Abstract
Dipyridamole inhibits phosphodiesterase 5 (PDE5) and adenosine re-uptake. The most prominent side-effect is headache. We examined the migraine-generating effects of dipyridamole as well as the cerebral blood velocity response in a single-blind study, including 10 patients with migraine without aura and 10 healthy subjects. Dipyridamole 0.142 mg/kg per min was administered intravenously. Headache intensity was scored on a verbal rating scale along with pain characteristics and accompanying symptoms. Blood velocity in the middle cerebral artery (V mca), blood pressure and heart rate were recorded repeatedly. Headache was induced in all migraine patients and in eight of 10 healthy subjects (P = 0.47) with no significant difference in headache intensity (P = 0.53). However, five patients but only one healthy subject experienced the symptoms of migraine without aura, according to ICHD-2 criteria, within 12 h (P = 0.14). Four patients reported photophobia after dipyridamole compared with no healthy subjects (P = 0.087). V mca decreased (P < 0.001) during and after dipyridamole infusion with no difference between groups (P = 0.15) coinciding with initiation, but not cessation of immediate headache. Thus, dipyridamole induces symptoms of migraine and an initial decrease in V mca in migraine patients, but not significantly more than in healthy subjects. This relatively low frequency of migraine induction, compared with nitric oxide donors and sildenafil, is probably due to the less specific action of dipyridamole on the cGMP signalling pathway as well as a possible bidirectional effect of adenosine on migraine induction.
Introduction
Oral dipyridamole (Persantin®) in combination with aspirin is widely used in the secondary prevention of stroke (1), whereas intravenous administration of dipyridamole is used as a cardiac stress test (2). Dipyridamole appears to work through two different signalling systems, the cAMP- and the cGMP-related pathways (3, 4). The most common side-effect in the clinical use of dipyridamole is headache occurring in approximately 38% after oral administration, causing discontinuation of treatment in 8% of patients (5) and occurring in 32–37% after routine intravenous administration (6, 7).
We have previously shown that intravenous administration of dipyridamole induces headache with a concomitant but not completely time-related minor dilation of the cerebral arteries in eight of 10 healthy subjects (8). Also, oral administration of another, but more selective inhibitor of phosphodiesterase 5 (PDE5), sildenafil (Viagra®) has recently been shown to induce headache in healthy subjects (9) with more severe headache, fulfilling the criteria for migraine in migraine patients (10). Sildenafil caused no significant changes in middle cerebral artery diameter or regional cerebral blood flow. In healthy subjects, circulating adenosine was recently found to induce a very mild headache with only extracranial vasodilation and, in a few subjects, a delayed headache was initiated (11). Whether dipyridamole is able to induce significantly more headaches, a larger arterial dilation and perhaps an attack of migraine in migraine patients has not previously been systematically investigated.
We wished to test the hypothesis that dipyridamole is able to induce symptoms of migraine in migraine patients and headache in healthy subjects, just like glyceryl trinitrate (GTN) and sildenafil.
The primary endpoint was to investigate whether dipyridamole can induce symptoms of migraine, in order to elucidate if dipyridamole may be used in the human migraine model. The secondary endpoints were to elucidate, first, if the induced headache was of higher intensity, and second, if a vascular response was more pronounced in migraine patients compared with healthy controls.
Compared with the oral sildenafil, the advantage of using dipyridamole in the human migraine model is that dipyridamole is available for both oral and intravenous use just like GTN, whereas sildenafil is available only for oral administration. The intravenous administration in the human migraine model helps to eliminate the potential variability in bioavailability after oral administration and makes it easier to time the intended investigations. The PDE5 inhibitor effects of dipyridamole make it a likely candidate for investigating the migraine-generating mechanisms downstream of GTN.
Methods
Design and material
The study was a single-blind study where dipyridamole 0.142 mg/kg per min was administered intravenously for 4 min in 10 healthy subjects, seven women and three men, mean age 29.7 years (range 22–45), body weight 69.8 ± 5 kg, and in 10 patients suffering from migraine without aura, eight women and two men, mean age 35.7 years (range 20–47), body weight 73.5 ± 3.5 kg. The healthy subjects served as a control group for the migraine patients. The groups were matched according to age (P = 0.15) and body weight (P = 0.56). Except for one subject, they were also matched for sex.
