Abstract
A new experimental human model of myofascial pain using intramuscular infusion of a combination of bradykinin, serotonin (5-hydroxytryptamine), histamine, and prostaglandin E2 was applied to patients with episodic tension-type headache (ETTH) in order to examine pain perception. Fifteen patients with ETTH and 15 healthy controls completed the randomized, balanced, double-blinded, placebo-controlled study. Pain intensity, punctate hyperalgesia and allodynia, and pain quality were recorded. The combination induced a moderate and prolonged pain in both patients (median 51 min) (P = 0.001) and controls (median 22 min) (P = 0.001). Patients reported more pain than controls both after the combination (P = 0.045) and after placebo (P < 0.001). The McGill pain score [PRI(R)] was significantly higher in patients (P = 0.002) and in controls (P = 0.001), whereas pain quality and hyperalgesia were similar after the combination compared with placebo in the two groups. Due to side-effects nine subjects did not complete the study. The increased pain response, but similar qualitative pain perception, in ETTH patients may be explained by sensitization of peripheral nociceptors even though central mechanisms may also be involved.
Introduction
Myofascial factors may play a major role in the pathophysiology of tension-type headache (TTH) (1, 2). Thus, the most prominent abnormal finding in patients with TTH is increased pericranial myofascial tenderness by palpation (3, 4), and this tenderness is positively correlated to intensity and frequency of headache (5). Only few experimental human models have been developed to examine myofascial mechanisms in TTH (6–11). Improved models of myofascial pain are needed to increase the present knowledge of its aetiology, evolution and treatment. TTH is extremely prevalent since 40–74% of the population has experienced this type of headache in the last year (12, 13). In addition to high personal costs, this disorder also results in substantial socio-economic costs as TTH mostly affects people of working age (14, 15).
Tenderness and pain may be due to release of endogenous substances in myofascial tissues sensitizing and exciting peripheral nociceptors (16–20). A characterization of the neurobiological basis of human muscle pain may therefore be of clinical importance in TTH.
Recently, we demonstrated that infusion of a combination of bradykinin (Bk), serotonin [5-hydroxytryptamine (5-HT)], histamine (His), and prostaglandin E2 (PGE2) into the trapezius muscle of healthy subjects produced a prolonged, local and moderate pain and tenderness while single substances or combinations of two or three substances induce only mild and momentary pain and tenderness (21). The trapezius muscle was chosen because this muscle is one of the most tender muscles in TTH (4, 5), and the localization and size of the muscle also make it suitable for examination. The aim of the present study was to apply this experimental human model to patients with episodic tension-type headache (ETTH) in order to examine the pain intensity and to elucidate possible pathophysiological mechanisms in TTH.
Material and methods
Subjects
Nineteen patients with a diagnosis of frequent ETTH and 20 healthy subjects were included (Table 1). The patients were recruited from the out-patient Headache Clinic at Glostrup Hospital and the headache was diagnosed according to the criteria of the Inter-national Headache Society (22) and verified by a 4-week headache diary prior to inclusion (23). Inclusion criteria for patients were 8–15 days of TTH/4 weeks, and a total tenderness score (TTS) (24) of at least 8 (maximum possible score of 48) and a local tenderness score (LTS) of at least 2 (on a scale from 0 to 3) on the non-dominant trapezius muscle on the day of inclusion. Healthy subjects were not allowed to have more than 1 day of TTH per 4 weeks. All subjects underwent a full physical and neurological examination including a screening for depression [Major (ICD-10) Depression Questionnaire (25)] prior to participation in the study. Exclusion criteria for participants were: history of migraine or other primary headaches; allergy; serious somatic or psychiatric disorders including depression; use of any kind of daily medication (except for oral contraceptives); excessive use of analgesics (use of > 50 g of aspirins per month or equivalent); pregnancy or breast feeding; abnormal physical or neurological examination. Examinations were performed on headache-free days and subjects were not allowed to take analgesics or any other medication except for contraceptives 24 h prior to examination.
