Abstract
We recorded deep pain and surface electromyographic (EMG) responses to stress in 22 migraineurs during headache-free periods, 18 patients with tension-type headache (TTH), and 44 healthy controls. Sixty minutes of cognitive stress was followed by 30 min relaxation. EMG and pain (visual analogue scale) in the trapezius, neck (splenius), temporalis and frontalis areas were recorded. TTH patients had higher pain responses in temporalis and frontalis (with similar trends for trapezius and splenius) and more potentiation of pain during the test than controls. Migraine patients developed more pain in the splenius and temporalis than controls. Muscle pain responses were more regional (more pain in the neck and trapezius compared with the temporalis and frontalis) in migraine than in TTH patients. TTH patients had delayed pain recovery in all muscle regions compared with controls, while migraine patients had delayed pain recovery in a more restricted area (trapezius and temporalis). EMG responses were not different from controls in headache patients, and EMG responses did not correlate with pain responses. TTH patients had delayed EMG recovery in the trapezius compared with controls and migraine patients. These results support the concept that (probably central) sensitization of pain pathways and the motor system is important in TTH. Less pronounced and more regional (either peripheral or central) trigeminocervical sensitization seems to be important in migraine. Surface-detectable muscular activation does not seem to be causal for pain during cognitive stress either in migraine or in TTH.
Introduction
Perceived stress is a triggering factor for headache in both migraine and tension-type headache (TTH) (1–4). It has been hypothesized that repeated stress, lack of habituation (or sensitization) to repeated stress, or a prolonged physiological response to the stressor (or insufficient recovery) may cause disease, chronic pain or other subjective health complaints (5, 6). One of the physiological responses to cognitive stress is increased muscle activation (7), but the pathophysiological role of muscle activity in headache has not been fully established. In patients with TTH, some studies have reported a higher electromyographic (EMG) activity compared with healthy controls (8, 9), while others have found normal EMG activity in such patients (10). Muscular activation can occur as a direct consequence of stress, or it may be linked to a change in pain modulation such as central sensitization (11). Central sensitization of pain pathways has previously been suggested as an important mechanism in both TTH (12) and migraine (13).
The aim of this study was to investigate pain and muscle activation during and after stress in migraine and TTH patients. We measured surface EMG and perceived pain during controlled low-grade cognitive stress for 1 h followed by 30 min relaxation. This procedure can induce EMG activation and pain in the shoulders, neck and head of patients with TTH (14), cervicogenic headache (15), fibromyalgia (16) and in healthy controls (17). A similar experiment has to our knowledge not been performed on International Headache Society (IHS)-classified migraineurs. By comparing the effects of stress in migraine, TTH and healthy subjects, we aim to contribute useful information about the mechanisms of pain development and pain modulation in these disorders.
Materials and methods
Subjects
Forty-four healthy control subjects, 35 women [mean age 39.7 (SD 12.3) years] and nine men [36.6 (12.4) years], and 40 patients with headache participated in this study. Twenty-two patients were diagnosed with migraine, 20 women [39.8 (14.4) years] and two men [45.0 (7.1) years], and 13 of these patients had aura. Eighteen patients were diagnosed with TTH, nine women [33.8 (11.4) years] and nine men [35.7 (12.3) years]. Twelve of the TTH patients had chronic TTH. All headache patients were recruited from the Department of Neurology, St Olavs University Hospital, and diagnosed according to the IHS classification of headache from 1988 (18) (Table 1). Among the control subjects no one had headache or musculoskeletal pain for more than 1 day per month. Exclusion criteria were: neoplastic disease, hypertension, infectious disease, metabolic, endocrine or neuromuscular diseases, connective tissue disorder, tendinitis, recent significant accident or injury, pregnancy, daily medication (e.g. neuroleptics, antiepileptics, Ca2+-blockers, β-blockers, antidepressants) and significant associated diseases affecting either the heart, lungs, cerebrovascular system, central or peripheral nervous system. Migraineurs with TTH more than 7 days per month were also excluded. Controls were recruited from workplaces in Trondheim. The project was approved by the Regional Ethics Committee. All participants gave written informed consent and received NOK 500 (£42) for transport expenses and inconvenience. The participants were provided with written information concerning the aim of the study prior to the day of the stress test. The aim of studying pain and headache was mentioned, but the information focused on the practical details of the procedure. Pain expectancy (19) therefore probably did not contribute much to pain and, if present, would be similar in patients and controls, since the laboratory personnel were blinded as to the diagnostic status.
Demographic and headache data on subjects included in the study

One migraine patient had some attacks of short duration.
VAS, Visual analogue scale.
