Abstract
A randomized, controlled, cross-over trial compared single doses of 50 mg diclofenac potassium sachets and tablets with placebo in 328 patients with migraine pain, treating 888 attacks. For the primary endpoint 24.7% of the patients were pain free at 2 h postdose with sachets, 18.5% for tablets and 11.7% for placebo. Treatment differences were significant for sachets vs. placebo (P < 0.0001), tablets vs. placebo (P = 0.0040) and for sachets vs. tablets (P = 0.0035). The numbers needed to treat compared with placebo to achieve pain free at 2 h were 7.75 [95% confidence interval (CI) 5.46, 13.35] for sachets and 15.83 (95% CI 8.63, 96.20) for tablets. Sachets were also statistically superior to tablets for sustained headache response, sustained pain free and reduction in headache intensity within the first 2 h postdose measured on a visual analogue scale (P < 0.05). Onset of analgesic effect was 15 min for sachets and 60 min for tablets. Fewer patients needed rescue medication, and there were marked improvements in accompanying symptoms and working ability with both sachets and tablets vs. placebo. No safety issues were identified. This study demonstrates that sachets offer patients suffering from migraine pain a more effective treatment with a faster onset of analgesia when compared with tablets.
Introduction
Migraine is a common painful disorder affecting approximately 10–14% of the population, predominantly women (1, 2). Migraine attacks are often treated with unspecific (symptomatic) agents including non-steroidal anti-inflammatory drugs (NSAIDs) and common analgesics (often in combination with an antiemetic). Migraine-specific agents include ergot alkaloids and the triptans. Diclofenac is a potent NSAID that has been in clinical use for many years, particularly for the treatment of inflammatory, degenerative and rheumatic diseases, and soft-tissue rheumatism. It is also effective in treating non-rheumatic painful and inflammatory conditions such as post-traumatic and postoperative pain (3–9) and migraine headache (10–12).
Diclofenac in a tablet formulation is available as both sodium and potassium salts. The potassium salt of diclofenac has a higher solubility in water than the sodium salt and is more rapidly absorbed (12). Therefore diclofenac potassium 50 mg tablets are indicated for the management of acute and chronic pain, and are especially well suited for treating conditions for which a fast onset of action is important, such as migraine. The clinical efficacy of diclofenac potassium 50 mg tablets for the treatment of migraine has been established in a series of randomized, double-blind trials (10–12).
In one clinical trial diclofenac potassium tablets (50 and 100 mg) were more effective than placebo 2 h after drug intake (10). Headache response occurred in 22%, 39% and 44% of patients on placebo and on diclofenac potassium tablets 50 mg and 100 mg, respectively (P < 0.05 vs. placebo). Tolerability was also comparable between diclofenac potassium tablets and placebo. Moreover, diclofenac potassium tablets (50 and 100 mg) displayed similar efficacy as the 5-HT1D receptor agonist sumatriptan (100 mg tablets), but had a faster onset of effect (11). Statistically significant pain relief occurred at 60 min postdose for diclofenac potassium tablets, compared with 90 min for sumatriptan. Diclofenac potassium 50 mg tablets were also more effective than ergotamine + caffeine and demonstrated a faster onset of action (12). At 1 h postdose statistically significant reductions in pain intensity were observed vs. placebo for diclofenac potassium tablets but not for ergotamine + caffeine. No advantage of the 100-mg dose of diclofenac potassium over the 50-mg dose was found in any clinical study.
Diclofenac potassium sachets are a new drinkable formulation containing 50 mg of diclofenac potassium that have been developed primarily for use in acute pain and migraine attacks. Each sachet contains a powder that is dissolved in water and drunk by the patient. The expected advantage of diclofenac potassium sachets is a faster onset of analgesic effect than observed with diclofenac potassium tablets as the sachet formulation is more rapidly absorbed (13–15).