Demographic data

Data observed on the day of inclusion to study.
Data observed on the day of examination.
Median values are given with quartiles within parentheses.
All participants gave written informed consent to participate in the study, which was conducted in accordance with the Declaration of Helsinki and approved by the local Ethics Committee.
Preparations
Combination of endogenous substances
The combination has previously been described in detail (21). Briefly, 1 ml of the combination contained 92 nmol of Bk (Sigma, Vallensbaek Strand, Denmark), 156 nmol of 5-HT (All Bright, Vallensbaek Strand, Denmark), 130 nmol of His (Unikem, Copenhagen, Denmark), and 1.95 nmol of PGE2 (Bie & Berntsen, Roedovre, Denmark). Initially, Bk, 5-HT, and His were diluted with isotonic saline (9 mg/ml), whereas PGE2 was dissolved in ethanol (0.043%) and then diluted in isotonic saline. Bk and PGE2 were stored at − 20 °C, and 5-HT and His were stored at 5 °C. Immediately before infusion Bk and PGE2 were thawed and mixed with 5-HT and His.
Placebo
Isotonic saline containing ethanol in the same concentration as in the combination (0.007%) was used as placebo.
Experimental design
In a balanced, double-blinded, and placebo-controlled trial subjects were randomized to receive intramuscular infusions of 1 ml of the combination and 1 ml of the placebo. Each infusion was given over 10 min, and the two sessions were separated by at least 2 weeks. Infusions were given in a standardized anatomical point at the centre of the descending part of the non-dominant trapezius muscle midway between the spinal process of the seventh cervical vertebra and the acromion. The infusions were carried out by a syringe pump (Graseby 3400) using a 5-ml plastic syringe (Becton Dickenson, Broendby, Denmark). A tube (Medex; Simonsen & Weel, Copenhagen, Denmark) was connected from the syringe to the disposable EMG-needle (27 G and 25 mm; Judex, Aalborg, Denmark), that was connected to an electromyograph (Counterpoint; Dantec, Copenhagen, Denmark) to ensure intramuscular infusion.
Pain assessment
The recordings were performed by the same observer (a trained technician, H.A.), who was blinded with respect to substances and with respect to patients and controls.
Pain
Pain intensity was assessed on a 100-mm visual analogue scale (VAS) where 0 corresponded to no pain and 100 mm to the worst imaginable pain (26). The pain intensity was recorded at baseline (0 min) and every minute during infusion and until 5 min after stopping infusion, then every 5 min until 30 min, and at 45, 60, 90, 120 min, and 24 h after start of infusion.
Cutaneous hyperalgesia
Punctate hyperalgesia was assessed by gently pricking the skin at approximately 5-mm intervals with a needle (Microlance, 18GI½; BD Medical Systems, Drogheda, Ireland), and defined as a change of sensation into more pain. Allodynia was assessed by gently stoking the skin with a cotton swap, and was defined as a change of perception of stimuli into pain. Boarders of hyperalgesia were determined by slowly moving along 8 radians from well outside the area of hyperalgesia towards the standard point. These borders were marked and the eight points were mapped on transparent plastic and connected, whereafter size of the area was calculated (27). If changes were not reported in all eight directions the area was defined as 0 in the statistical calculations. Recordings were made at 0, 15, 30, 60, 120 min, and 24 h after start of infusion.
Pain quality
Pain quality was scored on a Danish version of the McGill Pain Questionnaire (28). Pain rating index [PRI(R)] based on numerical values assigned to each word descriptor and calculated as outlined by Melzack (29), number of words chosen (NWC), and most frequently used words were determined (29). Pain index was scored on a 5-point scale where 1 corresponded to mild, 2 to discomfort, 3 to distressing, 4 to horrible, and 5 to excruciating (29). Pain quality was determined at 11 (for the period 0–11 min), 60, and 120 min, and 24 h after start of infusion.
