Abstract
We describe two adults with stroke-like migraine attacks after radiation therapy (SMART syndrome), propose revised diagnostic criteria, and review the previously reported patients. ‘SMART’ is an acronym for a newly recognized syndrome which occurs as a delayed consequence of cerebral irradiation and consists of prolonged, unilateral, migrainous neurological symptoms with transient, dramatic cortical gadolinium enhancement of the affected cerebral hemisphere and is sometimes punctuated by generalized seizures and ipsilateral EEG slowing. Although the neurological symptoms can last for weeks, full recovery occurs. An appropriate evaluation should exclude alternative explanations.
Introduction
In 1995, Shuper et al. reported four children with ‘complicated migraine-like episodes’ that began 1–3 years after receiving cranial irradiation (1). Magnetic resonance imaging (MRI) studies of these patients were interpreted as revealing no acute changes, but use of gadolinium contrast was not mentioned. Conventional angiography triggered attacks in three of the four, similar to migraine or hemiplegic migraine patients (2). Neurological symptoms lasted less than 1 h, while headache lasted 3–24 h, such that the attacks were consistent with migraine with aura (3).
In 2000, Friedenberg and Dodick reported a 51-year-old woman with a history of migraine with aura and a prolonged episode of right posterior hemisphere dysfunction accompanied by a generalized seizure, after which she had right parieto-occipital cortical gadolinium enhancement which was attributed to ‘migralepsy’ (4).
In 2002, Murthy and Cohen reported a 12-year-old boy with prolonged migraine with aura 2.5 years after surgical resection and radiation therapy for an incidentally discovered medulloblastoma (5). He had a 6-week episode of left-sided headache, confusion, aphasia, hemiparesis and MRI abnormalities attributed to seizure activity
In 2003, Bartleson et al. re-reported the Friedenberg and Dodick patient and a second patient and suggested the existence of a syndrome manifesting years after cranial irradiation (6).
The patients in this study were reported in part at the American Academy of Neurology Annual Meeting in Miami, FL, on 13 April 2005 (7).
Case reports
Patient 1
A 45-year-old right-handed, adopted woman with a history of a low-grade right cerebellar astrocytoma, treated with gross total resection and subsequent involved field radiation in 1972 (cGy dose unknown), presented in October 2003 with her fifth episode of unilateral hemispheric dysfunction associated with headache. She had subacute left hemineglect, confusion, a left homonomous hemianopsia, visual hallucinations and a throbbing right parietal headache which lasted 1 week.
Prior episodes had occurred in 1979, 1986, 1997 and 2001, each lasting days to weeks. Different attacks involved different sides of the body. Ictal EEGs had shown temporo-parieto-occipital focal polymorphic delta slowing without epileptiform abnormalities that alternated sides with different attacks.
Since radiation, she also had frequent migraines during the 1980s and 1990s consisting of severe, throbbing headaches lasting hours, occasionally preceded by an aura of scintillating scotomas. She also had a diagnosis of partial seizures starting after radiation, but these stopped after treatment with carbamazepine. While EEG findings at that time are not known, several subsequent EEGs have not shown epileptiform abnormalities.