The current clinical trial, conducted as part of a clinical development programme for diclofenac potassium sachets, evaluated the efficacy, time to onset of action and safety of diclofenac potassium sachets in comparison with diclofenac potassium tablets and placebo in treating the pain and associated clinical signs of migraine. The measurements used were consistent with other migraine trials, and with the guidelines of the International Headache Society (IHS) Committee for the conduct of clinical trials in migraine and with current European guidelines for clinical studies in migraine (16, 17).
Patients and methods
This was a double-blind, double-dummy, randomized, multicentre, cross-over trial comparing 50 mg diclofenac potassium sachets, 50 mg diclofenac potassium tablets, and placebo in patients with migraine pain. The study was performed in 21 centres in five European countries in 2003 (details of the study group are available in the Appendix). The protocol was consistent with the declaration of Helsinki and was approved by the local ethics committees at each site. The primary objective was to determine whether a single dose of diclofenac potassium sachets was superior to placebo and non-inferior to diclofenac potassium tablets in treating the pain and associated symptoms of migraine headache. Secondary objectives were to evaluate the time to onset of analgesic effect and the safety of diclofenac potassium sachets in comparison with diclofenac potassium tablets and placebo.
Patients
The trial population was selected based on the criteria recommended by the IHS 1988 guidelines for the diagnoses of migraine with or without aura and the IHS 2000 guidelines for the conduct of clinical trials in migraine (16, 18). Male or female patients, 18–65 years, suffering from migraine with or without aura, but without interval headaches between migraine attacks, were eligible. Patients had to have a disease duration of at least 1 year and two to six migraine attacks per month over the previous 3 months. Females who were pregnant, lactating or considered at risk of pregnancy were not eligible.
Each patient was informed about the nature and aim of the study, and written informed consent was obtained. Patients were excluded if they suffered from other types of migraine (e.g. menstrual), had received an acute headache medication within 48 h preceding study medication intake, or lithium, digoxine, methotrexate, cyclosporin, phenobarbital or any other long-acting analgesic formulations during the study. Prophylactic treatment for migraine was permitted if patients remained on a constant dosing regimen with no more than one prophylactic agent. Patients with a known hypersensitivity to NSAIDs, acetylsalicylic acid or other drugs with prostaglandin-synthetase inhibiting activity, with severe cardiac, liver or acute renal insufficiency, active peptic ulcer disease or a history of significant gastrointestinal disease were excluded.
Study procedures
Eligible patients were randomized to double-blind treatment and received study medication to treat three separate migraine attacks at home. For each attack study medication was self-administered by the patient as soon as they were certain they were experiencing symptoms of a migraine attack. Study medication was not to be used unless there had been a symptom-free period of at least 48 h. Also, the last 48 h prior to intake of study medication had to be completely free of any acute headache medication. A patient diary was used to record efficacy and safety parameters over an 8-h observation period [including predose ‘baseline’ assessments, and notes on adverse events (AEs)]. Patients had to return to the study clinic at least every 4 weeks for continued review and for a final study visit after three migraine attacks had been treated. At each visit completed patient diaries were collected and AEs were transcribed into the study case record forms. Maximum study duration for each patient was planned for 2 months.
Medication and the randomization scheme were prepared by Novartis Pharma AG. Diclofenac potassium sachets were developed using a patented drug delivery technology (Patent of APR Applied Pharma Research SA, Switzerland licensed to Novartis Pharma AG). Patients received three sets of study medication, designated as A, B and C where: A = diclofenac potassium sachet and placebo tablet; B = diclofenac potassium tablet and placebo sachet; C = placebo sachet and placebo tablet. Medication was taken in a predefined randomized treatment sequence (A/B/C, B/C/A, C/A/B).