Electromyography
Processing of the electrical activity was performed by the Counterpoint programs. The power spectrum of each of the 1-s measurement sessions was calculated for the frequency range 0–1 kHz. The signal was filtered by a 1-kHz filter and the sampling frequency was 2.56 kHz. By means of the disposable EMG needle used for infusion the root mean square (RMS) and mean frequency were measured at baseline and 11 min after start of infusion.
Other parameters
The localization of local pain, projected pain (pain in direct relation to pain at the side of infusion, i.e. local pain) and referred pain (pain at a distant side separated from local/projected pain) was outlined by participants on an anatomical map during the observation period (120 min). Development of wheal and flare were also registered during the observation period.
The presence of pain and tenderness in shoulder or neck region, referred pain, headache and intake of medication were requested in a 24-h diary.
Data analysis and statistics
The standard deviation of pain and tenderness was estimated to 50% and the clinically relevant difference in these parameters was estimated to 50%, power was set to be 80% and the level of significance to 5%. Number of subjects to participate was calculated at 15 in each group (30).
Results are presented as medians and quartiles. Primary efficacy parameter was pain presented by area under the pain curve [area under the pain (VAS)–time (60 min) curve] (AUCpain) (31) after the combination compared with placebo, and pain after the combination in patients compared with controls. The pain (AUCpain), maximum pain score (VAS peak) (Maxpain), duration of pain (painoffset − painonset) (Durpain), hyperalgesia [area under the hyperalgesia (area of hyperalgesia)–time (60 min) curve] (AUChyperalgesia), maximum area of hyperalgesia (Maxhyperalgesia), and duration of hyperalgesia (hyperalgesiaoffset − hyperalgesiaonsett), and pain quality [PRI(R), NWC, and pain index] for the combination were compared with placebo by means of Wilcoxon signed rank sum test (paired data). The Wilcoxon signed rank sum test was also used to compare EMG measurements (RMS and mean frequency) before and after infusions in patients and in healthy controls. The Mann–Whitney test (unpaired data) was used to compare data between patients and controls. The McNemar test was used to compare the frequency of development of pain, hyperalgesia, and headache after infusion of the combination to placebo within the two groups, while Fischer's exact test was used to compare these parameters between patients and controls. Five percent was accepted as level of significance. All data were analysed with SPSS software, version 10.0 (SPSS Inc., Chicago, IL, USA).
Results
Fifteen patients and 15 healthy controls completed the study; the demographic data are shown in Table 1. All patients completed the 24-h diary and 10 patients and 10 controls returned for assessment of pain parameters 24 h after the infusion.
Pain
The AUCpain was significantly increased after infusion of the combination compared with placebo both in the patient group (P = 0.001) and in the control group (P = 0.001). In patients the AUCpain was significantly higher than in controls after the combination (P = 0.045) and after the placebo (P < 0.001) (Table 2, Fig. 1). However, when the pain induced by the placebo was subtracted from the pain induced by the combination (AUCcombination −AUCplacebo) no significant difference in the reported pain could be detected between patients and controls (P = 0.367). The Maxpain and Durpain were significantly increased after infusion of the combination compared with placebo both in patients (P = 0.001 and 0.041, respectively) and in controls (P = 0.001, in both cases). After the combination the Durpain was significantly higher in patients compared with controls (P = 0.041), while there was no significant difference in Maxpain in patients compared with controls after the combination (P = 0.161). After placebo the Maxpain and Durpain were significantly higher in patients compared with controls (P < 0.001, in both cases) (Table 2, Fig. 1). Pain was present in all subjects (100%) after the combination and was reported within 1–4 min and reached its maximum, Maxpain, within 20 min (median 10 min), in both groups. After the placebo, all patients (100%) and five controls (33%) developed pain within 1–6 min and reached the Maxpain within 13 min (median 8 min in patients and 2 min in controls) in both groups. The frequency of reported pain was not significantly different after the combination compared with placebo in either group or after the combination between groups. No pain was present after 24 h in either group.