Subjects were recruited among hospital staff and medical students. Migraine patients were recruited mainly from the headache clinic.
The number of subjects included was based on previous studies of the GTN model (12, 13). In relation to applying dipyridamole as a migraine-provoking substance in the migraine model, it would be of interest only if dipyridamole induced migraine with the same frequency as GTN or sildenafil. In these studies, 10 and 12 subjects were included and thus 10 subjects in each group were chosen for the present study.
Inclusion of subjects was performed by one investigator (S.O.). Those performing the measurements and asking questions of headache, investigator (L.H.L.) and study technician (K.E.), were blinded to the status (patient or healthy subject) of the subjects investigated. Furthermore, the participants were not informed that the purpose of the study was to detect a difference in headache response between the two groups.
The dipyridamole dose used was similar to that used for routine cardiovascular performance tests (2) and in a previous study in healthy subjects (8). An intravenous formulation was chosen to avoid known variation in bioavailability after oral administration (3). Placebo was not included in this pilot study since the primary endpoint was to detect a difference between healthy subjects and migraine patients.
Exclusion criteria for all subjects were a medical history of pulmonary disorders, cerebro- or cardiovascular disorders, hypo- or hypertension, a history of alcohol or drug abuse or psychiatric disease. Only subjects with a normal ECG were included. The healthy subjects had no history of migraine and a headache frequency <5 days a month. They were without a family history of migraine, i.e. no first-degree relative with migraine.
The migraine patients had a migraine frequency ranging from 5 per year to 6 per month and tension-type headache <5 days a month. No prophylactic treatment for migraine was allowed. All subjects gave written informed consent before inclusion and a medical examination, including an ECG, was performed prior to the study. The study was approved by the ethics committee of Copenhagen County (KA93086) and the Danish health authorities and was conducted according to the Helsinki II declaration.
Headache recordings
Headache intensity was scored on an 11-point numerical verbal scale from 0 to 10 during the study, where 1 represents a very mild headache (including a feeling of pressing or throbbing), 5 a headache of moderate intensity and 10 the worst headache imaginable (12). Headache characteristics, headache location and other side-effects were also recorded. All subjects were asked to record their headache, the headache characteristics and any intake of medication every hour at home in a headache diary from 2 to 10 h following the dipyridamole infusion. The subjects were allowed to treat their headache at home with plain analgesics (paracetamols, aspirin, non-steroidal anti-inflammatory drug) or migraine medication (sumatriptan). Time for intake and drugs used were recorded in the headache diary. Due to the pharmacokinetic profile of dipyridamole, with a tri-exponential decline with the major decline within the first hour, immediate headache is defined as headache occurring within the first hour after infusion and delayed from 2 to 12 h (14).
Blood flow velocity
A time-averaged mean of the maximal blood velocities in the middle cerebral artery (V mca), comprising at least one cardiac cycle, was recorded bilaterally by transcranial Doppler (TCD) (2 Mhz, 3-D Transscan; EME, Uberlingen, Germany) using handheld probes (15) as previously described in a validated set-up (8, 16). A fixed point for measurements of V mca was chosen along the middle cerebral artery. Measurements of endtidal pCO2 (PetCO2) were performed simultaneously with the TCD measurements (POET; Criticare Systems Incorporated, Waukesha, WI, USA) using a mask placed lightly over the nose and mouth (16).
Procedures
The subject arrived non-fasting, headache free on the day of the study. No coffee, tea or other caffeine-containing drinks (no methylxanthine-containing compounds), alcohol, tobacco, chocolate, medication or smoking were allowed for at least 12 h prior to the study. Migraine patients were free of attack at least 48 h before the study day. The subjects were kept in a supine position during the study. After 30 min rest, baseline measurements of blood pressure, heart rate, PetCO2 and V mca were performed. Dipyridamole was infused for 4 min. V mca was recorded and headaches as well as headache characteristics were scored at 3 min, 7 min, 10 min and then every 10 min until 60 min after infusion of dipyridamole.
