Abstract

Thunderclap headache has an abrupt and maximal onset. Thunderclap headache can be secondary to subarachnoid haemorrhage, aneurysms without obvious rupture, pituitary apoplexy, venous sinus thrombosis, cervical artery dissection and intracranial hypotension (1–6). Primary thunderclap headache (PTH), a diagnosis to consider when these more serious causes have been excluded, has been described as a distinct entity. When associated with sexual activity, the headache can be classified as primary headache associated with sexual activity (PHSA). Vasospasm may complicate PTH (7). We present a patient with recurrent PHSA associated with cerebral infarction without evidence for vasospasm, vessel occlusion, dissection or other risk factors.
Case report
A 54-year-old male presented with a sudden onset headache during coitus but prior to orgasm. The headache was maximal at onset, rapidly generalized from its origin in the left occipital region to the entire head and was rated as 8/10 in severity. The patient described two prior coital headaches in the past year that were similar in severity and headache quality. His headache responded to narcotic analgesia and almost completely resolved within hours. He also reported some mild imbalance of gait and nausea, and he vomited once. These symptoms were present immediately after headache onset. There was no sense of vertigo or limb incoordination.
He had no significant previous medical conditions. There were no significant risk factors for cerebrovascular disease. He smoked a pipe infrequently and took no regular medications. His only recent medication exposure was to ‘Neo Citran’ (pheniramine maleate 20 mg, phenylephrine hydrochloride 10 mg, acetaminophen 325 mg), which he took 4 days prior to his symptoms for mild upper respiratory tract symptoms.
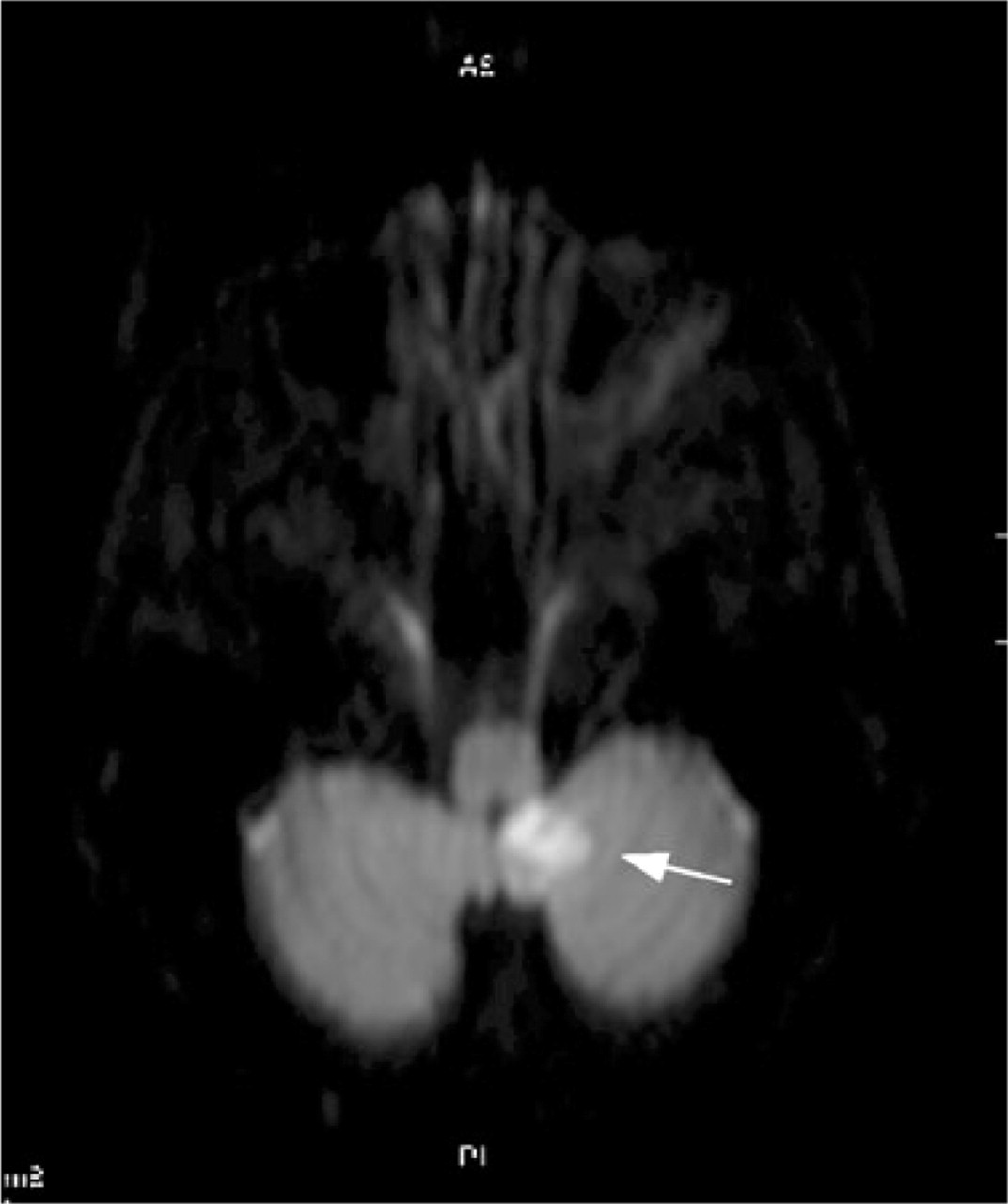
Examination showed mild ataxia of tandem walking. No nystagmus, dysarthria or other findings of cerebellar dysfunction were noted. The remainder of his neurological examination was normal. His blood pressure was 156/92 and there were no abnormalities on general exam. A computed tomographic scan of the brain was reviewed by a neuroradiologist and interpreted as normal. There were 925 red blood cells per mm3 in the first collected tube of cerebrospinal fluid, dropping to 29 on the last tube, suggesting a traumatic lumbar puncture as the cause. No xanthochromia, pleocytoisis or protein or glucose abnormalities were detected. Magnetic resonance imaging, completed 27 h after symptom onset, showed a left inferomedial posterior inferior cerebellar artery (PICA) infarct on FLAIR and diffusion-weighted sequences (Fig. 1). Apparent diffusion coefficient sequences confirmed the infarct was acute (Fig. 2). Time-of-flight MRA of the circle of Willis did not reveal any associated vascular dissection or occlusion. A contrast enhanced magnetic resonance angiography (MRA) of the neck and aortic arch was normal. Femoral catheter angiography on day 2 after admission was normal beyond mild irregularity of the proximal PICA (Fig. 3). Additional investigations included a normal EKG, transthoracic echocardiogram and 24-h holter monitor.
A magnetic resonance image, completed 27 h after symptom onset, identified a left inferomedial posterior inferior cerebellar artery infarct shown here on a diffusion-weighted sequence.