Median pain intensity (a) and area under the pain [visual analogue scale (VAS), mm)–time (60 min) curve (AUCpain) (b) after infusion of the combination of endogenous substances and placebo into the non-dominant trapezius muscle of patients with episodic tension-type headache (ptt) (n = 15) and healthy controls (contr) (n = 15). ∗P ≤ 0.05; ∗∗P ≤ 0.01; ∗∗∗P ≤ 0.001.
Pain after infusion of the combination of endogenous substances and placebo into the trapezius muscle of patients with episodic tension-type headache (n = 15) and healthy controls (n = 15)

Median values are given with quartiles within parentheses.
AUCpain, Area under the pain [visual analogue scale (VAS)]–time (60 min) curve; Maxpain, maximum pain score (VAS-peak); Durpain, duration of pain.
P ≤ 0.05;
P ≤ 0.01;
P ≤ 0.001.
Cutaneous hyperalgesia
No significant differences in the development of punctate hyperalgesia (AUChyperalgesia, Maxhyperalgesia, or Durhyperalgesia) were detectable when comparing the combination with the placebo or comparing patients with controls (P > 0.100). Ten patients (67%) and eight controls (53%) developed hyperalgesia to pin prick after the combination, and seven (47%) patients and four (27%) controls after placebo. There were no significant differences in frequency of hyperalgesia within or between the two groups. Hyperalgesia developed within 15–60 min (median 27 min) in both groups. No allodynia was found.
Pain quality
Pain was described by words as listed in Table 3. In the period from 0 to 11 min, the total PRI(R), NWC, and the pain index were higher for the combination than for placebo both in the patient group (P = 0.002, 0.002 and 0.001, respectively) and in the control group (P = 0.001, in all cases). Likewise, in PRI(R) subclasses (sensory, affective, miscellaneous, and evaluative) significant differences were found after the combination compared with placebo in patients (P-values 0.019–0.042) and in controls with regard to sensitive, miscellaneous, and evaluative classes (P-values 0.001–0.002), whereas in the affective subclass no significant difference could be detected (P = 