Headache intensity was recorded before taking study medication (baseline, time = 0) and at 1, 2, and 8 h postdose on a verbal scale assessed as ‘none’ (0), ‘mild’ (1), ‘moderate’ (2), or ‘severe’ (3). In addition to allow for comparison between different pain scales and to increase the overall validity of the results, headache intensity was also recorded on a 100-mm visual analogue scale (VAS) at 0, 15, 30, 45, 60, 90 min, 2, 3, 4, 6, and 8 h postdose (0 mm = no headache to 100 mm = excruciating headache requiring bed rest). The presence of accompanying symptoms (‘nausea’, ‘vomiting’, ‘photophobia’, ‘phonophobia’, ‘none’) was recorded at 0, 1, 2, 4, 6, and 8 h postdose. Working ability was evaluated on a verbal scale at 0, 2 and 8 h postdose as ‘normal’ (0), ‘mild impairment’ (1), ‘severe impairment’ (2), or ‘bed rest required’ (3). Use of rescue medication during the first 8 h, time of intake, and the time of attack recurrence within 48 h were also recorded.
Rescue medication was supplied to each patient where one dose was two tablets of paracetamol, 500 mg each tablet. Additional rescue medications could be prescribed by the investigator if paracetamol was considered inadequate. Patients were instructed not to take rescue medication within 2 h of treatment unless absolutely necessary. Patients who took rescue medication before the 8-h postdose time point had to record final efficacy assessments in their diary immediately after taking the rescue medication, and were to discontinue further efficacy measurements.
Safety assessments consisted of monitoring and recording all AEs and serious adverse events (SAEs).
Sample size determination and statistical analyses
Sample size was based on the percentage of pain-free patients at 2 h postdose assuming 10% on placebo and 30% on both diclofenac potassium sachets and diclofenac potassium tablets. With a power of 80%, a significance level of 2.5% one-sided and a non-inferiority margin of 10%, the demonstration of superiority over placebo required 80 patients to be analysed, whereas the analysis of non-inferiority required 236 patients (with an assumed proportion of discordant pairs of 0.3). It was planned to randomize a total of 300 patients.
The primary efficacy parameter was the percentage of patients pain free at 2 h postdose (headache intensity on the verbal scale =‘none’). Patients who took rescue medication within 2 h postdose were not pain free. The last postdose headache intensity data before 2 h and the first data after 2 h (VAS or verbal scale) were used to impute missing values.
The primary efficacy parameter was analysed for the intent-to-treat (ITT) population (all treated patients) and the per-protocol (PP) population. The PP population included ITT patients who treated three migraine attacks (separated by >48 h), did not use rescue medication within 2 h postdose for any attack, and had a baseline pain assessment within 1 h prior to taking study drug. A logistic regression analysis was used with the variables treatment, period, patient and baseline VAS headache intensity. A subgroup analysis included ITT patients with moderate or severe headache intensity at baseline. The same logistic regression analysis was applied to the endpoints: headache response at 2 h (pain free or pain reduction from moderate or severe at baseline to mild at 2 h), sustained headache response (headache response at 2 h and no recurrence or worsening of headache and intake of rescue medication within 24 h) and sustained pain free (pain free at 2 h and no recurrence of headache and intake of rescue medication within 24 h).
An analysis of covariance (
Results
Study dates
Patient recruitment began on 3 July 2003. The study was stopped before all randomized patients had completed three treated migraine attacks on 14 November 2003. This was performed as, by this date, sufficient numbers of patients for the statistical analysis had completed the study.
Characteristics of the patients
A total 328 patients were randomized (from 21 centres) and 317 received at least one treatment [291 diclofenac potassium sachets, 298 diclofenac potassium tablets and 299 placebo (888 attacks were treated)], and 274 patients received all three treatments (Fig. 1). A total of 43 treated patients discontinued the study.

Disposition of patients.
All 317 patients receiving treatment were included in the ITT population and were analysed for safety. Deviations from the protocol led to the exclusion of 92 patients from the PP population (n = 225) (Fig. 1). Demographic and background characteristics are summarized for all treated patients in Table 1. There were only slight differences between treatments regarding demographic data and baseline migraine attack data. Data were similar for the PP population (data not shown).
Demographic and background characteristics (treated patients)

SD, standard deviation; VAS, visual analogue scale.
Baseline data are for each individual treated attack.
