Abstract

Cluster headache (CH) is a distinct, clinical and epidemiological entity. Attacks are stereotypical. Episodes of severe unilateral headache are accompanied by symptoms of autonomic dysfunction. Both aetiology and pathophysiology of CH remain unknown (1).
Some types of headache are considered neurovascular diseases. Selective inhibitors of phosphodiesterase (PDEi) have been included in the 2nd edition of the International Classification of Headaches (2) as headache triggers. PDE are enzymes involved in cAMP and cGMP degradation. When they are inhibited, nucleotide levels increase. This is the mechanism used by sildenafil, a highly selective inhibitor of intracellular cGMP degradation (3). Sildenafil and dipiridamol are the best studied PDE-5i and both trigger headaches as a secondary effect (3, 4).
We now report the first observation, to our knowledge, of isolated attacks of CH after sildenafil administration during the remission period in a patient with a personal history of this particular type of headache.
Case report
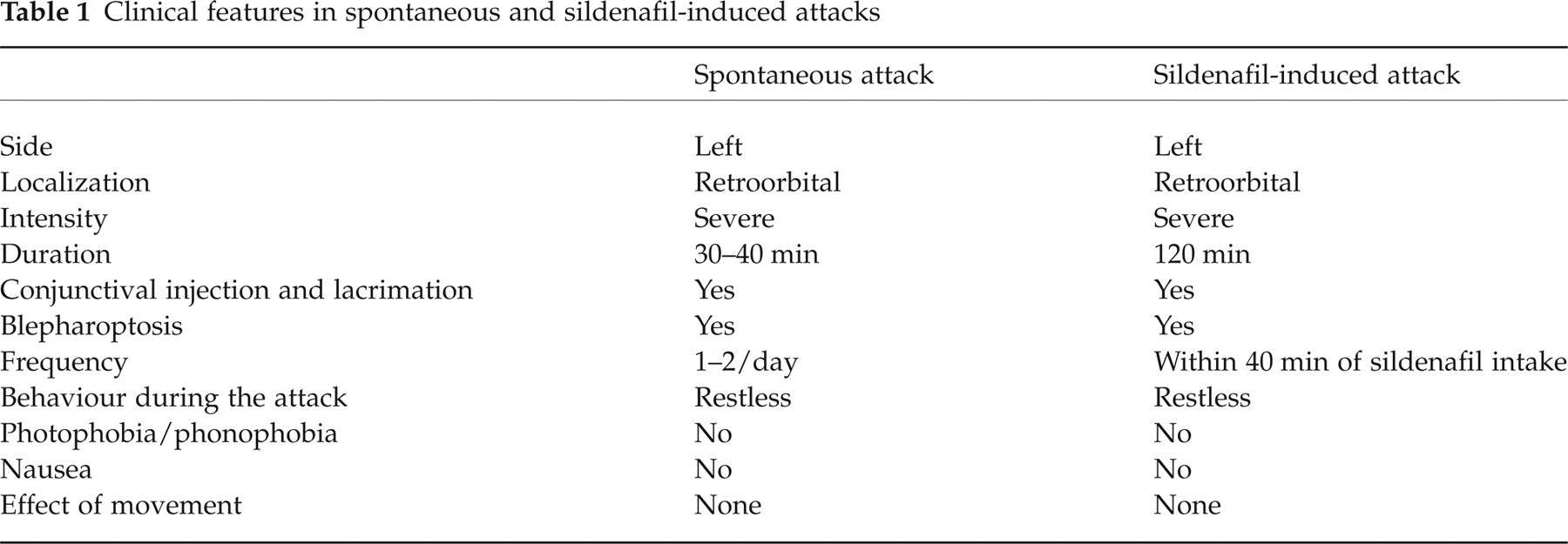
A 54-year-old male patient had suffered from an episodic CH subtype throughout a 27-year period. Cluster ‘bouts’ occurred once or twice a year and lasted 20–40 days. Each individual attack lasted 30–40 min and occurred once or twice a day. During the attack, he presented a severe drilling pain in the left retroorbital area, associated with conjuctival injection, tearing and blepharoptosis on the ipsilateral side, and the pain was so excruciating that the patient was unable to lie down. Pain attacks were never provoked by alcohol. Neuroimaging [magnetic resonance imaging (MRI) and angio MRI] was normal. In our case, subcutaneous sumatriptan was effective in stopping acute attacks and a daily 800-mg lithium dose was effective in reducing the length of the symptomatic cluster period.