0.010). No significant differences in total PRI(R), subclasses of PRI(R), NWC, or pain index were detected after the combination between the two groups. After the placebo significantly higher values of total PRI(R), PRI(R) of the sensitive subclass, NWC, and pain index were found in patients compared with controls after placebo (P = 0.004, 0.002, 0.004 and < 0.001, respectively); no significant differences were found in the remaining subclasses of PRI(R) (Table 3). At 60 min the PRI(R) and NWC were higher in the patient group after infusion of the combination compared with placebo (P = 0.039, and 0.042, respectively) while no differences in pain index were detectable. No further differences were identified within or between the two groups at 60 and 120 min.
Pain quality after infusion of the combination of endogenous substances and placebo into the trapezius muscle of patients with episodic tension-type headache (n = 15) and healthy controls (n = 15)
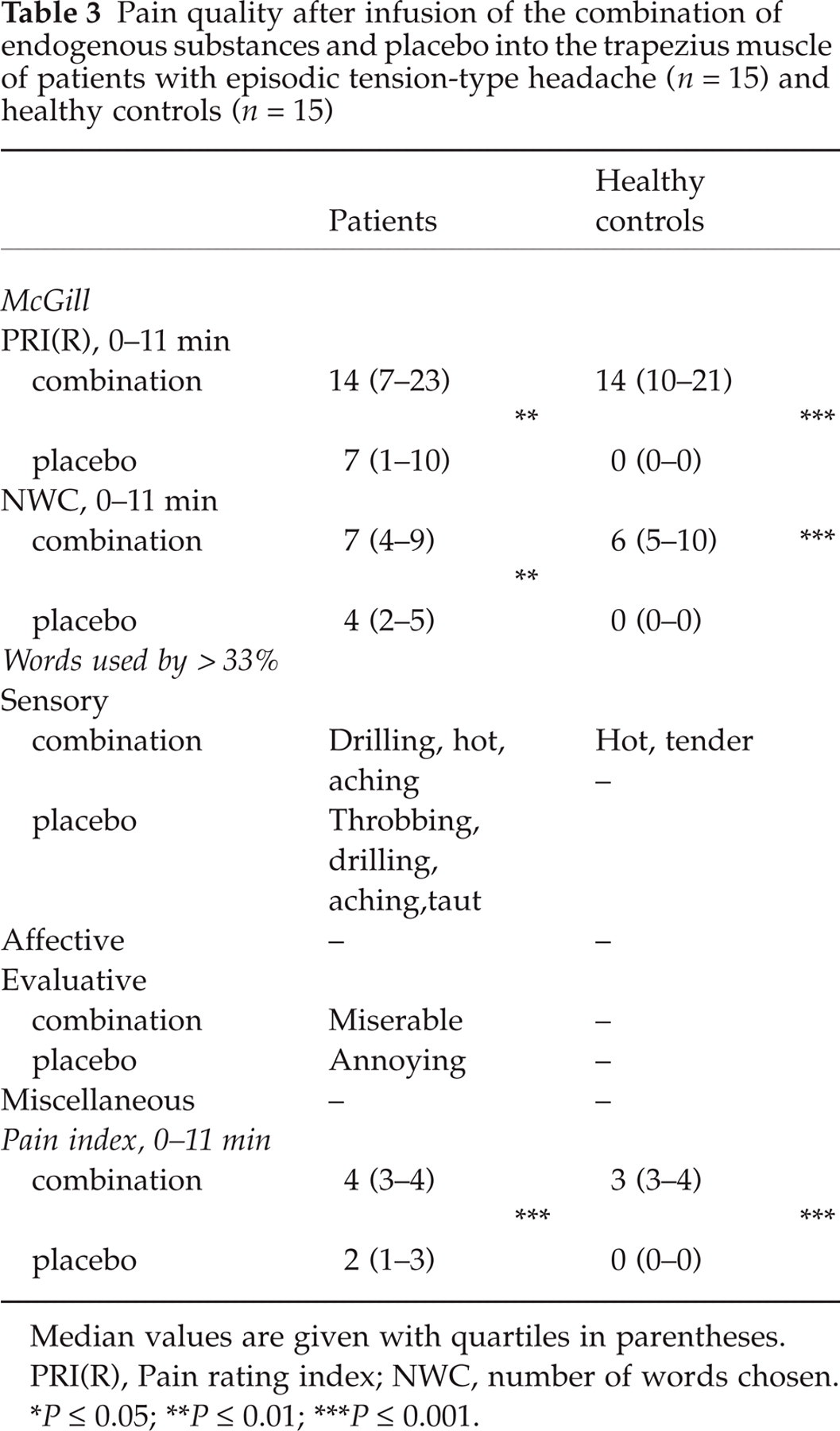
Median values are given with quartiles in parentheses.
PRI(R), Pain rating index; NWC, number of words chosen.
P ≤ 0.05;
P ≤ 0.01;
P ≤ 0.001.
Electromyography
In patients, there were no significant changes in RMS and mean frequency after infusion of the combination (P > 0.750) or placebo (P > 0.800). In controls, the RMS was significantly increased (P = 0.031) after infusion of the combination, but not after placebo (P = 0.397). No changes in mean frequency were detected, although a trend to decreased mean frequency (P = 0.064) after the combination compared with baseline was found in controls.
Other parameters
0–2-h observation
All participants developed pain after the combination, and significantly more patients developed pain after the placebo compared with controls (P ≤ 0.001) (Table 4).
Percentage distribution of reports of pain, referred pain and tension-type headache after infusion of the combination of endogenous substances and placebo into patients with episodic tension-type headache (n = 15) and healthy controls (n = 15)

TTH, Tension type headache; n.s., not significant.
P ≤ 0.05.