Pain-free and headache response
For the ITT population, 2 h after intake of study medication 24.7% (72/291) of the patients were pain free with diclofenac potassium sachets, compared with 18.5% (55/298) for diclofenac potassium tablets and 11.7% (35/299) for placebo (Table 2). Differences from placebo were statistically significant for both diclofenac potassium sachets and diclofenac potassium tablets. Diclofenac potassium sachets were also statistically significantly more effective than diclofenac potassium tablets (Table 2). The PP analysis confirmed the results of the ITT analysis (data not shown).
Treatment differences in pain free, headache response, sustained headache response, and sustained pain free based on the verbal scale (intention-to-treat population)
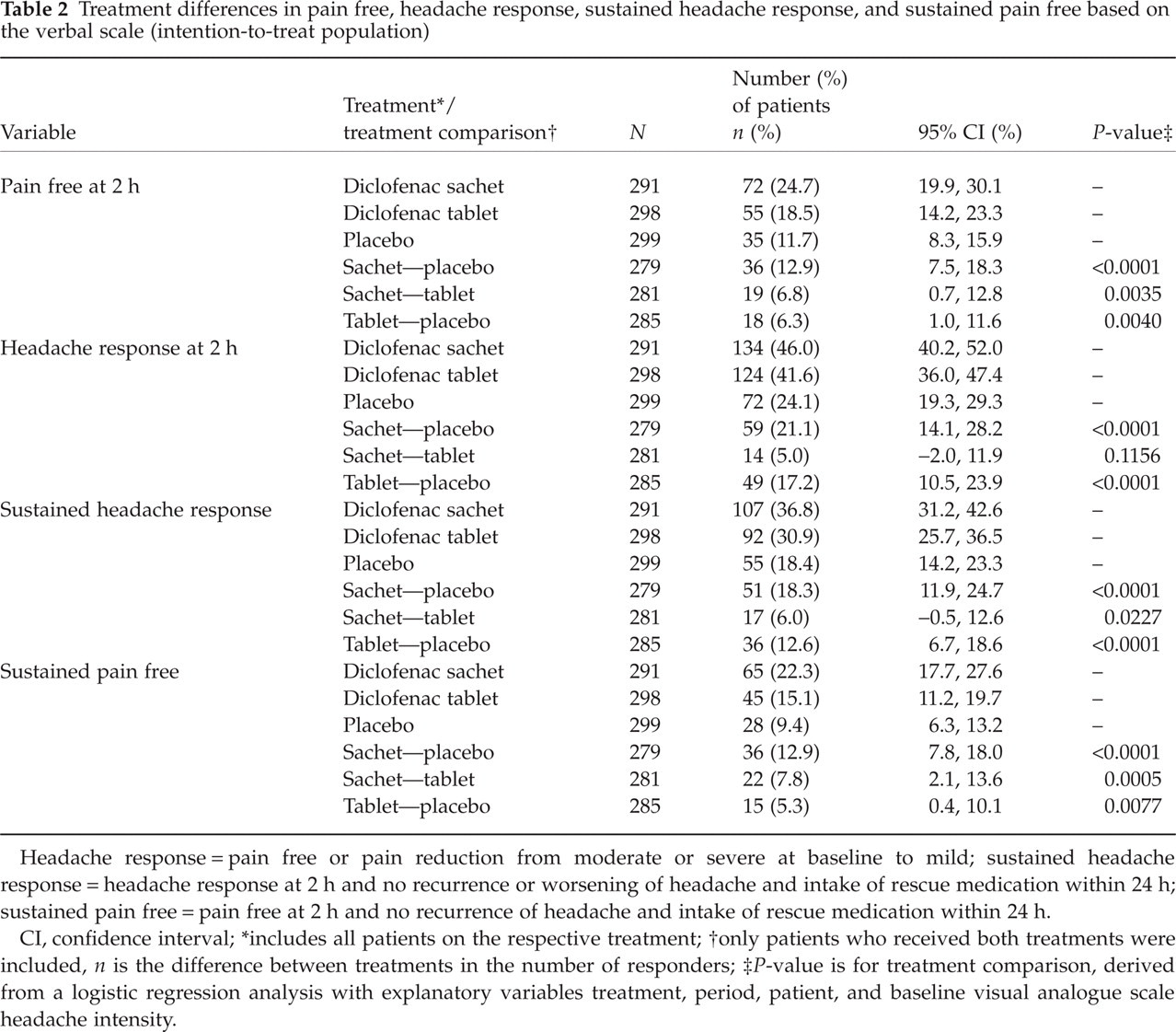
Headache response = pain free or pain reduction from moderate or severe at baseline to mild; sustained headache response = headache response at 2 h and no recurrence or worsening of headache and intake of rescue medication within 24 h; sustained pain free = pain free at 2 h and no recurrence of headache and intake of rescue medication within 24 h.
