Abstract
The presence of cutaneous allodynia may predict response to triptans. Identical randomized double-blind studies were conducted comparing the efficacy of rizatriptan 10 mg or placebo administered within 1 h of headache onset, while pain was mild. The primary endpoint was freedom from pain at 2 h. Presence of symptoms suggesting cutaneous sensitivity (SCS) at baseline and at 2 h post-treatment was recorded. Before treatment, 29% of rizatriptan patients and 22% of placebo patients reported SCS. At 2 h, the percentage of patients with SCS was significantly decreased with rizatriptan. The presence of SCS pre-treatment was not predictive of response to rizatriptan. Most patients with SCS at 2 h were non-responders. Early treatment with rizatriptan significantly reduced the percentage of patients with SCS at 2 h. The presence of SCS at baseline did not predict pain-free response, but presence of SCS at 2 h correlated with lack of a 2-h pain-free response.
Introduction
Symptoms of cutaneous allodynia, described as pain resulting from the application of a non-noxious stimulus, such as heat, cold or pressure, to normal skin (1), appear during the course of a migraine attack in up to 79% of patients (2). Cutaneous allodynia represents the sensitization of central trigeminal neurones that receive convergent input from the dura and skin and typically develops 20 min to 2 h after the onset of the migraine attack (3). The presence of cutaneous allodynia has been demonstrated to decrease the efficacy of triptan therapy, and studies of early treatment during the migraine attack before the development of allodynia have yielded superior results to those involving late treatment when the pain is moderate to severe (4).
A typical assessment of cutaneous allodynia includes intensive quantitative sensory testing using specialized equipment to measure a patient's pain thresholds to thermal and mechanical stimulation of the skin during a migraine-free period and again during a migraine attack. Patient-reported symptoms of cutaneous sensitivity (SCS) may represent less intensive, but useful, surrogates of cutaneous allodynia. It is not currently known whether patient-reported symptoms of cutaneous sensitivity respond to effective therapy, like other associated symptoms, nor if their presence predicts response to acute migraine therapy. We examined these questions as part of the rizatriptan Treat A Migraine Early (TAME) studies, in which patients were instructed to treat a migraine attack within 1 h of onset, while the pain was still mild (5). The results of both studies are presented separately; data were not pooled for these analyses.
Methods
The rizatriptan TAME (Treat A Migraine Early) studies (Protocols 065 and 066) were multicentre, randomized, double-blind studies in which migraineurs were instructed to treat their headache with rizatriptan 10 mg or placebo within 1 h of headache onset, while pain was mild. Study design details have been published (3) and will be summarized here. The studies tested the hypotheses that rizatriptan was superior to placebo, as measured by the proportion of patients with freedom from pain at 2 h after treatment and the proportion of patients with 24-h sustained freedom from pain.
Patients were 18 years of age or older with at least a 6-month history of migraine with or without aura by International Headache Society criteria and with a history of attacks that were typically mild at onset. The primary endpoint was 2-h freedom from pain. A single dose of study medication was used to treat the headache. Rescue medication was permitted 2 h after study medication for non-responsive or recurrent headache. Patients who participated in TAME 1 were not eligible to participate in TAME 2.
Patients assessed pain severity and the presence or absence of SCS at baseline (immediately before taking study medication) and at 1 h and 2 h after treatment and recorded this information in the patient diary. Symptoms of cutaneous sensitivity were defined as: sensitivity while brushing hair; sensitivity while showering; sensitivity while shaving or with any facial contact; discomfort from rings; discomfort from earrings; discomfort from necklaces; discomfort from eyeglasses; and discomfort from contact lenses.
