF and serum NSE concentrations evaluated during a short time interval from headache onset were within normal range compared with a normal population. Based on these findings, we conclude that acute benign headache is not associated with neuronal damage as determined by means of CSF and serum NSE concentrations.