Blood pressure and heart rate were measured repeatedly using an automatically inflated arm-cuff (Tonoprint; Speidel + Keller GmbH, Jungingen, Germany). ECG was recorded continuously during the study.
Because of the blinding procedures all subjects received sumatriptan 100 mg for rescue medication.
Statistics
All values are presented as means ± standard error of the mean (SEM). P < 0.05 was considered significant.
The area under the curve (AUC) and the peak response for each variable, V
mca, blood pressure, heart rate and PetCO2 on the separate days was chosen as summary measure when analysing difference in response between study groups and compared using an unpaired t-test. Changes over time for the variables were analysed with two-way analyses of variance with factors time and subjects. The
Results
Headache and accompanying symptoms
Headache scores over time are shown in Fig. 1a,b, healthy and migraine patients, respectively. Headache was induced in all migraine patients and in eight of 10 healthy subjects (P = 0.47) (Table 1 and Fig. 2). The headache in five patients but only in one healthy subject fulfilled the symptoms of the ICHD-2 criteria for migraine without aura within 12 h (P = 0.14) (Fig. 2). The latter only in the first 10 min from infusion corresponded to a steep increase in heart rate, from 75 to 129 beats/min within the first 7 min. One patient reported a mild headache with a similar location to usual migraine, but with no accompanying symptoms (Table 2). There were no significant differences in headache intensity between groups (P = 0.53). Headache characteristics are shown in detail for migraine patients in Table 2.

(a,b)Median headache score over time. Individual headache scores are shown as thin lines. A thick line indicates the median headache score over time in healthy subjects (a, •) and in migraine patients (b, ▪). There were no differences in headache intensities between groups, but five of 10 migraine patients fulfilled the ICHD-2 criteria for migraine without aura postdose.
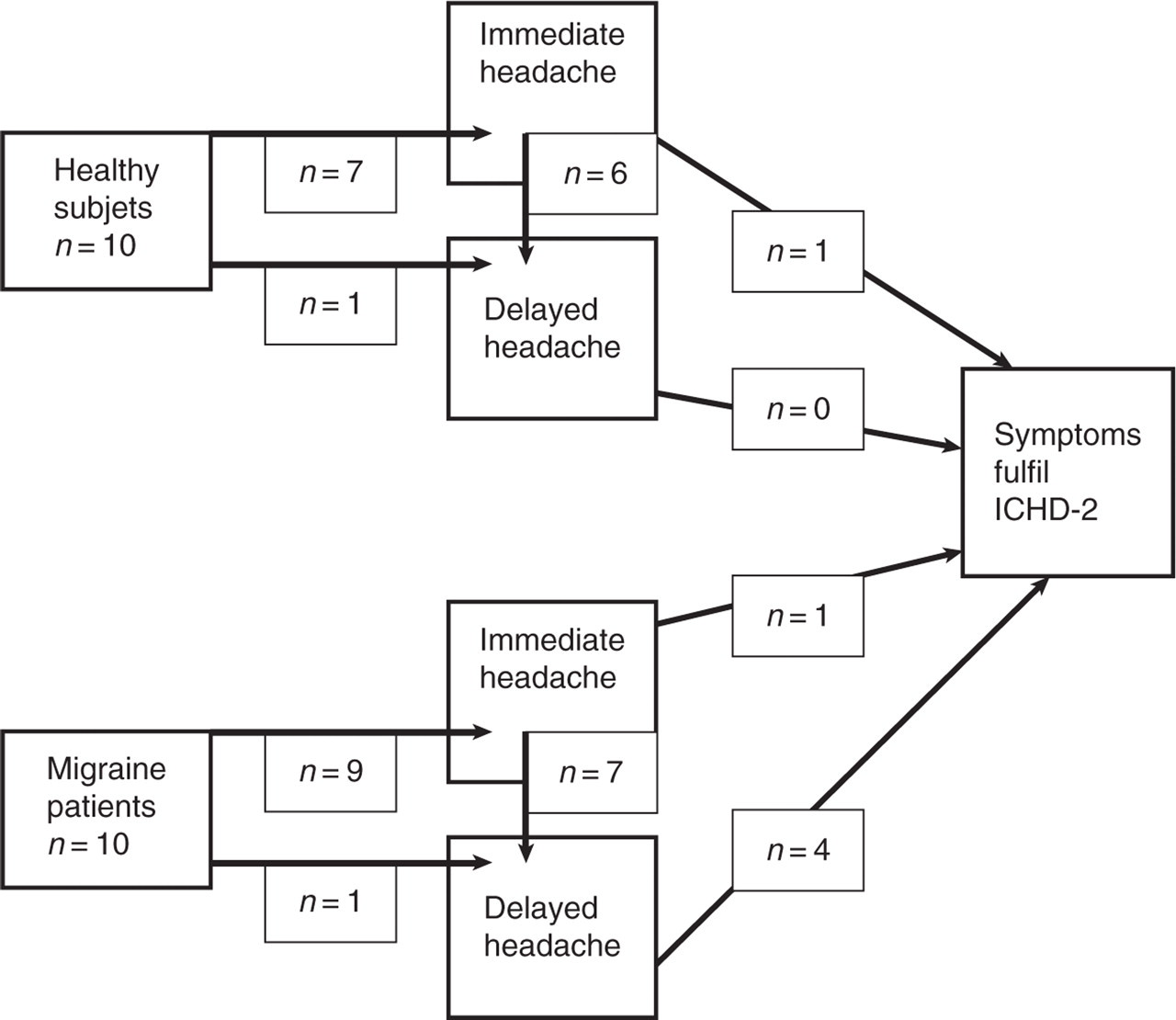
Flow diagram showing headache induction. An overview of the number of subjects from either the healthy subjects or the migraine patients who developed immediate and delayed headache, respectively. Furthermore, the number of subjects from each group who developed symptoms fulfilling the criteria for migraine without aura according to ICHD2 is given.