CI, confidence interval;
includes all patients on the respective treatment;
only patients who received both treatments were included, n is the difference between treatments in the number of responders;
P-value is for treatment comparison, derived from a logistic regression analysis with explanatory variables treatment, period, patient, and baseline visual analogue scale headache intensity.
The predefined subgroup analysis of patients with moderate to severe headache at baseline also showed similar results as for the full ITT analysis. A total of 24.2% (64/265) of the patients were pain free with diclofenac potassium sachets, compared with 17.0% (45/265) for diclofenac potassium tablets and 12.5% (32/257) for placebo. Again diclofenac potassium sachets were statistically significantly more effective than placebo (P < 0.0001, logistic regression) and diclofenac potassium tablets (P = 0.0214, logistic regression).
The numbers needed to treat to achieve pain free at 2 h within the full ITT analysis were 7.75 [95% confidence interval (CI) 5.46, 13.35] for diclofenac potassium sachets and 15.83 (95% CI 8.63, 96.20) for diclofenac potassium tablets, both compared with placebo.
The number of patients with headache response at 2 h was 46.0% (134/291) with diclofenac potassium sachets, compared with 41.6% (124/298) for diclofenac potassium tablets and 24.1% (72/299) for placebo (Table 2). These rates were also statistically significant over placebo for both diclofenac potassium sachets and diclofenac potassium tablets, but the difference between diclofenac potassium sachets and tablets was not statistically significant (Table 2).
Similar results were observed for sustained headache response and sustained pain free. For both parameters diclofenac potassium sachets and diclofenac potassium tablets were more effective than placebo (Table 2). Moreover, diclofenac potassium sachets were statistically significantly more effective than diclofenac potassium tablets regarding rates of sustained headache response and sustained pain free.
Headache intensity and onset of effect
For the ITT population, during the first 2 h after intake of study medication the
average reduction in headache intensity on the VAS was 16.6 mm, 11.5 mm and 7.0 mm
for diclofenac potassium sachets, diclofenac potassium tablets and placebo,
respectively (Table
3). Diclofenac potassium sachets and diclofenac potassium tablets were
statistically significantly more effective than placebo, and diclofenac potassium
sachets were also more effective than diclofenac potassium tablets. Similar results
were observed over the first 4 and 8 h, with both diclofenac potassium sachets and
diclofenac potassium tablets being more effective than placebo in average reduction
in VAS headache intensity (P < 0.0001,
Average reduction of headache intensity on the visual analogue scale (VAS) over the first 2 h (intention-to-treat population)

LS, least squares; SE, standard error of the mean; CI, confidence interval.
All statistics for treatment effects and treatment comparisons are based
on the
The mean VAS headache intensity at different time points is plotted in Fig. 2, which shows a discernable difference in favour of diclofenac potassium sachets. This was maintained during the entire 8-h observation period (Fig. 2, insert). The time to onset of analgesic effect was at 15 min for diclofenac potassium sachets and at 60 min for diclofenac potassium tablets. For both treatments, VAS headache reductions were statistically significantly greater than on placebo at all time points from their onset of analgesic effect onwards to 8 h. In addition, diclofenac potassium sachets were also statistically significantly more effective than diclofenac potassium tablets regarding VAS headache reduction between 15 and 90 min postdose, but not at 2 h postdose.