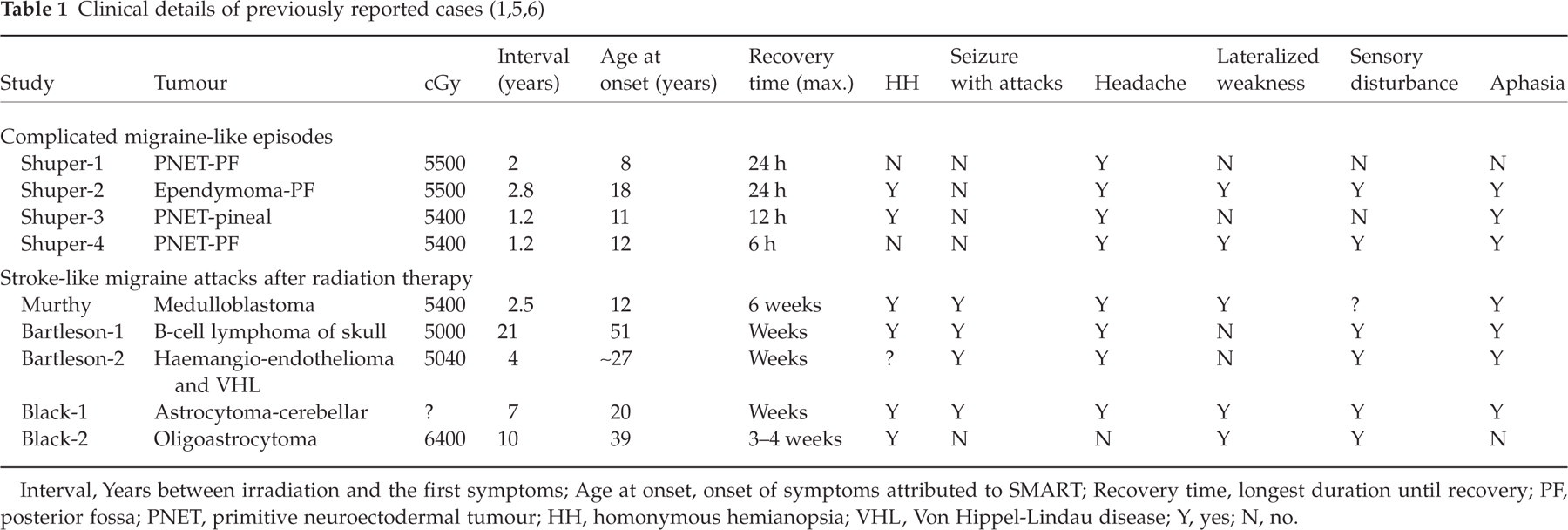
An MRI during the prior spell in 2001 had demonstrated intense gyriform enhancement in the left hemisphere, without T2 signal changes and without respecting any specific vascular territories (see Fig. 1a,b).

(a,b) Axial T1 magnetic resonance image (MRI) in 2001 showing thick gyral enhancement of the left posterior hemisphere. (c,d) During a subsequent attack in 2003, axial T1 MRI reveals gyral enhancement occurring this time in the right posterior hemisphere.
Diffusion weighted images were normal. Magnetic resonance angiography (MRA) and conventional cerebral angiography at that time showed no significant pathology.
MRI on presentation in October 2003 revealed the identical unusual pattern of intense cortical ribbon enhancement, this time corresponding to the involved right hemisphere without mass effect (see Fig. 1c,d). Again the diffuse cortical enhancement did not conform to vascular territories. Fluid attenuated inversion recovery (FLAIR) images showed mildly increased cortical T2 signal. Diffusion-weighted imaging was normal. After complete recovery, repeat neuroimaging revealed resolution of all MRI abnormalities observed 3 weeks before. Another ictal EEG, her third in as many attacks, revealed slowing corresponding to the MRI abnormalities but no epileptiform abnormalities. Verapamil therapy correlated with migraine headache improvement.
Other studies from 2001 and 2003 were normal, including cerebrospinal fluid (CSF) and extensive blood tests for inflammatory and infectious aetiologies.
Patient 2
A 44-year-old right-handed woman with a history of a grade 3 mixed oligoastrocytoma in the right temporo-parietal region post cranial irradiation and chemotherapy in 1990, presented in March 2005 with recurrent subacute right hemispheric dysfunction without headache. She had received 6400 cGy of cranial irradiation in 36 fractions followed by six cycles of BCNU and interferon. Subsequent yearly MRIs showed stable T2 signal changes consistent with encephalomalacia within the radiation field. She had no personal or family history of migraine.
Recurrent episodes began in 2001 with brief spells of dysarthria and left hand clumsiness lasting minutes. EEG revealed right temporal sharp waves. With phenytoin use, no further spells occurred until March 2005, when she presented with neurological dysfunction in association with fever, chills, sweats, myalgias, piloerection, nausea, vomiting and abdominal cramping. She was hospitalized with presumed viral gastroenteritis. Neurological examination demonstrated a left homonomous hemianopsia and dysarthria. An MRI, including contrast enhanced images, was unremarkable at this time. Her symptoms resolved and her neurological examination returned to baseline.