Questionnaire and interview
A structured interview concerning headaches and musculoskeletal complaints (distribution, severity and duration) was performed. One of these questions was: ‘Please state the level of general tension you have felt during the last 2–3 months’. The response was scored on a visual analogue scale (VAS) with endpoints: not tense to very tense. Thirteen of the 22 migraineurs reported a migraine attack within 2 days before the stress test, while 11 patients reported an attack within a day after the stress test. Demographic data are given in Table 1. All subjects answered a questionnaire on marital status, weight, medication, stimulant use, exercise habits and sleep problems (data not shown). Subjects also reported headache intensity before and after the test was finished.
Electromyographic recordings
Muscle activity was measured by bilateral recorded surface EMG (electrode diameter 6 mm, fixed distance between the two poles 20 mm) (17). The system noise was ± 1.5 µV (root mean square, RMS). EMG was filtered (10–1250 Hz), stored on a digitizing tape recorder (Earth Data 128), and A/D converted (Powerlab 16S, sampling rate 2 kHz; ADInstruments Ltd, Chalgrove, UK) for calculation of RMS values (100 ms running time window). Sharp transients and ECG artefacts were eliminated with a median filter. Bilateral recordings were performed over the frontalis, temporalis, neck (splenius captis), and trapezius muscles. Frontalis electrodes were placed on a vertical line, crossing the pupil with the inferior electrode 10 mm above the upper border of the eyebrow. The inferior temporalis electrode was placed 10 mm lateral to the external angle of the orbit, while the second electrode was placed vertically above the first. The neck electrode was placed at the C2 level just at the edge of the trapezius muscle about 35–40 mm laterally, while the second electrode was placed directly below. The medial trapezius electrode was placed 10 mm lateral to the midpoint of a line connecting the acromion and the spinous process of C7, while the lateral electrode was placed on the same line. The ground electrode was placed on the C7 spinous process (17).
Procedure
The subjects were seated in an ordinary office chair without armrests and performed a two-choice reaction-time test presented on a PC monitor for 60 min (17). The test involved a grid (7 columns by 5 rows) in which a large and a small square were placed randomly (20). The subject was then presented with a suggestion on how to move the small square to superimpose it on the large square (for instance, ‘two up, four left’), and the subjects responded by pressing either ‘right’ or ‘wrong’ on a panel before them with their right index or ring fingers, respectively. Then the positions of the squares were changed, and a new suggestion was displayed. The subjects were instructed to carry out the assignment as fast and correctly as possible, and the computer provided feedback on performance by informing whether the answer was correct or not, and how fast the trial was performed (very slow, slow, normal, fast or very fast) (21). The ‘normal’ response for each subject was determined as the mean response time during a 5-min trial period.
Before the stress test was started, the procedure was explained and electrodes were fixed in a relaxed atmosphere for about 30 min. A mini trial was also performed to familiarize the subjects with the procedure. The laboratory temperature was kept at 24.5 ± 1 °C. Short maximal voluntary contractions were performed on each pair of muscles twice (frontalis muscle – raising eyebrows, temporalis – clenching teeth, neck – pushing head back against resistance, trapezius – pushing extended arms upwards against resistance at a 45° angle out from the body). The recording proceeded with 5 min uninstructed rest (UIR) followed by 5 min active, instructed rest with visual EMG feedback (FB). The cognitive task was then performed for 1 h (800–1500 trials), followed by 30 min recording during rest. The patients were asked to relax while seated and to move as little as possible during the recovery period. After the UIR and FB periods, at 10-min intervals during the cognitive task and at 10-min intervals during the recovery period, the subjects were asked to mark on a VAS scale their level of pain (no pain to worst bearable pain). The different locations of pain corresponded to the positions of the EMG electrodes (17). No patient had to be excluded because of headache attacks during the test. Levels of tension, fatigue, and sleepiness were also scored (VAS) by the patient during the test. Venous blood was sampled before the test and immediately after the stress period (after 60 min). These data as well as cardiovascular stress responses will be reported in a separate paper.
Data analysis
Mean EMG RMS values in µV were calculated for UIR, FB and each 10-min interval during the cognitive test and during recovery. From these values, four main variables were produced: mean EMG before test (UIR period), mean EMG response during the test (average of 60 min during stress–UIR), early and late EMG recovery responses after the test. Early recovery response was defined as the difference between the mean value during the 65–75-period and the UIR baseline value, late recovery response was defined as the difference between the mean during the 85–95-period and UIR baseline.
Pain responses were calculated for each location as the difference between maximal reported pain during the test (irrespective of right or left side) and baseline pain. To evaluate the patients’ recovery from pain, the number of subjects who did not return to their starting pain were counted for each location at 75 and 95 min (early and late recovery, respectively).
