Abstract
No evidence is available to show that nasal congestion is a manifestation of exposing an individual to high altitude and hypoxia. Since both nasal congestion and high-altitude headache are vasogenic, we explored whether there is a coincidence between these two symptoms. A prospective observational study was carried out on a cohort of 118 adults (>18 years old) in a mountain clinic at 3450 m. After 24 h of ascent, an interview was held to ask if each individual experienced acute mountain sickness symptoms (headache, etc.) and nasal congestion. Sixty-six (55.9%) individuals mentioned headache within 24 h after ascent and nasal congestion was reported by 34 (28.8%) individuals. There was a reverse association between headache and nasal congestion (P < 0.001). In conclusion, there is a reverse association between altitude headache and nasal congestion, probably as result of contradictory autoregulation effects or exaggerated sympathetic activity.
Introduction
Altitude illness results from hypobaric hypoxia at altitudes greater than 2000 m above sea level (1). The severity of acute mountain sickness (AMS), the most prevalent form of altitude illness, correlates with altitude and rate of ascent (2). Initial manifestations include headache, lassitude, dizziness, nausea and dyspnoea (1). Later, there is anorexia, insomnia, increased dyspnoea and shortness of breath at rest, and social withdrawal. Ascending too rapidly, overexertion within 24 h of ascent, inadequate fluid intake, hypothermia, tobacco and consumption of alcohol or other sedatives can trigger and exacerbate AMS (1).
Headache is the most prevalent symptom of AMS (3, 4). Older age appears to offer some protection, whereas headaches are more severe in women and persons with headaches in daily life (5). Why headache is developed is not fully understood but brain oedema and cerebral vasodilation due to impaired autoregulation have been suggested as the probable causes (6). Alteration in permeability of the blood–brain barrier because of hypoxia-induced chemical mediators such as bradykinin, histamine, nitric oxide, arachidonic acid and vascular endothelial growth factor also might be included as a contributing factor (7, 8).
There is no evidence showing that nasal congestion is one of the manifestations of exposing an individual to high altitude and consequent hypoxia. In contrast, in our experience in a mountain clinic at 3450 m above sea level, nasal congestion has been one of the most frequent complaints. This is a prospective study to estimate the prevalence of nasal congestion during 24 h after ascent of healthy non-professional individuals. Furthermore, in view of the fact that both nasal congestion and high-altitude headache are vasogenic, we wished to determine whether there is a coincidence between these two symptoms.
Methods
Study setting
An observational study was carried out on a cohort of 118 adults in Tochal mountain hotel clinic, Tehran, Iran, from 21 July to 2 October 2006. Tochal hotel is located at an altitude of 3450 m above sea level and designed to host ordinary non-professional hotel guests. Individuals were transferred from a height of 1700 m to 3450 m by cable car within 45 min. The temperature was 22–27°C in different areas of the hotel within the days of the study performance.
Participants
All individuals were >18 years of age and had been living at an altitude of 1400–1650 m for at least 1 week before coming to the hotel. Guests with documented coronary artery disease (CAD), ischaemic heart disease, cerebrovascular disease, chronic obstructive pulmonary disease, asthma, migraine or pregnancy were excluded from the study. Both current upper respiratory tract infection and history of relapsing allergic rhinitis were also exclusion criteria. All study individuals gave oral consent to participate. Of the 132 guests accommodated in the hotel within the study period, 10 were <18 years old, two had CAD, one had migraine and one refused consent, leaving 118 to participate in the study. Participants were asked not to drink alcohol, smoke cigarettes or take self-prescribed drugs and analgesics during the study.
Assessments
Age, gender, social habits and any previous serious or chronic disease were recorded. Vital signs including heart rate, respiratory rate and blood pressure were measured by a general physician during the first hour of ascent after letting any individual rest in a supine position for at least 10 min. After 24 h of ascent, a face-to-face interview was held to ask each individual if they experienced AMS symptoms including headache, dizziness and light headedness, anorexia, nausea and vomiting, insomnia, fatigue and weakness, regardless of the fact that they had come to the mountain clinic during their 1-day stay in. Moreover, nasal congestion and blood-stained nasal discharge were recorded for each participant. AMS incidence was set at a Lake Louise acute mountain sickness score of ≥3 with headache and at least one other symptom (1991 International Hypoxia Symposium).
Statistical analysis
Data were entered into the SPSS 13.0 statistical package (SPSS Inc., Chicago, IL, USA). Using χ2 test (Fisher's exact test if appropriate), possible associations were analysed between categorical variables and dichotomous variable of nasal congestion incidence. To apply parametric tests dealing with continuous variables, normality of distributions was evaluated using the one-sample Kolmogorov–Smirnov test. Independent sample t-test (Mann–Whitney if appropriate) was used to determine any differences of mean between two groups of headache and nasal congestion occurrence. Stepwise logistic regression analysis was performed for factors associated with the altitude headache to calculate related odds ratios (OR). Mean values were reported as ±1 SD and 95% confidence interval (CI) and median was reported with interquartile range. Significance was assigned to values of P < 0.05.