Induced headache and migraine.

Number of healthy subjects (n = 10) and migraine patients (n = 10) who develop headache and migraine fulfilling the symptoms of migraine without aura according to ICHD−2, in association with the dipyridamole infusion during the first hour (immediate headache) and during the subsequent 11 h (delayed headache). One migraine patient fulfilled the ICHD-2 criteria during both immediate and delayed headache.
Headache characteristic in migraine patients.

Headache characteristics for patients with migraine without aura after infusion of dipyridamole. Time period A is 0–60 min (immediate headache) and B is 60 min to 12 h (delayed headache). NC, Not characterized; press, pressing headache; puls, pulsating headache; bilat, bilateral. Migraine assessed as migraine fulfilling the ICHD-2 criteria for migraine without aura. The intensity of headache response was generally reported to be lower than during the patients’ usual migraine attacks.
Not all symptoms at the same time.
Self-assessed as similar location as usual migraine but only mild headache.
Time to fulfilment of the symptoms of migraine without aura according to ICHD-2 varied between 10 min and 11 h. The immediate median peak headache scores (0–60 min) were 2 (range 0–5) in migraine patients and 1.5 (range 0–5) in healthy subjects (P = 0.85) and delayed median peak headache scores (2–12 h) were 1.5 (range 0–8) and 2 (range 0–7) in migraine patients and healthy subjects, respectively (P = 0.97) with no significant difference in the AUC (P = 0.90 and P = 0.40, immediate and delayed headache, respectively).
Four migraine patients reported photophobia following dipyridamole infusion, three of them already within the first 60 min. The intensity of the photophobia was reported to be mild in two, moderate in one and severe in one patient (Table 2). No healthy subjects experienced photophobia (P = 0.087). The occurrences of phonophobia (three in both group) and nausea (five healthy subjects and four migraine patients, with more patients reporting nausea of moderate intensity) were not significantly different between groups (P = 1).