Statistical analysis
The Mann–Whitney U-test was used for intergroup comparisons. Wilcoxon's signed-rank test was used for intragroup comparisons of responses at different times or locations. Differences in pain recovery rates were analysed with Pearson's χ2 test. In order to test if pain or EMG increased or decreased within the test (i.e. from 10 to 60 min), repeated measures
Results
Pain responses to cognitive stress compared with baseline pain
Pain increased significantly compared with baseline during the test for all three groups (Fig. 1). TTH patients reported the highest pain scores in general. Pain responses did not differ significantly between migraine and TTH patients (Table 2).
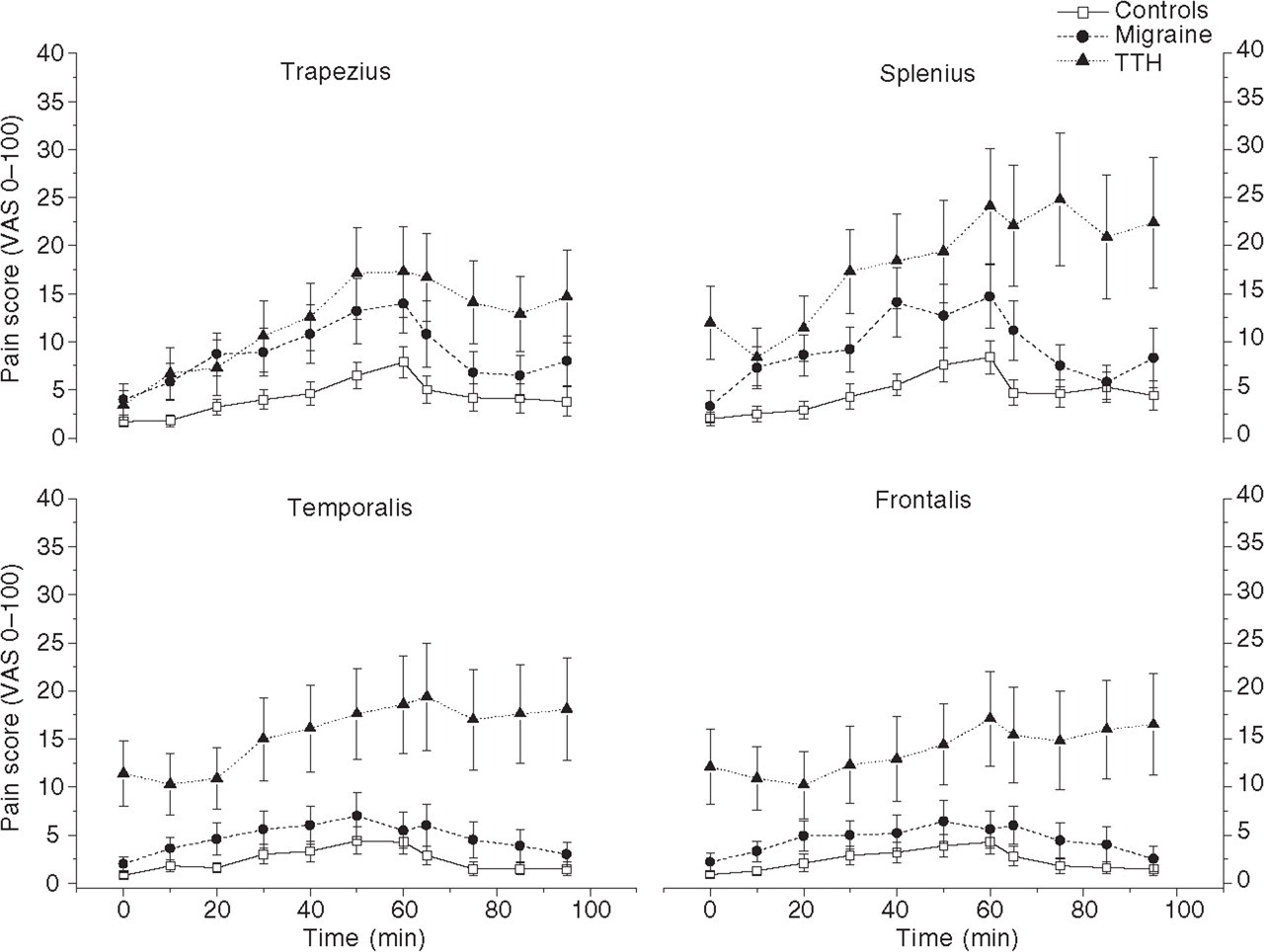
Mean pain (SEM) development as a function of time in the four muscle regions for the three subject groups. The group means were calculated from the average of left and right values for each subject in the group. These graphs deviate accordingly slightly from the median values reported in Table 2. T = 0 min, baseline pain; T = 10–60 min, during the cognitive stress test; T = 65–95 min, relaxation period after the test. Migraineurs had more baseline pain in the temporalis (P = 0.073) and frontalis (P = 0.038) than controls, while tension-type headache patients had more baseline pain in the neck (P = 0.014), temporalis (P < 0.0005) and frontalis (P < 0.0005) compared with controls, and more baseline pain in the temporalis (P = 0.003) and frontalis (P = 0.048) compared with migraineurs. Both patients and controls had significantly increased pain during the test compared with baseline values (P ≤ 0.05).
