Abstract
This paper describes the differences in the presence of myofascial trigger points (TrPs) in the upper trapezius, sternocleidomastoid, temporalis and suboccipital muscles between unilateral migraine subjects and healthy controls, and the differences in the presence of TrPs between the symptomatic side and the nonsymptomatic side in migraine subjects. In addition, we assess the differences in the presence of both forward head posture (FHP) and active neck mobility between migraine subjects and healthy controls and the relationship between FHP and neck mobility. Twenty subjects with unilateral migraine without side-shift and 20 matched controls participated. TrPs were identified when there was a hypersensible tender spot in a palpable taut band, local twitch response elicited by the snapping palpation of the taut band and reproduction of the referred pain typical of each TrP. Side-view pictures were taken in both sitting and standing positions to measure the cranio-vertebral angle. A cervical goniometer was employed to measure neck mobility. Migraine subjects showed a significantly greater number of active TrPs (P < 0.001), but not latent TrPs, than healthy controls. Active TrPs were mostly located ipsilateral to migraine headaches (P < 0.01). Migraine subjects showed a smaller cranio-vertebral angle than controls (P < 0.001), thus presenting a greater FHP. Neck mobility in migraine subjects was less than in controls only for extension (P = 0.02) and the total range of motion in flexion/extension (P = 0.01). However, there was a positive correlation between the cranio-vertebral angle and neck mobility. Nociceptive inputs from TrPs in head and neck muscles may produce continuous afferent bombardment of the trigeminal nerve nucleus caudalis and, thence, activation of the trigeminovascular system. Active TrPs located ipsilateral to migraine headaches might be a contributing factor in the initiation or perpetuation of migraine.
Introduction
Migraine is a common disorder with a lifetime prevalence of 16% worldwide and a last-year prevalence of 10% (1, 2). Over recent decades there have been major advances in the understanding of migraine pathophysiology. Much attention has been paid to central mechanisms of migraine, particularly to the activation of the trigeminovascular system (3, 4). On the other hand, some authors have proposed that headache may be due to an excess of nociceptive inputs from peripheral structures converging upon the same neurons and being integrated with supraspinal effects (5). Simons et al. (6) and Gerwin (7) have claimed that pain from pericranial head, neck and/or shoulder muscles may be referred to the head, thus being experienced as headache. In their comprehensive text, Simons et al. (6) described the referred pain patterns from different myofascial trigger points (TrPs) in head and neck muscles which might potentially contribute to some head and neck symptoms found in subjects with migraine. A TrP is a hyperirritable spot associated with a taut band of a skeletal muscle. It is painful on compression and on stretch and usually gives rise to a typical referred pain pattern (6). Hu et al. have reported that noxious stimulation of muscle afferents increases the excitability of spinal cord neurons (8). Boquet et al. found that upper cervical dysfunctions were located ipsilateral to migraine headaches in 24 subjects presenting with strictly unilateral migraine (9). Based on these findings, it seems plausible that TrPs in head and neck muscles might be an initiating or perpetuating factor for some migraine headaches.
Cervical musculoskeletal abnormalities have traditionally been linked to different headaches (10, 11). One frequently noted abnormal posture is an excessive forward head position, or forward head posture (FHP) (12). FHP has already been related to cervicogenic headache (13) and we have recently found FHP in association with chronic tension-type headache (CTTH) (14). On the other hand, limited neck mobility has been considered a major feature of cervicogenic headache (CeH) (15) and we have also found a restriction of neck mobility in CTTH (14). The only study assessing neck mobility in migraine subjects found no significant difference in the range of motion between migraine and control subjects (16). However, as neck symptoms are commonly associated with migraine headache (17), further studies of neck mobility and head posture in subjects with migraine are required.
To our knowledge, there has been no blinded, controlled study in the peer-reviewed literature analysing differences in the presence of TrPs between migraine and control subjects. This paper describes the differences in the presence of TrPs in head and neck muscles between subjects presenting with strictly unilateral migraine and healthy controls and the differences in the presence of TrPs between the symptomatic and non-symptomatic sides in migraine subjects. In addition, we assess the differences in the presence of both FHP and active neck mobility between subjects with unilateral migraine and healthy controls and the relationship between FHP and active neck mobility.