A mild degree of flushing and, in some, a feeling of heaviness in the body were reported during and immediately after infusion of dipyridamole in both groups. Rescue medication was taken at home by two migraine patients (both oral sumatriptan 100 mg) and three healthy subjects (one took ibuprophen 200 mg, one paracetamol 1 g and one the tablet of sumatriptan 100 mg received at discharge from the study site).
Middle cerebral artery blood velocity (Vmca)
Because of no significant side difference in V
mca (AUC P = 0.42 and P = 0.2, patients and healthy, respectively) the data were analysed as a mean between the two sides. In both groups dipyridamole induced a significant decrease in V
mca lasting 60 min following the start of infusion (P < 0.001,

Mean blood flow velocity (V mca) in the middle cerebral artery and PetCO2 changes. Mean values in percent of baseline ± SEM for V mca (filled symbols) and PetCO2 (open symbols) for healthy subjects (circle) and migraine patients (square) are shown above.
Blood pressure and heart rate
Absolute mean values over time of systolic blood pressure, diastolic blood pressure and heart rate are shown in Table 3. There were no overall differences in systolic (P = 0.36 peak and P = 0.51 AUC) and diastolic blood pressure (P = 0.98 peak and P = 0.22 AUC) or mean blood pressure (P = 0.54 peak and P = 0.21 AUC) between healthy subjects and migraine patients. Heart rate increased significantly (P < 0.001) within the first 30 min from the start of dipyridamole infusion with no difference between groups (P = 0.54 and P = 0.91, AUC and peak, respectively). No abnormal ECG recordings were detected during the study. The subjects showed no change in behaviour and made no complaint of any increase in heart rate.
Blood pressure and heart rate during and after dipyridamole infusion
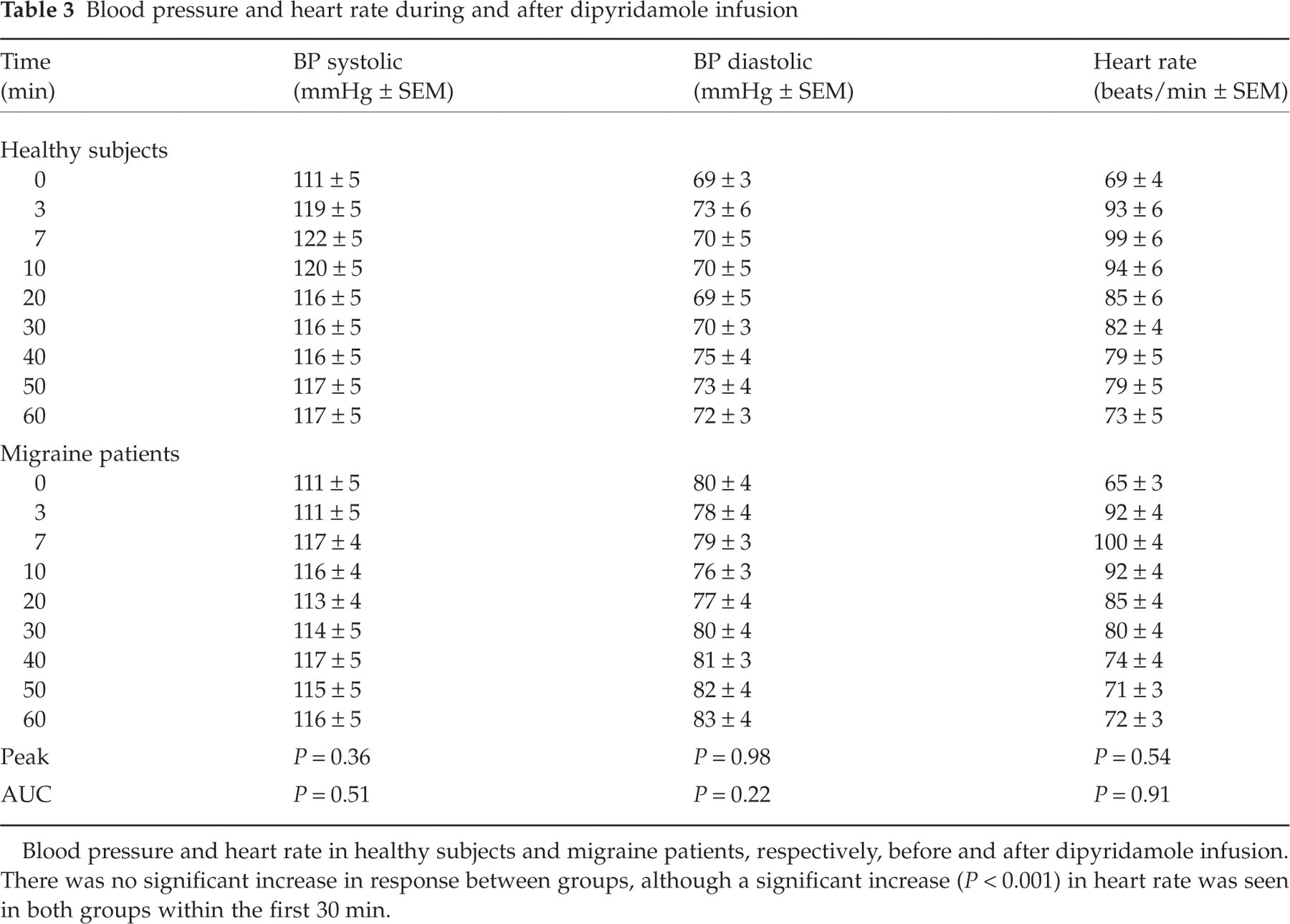
Blood pressure and heart rate in healthy subjects and migraine patients, respectively, before and after dipyridamole infusion. There was no significant increase in response between groups, although a significant increase (P < 0.001) in heart rate was seen in both groups within the first 30 min.
Discussion
Dipyridamole induced headache in eight of 10 healthy subjects and in all migraine patients. In four migraine patients dipyridamole was also able to induce a delayed headache fulfilling the symptoms of migraine without aura according to ICHD-2 between 2 and 11 h postdose. Furthermore, within the first 10 min from the start of infusion, one migraine patient and one healthy subject reported a transient immediate headache fulfilling the ICHD-2 criteria for migraine. Migraine