During the remission period the patient received 50 mg of sildenafil. Twenty minutes later he developed a typical CH attack, starting in the left retroorbital area and associated with lacrimation, conjuctival injection and ptosis, but lasting 120 min in spite of the sumatriptan intake. He did not have other attacks on the following days.
A month later, he was administered another dose of sildenafil presenting a new isolated attack of CH in the same location, the left retroorbital area, with the same autonomic symptoms and also lasting around 120 min. His behaviour during the sildenafil-induced attacks was identical to that during spontaneous attacks (Table 1).
Clinical features in spontaneous and sildenafil-induced attacks
Three months later and without having taken any drugs he developed a new cluster period with the typical pattern.
In a new remission period he agreed to try sildenafil again. Written consent was obtained. Administration of sildenafil triggered an attack within the following 30 min while the spontaneous attacks the patient used to have at about 14.00 h and/or 03.00 h were also present. We repeated the administration of a 50-mg sildenafil dose three times more on three different days with identical results.
During the following cluster period we tried to trigger pain attacks with sildenafil. New written consent was obtained. He was administered the active drug three times on three different days and a placebo (single blind) twice on different days. There was a pain episode within the following 40 min each time he received the active drug. All pain attacks were on the left side, retroorbital and associated with lacrimation, conjuctival injection and ptosis, lasting 100–130 min under sumatriptan treatment. When the patient was administered the placebo, there were no pain attacks and he did not experience a sexual response. The characteristics of the pain regarding localization, quality, associated phenomena and behaviour were similar in all attacks, either spontaneous or sildenafil induced, but the former were shorter (30–45 min) than the latter (100–130 min), although in all cases the patient received sumatriptan subcutaneously in the first 15 min of pain onset. In this bout, as in the preceding ones, the patient responded to an 800-mg lithium dose a day.
Discussion
Glyceryl trinitrate (GNT), a prodrug for nitric oxide (NO), induces migraine attacks indistinguishable from spontaneous attacks in around 80% of migraine suffers (5) and headaches in CH patients and normal volunteers (6). In CH patients, pain attacks occur 1 h after GNT administration but only if the patient is within the active cluster period (7).
One the other hand, PDEi produce a monophasic pain within 5 h after administration. Sildenafil induces a headache, usually as a side-effect. In migraine sufferers, attacks mimic migrainous pain (3). In healthy volunteers attacks may mimic a tension-type headache (8).
CH attacks induced by sildenafil have not previously been mentioned anywhere either during active or remission periods.
In our patient, we observed a repeated painful response after sildenafil intake during both active and remission periods. The only difference between spontaneous and induced attacks was the length of the crisis. This response was not present when the patient received placebo.
The difficulty in searching for causative mechanisms involved in ‘vascular headaches’ is that this condition depends on studies performed on patients, since no validated animal models exist.
Classically it has been thought that PDEi produce headaches by vasodilation, considering the ‘vascular’ characteristics of the pain. However, Olesen et al. demonstrated that headaches appear without any changes in middle cerebral artery diameter (3). They also reported that plasma levels of calcitonin gene-related peptide, cAMP and cGMP after sildenafil administration were similar to those found after placebo administration; they were not increased as would have been expected (9). However, it cannot be excluded that these second messengers may play a role in the pathophysiology of headaches, acting inside the cells, the perivascular space or local brain areas.
We believe that CH patients should be alerted to the possibility of experiencing a pain attack after sildenafil intake during an active or even a remission period.