Materials and methods
Subjects
Twenty subjects with unilateral migraine participated in this study from September 2004 to March 2005. Migraine was diagnosed according to the International Headache Society (18) criteria by an experienced neurologist. Key elements of headache history were ascertained, including family history, headache features, temporal profile and current and past medications. To be included, subjects had to describe strictly unilateral headaches without side-shift. Secondary conditions were ruled out by means of systematic neurological examination and ancillary tests. Routine blood analyses with erythrocye sedimentation rate and urine analyses were always obtained. In addition, computed tomography or magnetic resonance imaging of the brain were invariably performed. The health status of all subjects was clinically stable, without current symptoms of any other concomitant disease. The evaluation was held when all migraine subjects were headache free and when at least 1 week had elapsed since the last migraine attack in order to avoid migraine-related allodynia (19, 20). Twenty healthy age- and sex-matched subjects without headache during the prior year served as controls. All subjects signed an informed consent prior to inclusion. This study was supervised by the Departments of Physical Therapy and Neurology of Rey Juan Carlos University and Fundación Hospital Alcorcón, Spain. The research project was approved by the local human research committee.
Musculoskeletal examination
A picture of the lateral view of each subject was first taken to assess FHP objectively. The base of the camera was set at the height of the subject's shoulder. The tragus of the ear was clearly marked and a plastic pointer was taped to the skin overlying the spinous process of the seventh cervical vertebra (C7). Once the picture was obtained, it was used to measure the cranio-vertebral angle: the angle between the horizontal line passing through C7 and a line extending from the tragus of the ear to C7 (21, 22) (Fig. 1). A smaller cranio-vertebral angle indicated a greater FHP. Raine and Twomey have reported the reliability of this procedure as high [intraclass correlation (ICC) = 0.88] (23). FHP was assessed in two different positions: a relaxed sitting position on a high-backed chair and a relaxed standing position in a standard protocol. While maintaining each position, subjects were instructed to focus visually on a point directly ahead on the wall in order to minimize the tendency towards flexion or extension of the neck. Then a picture of the lateral view of each subject was taken at both positions. Details of this protocol can be found elsewhere (14).

Measurement of the cranio-vertebral angle. The angle was assessed directly from a side-view picture using a protractor image and a straight edge .
Second, active neck mobility was assessed with a cervical goniometric device manufactured by Performance Attainment Associates (St Paul, MN, USA). The cervical goniometer has already obtained an intratester reliability (ICC) ranging from 0.7 to 0.9 and an intertester reliability (ICC) ranging from 0.8 to 0.87 (24, 25). Neck mobility was measured in a relaxed sitting position. It was recorded as the total range of motion for different types of movement, i.e. flexion/extension, lateral flexion and rotation, as well as for half-cycles, namely movements in a single direction, i.e. flexion and extension, and lateral flexion and rotation to each side (right and left in controls; symptomatic and non-symptomatic in patients). For this purpose, all subjects were asked to sit comfortably on the chair with both feet flat on the floor, hips and knees positioned at 90° angles and buttocks positioned against the back of the chair. Then, the goniometer was placed at the top of their head. Once the goniometer was set in neutral position, they were asked to move the head actively as far as possible in a standard protocol: forwards (flexion), backwards (extension), right lateral flexion, left lateral flexion, right rotation and left rotation. Two measurements were recorded for each type of movement. Since no significant differences were found between the two measurements (paired Student's t-test), data for further analysis were derived from the average of both values. Moreover, the intraexaminer repeatability of neck mobility (ICC) ranged from 0.91 to 0.94, which indicated a high consistency of these measurements.