patients, but no healthy subjects, experienced photophobia (P = 0.087), accompanied by slightly more severe nausea compared with healthy subjects. Concurrent with immediate headache, dipyridamole caused a partly PetCO2-dependent decrease in blood velocity in the middle cerebral arteries of similar magnitude in both healthy subjects and migraine patients. A dipyridamole-induced decrease in end-expiratory PetCO2 and in blood flow has been reported previously (8, 18).
The induction of migraine has not previously been considered, although a possible migraine-generating effect was suggested earlier (19). A trial, planned to include 50 migraine patients receiving oral dipyridamole 100 mg four times a day for 12 weeks, had to be terminated after inclusion of only nine patients, because of severe headaches in the subjects within the first days of treatment (19).
What may be the mechanisms of dipyridamole-induced headache and migraine? The exact mechanism of action is not fully understood. Dipyridamole may work through two different pathways. It is an unspecific inhibitor of cGMP degrading enzyme (PDE5) (4), thus increasing cGMP concentrations (20, 21). It furthermore inhibits adenosine re-uptake, thus increasing the circulating plasma adenosine as well as intercellular adenosine, which leads to an increased intracellular cAMP (3). Dipyridamole has also been associated with the release of calcitonin gene-related peptide (CGRP), which may act through cAMP (22). Both the cGMP (10, 13) and the cAMP pathways (11, 23) affected by dipyridamole appear involved in headache and migraine generation, and the former is not necessarily related to cerebral vasodilation (9, 10). Nitric oxide (NO) donors increase cGMP and generate headache resembling the patients’ usual migraine attacks by yet unknown mechanisms (24). Furthermore, an inhibitor of NO synthase, L-NMMA, is effective in acute treatment of migraine attacks (25). Plasma concentrations of CGRP are increased during migraine headache (26) and a new interesting proof of concept in migraine treatment is the use of CGRP receptor antagonists (27). Increased concentrations of circulating adenosine (28) as well as of plasma cAMP in the jugular vein (29) have also been associated with migraine. Adenosine may, however, have a dual and opposing effect in migraine pathophysiology which could counteract the cGMP migraine-inducing effects of dipyridamole. Apart from increasing cAMP levels which can induce headache, adenosine may also stimulate the neuronal adenosine A1 receptors, which appear to inhibit neuronal signalling (30), an effect suggested to be involved in migraine relief (31).