One week later the left homonymous hemianopsia, confusion and dysarthria returned without any associated constitutional symptoms. She gradually developed left hemiparesis and paraesthesias lasting several days. Examination revealed flaccid dysarthria, dense left homonymous hemianopsia, upper motor neuron weakness of the left face, arm and leg with hyperreflexia and mild left-sided cortical sensory loss. Babinski signs were absent. The dysarthria and visual deficit resolved over 4 weeks.
During the height of her symptoms in March 2005 an MRI revealed intense, full-thickness cortical enhancement of the right parieto-temporo-occipital regions and posterior frontal lobe (see Fig. 2a,b) with increased signal of the cortical ribbon on FLAIR.
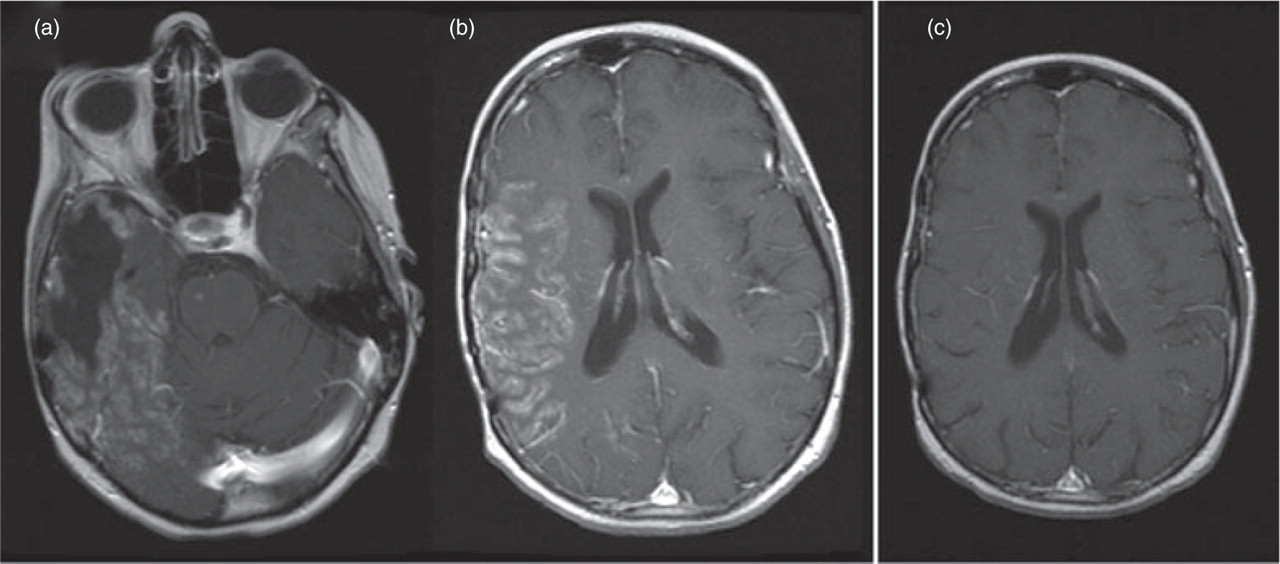
(a,b) Axial T1 magnetic resonance image (MRI) with gadolinium showing thick gyral enhancement of the right hemispehere (with postoperative changes from a previously resected anterior temporal oligoastrocytoma). (c) Three weeks later, axial T1-enhanced MRI shows interval resolution of enhancement.
A small focus of restricted diffusion within this territory was interpreted as less than expected given the widespread T2 changes, suggesting ischaemia more than infarction.
MRA showed mild right middle cerebral artery narrowing just proximal to the trifurcation. Conventional angiography confirmed good flow across a 50% stenosis of the distal M1 with a subtle capillary blush corresponding to the cortical abnormalities on MRI. Prolonged ictal video EEG monitoring showed right temporo-occipital slowing without any epileptiform discharges or clinical signs of subtle seizures. Complete blood count, sedimentation rate, chemistries, thyroid stimulating hormone, blood pressure monitoring and urinalysis were normal.