Patients were classified as having SCS if they answered ‘yes’ to having one or more of the symptoms listed above; individual symptoms were not captured. Treatment groups were compared with respect to the proportion of patients with a presence of SCS at 1 and 2 h post-dose via a logistic regression model with factors for treatment group and centre. The consistency of treatment effect of rizatriptan vs placebo with respect to freedom from pain at 2 h post-dose by absence or presence of SCS at baseline was also explored and tested via a logistic regression model with factors for treatment group, SCS absence or presence at baseline, and treatment by SCS absence or presence at baseline interaction. To assess the predictive value of SCS at baseline, the proportion of patients with pain freedom at 2 h post-dose by the presence or absence of SCS at baseline was examined and tested for its predictive value (independent of treatment group) in the context of a logistic regression model with factors for treatment group, centre, and SCS absence or presence at baseline. Additionally, for all binary outcome measures, percentages of patients and corresponding 95% confidence intervals were calculated and tabulated by treatment group. The results of both studies are presented separately; data were not pooled for these analyses. The percentage of patients with SCS and other migraine-associated symptoms at 2 h who also had pain freedom at 2 h is summarized for both studies individually (SCS) and pooled; no statistical testing was performed.
Results
Baseline characteristics are shown in Table 1. Symptoms of cutaneous sensitivity were present pre-treatment in 29% of patients in both rizatriptan and placebo groups in TAME 1 and in 22% of both groups in TAME 2.
Patient baseline characteristics

The percentage of patients with SCS after treatment was significantly decreased for rizatriptan vs placebo at 2 h in TAME 1 and at 1 and 2 h in TAME 2 (Fig. 1). Pain freedom at 2 h was significantly greater with rizatriptan than with placebo in both studies (Fig. 2). The treatment effect was similar regardless of the presence of SCS at pre-treatment baseline. However, most patients who had SCS at 2 h were non-responders in terms of pain freedom (TAME 1, R 7/47(14.9%) and P 4/39(10.3%); TAME 2, R 8/42(19.1%) and P 2/37(5.4%); pooled, R 15/89(16.8%) and P 6/76(7.9%) were pain free at 2 h), as were patients who had other migraine-associated symptoms present at 2 h (Fig. 3). The presence of SCS pre-treatment was not predictive of response to treatment with respect to pain freedom at 2 h (TAME 1, OR = 1.25, 95% CI = 0.81, 1.93, P = 0.307; TAME 2, OR = 0.85, 95% CI = 0.55, 1.32, P = 0.479; Fig. 4).