Could the dipyridamole headache be a direct consequence of the vascular changes? The decrease in V mca induced by dipyridamole was approximately 20% without correction for changes in PetCO2 and less when correcting. PetCO2 returned to baseline before the blood velocity changes. Thus, the persistent decrease in V mca may indicate a minor vasodilatory response in the middle cerebral arteries, as previously described (8). For comparison, GTN induced an approximately 25% decrease in V mca in previous studies (8) with a more pronounced decrease in migraine patients, which was not found in the present study. The decrease in V mca outlasted the change in heart rate, indicating that the heart rate changes do not fully explain the V mca changes. Despite these small changes in V mca, a pure vascular mechanism of headache induction appears unlikely: (i) the dipyridamole-induced V mca changes are only time-related to the initiation of the immediate headache but not the cessation, as previously shown, thus the immediate headache declined before normalization of blood flow velocity (8); (ii) the peak increase in heart rate of 43% in healthy subjects and 53% in migraine patients is similar to that reported after adenosine infusion (11), which caused only very mild headache compared with the present study and no symptoms of migraine.
All subjects were asked to refrain from intake of caffeine-containing beverages 12 h prior to the study, and a caffeine withdrawal headache might bias the current results. However, the reported immediate headache was seen in close association with infusion of dipyridamole, and the subjects were allowed intake of caffeine after discharge, rendering it unlikely that the migraine was due to caffeine withdrawal alone. Other environmental factors affecting the subjects after discharge might also influence the induction of delayed headache; however, for practical purposes it is not possible to keep the subjects in hospital during the following 24 h and, also, it cannot be excluded that the stress of being hospitalized also increased the likelihood of headache induction.
In the present study dipyridamole was not as potent as other cGMP elevating compounds, used for a human migraine model, in inducing migraine in migraine patients. The low frequency of migraine induction by dipyridamole renders it unsuitable for use in a human headache and migraine model. Thus, dipyridamole is not a good replacement for or supplement to the GTN migraine model (12), where eight of 10 reported a migraine attack postdose (13). Nor can it replace the cGMP-elevating PDE5 inhibitor sildenafil, which induced migraine in 10 of 12 migraine patients (10, 13) despite the advantages of intravenous administration.
The frequency of migraine induction by dipyridamole appears more similar to the frequency of migraine induction by the cAMP-elevating compounds such as human α-CGRP, where three of nine patients reported fulfilment of ICHD-2 criteria for migraine without aura (23). Intravenous administration of adenosine (11), however, was not as effective as the other cAMP-related mechanisms in headache induction in healthy subjects, perhaps because of a poor penetration of the blood–brain barrier (22, 23, 32) or effect on adenosine A1 receptor associated with headache relief (33, 34).
In conclusion, dipyridamole induces headache in both healthy subjects and migraine patients with no difference in headache intensity. Five of 10 migraine patients met the criteria of migraine without aura after dipyridamole, making dipyridamole a less potent migraine-inducing compound than GTN or sildenafil. The dual actions of dipyridamole may explain this difference in migraine frequency, since the cGMP effects through PDE5 inhibition may be counteracted by the adenosine effects on A1 receptors.
The relatively low frequency of migraine induction makes it less useful in a human migraine model than, for example, GTN or sildenafil.
Acknowledgements
We thank laboratory technician Kirsten Enghave for skilful assistance and also the Hoerslev Foundation and the Danish Research Counsel for financial support.