Significantly more patients developed pain projection after the combination compared with the placebo (P = 0.016). No other significant differences were detected in controls or between the two groups (Table 4). The projection was more often proximal in patients compared with controls (Fig. 2). After infusion of the combination 10 patients reported pain projection ipsilaterally: in proximal direction to the neck (n = 9) and retro-auricular region (n = 7) and in distal direction to the shoulder (n = 5). Six controls reported ipsilateral pain projection: in proximal direction to the neck (n = 6) and the retro-auricular region (n = 2) and in distal direction to the shoulder (n = 5). After infusion of the placebo, three patients reported pain projection in proximal direction to the retroauricular region, and two distal to the shoulder.

Distribution of pain after infusion of the combination of endogenous substances in patients with episodic tension-type headache (a) and in healthy controls (b).
No differences in frequency of referred pain were detected within or between the two groups. In two patients referred pain was experienced ipsilaterally after the combination; one subject to the neck and retroauricular region, and in another to the frontal region, whereas three controls reported referred pain ipsilaterally to the neck region. After the placebo, three patients reported referred pain to the ipsilateral retroaucicular region (n = 2) and to the frontal region bilaterally (n = 1), while none in the control group reported projection of the pain or referred pain (Table 4, Fig. 2).
2–24-h diary
After the combination there was no increase in reports of TTH in patients compared with controls (P = 0.165), whereas more patients reported TTH after the placebo compared with controls (P = 0.003). No further significant differences in frequency of TTH or pain and tenderness after infusions were detected within or between the groups (Table 4). Headache in all cases fulfilled the criteria of TTH and was present 3–24 h after the infusions and with a median intensity of 16 mm (6–50 mm) on the VAS scale. Pain and/or tenderness were reported 2–24 h after infusions.
Adverse events
Seven subjects (three patients and four healthy controls) were excluded during infusion of the combination and one subject (one healthy control) during infusion of the placebo due to vaso-vagal symptoms appearing between 3 and 6 min after start of the infusions. During infusion of the combination these symptoms appeared in association with a median pain score of 78 mm on VAS (10–100 mm), but during the placebo infusion no pain was reported. Four subjects developed symptoms during their first infusion, and four subjects during their second infusion. Symptoms were sense of heat, dizziness, and nausea, which disappeared spontaneously in all cases within a few minutes after cessation of infusions. One subject was excluded because of universal erythema 14 days after infusion of the placebo and 7 days after initiation of antibiotic treatment of a nail inflammation.
In all subjects redness, but no weal, was observed locally around the needle in direct relation to infusion of both the combination and the placebo, but disappeared completely in all cases within 1 h.
Discussion
In the present study the combination of Bk, 5-HT, His, and PGE2 induced a prolonged and moderate pain in both ETTH patients (median 51 min) and controls (median 22 min). The induced pain after this combination was significantly more pronounced than the pain induced after placebo in both groups. The results thereby confirm previous results from a pilot study in healthy controls (21). To our knowledge, only one human model of myofascial pain using naturally occurring endogenous substances has previously been applied to patients (20). In this study injections of 2000 nmol of 5-HT induced mild and brief pain in healthy subjects, but in contrast to the present study injections of 0.2–2000 nmol of 5-HT into the painful masseter muscles of fibromyalgia patients did not produce more pain compared with either baseline or saline. Our present and previous (21) findings indicate that a combination of several substances, i.e. an ‘inflammatory soup’ (32), is needed to induce a clinically relevant and prolonged pain in both myofascial pain patients and healthy controls.
The ETTH patients reported more pain than controls not only after the combination but also after the placebo. The pronounced pain sensitivity in patients may reflect increased excitability or a prior sensitization of peripheral myofascial afferents, possibly caused by local release of endogenous substances in tender muscles (18). Impaired supraspinal descending inhibition (33) or increased excitability at the spinal/trigeminal level because of prolonged activation of primary sensory afferents in the tender muscles may, however, also contribute to the increased sensitivity in patients (34). This is supported by studies in animals showing that decreased supraspinal descending inhibition (35) and central sensitization (36) may contribute to increased pain perception.
