Abstract

Migrainous vertigo (MV) is a new type of headache that has not yet been recognized by the International Classification of Headache Disorders (ICHD-2) (1, 2). In MV patients, vertigo is an independent migrainous symptom that does not usually correspond to aura and may be dissociated from headache. These episodes of spontaneous or positional vertigo last from a few seconds to several hours, sometimes even for days: attacks that last less than migraine aura are not unusual (20–30%), those lasting hours or days occur in 20–50%, while those that last as long as typical migraine aura (5–60 min) occur in 10–30% of patients. Benign paroxysmal childhood vertigo often precedes by months or years MV symptoms in affected patients. Although there are as yet no internationally approved MV diagnostic criteria, symptoms which may help make a diagnosis (Table 1) and a structured diagnostic interview (3) have recently been proposed (4). Numerous hypotheses have been suggested in an attempt to clarify the pathophysiology of MV, but the underlying mechanisms remain unclear (4).
Diagnostic criteria for MV proposed by Neuhauser H. & Lempert T. (4)

To our knowledge, a lesion-related origin of MV has not previously been suggested. We describe a child with MV, a normal neurological examination, no family history of headache nor personal history of benign paroxysmal childhood vertigo, in whom MRI revealed a right cerebellar arachnoid cyst adjacent to the flocculo-nodular region.
Case report
A 13-year-old, otherwise healthy, Italian boy, born uneventfully as the first child of two nonconsanguineous healthy parents, was referred to us because of headache and vertigo. There was no family history of headache, nor personal history of benign paroxysmal childhood vertigo. Weight at birth was 3.370 kg, head circumference 35 cm, and length 51 cm. Neurological and psychomotor development were reported to be normal until the age of 6 years, when the first clinical episode occurred. Symptoms referred by the parents and child were episodes of spontaneous vertigo (never positional vertigo), a vegetative alteration (pallor and tachycardia), and a slight imbalance and head motion intolerance, all of which were always associated with a severe migraine that started simultaneously. The vertigo attacks occurred exclusively when the boy was awake, their onset was rapid (developing in < 5 min), they lasted from 30 min to several days and occurred at a frequency of 1–2 per month. The migraine attacks associated with vertigo were ‘moderate’ in intensity (they interfered with, but did not prevent daily activities); these headache attacks, which always occurred in the morning or early afternoon, displayed the clinical characteristics of migraine in more than 75% of the attacks (pulsating, unilateral alternant, fronto-temporal location, photophobia and phonofobia, rarely nausea and vomiting).
The patient sometimes also suffered episodes of more severe migraine (all accompanied by photophobia and phonofobia) without vertigo; these episodes were pulsating, strictly unilateral alternant, fronto-temporal and of continuous, severe intensity; pain was invariably exacerbated by routine physical activity, often preventing daily activities from being performed. These migraine without aura (MoA) attacks lasted from 30 min to several days.
From the age of 6 to when the patient came to our attention (age of 13), the semeiology of the attacks remained the same, while the frequency and intensity of the episodes increased slightly (2–3 attacks per month of vertigo associated with migraine episodes, and 1 per week of severe migraine without aura not associated with vertigo). The child had tried various types of analgesic therapy (paracetamol, ibuprofen, acetilsalicilate), without ever achieving any form of control over the attacks. Several EEG recordings performed over the years were reported to be normal.
Upon admission to our paediatric headache centre, the patient underwent a thorough haematological examination, including a complete blood cell count, electrolytes, glucose, erythrocyte sedimentation rate, and liver and kidney function tests, all of which were normal; metabolic investigations were also normal, as was cognitive function. Intellectual functioning was assessed by means of the Wechsler Intelligence Scale for Children-Revised (WISC-R); while the patient's overall score fell within the average range (IQ 112), a significant difference was observed between Verbal and Performance IQ scores (Verbal IQ > Performance IQ of 24 points); the patient scored relatively poorly for his age in the Comprehension subtest (designed to investigate social and practical judgement) and in the Symbol Search subtest (a measure of visual motor velocity and coordination).
The paediatric, neurological and otological (eardrums, tympanograms and pure tone audiograms) examinations and the vestibular tests both in the symptom-free period and during the MV attacks (the child was examined during two MV episodes) were unremarkable; vestibular function tests consisted of positional and positioning tests, the alternate binaural bithermal caloric test and the rotation chair test. The neuroradiological investigation (MRI) revealed a right cerebellar arachnoid cyst adjacent to the flocculo-nodular region (Fig. 1a,b); EEG, performed while the patient was both asleep and awake, was normal.