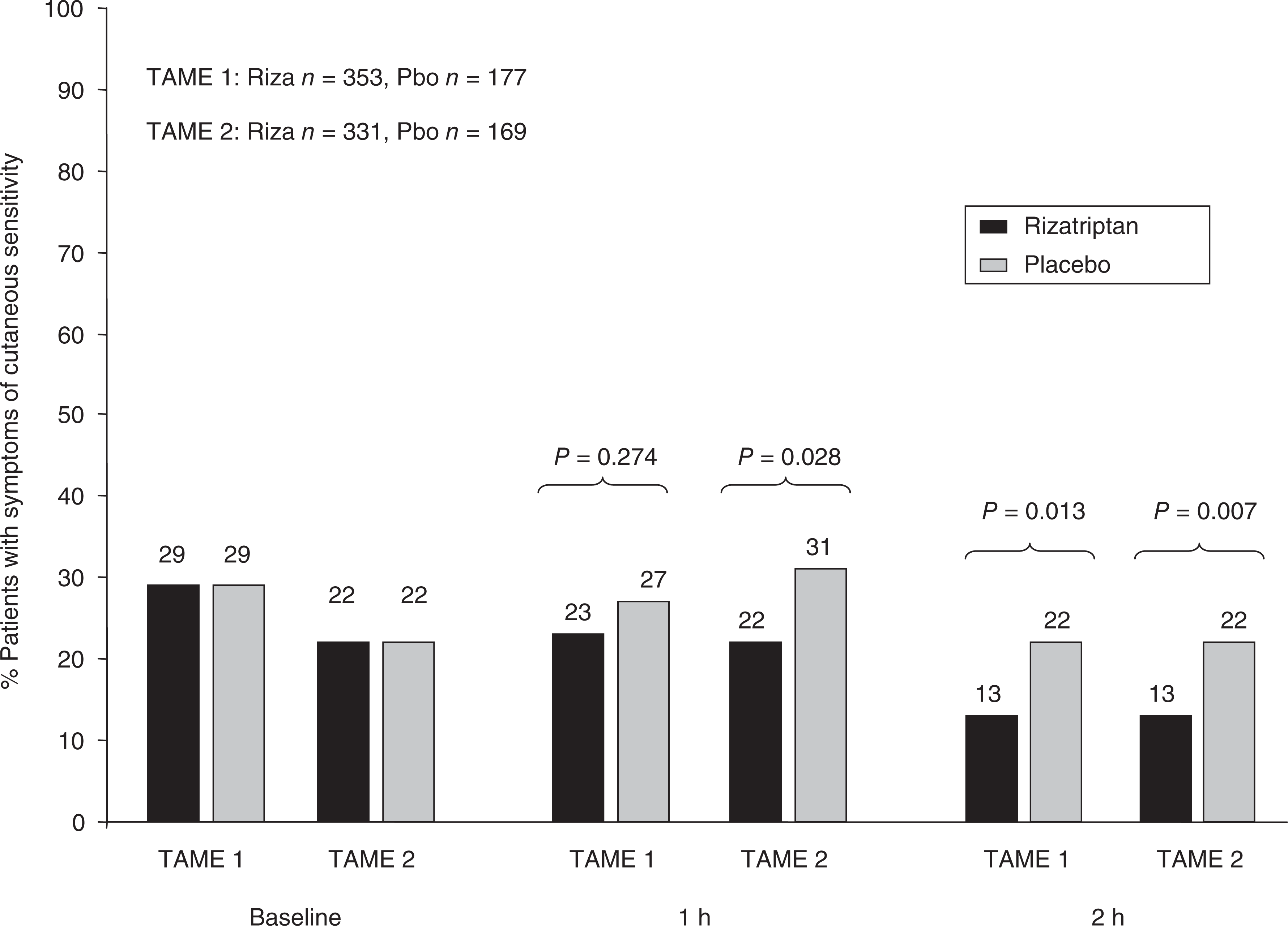
Presence of symptoms of cutaneous sensitivity. Bar graph showing the percentage of patients who reported having symptoms of cutaneous sensitivity (SCS) at baseline and at 1 and 2 h after taking rizatriptan 10 mg (black) or placebo (grey) in both TAME 1 and TAME 2.

2-h pain freedom overall and by symptoms of cutaneous sensitivity (SCS). Bar graph showing the percentage of patients who reported being pain-free at 2 h after taking rizatriptan 10 mg (black bars) or placebo (grey bars) in the overall population and in those with or without SCS at baseline.

2-h pain freedom in patients with or without migraine-associated symptoms at 2 h. Bar graph of pooled TAME 1 and TAME 2 data showing the percentage of patients who reported being pain-free at 2 h after taking rizatriptan 10 mg (black bars) or placebo (grey bars) when the indicated migraine-associated symptom was present (yes) or absent (no) at 2 h after treatment.

2-h pain freedom by baseline symptoms of cutaneous sensitivity (SCS) independent of treatment group (treatment groups pooled). Bar graph showing the percentage of patients who reported being pain-free at 2 h post-dose in those with or without SCS at baseline.
As reported previously, the incidence of adverse experiences (AEs) in TAME 1 and TAME 2 was greater with rizatriptan compared with placebo, with the most common drug-related AEs being dizziness, dry mouth and paraesthesia. These AEs are consistent with those previously reported for rizatriptan and the triptan class (5).
Discussion
Identification of migraine patients who experience cutaneous allodynia as part of their migraine attacks may be important to optimize acute therapy. In patients with allodynia, an increased interval between headache onset and triptan administration has been associated with decreased pain-freedom rates (4). Although true cutaneous allodynia is best established through objective, clinician-centred measures, these methods are expensive, time-consuming, and not amenable to patient self-assessment at the time of an attack. Thus, other assessment methods, such as questionnaires, are being pursued as surrogate markers for cutaneous allodynia (6).