Results
Of 118 individuals, 60 (50.8%) were females and 58 (49.2%) males. Mean age was 35.21 ± 10.21 years (95% CI 33.35, 37.07, range 21–65) and there was no significant difference between the mean ages of females and males. None of the participants had been acclimatized before ascent. Fifty-two (44.1%) individuals did not experience headache during the 24 h after ascent and stay in, whereas 66 (55.9%) individuals mentioned mild to severe headache at the moment. Age, gender, social habits (alcohol consumption and/or smoking cigarettes) and past serious or chronic disease were not associated with headache. Higher primary measured systolic blood pressure (P = 0.045) and heart rate (P = 0.001) were associated with a higher risk of developing headache (Table 1).
Vital sign differences in headache
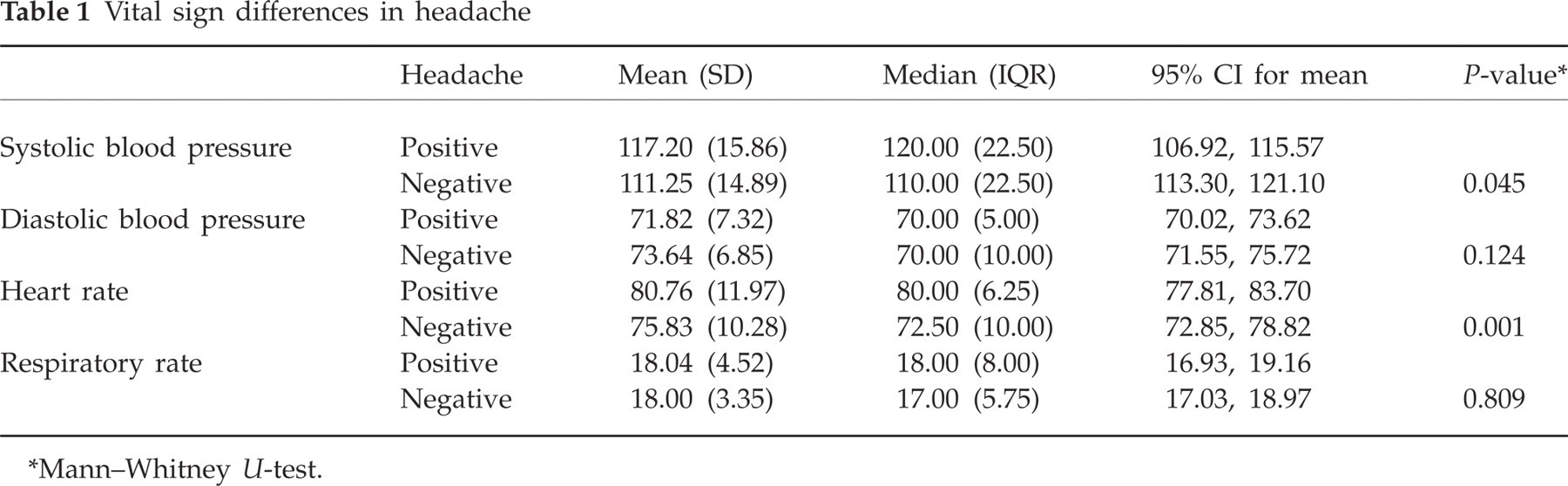
Mann–Whitney U-test.
Nasal congestion was reported by 34 (28.8%) individuals and 84 (71.2%) did not suffer from nasal congestion. All cases of nasal congestion was responsive to phenylephrine. Age, gender, social habits (alcohol consumption and/or smoking cigarettes) and past serious or chronic disease were not associated with nasal congestion. Blood-stained nasal discharge was reported in eight (6.78%) participants, who were all also nasal congestive. Other than headache, higher primary measured systolic blood pressure (P = 0.024) and heart rate (P = 0.002) were associated with a lower risk of developing nasal congestion, while diastolic blood pressure and respiratory rate showed no difference (Table 2).
Vital sign differences in nasal congestion

Mann–Whitney U-test.
Twenty-four (70.6%) participants with nasal congestion did not have headache, whereas 28 (33.3%) nasal congestion-free individuals did not indicate any headaches within 24 h of ascent, showing a significant reverse association between headache and nasal congestion (P < 0.001). Heart rate, systolic blood pressure and nasal congestion were entered into the regression analysis as independent variables. On logistic regression, only nasal congestion was detected to be an independent predictor of altitude headache (OR 0.19, 95% CI 0.07, 0.50; P = 0.001), whereas both heart rate and systolic blood pressure were excluded. An overall correctly predicted value of 67.3% was calculated.