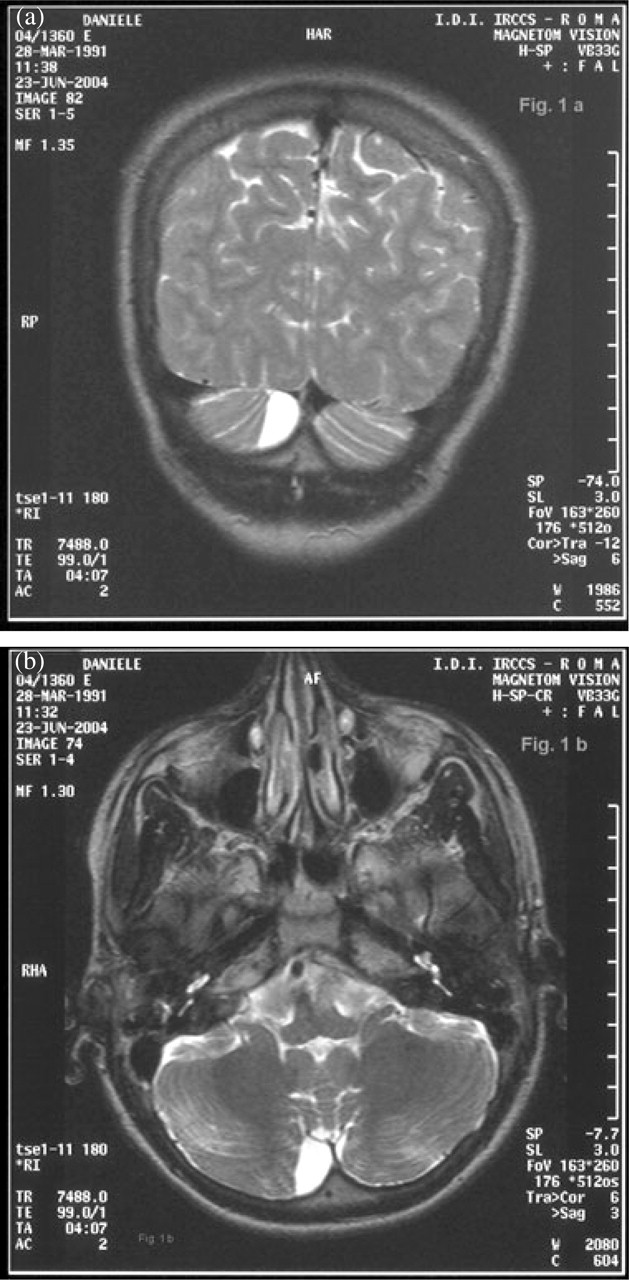
(a) Coronal and (b) axial magnetic resonance imaging; right cerebellar cyst adjacent to flocculo-nodular region.
Following the diagnosis of MV, informed consent was obtained from the boy's parents to start antimigraine prophylactic therapy, which did not, however, modify the clinical picture; in particular, the patients was unresponsive to both flunarizine (5 mg/day) and, three months later, verapamil (80 mg/day) prophylactic therapy. Ten months after having come to our paediatric headache centre, the patient's clinical pictures has substantially remained the same, though there is a mildly worsening trend (slight increase in both types of episode described above).
Discussion
In migrainous vertigo (MV), vertigo is causally related to migraine; this clinical condition may vary considerably, and its connection to migraine may be subtle.
As migraine and vertigo are often associated, a differential diagnosis can, in most cases, be made or causal relationship identified between these two conditions; however, a grey area remains because in some cases both conditions are statistically associated as a result of a correlation with a third factor, or because migraine and vertigo sometimes just happen to coexist in the same patient.
MV is a new type of headache that has not yet been recognized by the International Classification of Headache Disorders (ICHD-2) (1–4). The subdivision of migraine with aura (MA) in the ICHD-2 is different from that in the first edition (ICHD-1). The new diagnostic criteria for MA are defined according to empirical findings that have a higher sensitivity and specificity than the ICHD-1 criteria (5). According to the new ICHD-2 criteria, as an aura symptom of basilar-type migraine (5), vertigo should last between 5 and 60 min and be followed by migrainous headache; furthermore, to fulfil the ICHD-2 criteria, there should be a second aura symptom from the cerebral posterior circulation (double vision, dysarthria, tinnitus, hypoacusia, simultaneous visual symptoms in both temporal and nasal fields of both eyes, ataxia, decreased level of consciousness, simultaneous bilateral paresthesias).
In our child, both the episodes of vertigo, which occurred exclusively when he was awake, and the migraine, whose onset was rapid (<5 min) and coincided with the vertigo, lasted from 30 min to several days and were not associated with any other symptoms ascribable to the cerebral posterior circulation; thus, a diagnosis of basilar-type migraine was ruled out in our case because the ICHD-2 diagnostic criteria were not fulfilled (5). Migraine without aura episodes not associated with vertigo in our patient did, instead, fulfil the ICHD-2 criteria for the diagnosis of MoA (5).