Apparent diffusion coefficient sequences of magnetic resonance imaging confirms the acute nature of the infarct as observed in the diffusion-weighted sequence.

Conventional catheter angiogram on day 2 after admission was normal beyond mild irregularity of the proximal posterior inferior cerebellar artery.
Discussion
Our patient experienced an explosive thunderclap type headache prior to orgasm. This was not preceded by another headache and might be classified as PHSA without further subclassification if no cerebral infarction had occurred (8). His history of prior, presumably benign, events suggested that PHSA may have included this rare complication. The aetiology of his PICA territory infarction may have been secondary to transient vasospasm, although none was evident on MRA the day following the event or on catheter angiography on day 2 after admission. Thunderclap headache with vasospasm has been associated with cerebral infarction and other focal neurological events (9–11). The prior use of adrenergic agonists in ‘Neo Citran’ could have contributed to vasospasm as outlined in a previous case report (12). The data associating this medication with stroke are limited. We believe this is unlikely to be a significant contributor to our case given the long period between ingestion and the clinical event. Vasospasm was also not observed in our patient, unlike the previous case report. The lack of radiographic evidence for vasospasm prevents the classification of this event as being secondary to angiopathy.
Headache can be a common feature of primary cerebral infarction, although it is not typically a thunderclap headache (13). Nociceptive innervation of the first few centimeters of cerebral vessels may be the source of such pain (14–16). Proposed mechanisms also include dysfunctional sympathetic activity, although relationships between sympathetic axons and pain remain controversial in other parts of the nervous system (17). However, our patient's recurrent PHSA prior to infarction probably excludes this explanation. PHSA, like PTH, may reflect a vascular disorder that may not have demonstrable angiographic vasospasm (9–11).
The natural history of PHSA appears to be largely benign. However, our case highlights a risk of infarction in PHSA in which demonstrable vasospasm may not be present. The pathophysiology of PHSA with or without vasospasm is not understood.