Finally, TrPs were sought in both upper trapezius, both sternocleidomastoid, both temporalis and the suboccipital muscles by an assessor who had more than 4 years’ experience in TrP diagnosis. TrP diagnosis was performed following the diagnostic criteria described by Simons et al. (6) and Gerwin et al. (26): (i) presence of a palpable taut band in a skeletal muscle; (ii) presence of a hypersensible tender spot in the taut band; (iii) local twitch response elicited by the snapping palpation of the taut band; and (iv) reproduction of the typical referred pain pattern of the TrP in response to compression. These criteria were adapted for the suboccipital muscles, following the guidelines formerly described by our own research group (27). Specifically, the diagnosis of TrPs in the suboccipital muscles was made when there was tenderness in the suboccipital region, referred pain evoked by maintained pressure (Fig. 2) and increased referred pain on muscle contraction with extension of the upper cervical spine (27). A TrP was considered active if the subject recognized the evoked referred pain as familiar, i.e. similar to other sensations that they were used to perceiving; conversely, a TrP was considered latent if the subject did not recognize the evoked referred pain as familiar (6, 26). Figure 2 details the location and the referred pain patterns evoked by TrPs in the examined cervical muscles based on Simons et al. (6)

Referred pain from myofascial trigger points (TrPs) in the suboccipital, sternocleidomastoid, upper trapezius and temporalis muscles according to Simons et al. Reprinted with permission from Simons D, Travell J, Simons L. Myofascial pain and dysfunction: the trigger point manual, Vol. 1, 2nd edn. Baltimore: Williams and Wilkins 1999.
The whole examination was performed by the same assessor, who was blinded to the subjects’ condition. After TrP assessment, all subjects were asked if the TrP was familiar or reproduced a familiar pain. Since control subjects could have had some head pain without being current headache sufferers, the assessor remained blinded through the entire procedure.
Statistical analysis
Data were analysed with the SPSS statistical package (version 12.0; SPSS Inc., Chicago, IL, USA). Mean values of the cranio-vertebral angle and all cervical motions and the number of active and latent TrPs were calculated. A normal distribution of quantitative data was assessed by means of the Kolmogorov–Smirnov test (P > 0.05). Those data without a normal distribution (i.e. number of active or latent TrPs) were analysed with non-parametric tests, whereas data with a normal distribution (i.e. cranio-vertebral angle, neck mobility and age) were analysed with parametric tests. Differences in the number of either latent or active TrPs between both study groups were assessed with the Mann–Whitney U-test. In the migraine group, differences in the number of either latent or active TrPs between the symptomatic and non-symptomatic sides were assessed with the Wilcoxon signed rank test. Also, the distribution of latent and active TrPs on each side of migraine subjects and healthy controls was analysed with the χ2 test. Differences in both FHP and neck mobility between both groups were assessed with the unpaired Student's t-test. Within each study group, differences in both lateral-flexion and rotation ranges between sides (symptomatic and non-symptomatic in migraine patients, right and left in controls) were assessed with the paired Student's t-test. Since no difference was found between right and left motions in healthy subjects, data for comparison with the patient group were derived from the average of right and left values. The unpaired Student's t-test was used to assess possible gender differences in both neck mobility and FHP. Pearson's correlation test (r) was used to analyse the association between the cranio-vertebral angle (FHP) and neck mobility for each cervical motion. Finally, Pearson's correlation test (r) was also used to assess the association between age and both neck mobility and FHP within each study group. In general, a P-value <0.05 was considered statistically significant; however, when two related comparisons were performed (i.e. cervical flexion and extension, right and left lateral flexions, and right and left rotations) a corrected P-value <0.025 was taken (Bonferroni correction).
Results
A total of 20 migraine subjects, seven men and 13 women, aged 17–57 years (mean 33 ± 10 years), and 20 healthy volunteers, eight men and 12 women, aged 19–55 years (mean 30 ± 8 years), were studied. No significant differences were found in gender or age between the two study groups. Eighteen migraine subjects recalled a positive family history of migraine in first-degree relatives. Headache attacks had typical migraine features and were usually severe and incapacitating. In all cases, migraine was strictly unilateral, without side-shift. All had taken several prophylactic drugs, but only five maintained the prophylactic therapy. All patients were examined on a headache-free status, at least 1 week after their last migraine attack. Demographic data and headache features are given in Table 1.