Pain test responses [VAS scale (mm), median (range)], and the number (%) of patients who developed pain during the cognitive stress test
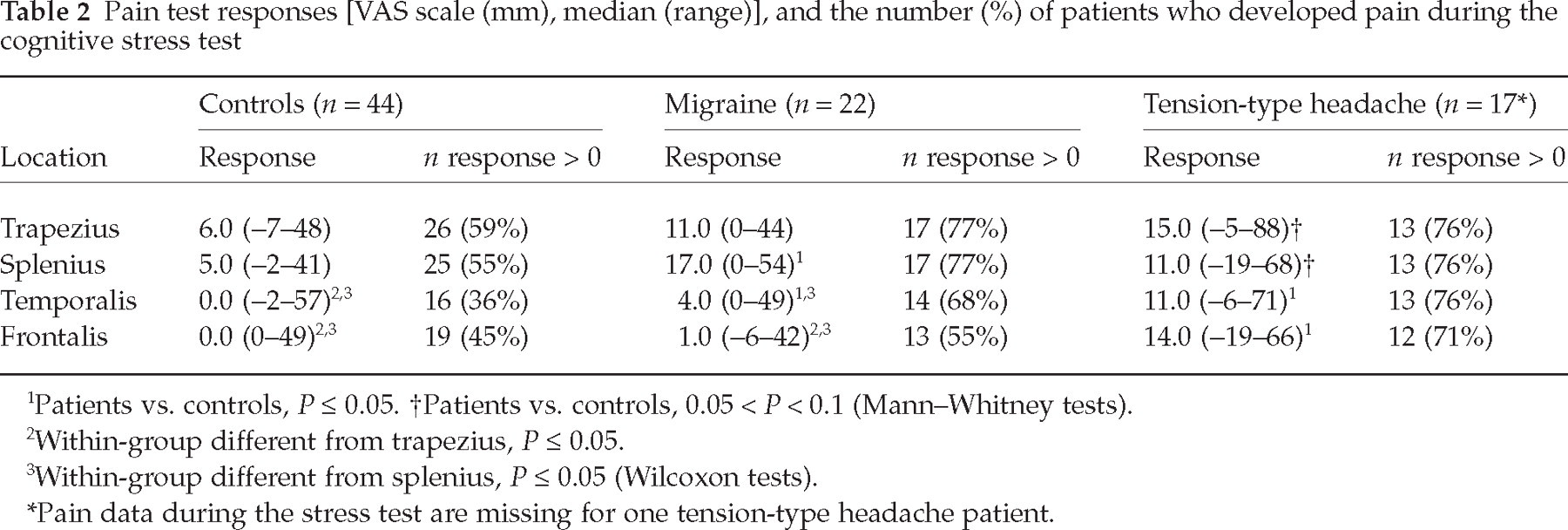
Patients vs. controls, P ≤ 0.05.
Patients vs. controls, 0.05 < P < 0.1 (Mann–Whitney tests).
Within-group different from trapezius, P ≤ 0.05.
Within-group different from splenius, P ≤ 0.05 (Wilcoxon tests).
Pain data during the stress test are missing for one tension-type headache patient.
Compared with controls, migraine patients reported a significantly higher pain response in the splenius and temporalis muscles. TTH patients had significantly higher responses in the temporalis and frontalis, and they showed a trend towards more pain in the trapezius (P = 0.06) and splenius (P = 0.06). Both controls and migraineurs reported significantly lower responses in the frontalis compared with the trapezius and splenius. Controls also experienced less pain in the temporalis compared with the trapezius and splenius (Table 2).
Both migraineurs and TTH patients had generally higher baseline pain in the head compared with controls (Fig. 1). TTH patients also had higher baseline pain in the neck compared with controls, and higher baseline pain than migraineurs in the head.
Pain recovery after cognitive stress
TTH patients needed more time than controls to recover from the pain (Fig. 1). Indeed, many patients reported increased pain instead of recovery throughout the rest period. More TTH patients than controls did not recover from pain after the first 10 min of rest. This difference was observed in all regions (Table 3). After 30 min of rest, most TTH patients still had not recovered to baseline pain in the trapezius, temporalis and frontalis. More migraineurs than controls did not recover after 10 min of rest in the trapezius and temporalis. After 30 min of rest, pain recovery in migraineurs differed significantly from controls in the temporalis, while a similar trend was observed in the trapezius (P = 0.07).