In MV patients, vertigo is an independent migrainous symptom which does not usually correspond to aura and which may be dissociated from headache; head motion intolerance (illusory motion aggravated or provoked by head movements), nausea and imbalance are frequent additional symptoms. Many patients with MV experience attacks of vertigo both with and without headache, while in some cases vertigo and headache never occur together (4). Migraine attacks (migraine without aura) without vertigo, like those referred by our child, have however, also been reported (3, 4). In our patient, the episodes of vertigo were invariably associated with migraine attacks, a finding reflected in the literature, in which the association of these two conditions is reported in 45% of patients (4); it is noteworthy that in our child the episodes of MoA attacks may occur without vertigo. The clinical characteristics and the investigations performed in our child pointed to a diagnosis of MV; indeed, the clinical picture in our patient fulfils the diagnostic criteria for ‘definite migrainous vertigo’ recently proposed by Neuhauser and Lempert (4) (Table 1).
The most common cause of vertigo in teenage children is migraine. One recent paper (6) reported that 47.2% of paediatric patients with vertigo complained of nausea and vomiting, 23% only had nausea, 65.5% had a concomitant headache, 49% had tinnitus, 27% had aural fullness, 25.5% had benign paroxysmal childhood vertigo, 14.5% had hearing loss, 9.1% had loss of consciousness, 9.1% had photophobia and 5.5% had phonophobia. When making a differential diagnosis, the most common causes of recurrent vertigo (such as Meniere's Disease, benign paroxysmal positional vertigo, vertebrobasilar TIA, vascular compression or Schwannoma of the 8th nerve, perilymph fistula, autoimmune inner ear disease, unilateral vestibular loss, central positional vertigo and seizures) must be ruled out; these pathologies are, however, rare in children.
Although numerous hypotheses have been made regarding the pathophysiology of MV, the underlying mechanisms remain unclear: spreading depression with involvement of vestibular central cortical areas, imbalance of neurotransmitters that are be able to modulate the activity of vestibular neurons, a dysfunction of the network that connects the vestibular nuclei, trigeminal system and thalamocortical pathways and genetic defects of ion channels have all been proposed, but none of these potential pathophysiological mechanisms has been documented. The etiopathogenetic mechanisms of MV therefore remain highly speculative.
All the afore-mentioned hypotheses suggest a primary etiopathogenic origin of MV, ignoring the possibility of a lesion-related origin of this condition as a result of structural interference, at either the cortical or subcortical level, on the central vestibular network processes. It is, indeed, noteworthy that the overall findings in acute MV report a central vestibular dysfunction in 53% of the patients observed (7).
It has recently been suggested that migraine may be an indicator of subclinical brain lesions (8), particularly in certain brain areas (e.g. posterior fossa cranica), with clinical evidence (9) in migraine patients of a persistent dysfunction in the brainstem and certain cerebellar structures (e.g. ventrolateral cortex and caudal fastigial nucleus). Reduced cerebellar choline values in migraine patients, as determined by MRI-spectroscopy, have also been reported (10).
As subtle subclinical cerebellar signs in patients with migraine with or without aura have recently been reported (8–13), we stress the importance in our patient of the neuroradiological findings (i.e. cerebellar arachnoid cyst), and believe that both the vertigo and migraine in our MV-affected child represent symptoms that are ascribable to cerebellar involvement. Another intriguing finding in our child was the discrepancy between Verbal and Performance IQ scores at the WISC-R (Verbal IQ > Performance IQ of 24 points); we believe that this finding may be due to interference in the fronto-ponto-cerebello-thalamo-cortical pathway, which is involved in visual motor velocity and coordination. The clear clinical expression of the arachnoid cyst in this child may, indeed, be related to a ‘crowded’ posterior fossa (14) and/or transient dysfunction in some of the brainstem and cerebellar structures (cerebellar cortex, flocculo-nodular region and vestibular nucleus) involved in central vestibular function control.
Should the clinical picture in our patient worsen in the future, a neurosurgical operation to remove the arachnoid cyst is one therapeutic hypothesis that would be considered.
Thus, we believe that the association of vertigo and migraine warrants a thorough neuroradiological investigation (MRI is to be preferred to study the posterior fossa cranica), especially if antimigraine therapy proves to be ineffective, as it did in our child.
Conclusion
Patients presenting with MV often pose a diagnostic challenge, particularly if there are no other associated neurologic symptoms or poor, aspecific signs. We believe that migraine and vertigo in MV may, in some cases, both be symptoms of brainstem or cerebellar involvement as a result of a stroke or other more benign lesions, such as atrophy or arachnoid cysts. Moreover, in selected patients (especially if neither a family history of headache nor a previous personal history of benign paroxysmal childhood vertigo are reported), a brain MRI should be performed, particularly in children with a poor or no response to antimigraine therapy. Lastly, we do not think that the inclusion of MV in any revised IHS classification in the future will be an easy task: is MV a primary or symptomatic headache? … Or both?