Demographic data and headache features
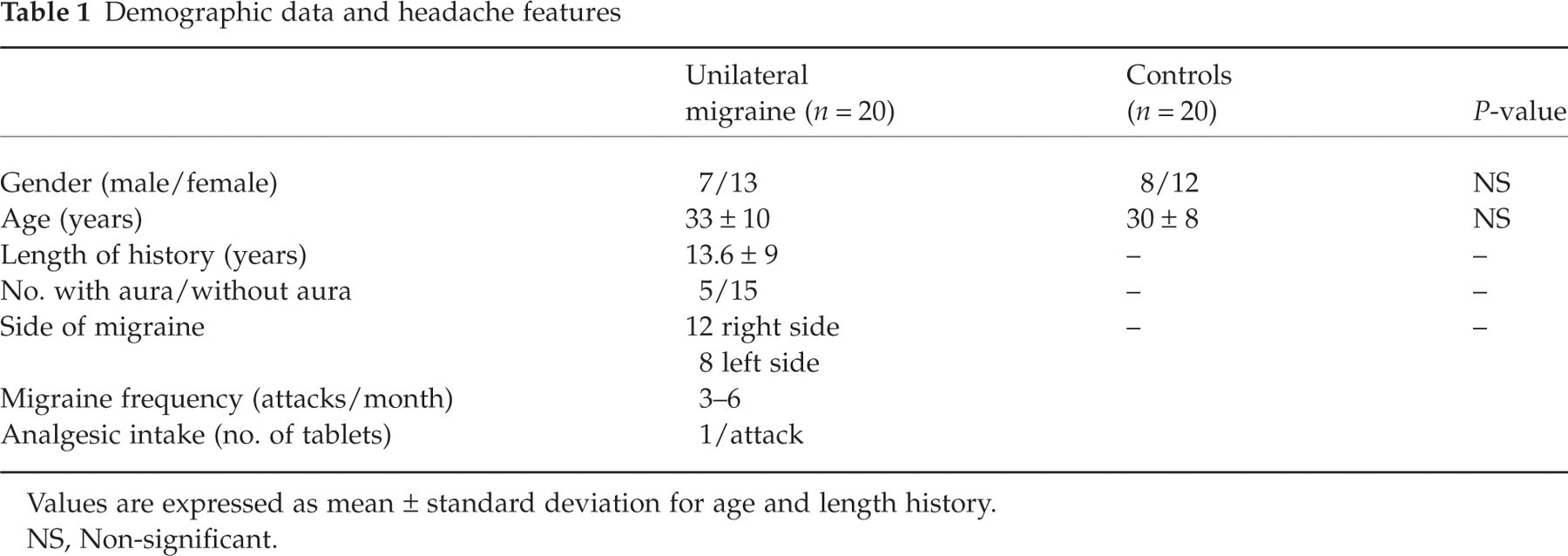
Values are expressed as mean ± standard deviation for age and length history.
NS, Non-significant.
Each of the 20 migraine subjects exhibited at least three TrPs in the explored muscles. The mean number of TrPs in migraine patients was 3.6 ± 1.5, of which 1.9 ± 1.1 were active and 1.7 ± 1.2 were latent. However, all subjects in the control group also had TrPs (mean no. 1.7 ± 0.9), which were constantly latent. Differences in the total number of TrPs and the number of active TrPs between both study groups reached statistical significance (P < 0.001), while the number of latent TrPs was not significantly different. Within the migraine group, the mean number of both active and latent TrPs was greater on the symptomatic side (total no. 2.4 ± 0.5; active TrPs 1.3 ± 1; latent TrPs 1.1 ± 0.8) than on the asymptomatic side (total no. 0.5 ± 0.6; active TrPs 0.1 ± 0.3; latent TrPs 0.4 ± 0.6) (P < 0.01). Conversely, there were no asymmetries in the number of TrPs on either side within the control group.
Active TrPs in migraine patients were almost always located ipsilateral to migraine headaches and the induced referred pain reproduced a sensation that was usually felt during migraine attacks. This was true for each of the explored muscles (upper trapezius, sternocleidomastoid and temporal muscles), except for the suboccipital muscles. Active suboccipital TrPs were present in only five patients and were located bilaterally. Data referring to the distribution of either latent or active TrPs in both study groups are shown in Table 2.