Number (%) of patients who did not return to baseline pain during the early (after 15 min of rest, t = 75 min) and the late (after 35 min of rest, t = 95 min) recovery phases
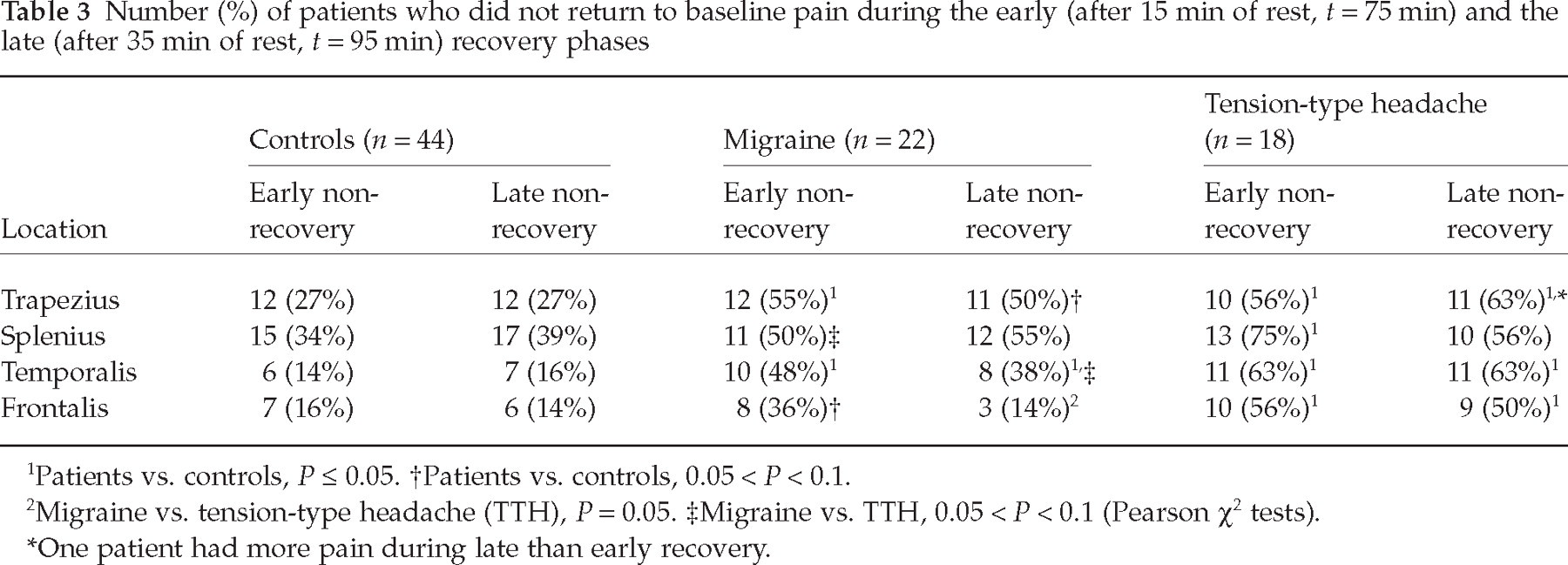
Patients vs. controls, P ≤ 0.05.
Patients vs. controls, 0.05 < P < 0.1.
Migraine vs. tension-type headache (TTH), P = 0.05.
Migraine vs. TTH, 0.05 < P < 0.1 (Pearson χ2 tests).
One patient had more pain during late than early recovery.
EMG responses during cognitive stress compared with baseline EMG
We found a significant difference in temporalis EMG response in migraineurs compared with controls. Apart from that, no significant group differences in EMG responses were found (Fig. 2, Table 4). EMG increased most in the trapezius and frontalis, while mean activity was slightly reduced in the splenius (Table 4). With the exception of the temporalis, all EMG group responses were significantly different from zero for both the TTH and control groups. Migraineurs had EMG responses significantly different from zero in the trapezius, temporalis and frontalis.