Mean visual analogue scale (VAS) headache intensity at different time points (intention-to-treat population). Headache intensity according to a VAS (0 = no headache to 100 = excruciating headache requiring bed rest). Values were recorded at 0, 15, 30, 45, 60, 90 min, 2, 3, 4, 6 and 8 h postdose. ∗P ≤ 0.05 for diclofenac sachet vs. placebo; †P ≤ 0.05 diclofenac sachet vs. diclofenac tablet; ‡P ≤ 0.05 for diclofenac tablet vs. placebo. •, Diclofenac sachet; ▪, diclofenac tablet; ▵, placebo.
Additional results for headache intensity on the verbal scale are presented in Table 4. For this analysis data after taking rescue medication were excluded, but at 2 h postdose the percentages of pain-free patients (headache intensity on the verbal scale =‘none’) were almost identical to the primary analysis: 24.7% (63/255) of the patients were pain free with diclofenac potassium sachets, compared with 18.6% (48/258) for diclofenac potassium tablets and 12.7% (34/267) for placebo.
Headache intensity on the verbal scale (intention-to-treat population)

Accompanying symptoms and working ability
Diclofenac potassium sachets and diclofenac potassium tablets were more effective than placebo in reducing the percentage of patients experiencing nausea, vomiting, photophobia and phonophobia. However, differences between the two active treatments were not meaningful, and at 2 h postdose the percentage of patients who were free of any accompanying symptoms was 41.9% (106/253) for diclofenac potassium sachets, 40.5% (105/259) for diclofenac potassium tablets and 29.6% (80/270) for placebo.
Marked improvements were seen in working ability at 2 h postdose when compared with baseline values for diclofenac potassium sachets and diclofenac potassium tablets, with the worst treatment results seen on placebo. At 2 h postdose the percentage of patients who had improved from baseline was 50.0% (123/266) for diclofenac potassium sachets, 42.7% (109/255) for diclofenac potassium tablets and 26.5% (69/260) for placebo. In addition, the percentages of patients with mild impairment, severe impairment and bed rest required were all reduced from baseline with both diclofenac potassium sachets and diclofenac potassium tablets at 2 h postdose. Notably, there was a substantial difference in patients requiring bed rest, and only 7.6% (19/251) of patients taking diclofenac potassium sachets were still bed bound compared with 15.2% (39/257) and 20.8% (55/264) of patients on diclofenac potassium tablets and placebo, respectively.
Rescue medication and recurrence of headache
Rescue medication use was restricted voluntarily during the first 2 h, but up to 3 h after dosing 27.8% (83/299) of patients on placebo reported rescue medication intake, compared with 17.2% (50/291) and 21.1% (63/298) of patients taking diclofenac potassium sachets and diclofenac potassium tablets, respectively. By the end of the 8-h observation period 50.2% (150/299) of patients on placebo had remedicated, compared with 35.1% (102/291) and 36.2% (108/298) of patients on diclofenac potassium sachets and diclofenac potassium tablets, respectively. The median time to using rescue medication was similar for placebo (3.0 h) and diclofenac potassium sachets (3.05 h) and slightly lower for diclofenac potassium tablets (2.87 h), when considering only those patients who took rescue medication.
The percentages of patients with recurrence of attack within 48 h after attack resolution were 15.5% (45/291) for diclofenac potassium sachets, 21.8% (65/298) for diclofenac potassium tablets and 21.1% (63/299) for placebo. Median time to attack recurrence was slightly higher for diclofenac potassium sachets (14.0 h) than for diclofenac potassium tablets (12.7 h) and placebo (12.5 h).