Distribution of myofascial trigger points (TrPs) (active or latent) on the symptomatic and non-symptomatic side in unilateral migraine subjects and either side (left or right) in controls


NS, Non-significant; n, number of subjects; NA, not applicable.
P-values are derived from the χ2 test.
In the control group there was a negative correlation between age and cervical flexion (r = −0.5; P < 0.01), extension (r = −0.4; P < 0.05), both lateral flexions (r = −0.4; P < 0.01 on the left, and r = −0.3; P < 0.05 on the right) and right rotation (r = −0.4; P < 0.01), although there was no correlation between age and left cervical rotation. Conversely, neck mobility was not correlated with age in migraine subjects. Similarly, FHP was correlated with age in the control group (r = −0.55; P < 0.01 for the sitting position, and r = −0.45; P < 0.05 for the standing position), but not in the migraine group. In each group a positive correlation was found between FHP in both positions (r = 0.78; P < 0.001 in migraine subjects; r = 0.8; P < 0.001 in controls): the greater the FHP in the sitting position, the greater the FHP in the standing position. There were no significant differences in either neck mobility or FHP between males and females in either group.
The cranio-vertebral angle was smaller, i.e. there was a greater FHP, in migraine patients than in healthy controls for both sitting and standing positions (P < 0.001). Otherwise, migraine subjects showed less neck mobility than healthy controls in extension (P = 0.02) and the total range of motion in flexion-extension (P = 0.01), but not in the remaining movements. In the migraine group, neck mobility in both lateral-flexion and rotation motions did not differ between the symptomatic and non-symptomatic sides. Table 3 details both neck mobility and FHP parameters of each study group.
Head posture and neck mobility in both study groups

Values are expressed as means ± standard deviation.
Average of the values corresponding to both sides (right and left) in the control group.
P-values are derived from the unpaired Student's t-test (P-value <0.025 was considered statistically significant).
NS, Non-significant.
A positive correlation was found between the cranio-vertebral angle in the sitting position and neck mobility in all cervical motions, except for right lateral-flexion: flexion (r = 0.4; P = 0.02), extension (r = 0.4; P = 0.03), left lateral-flexion (r = 0.5; P < 0.001), left rotation (r = 0.4; P < 0.01) and right rotation (r = 0.5; P < 0.01); the smaller the cranio-vertebral angle, i.e. the greater the FHP, the less the neck mobility. These correlations did not differ between the two study groups. The cranio-vertebral angle in the standing position showed similar correlations with different cervical motions: extension (r = 0.5; P < 0.01), left lateral-flexion (r = 0.6; P < 0.001), left rotation (r = 0.6; P < 0.001) and right rotation (r = 0.6; P < 0.001).
No significant differences in the distribution of TrPs, FHP or active neck mobility were found between migraine subjects with and without aura, or between those on prophylactic treatment and those without medication.
Discussion
In this study all subjects presenting with unilateral migraine plus all healthy subjects showed evidence of TrPs in neck and head muscles, which had the potential to refer pain to the head. The presence of TrPs in our control group was not unexpected, as latent, i.e. non-active, TrPs have been commonly observed in healthy, normal subjects (28). Yet the total number of TrPs was significantly greater in patients with migraine. Moreover, active TrPs were found only in the migraine group, while they were absent in the control group. Active TrPs are those that reproduce a familiar or usual pain, in this case headache (6, 26). In our group of migraine subjects, active TrPs were mostly located ipsilateral to the side of headache. In fact, the induced referred pain reproduced a sensation that was usually felt during spontaneous migraine attacks.
Our findings support the hypothesis that TrPs in head and neck muscles may play an important role in the origin, maintenance or perpetuation of migraine. A link between TrPs in head-neck muscles and migraine headache might be the activation of the trigeminal nerve nucleus caudalis, and thence the activation of the trigeminovascular system. Olesen (5) has proposed that headache may be due to an excess of nociceptive inputs from peripheral structures. According to his model, perceived headache intensity would be the sum of nociception from cranial and extracranial tissues converging on the neurons of the trigeminal nucleus caudalis. In some subjects with migraine, nociceptive inputs from TrPs in these muscles could produce a continuous afferent bombardment of the trigeminal nerve nucleus caudalis. This would result in temporal and spatial summation of neuron signals, a decrease in pain threshold and, finally, a tendency to a progressive increase of headache. Such repeated nociceptive stimulation of the nucleus caudalis may possibly facilitate the activation of the trigeminovascular system. If an association between TrPs in head and neck muscles and migraine headache is eventually confirmed, a therapeutic approach based on TrP management should be evaluated in further studies.