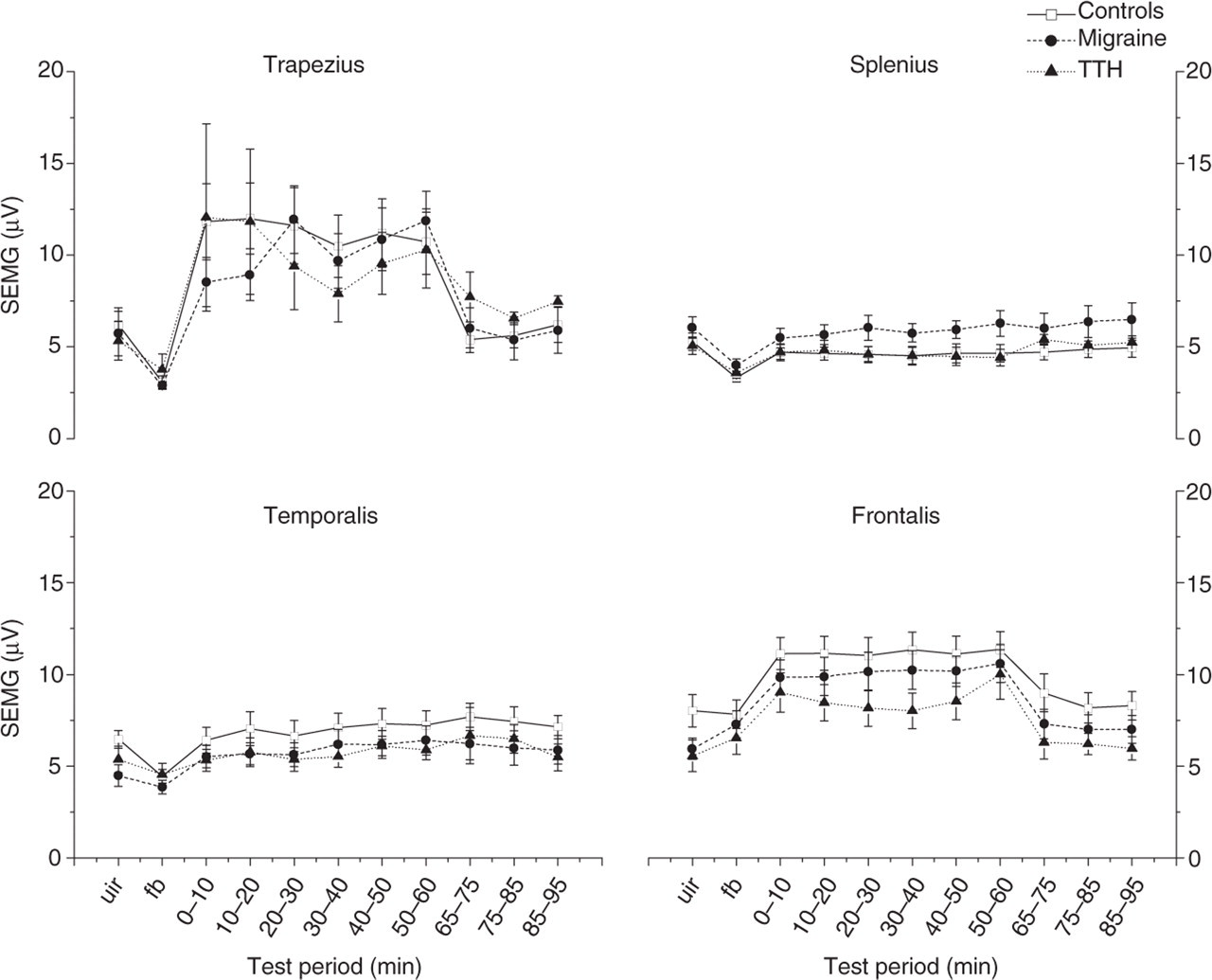
Electromyographic (EMG) activity as a function of time in the four muscle regions for the three subject groups. Values are given in mean (SEM). UIR, Uninstructed rest period (baseline EMG); FB, EMG feedback-aided rest period. 0–65, During the cognitive stress test; 65–95, relaxation period after the test.
Surface electromyographic responses (root mean square differences relative to uninstructed rest period baseline values in µV) during test (0–60 min mean), early recovery (65–75 min) and late recovery (85–95 min)

Values reported are mean (SD), using the average of left- and right-side measurements.
Patients vs. controls, P ≤ 0.05.
Patients vs. controls, 0.05 < P < 0.1.
Migraine vs. TTH, P ≤ 0.05 (Mann–Whitney tests).
EMG recovery after cognitive stress
EMG activity decreased most in the trapezius and frontalis when the stress stimulus was stopped (Fig. 2), and mean EMG activity was generally stable during the 30 min of recovery in all groups. TTH patients had significantly delayed EMG recovery in the trapezius compared with both controls and migraineurs (Table 4).
Pain and EMG change during sustained stress
Pain also increased significantly during the test, from the 10th to the 60th minute, for all groups (Fig. 1), with the exception of the frontalis area in migraineurs, where only a trend was observed (P = 0.055;
Correlations between pain and EMG
Correlation analyses were done in order to establish if EMG responses could predict pain responses. Similar analyses were done for baseline EMG vs. pain responses and baseline pain vs. EMG responses.
EMG responses were not significantly correlated with pain responses either for migraine or TTH. For patients with TTH, it was found that pain responses correlated negatively with baseline EMG in the frontalis (r s =−0.589, P = 0.013). A trend was also found towards a negative correlation between baseline EMG and baseline pain in the trapezius for these patients (r s =−0.434, P = 0.072). In migraineurs, a significant correlation between baseline pain and EMG response in the trapezius was found (r s = 0.445, P = 0.038).