Tolerability
No patient had an SAE, and no major safety issues arose during the study. A total of 83 AEs were reported: 20 were assigned to diclofenac potassium sachets, 17 to diclofenac potassium tablets and 32 to placebo. Fourteen AEs were not attributed to any treatment (13 occurred ‘between two drugs’ and one AE ‘post last drug’). Gastrointestinal complaints were the most frequent AEs reported (Table 5).
Most common treatment assigned adverse events (intention-to-treat population)

Numbers are adverse events (AEs) reported. Most frequent = incidence ≥ 0.5% of any treatment. AEs are assigned according to their date of onset to the last treatment taken within 72 h postdose. Accompanying symptoms (nausea, vomiting, photophobia, phonophobia) experienced within 8 h postdose were not recorded as AEs, only if they continued over the 8-h time period.
Of the 69 assigned AEs, only 10 were of severe intensity (two diclofenac potassium sachets: vomiting and oral paraesthesia; three diclofenac potassium tablets: dizziness, vertigo and haematuria; five placebo: upper abdominal pain, diarrhoea, nausea, abdominal pain and rigors). A total of 38 AEs (13 diclofenac potassium sachets, six diclofenac potassium tablets, 19 placebo) were suspected to be causally related to the study drug. The most frequent related AEs were diarrhoea, dyspepsia, abdominal pain, dry mouth and nausea. All 14 unassigned AEs were not suspected to have any relationship to study drug. Six patients had an AE leading to study discontinuation (diclofenac potassium sachet: urticaria, vomiting; diclofenac potassium tablet: vomiting, urticaria and haematuria; placebo: eye swelling). One case of urticaria and the haematuria were suspected as possibly drug-related.
Discussion
The results of this double-blind, randomized, cross-over study clearly demonstrate the efficacy and tolerability of the 50 mg diclofenac potassium sachet formulation in treating migraine pain. Diclofenac potassium sachets were superior to placebo and were even more effective than diclofenac potassium tablets for the primary endpoint of pain free at 2 h postdose and the secondary endpoints of sustained pain free and sustained headache response. The number of patients achieving headache response was not significantly different between the two active treatments, but this can be expected as it is likely that patients will have experienced at least some pain reduction at 2 h postdose with either diclofenac potassium sachets or diclofenac potassium tablets. A clear difference in onset of analgesic effect was also seen in favour of diclofenac potassium sachets when compared with diclofenac potassium tablets and the reduction of headache pain seen with diclofenac potassium sachets was maintained over the 8-h postdose period.
The superior clinical efficacy of diclofenac potassium sachets regarding pain free at 2 h postdose was also observed in a subgroup analysis of patients who had moderate to severe headache and in a PP analysis population, indicating a robust and clinically significant treatment effect. Moreover, the results obtained for headache intensity using the VAS (with different descriptive terms) also supported the primary efficacy findings.
Clinical trials using diclofenac potassium tablets in migraine have already established their efficacy and a faster onset of action than sumatriptan or ergotamine plus caffeine (10–12). The current trial provides convincing evidence that diclofenac potassium sachets will be an even more rapid and effective treatment for migraine than diclofenac potassium tablets. The potential for a faster onset of action for diclofenac potassium in a drinkable formulation has also been demonstrated in pharmacokinetic studies using sachets, tablets and oral drops (13, 14). In the current trial a new diclofenac potassium sachet formulation—which compared with diclofenac potassium tablets has a significantly shorter time to higher peak plasma concentrations (15)—has been tested and the results provide convincing evidence of its clinical utility and fast onset of action.
The primary endpoint of patients pain free at 2 h obtained with diclofenac potassium sachets (24.7%) and the associated numbers needed to treat (7.75) can be compared indirectly with those for the widely prescribed triptans. In a recent meta-analysis of 37 published clinical trials covering almotriptan, eletriptan, frovatriptan, naratriptan, rizatriptan, zolmitriptan and sumatriptan, the number of patients pain free within 2 h ranged from 11.6% (with 2.5 mg frovatriptan) to 40.8% (with 10 mg rizatriptan) and the placebo-corrected numbers needed to treat ranged from 11.3 (with 2.5 mg frovatriptan) to 3.2 (with 10 mg rizatriptan) (19). As diclofenac potassium sachets have not been compared with any triptan in a controlled clinical trial, direct comparisons cannot be made, but comparable analgesic efficacy has already been established between diclofenac potassium tablets 50 mg and sumatriptan 100 mg (11). Moreover, it remains to be shown definitively (using controlled clinical studies) if the triptans are superior to common aspirin or NSAIDs regarding analgesic efficacy in migraine (20–26).