Migraine patients in this study showed a greater FHP, i.e. a smaller cranio-vertebral angle, than control subjects in both sitting and standing positions. FHP has been previously related to other headaches. For example, Watson and Trott found that subjects presenting with cervicogenic headache showed a lesser cranio-vertebral angle than controls (13). Our own research group has found a reduced cranio-vertebral angle in CTTH sufferers in comparison with healthy controls (14). In addition, Treleaven et al. (29) found that patients with postconcussional headache had a lesser cranio-vertebral angle than healthy subjects, though the differences were not significant. In contrast to our findings, Zito et al. (30) have recently reported no significant differences in FHP between migraine and healthy subjects. The cranio-vertebral angle in our migraine group was similar to that found in CeH (13) and CTTH subjects (14), whereas the mean cranio-vertebral angle in our non-headache group was similar to that found in healthy subjects in the aforementioned studies. Therefore, we can regard our healthy control group as having a normal posture, whereas our migraine group presented an altered posture. FHP appears to be a feature common to several headache syndromes. However, FHP may be a consequence, i.e. an antalgic posture trying to reduce pain, rather than an aetiological factor in headache. Whether FHP contributes to the origin or the perpetuation of migraine must be verified by future research. Determination of the clinical significance of FHP in different headaches, including migraine, would require the development and testing of specific physical therapy programmes.
Finally, neck mobility was not generally altered in migraine patients, albeit extension and the total range of motion in flexion/extension were significantly reduced in the migraine group compared with the control group. Moreover, neck mobility in lateral movement did not differ between the symptomatic and non-symptomatic sides in migraine subjects. These findings are in agreement with those of Zwart (16) and Zito et al. (30), who likewise found similar neck motions in migraine and non-headache control groups. Cervical mobility has been shown to decrease with age (31, 32). Our study confirmed that situation in the control group, but not in the migraine group. A positive correlation was found between FHP, i.e. the cranio-vertebral angle, and neck mobility in almost all cervical motions. Therefore, subjects with a greater FHP, i.e. a smaller cranio-vertebral angle, showed lesser neck mobility. Our results are consistent with those reported by Walmsley et al. (33) and Ordway et al. (34), who also showed that FHP could dramatically affect neck mobility. This is not surprising, as FHP may lead to excessive compression on the facet joints and posterior surfaces of the vertebra bodies, thus affecting the biomechanics of the head and the neck. Based on these findings, we may conclude that differences in neck mobility are most likely a consequence of the abnormal head posture, the pain, or both, rather than a causative factor for headache.
There are some limitations to our study. First, only patients with strictly unilateral migraine were included. Hence, our results cannot be extrapolated to bilateral migraine of other headache disorders, such as tension-type headache. It would be interesting to assess the relevance of TrPs in head and neck muscles in subjects presenting with different headaches. The second limitation was the sample size. It would be necessary to repeat the same procedure with a greater number of subjects to establish definitely a link between active TrPs and migraine headaches.
In conclusion, subjects with unilateral migraine had a greater number of TrPs than healthy subjects. Active TrPs were mostly located ipsilateral to migraine headaches. The presence of active TrPs may be a contributing factor in the initiation or perpetuation of some migraine headaches. Migraine subjects showed a lesser cranio-vertebral angle, but not lesser neck mobility, than healthy controls. Nevertheless, neck mobility was correlated with FHP: the greater the FHP, the lesser the cervical range of motion. The clinical significance of TrPs and FHP in migraine remains to be investigated. The assessment of some physical therapy programmes might eventually help elucidate the influence of TrPs and neck posture in the clinical course of migraine.
Acknowledgements
We thank Dr David Simons and Dr Robert D. Gerwin for their kind encouragement.