The majority of EMG–pain correlations in patients were accordingly not significant. EMG and pain variables were not significantly correlated in control subjects.
Background variables vs. test responses
General tension during the last 2–3 months was correlated with pain responses in the shoulders (r s = 0.37, P = 0.015) and neck (r s = 0.36, P = 0.016) in controls. Similar correlations were not found in the patient groups. We found no differences in tension responses during the test (data not shown) between the three groups. No correlations between tension and pain responses were found for controls or migraineurs. One significant correlation between neck pain response and tension (r s = 0.544, P = 0.024) was found for patients with TTH. There were no statistical differences in pain responses between episodic and chronic TTH. Consistent differences between migraine subgroups with and without aura were not found. Headache duration did not correlate with pain of EMG responses. Comparing headache intensity before and after the stress test (paired Wilcoxon tests) did not show a significant increase for any of the groups (Table 1).
EMG and pain responses related to the migraine cycle
Migraineurs who reported an attack within the next day had significantly lower EMG response [2.6 (SD 2.1) µV vs. 5.8 (4.9) µV, P = 0.028] in the frontalis region and early EMG recovery response [1.1 (2.9) µV vs. 2.4 (2.1) µV, P = 0.047] in the temporalis region, compared with those who did not have an attack within the next day. Migraine patients who had experienced an attack within 2 days before the stress test had a higher EMG response in the trapezius during the test [7.4 (4.9) µV vs. 0.5 (6.2) µV, P = 0.004]. Pain responses did not differ between attack-related subgroups.
Discussion
Migraineurs had significantly higher neck (splenius) pain responses compared with controls. Neck pain has previously been reported in 75% of migraine patients associated with either the prodrome, headache or postictal phase (22). Because higher neck pain responses occurred with slightly reduced EMG activation, pain can not have been caused by muscular activity, and we suggest that migraineurs may be sensitized to pain in upper cervical as well as trigeminally innervated regions. Either peripheral nociceptor senistization or upper cervical central pain sensitization may be present. The lack of EMG abnormalities in migraineurs are in concordance with old results published by Anderson and Franks (23), but since their study design (e.g. measuring stress responses to serial sevens), as well as the designs of other old EMG studies in migraine, were different from ours the results are probably not comparable.
Neck pain may be the first step towards a migraine attack because 50% of our patients reported an attack within the day following the stress test. This is in concordance with Kaniecki (22). Another important observation is that migraineurs developed more pain (headache) in the temporalis area than healthy subjects, but not in the frontal region. This pattern differs from the most usual frontotemporal location of initial headache during the migraine attack (24), suggesting that stress-induced pain in migraineurs evolves through a mechanism different from the migraine attack.
TTH patients had pain responses of equal magnitude in all locations, while controls (and migraineurs) had less pain in the head compared with the shoulder and neck. Pain increased more during the test in TTH patients compared with controls. Because we kept the stress stimulus level constant, it is possible that this ‘pain potentiation’ reflects temporal summation, a mechanism which has been associated with central sensitization (25).
In addition, TTH patients (and migraine patients to a lesser degree) failed to recover from pain during the rest period. Aftersensations, defined as pain perceived after a stimulus has ended, have been found in experimental conditions which induce central sensitization (26). Prolonged pain in TTH after stress may reflect such long-lasting aftersensations. This interpretation is also compatible with studies that suggest that central sensitization is important in TTH (12, 27) and migraine (28, 29).
To summarize, the widespread pain response (suggesting muscular hyperalgesia), the pain potentiation during sustained stress (accompanied by largely constant EMG levels) and the slow pain recovery support the role of central pain sensitization in TTH (30). The less pronounced, more localized increased muscular pain response and the delay in pain recovery with less potentiation in migraine are more difficult to interpret. Sensitization of muscular nociceptors in the neck and temple or partial central cervical and trigeminal segmental sensitization are possible mechanisms.
Pain responses were associated with increased general tension only in headache-free subjects, indicating that the putative central sensitization state in headache is not necessarily experienced consciously, at least not as general tension, perhaps because mainly subcortical structures are involved. We did not measure perceived stress in the present study. Subjective tension was scored during the stress test, however, and this variable may partly reflect perceived stress as well as muscular factors (31). The correlation between neck pain and tension during the test in TTH patients indicates that muscular pain also may contribute to perceived tension. Because no tension response group differences were found, we conclude that the level of test-related stress were probably similar between groups in the present study.