The TAME studies used a patient-centred questionnaire as a surrogate for more objective measures of cutaneous allodynia. We asked patients to indicate the presence or absence of symptoms of cutaneous allodynia at pre-treatment baseline and post-treatment to assess these symptoms as additional migraine-associated symptoms, like photophobia or nausea, and to assess these symptoms as predictors of treatment response to rizatriptan or placebo therapy. Our questionnaire included seven of the 12 questions tested by Jakubowski et al. (6) to identify patients with allodynia; their questionnaire also included questions on sensitivity to pulling hair back, tight clothes, heat, cold, and resting your face on the pillow on the side of the headache. Jakubowski et al. (6) used one or more positive responses to the questionnaire as the presence of symptoms of cutaneous allodynia, as we did in the current study. In the Jakobowski study, however, patients answered the questionnaire based on recall of cutaneous allodynia symptoms during past migraine attacks, and whether or not they actually had symptoms of allodynia during a migraine attack was verified with quantitative sensory testing during a subsequent migraine headache. In the current study, we asked patients to answer the questionnaire when they were actually experiencing the symptoms immediately before they took their study medication and again 2 h after treatment to see if these symptoms resolved with treatment. As Jakubowski et al. (6) showed that most of the patients with positive responses to their questionnaire were later shown to be allodynic by quantitative sensory testing during a migraine attack, we have confidence that our questionnaire also correctly identified similarly allodynic patients.
Early treatment with rizatriptan significantly reduced the percentage of patients with SCS at 2 h. In this way, SCS responded to treatment with rizatriptan in the same way as other migraine-associated symptoms studied in the TAME studies, such as photophobia (from 67% to 23% vs 65% to 44% with placebo in TAME 1, with similar values seen in TAME 2) and phonophobia (51% to 18% vs 49% to 35% with placebo in TAME 1). The presence of SCS at baseline, as a surrogate for cutaneous allodynia, did not predict response to acute therapy in this study. With the sample sizes from TAME 1 and TAME 2, there was at least 80% power to show treatment differences of 15% or more with respect to 2-h pain freedom comparing patients with and without SCS before treatment. As seen with other migraine-associated symptoms, the presence of SCS at 2 h after treatment, however, was correlated with lack of response to acute therapy in terms of pain freedom: most patients with SCS at 2 h did not achieve pain freedom at 2 h.
In the current study, considerable attention has been directed to interpreting the patient-reported symptoms studied as surrogate markers for cutaneous allodynia and, as such, as markers of clinical response to triptans. This study casts doubt on the assertion that triptan response can be predicted by questioning patients about symptoms suggesting cutaneous allodynia. In fact, many of these symptoms occur early in migraine and appear to respond to triptan interventions in much the same way as other migraine-associated symptoms. Although it cannot be ruled out that the clinical resolution of these symptoms is a unique response to rizatriptan, the authors believe that this response more likely reflects the action of the class of triptans, which supports the utilization of triptans in the ‘early’ treatment of migraine regardless of whether symptoms suggesting cutaneous allodynia are present or not.
As an initial attempt to use patient-centred assessments of cutaneous allodynia, there exist several limitations. First, patients were presented with a list of symptoms commonly associated with cutaneous allodynia; a patient was considered to have allodynia if any symptom was present at pre-treatment baseline. We weighted all the symptoms the same on the questionnaire when, in reality, some individual symptoms may be more predictive than others if we had analysed them within a logistic regression analysis. Also, using different cut-off points for SCS (e.g. two or more positive constitutes a positive response, three or more, etc.) may have yielded different results. In addition, no attempt was made to associate the presence of SCS with objective, clinician-centred assessments or neurophysiologic testing for cutaneous allodynia; however, patients were asked to complete the questionnaire while they were experiencing these symptoms, and, as discussed above, patients with positive answers to the Jakubowski questionnaire subsequently tested positive for allodynia by sensory testing (6). Future investigations can be strengthened by a more detailed patient assessment (e.g. patient scoring of individual symptoms, as opposed to the aggregate approach used in this study) and by the addition of clinician-centred assessments of allodynia in those patients reporting these symptoms by history at study entry.