Based on Lake Louise criteria, AMS diagnosis was made in 50 (42.4%) participants within 24 h. Clinical examination revealed no symptoms or signs of high-altitude pulmonary oedema, ataxia or impaired consciousness (high-altitude cerebral oedema).
Discussion
The term ‘high-altitude illness’ is used to describe the cerebral and pulmonary syndromes that can develop in unacclimatized persons shortly after ascent to high altitude (6). Nasal congestion has not previously been shown to be a manifestation of exposure to high altitude and consequent hypoxia. In our investigation, nasal congestion involved 28.8% of individuals who had recently come to a high altitude of 3450 m. Since high-altitude hypoxia leads to conscious or unconscious hyperventilation, nasal congestion might simply relate to hypoxia, hyperventilation and subsequent hypocapnia. One possible explanation is increased nasal resistance secondary to low arterial CO2 pressure levels. Another possible explanation is reduced alae nasae muscle activity secondary to the reduced activity of serotonin-containing raphe neurons (9). While it is well known that environmental conditions (such as cold and dry air) are responsible for nasal congestion (10), the dry (but not cold) weather of Tochal mountain within months of the study might lead to nasal congestion.
In addition, we tested the hypothesis that high-altitude headache and nasal congestion might be associated, so as to generalize the pathophysiology of this sort of headache to high-altitude nasal congestion, whereas both symptoms are attributed to vasodilation. Surprisingly, we found out that there is a reverse association between nasal congestion and headache at high altitude, leading us to hypothesize that parallel autoregulation and pathophysiological mechanisms probably result in contradictory vasoreactivity inside the brain and upper respiratory tract.
Neuroimaging has demonstrated vasogenic oedema in individuals with moderate-to-severe acute mountain sickness or high-altitude cerebral oedema (11). Haemodynamic factors such as sustained vasodilation, impaired cerebral autoregulation and elevated cerebral capillary pressure probably contribute to the formation of oedema (6). Furthermore, these changes are not confined to those with AMS, and current evidence confirms that all people have some degree of brain swelling on ascent to high altitude which can be a potential source of altitude headaches (12). Hypoxia-induced biochemical alteration of the blood–brain barrier may also be important. Possible mediators, some triggered by endothelial activation, include vascular endothelial growth factor, inducible nitric oxide synthase (iNOS) and bradykinin (6). High-altitude headache may develop when the trigeminovascular system is activated at high altitude by both chemical and mechanical stimuli [e.g. nitric oxide (NO) and vasodilation] (13). In support of a role for NO in the genesis of headache there is a report showing that inhibition of NOS improves tension-type headache (14).
Treg-produced immunosuppressive cytokines interleukin (IL)-10 and transforming growth factor(TGF)-β and Th1-produced interferon (IFN)-γ seem to be the probable agents which inhibit allergic nasal congestion (15). Both IL-10 and TGF-β have been shown to have protective characteristics against vasodilation and brain oedema (16–20). In contrast, IFN-γ induces a marked increase in release of arterial-derived NO, which contributes to vasodilation and possible brain oedema (21). In addition, current evidence shows significant changes in IFN-γ levels during acute and chronic exposure to high altitude (22).
Similar to previous studies (23–25), our findings show an association between heart rate, systolic blood pressure and high-altitude headache (Table 1), but, in contrast, a reverse association between heart rate, systolic blood pressure and nasal congestion (Table 2). Increased sympathetic activity during acute exposure to hypobaric hypoxia seems to be the main reason for higher heart rate and systolic blood pressure (23). Autonomic cardiovascular dysfunction and an exaggerated individual chemoreflex vasoconstrictive response to hypoxia that might prevent nasal resistance (25) have been hypothesized as having a potential role in AMS and development of altitude headache (26). In view of the fact that imbalance of the sympathetic and parasympathetic nervous systems, with parasympathetic hyperactivity and sympathetic hypoactivity, results in nasal congestion (25), hypoxic altitude nasal congestion might be considered as a non-allergic/non-infectious rhinitis.
In conclusion, nasal congestion may develop at high altitude, probably as a consequence of hypoxia, whereas it is reverse-associated with altitude headache. Similar autoregulation might play a contradictory vasoreactive role within the upper respiratory tract and brain. Further studies are needed therefore to clarify cytokines' role in AMS and high-altitude headache. Moreover, hypoxia-induced sympathetic activity may explain the reverse association between altitude headache and nasal congestion.