In TTH patients there was also a lack of EMG recovery in the trapezius after stress. Conflicting results concerning EMG recovery have been published, but older studies have methodological weaknesses (reviewed by Flor and Turk (32)). The response to stress seems accordingly to be stronger, less localized, more prolonged, and also involving the motor system to a larger extent in TTH compared with migraine. Both pain processing, possibly including the muscle-specific part of the homeostatic lamina 1 system (33), and central motor control may accordingly be sensitized in TTH. The so-called emotional motor system, causing involuntary modulation of motor neuron excitability as a response to stressful stimuli or emotional reactions, possibly also modulating pain, is of particular relevance in this respect (11).
The lack of difference in muscular activity between controls and patients with TTH in the present study is in concordance with a study published by Schoenen et al. (8). They found that EMG activity in pericranial muscles during postural changes and mental activity in patients with chronic TTH was not different from healthy controls. Our results are, moreover, partly in concordance with the results published by Bansevicius et al. (14), who used the same cognitive stressors as in the present study. They found a higher EMG response for patients with TTH than in controls in the splenius and trapezius, but it turns out that the discrepancy is apparently related mainly to a lower control group EMG response than in our study. It is also possible that patients recruited from our tertiary headache department were more severely affected than TTH patients recruited mainly from primary care physicians in the previous study.
Several other studies also suggest that muscular activity is normal or only slightly increased, being hardly sufficient to explain pain in TTH (34). Clark et al. (35) recorded temporalis muscle EMG and level of stress and pain every 30 min for 6 days, and found no correlation between pain and EMG. Mørk et al. (36) found no increase in EMG related to experimentally induced muscle pain in episodic TTH. It is nevertheless possible that low-level single motor unit exhaustion can contribute to chronic muscle pain generation (37). If so, one would not expect to find a correlation between cognitive stress-induced surface EMG and induced pain. Anderson and Franks (23) reported a negative association between pain and frontalis EMG in TTH. The presence of pain-induced EMG agonist inhibition (38) may obscure a tentative causal relationship between muscular activity and pain.
Our results suggest that central motor excitability to stress may decrease before a migraine attack and increase after the attack. Most transcranial magnetic stimulation studies of motor cortex in migraine report reduced interictal motor cortex excitability (TMS) (39), but motor excitability studies have apparently not yet been performed through the complete migraine cycle. Cyclic variation has also been found in cognitive evoked responses, i.e. increased P300 (40) and contingent negative variation (41) amplitudes before the attack. It remains to be explained how increased responsitivity to cognitive tasks may be linked to decreased motor excitability.
Pretest stress could possibly influence the baseline state of the central (homeostatic) pain and motor systems. However, baseline tension, fatigue and sleepiness were not significantly different between any of the three groups (data not shown). The patients followed a standardized procedure, including information and familiarization with the laboratory and the procedure before the test started. For these reasons, we believe that pretest stress was similar in the three groups. In addition, pain and EMG response variables are probably less influenced by pretest conditions compared with absolute pain and EMG levels used in several previous studies (8, 9).
The proportion of men was higher among the TTH patients than the other groups. No statistical differences in pain responses between healthy men and women were found, and we accordingly do not believe that the gender distribution invalidates our results. Because men generally report less pain than women (42), it seems likely that even higher levels of pain would have been reported for TTH patients if there had been more women in this group. Indeed, male TTH patients reported significantly less pain than women with TTH in the trapezius and temporalis during the rest period (results not shown).
We have not focused on the difference between pain in the specified head regions and headache as such in our study (a distinction which may be difficult in any case). We had no control group not exposed to stress, and we therefore do not know the spontaneous ‘2-h incidence’ of head or neck pain. As far as we know, there are no data on this in the literature. Since the overall point prevalence of headache in the general population has been found to be as high as 16% (43), one cannot exclude the possibility that some patients may have had random spontaneous headache fluctuations during the test. However, since no patients had to abort the study because of a new headache attack, headache intensity before and after the test was not significantly different in any group, and pain tended to return towards the baseline after the stress period (Fig. 1), it may be assumed that the pain in most subjects was related to the stressor.
In conclusion, migraineurs as well as patients with TTH respond to cognitive stress with more pain than headache-free subjects. Delayed pain recovery was observed mainly in TTH, but also in migraine patients, possibly indicating long-lasting aftersensations and central sensitization. Patients with TTH developed a more generalized pain in the shoulders, neck and head, possibly suggesting a more widespread central sensitization in TTH than in migraine. Migraineurs failed to report significant stress-induced frontal headache, but they did experience muscular hyperalgesia in the neck and temple. No marked differences were found between the three groups in EMG responses during stress, and pain responses could not be explained by EMG activation. Prolonged EMG activity was observed after stress in TTH patients, possibly suggesting that defective inhibition involves both pain and the motor system. Our data suggest, but do not prove, that motor excitability is modulated through the migraine cycle. Further studies of the link between pain and central motor systems in TTH and migraine are needed.