The recommended dosing schedule for 50 mg diclofenac potassium tablets for analgesia allows a repeated dosing up to three times per day (27, 28). As expected for a single dose trial, the use of rescue medication increased after 2 h postdose for all treatments but was higher for placebo when compared with both diclofenac potassium sachets and diclofenac potassium tablets. For certain endpoints (verbal headache intensity, accompanying migraine symptoms and working ability) the use of rescue medication led to significant reductions in the number of patients contributing to the analysis, and reduced the reliability of the interpretation for time points after 2 h. Hence, for these endpoints results after 2 h postdose were not presented in the current analysis.
The safety profile of diclofenac is similar to other NSAIDs and is well documented. In the present study, a single dose of both diclofenac potassium sachets and diclofenac potassium tablets was well tolerated and no major safety issues were identified. Gastrointestinal complaints appeared the most frequent AEs reported. However, digestive AEs are known side-effects of diclofenac (27) and they were not more frequently reported for the active treatments than for placebo. Moreover, events which were considered as causally related to the study drug or were severe were all more frequent with placebo than with either diclofenac formulation. Other events leading to discontinuation of study included two events of urticaria and one event of haematuria. Rarely occurring urticaria and isolated cases of haematuria are recognized side-effects of diclofenac and have been reported with an incidence of <1% (27). Therefore, in this clinical trial no evidence was provided that this new formulation of diclofenac potassium differs from other diclofenac potassium formulations with regard to safety.
In conclusion, the results of this study demonstrate that diclofenac potassium sachets offer patients suffering from migraine pain benefits of clinical importance by providing a more effective treatment with a faster onset of analgesia when compared with diclofenac potassium tablets.
Footnotes
Acknowledgements
The study was sponsored by Novartis Pharma AG, Basel, Switzerland. The lead author had free access to all data and analyses. Statistical analysis was provided by Lilienthal J, DATAMAP GmbH, Freiburg, Germany.
Appendix: study details
The study group consisted of the following principal investigators at 21 centres: Diener H-C., Gendolla A., University of Essen, Essen, Germany; Flöter T., Pain Relief Centre Frankfurt, Frankfurt, Germany; Owega A., Facharztpraxis für Neurologie, Psychiatrie und Psychotherapie, Köln, Germany; Schumann G., Bochum, Germany; Zöller B., Heidelberg, Germany; Csányi A., Petz Aladár County Hospital, Gyõr, Hungary; Fazekas A., St Istvan Hospital, Budapest, Hungary; Gács G., Péterfy Sandor Hospital, Budapest, Hungary; Harcos P., St Imre Hospital, Budapest, Hungary; Varga J., University of Pécs, Pécs, Hungary; Colombo B., Comi G., San Raffaele Hospital, Milano, Italy; Gallai V., University of Perugia, Perugia, Italy; Montagna P., University of Bologna, Bologna, Italy; Pinessi L., University of Turin, Torino, Italy; Keizer K., Catharina Hospital, Eindhoven, the Netherlands; Mulder L.J.M.M., Ikazia Hospital, Rotterdam, the Netherlands; Czapiñski P., Epilepsy and Migraine Treatment Centre, Krakow, Poland; Klimek A., Kopernik Hospital, Lódz, Poland; Kotowicz J., Military Medical Institute, Warsaw, Poland; Lyczak P., Navy Hospital, Gdañsk-Oliwa, Poland; Stepien A., Aviation Institute of Medicine, Warsaw, Poland.