However, punctate hyperalgesia was found only in some individuals and no statistical differences were found after the combination or between the groups. Likewise, no allodynia was found. These results indicate that the hypersensitivity to the present infusions in patients may be a peripheral phenomenon (37–41). Furthermore, unchanged mechanical pain thresholds on the finger and in the temporal region in either group after infusion of the combination (42) indicate that peripheral mechanisms may be responsible for the increased pain response in patients.
When we subtracted the pain induced by the placebo procedure from the pain induced by the combination no differences between the two groups could be detected. Thus, patients presented a stronger reaction to both the endogenous substances (chemical stimuli) and to the non-specific stimuli (the placebo and mechanical stimuli) compared with healthy controls. In agreement with this, we found lower pressure pain thresholds in the patients than in controls at baseline, as reported elsewhere (42). In line with the present results, infusion of hypertonic saline has been reported to produce more pain in patients with chronic myofascial pain (fibromyalgia and whiplash) than in healthy controls (43, 44), although there was no placebo control in these studies.
Both patients and controls used ‘stronger’[PRI(R) (total and subclasses) and pain index] and more words after the combination compared with placebo. The values of the PRI(R) (in total and in subclasses), pain index, and number of words used were nevertheless similar after the combination in the two groups. Moreover, the PRI(R) was similar to the PRI(R) in previous acute experimental pain studies (45), and to menstrual pain (29), while it was less pronounced compared with the chronic low back pain and masseter pain or cancer pain (29, 46, 47). The present data may therefore indicate that ETTH patients and healthy controls have a qualitatively similar experience of pain, in contrast to the quantitative experience of pain, which was more pronounced in patients.
A tendency to more proximal distribution of pain in patients compared with healthy subjects was observed. This observation is in agreement with observations in chronic myofascial pain patients (48, 49) and experimental controlled pain studies of chronic pain patients (43, 44). Proximal distribution of pain possibly reflects central sensitization (50, 51) but the number of patients in the present study was too low to make firm conclusions. However, central sensitization may play a role in these patients since their mechanical pain thresholds were decreased compared with thresholds in controls at baseline (42), and the finding may therefore not be a specific reaction to the combination. Referred pain was seen in 13–20% of patients both after the combination and after placebo, as well as in controls after the combination, whereas no referral of pain was seen in controls after placebo. The frequency of referral of pain is similar to reports of referred pain in healthy controls after stimulation with endogenous substances (19), but less pronounced compared with reports of evoked referred pain in other models of myofascial pain (52–54). Differences in quality and intensity of evoked pain responses in the different models might explain these variable results (55, 56). In TTH prolonged myofascial pain and tenderness are supposed to be responsible for the pain. After 10 min of intramuscular infusion of nociceptive substances referred pain and post-experimental headache were similar in patients and controls. Since for ethical reasons more prolonged infusions are not possible, the present model is probably not an experimental model of headache but rather an experimental model of myofascial pain.
In the present study EMG parameters were not affected in the patients after infusion of the combination, and it is therefore unlikely that concomitant electrical muscular activity was responsible for their increased pain response. In contrast, the increase in amplitude in healthy controls may indicate a normal muscular reaction which may be abolished or suppressed in ETTH patients, possibly as a long-term adaptation to myofascial pain (57).
The present experimental human model of myofascial pain using a combination of endogenous substances induces a prolonged (median 51 min in patients and 22 min in healthy controls) and moderate pain in both patients with ETTH and healthy subjects, and thereby represent a very valuable experimental human pain model which can be used for future pharmacological testing. Patients with ETTH showed a higher sensibility to applied stimuli, but pain quality was similar compared with healthy controls. Moreover, referred pain and punctate hyperalgesia and allodynia were no more frequently found in patients compared with controls. These results may indicate a peripheral increased excitability or sensitization in ETTH patients. However, involvement of central mechanisms may also be present, since clinical signs and pain thresholds were lower in ETTH compared with healthy controls.
Footnotes
Acknowledgements
We thank technician Hanne Andresen for helpful technical assistance during the data collection, and pharmacist Trine Schnor (The Central Pharmacy, Copenhagen Community Hospital Services) for preparation and testing of substances.