Her symptoms gradually resolved over 1 week. Three weeks later, MRI with contrast and diffusion-weighted images had returned to her baseline as described above (see Fig. 2c).
Discussion
We believe that the episodes of migraine with aura reported in Shuper et al.’s four patients and the migraine with aura and subsequent development of the full SMART syndrome with migraine-like symptoms in Murthy and Cohen's patient, Bartleson's two patients and the two patients reported here are all complications of remote cranial irradiation. These cases suggest a spectrum of migraine-like attacks ranging from migraine with aura to probable migraine with aura with prolonged duration of symptoms to the SMART syndrome. SMART is a distinctive syndrome involving transient, reversible neurological dysfunction which may include migrainous headache at times preceded by aura, prolonged hemispheric neurological impairment and sometimes seizure activity associated with characteristic, temporary, diffuse, unilateral cortical gadolinium enhancement on MRI. We do not believe the MRI changes are caused by seizure activity. Patient 1 demonstrated that attacks may alternate between the left and right hemispheres and patient 2 demonstrated that headache is not a required feature of SMART. Table 1 describes the major features of the four young patients reported with migraine with aura and the five patients with SMART syndrome currently in the literature. Table 2 lists our proposed criteria for diagnosis.
Interval, Years between irradiation and the first symptoms; Age at onset, onset of symptoms attributed to SMART; Recovery time, longest duration until recovery; PF, posterior fossa; PNET, primitive neuroectodermal tumour; HH, homonymous hemianopsia; VHL, Von Hippel-Lindau disease; Y, yes; N, no.
Proposed revisions to Bartleson et al.’s diagnostic criteria for SMART syndrome (6)

The MRI changes are transient and may not be found in every attack. As in our second case, it may take days to a week or more before the imaging findings are evident and they are transient, lasting perhaps days at a time.
There does not appear to be an association with a particular tumour type or any chemotherapeutic agent. All of the patients received a substantial dose of radiation therapy, but the minimum dose of radiation is uncertain.
SMART appears to be a late delayed consequence of radiation therapy that occurs in discrete spells. The pathophysiology is unknown but irradiation could affect primarily the vasculature or the trigeminovascular system and engender a transient, recurrent, self-limited yet dramatic disturbance of homeostasis that manifests years after the injury. Endothelial dysfunction may produce a disruption of the blood–brain barrier leading to the marked cortical enhancement. Radiation and the body's response to it could alter ion channel reactivity with a lowered threshold for cortical spreading depression such as that seen in the episodic yet severe attacks of hemiplegic migraineurs.
SMART must be considered in the differential diagnosis of spells associated with MRI changes as listed in Table 3. Status epilepticus can produce transient neurological deficits and can cause unilateral, cortical MRI changes that do not respect vascular territories with cortical T2 hyperintensity and a low apparent diffusion coefficient, including mild gyral enhancement (8, 9). Most cases of gyral enhancement after partial status epilepticus reveal superficial, leptomeningeal enhancement usually with prominent vasculature and T2 cortical hyperintensity (9, 10). The degree of gyral enhancement in our patients seems to be disproportionate to previously reported seizure-related changes. Also, our patients had ictal EEGs that did not reveal epileptiform abnormalities. Hormigo et al. reported four patients who presented with T2 cortical hyperintensity and mild cortical enhancement quite unlike our patients (11). These patients were found to be in non-convulsive status epilepticus by EEG and did not have prior irradiation or focal neurological deficits.
Differential diagnosis of transient spells with magnetic resonance imaging abnormalities

Beyond a careful history and physical and neurological examination, the work-up must be individualized. A gadolinium enhanced MRI scan of the head is required and the following should be considered: CSF examination, EEG, MRA, serum lactic acid level and blood pressure monitoring. Because conventional angiography triggered migraine attacks in some of Shuper et al.’s cases, MRA is preferred in suspected cases of SMART.
Because of the small number of patients reported with either the SMART syndrome or migraine with aura following radiation therapy, presentations will no doubt vary across a spectrum. Recognition of this syndrome can reassure patients, families and providers and help avoid some unnecessary and potentially hazardous diagnostic tests.