Abstract
Systemic administration of nitroglycerin (NTG), a nitric oxide (NO) donor, in migraineurs triggers after several hours an attack of which the precise mechanisms are unknown. We found previously in rats that nitroglycerin (10 mg/kg s.c.) is able to increase significantly after 4 h the number of neuronal nitric oxide synthase (nNOS)-immunoreactive neurones in the cervical part of trigeminal nucleus caudalis. In the present experiments, we demonstrate that the 5-HT1B/D agonist sumatriptan (0.6 mg/kg s.c.) does not alter this phenomenon when given before NTG. By contrast, pretreatment with lysine acetylsalicylate (50 mg/kg i.m.) attenuates the NTG-induced nNOS expression in the superficial laminae of trigeminal nucleus caudalis. These findings suggest that effect of NTG on nNOS at a high dosage may involve the cycloxygenase pathway and that activation of the peripheral 5-HT1B/D receptors is not able to modify this effect. These data could help to better understand the role of NO in the pathogenesis of headaches and the action of antimigraine drugs.
Keywords
Introduction
Systemic administration of nitroglycerin (NTG), a nitric oxide (NO) donor, in migraineurs can trigger attacks without aura after a delay of several hours (1, 2). Like spontaneous migraine attacks, the NTG-induced attacks can be interrupted by the 5-HT1B/D agonist sumatriptan (3), but their precise neurobiological mechanisms are still unknown. NTG was recently reported to induce a also delayed tension-type headache in patients suffering from chronic tension-type headache (4).
There is no doubt that NO and neuronal nitric oxide synthase (nNOS) play an important role in the processing of nociceptive information, and especially in sensitization of spinal second order nociceptors (5, 6). nNOS is present in the trigeminal system which suggests that it is similarly involved in the processing of cephalic pain (7). NOS inhibitors reduce c-fos activation by nociceptive stimuli in rat dorsal horn (8, 9) and can antagonize neurogenic activation of the trigeminovascular sytem (10). According to preliminary studies, they are also able to reduce pain in migraine attacks (11) and chronic tension-type headache (12).
NTG increases c-fos (13) and nNOS (14) expression in the spinal portion of trigeminal nucleus caudalis (TNC) in rats. In the latter study we hypothesized that the increased nNOS expression in second order trigeminal nociceptors might initiate a self-amplifying process of NO production at the basis of central sensitization. The increased nNOS expression could be due to a direct effect of NO on TNC neurones, but, because of its regional selectivity, it is more likely secondary to activation of primary trigeminal afferents. 5-HT1D receptors are located presynaptically on the distal and proximal portions of these afferents (15, 16) and their activation is thought to contribute to the antimigraine effect of 5-HT1B/D agonists, also called triptans (17). Non-steroidal anti-inflammatory drugs (NSAIDs), such as acetylsalicylic acid (Aspirin), are also effective in the treatment of acute migraine headache (18) and tension-type headache (19). This effect could to be due to their inhibitory action on cyclo-oxygenase 2 (COX-2) and prostaglandins in the spinal trigeminal complex (20), but they could also inhibit NOS activation by reducing the induction of transcription factor NF kappa β (21). Concordantly, it was shown that pretreatment with indomethacin reduces the NTG induced c-fos activation in the caudal trigeminal nucleus (22).
The aim of the present study was to determine if NTG-induced nNOS expression in rat trigeminal nucleus caudalis, as shown in our previous study (14), is altered by pretreatment with the 5-HT1B/D agonist sumatriptan or with lysine-acetylsalicylate (lys-ASA), a NSAID, both of which are effective in the acute treatment of migraine attacks (23).
Methods
The procedures of this study followed the guidelines of the International Association for the study of Pain and the European Communities Council (86/609/ECC). They were approved by the Ethics Comittee of the Faculty of Medicine, University of Liège. Seventy-eight male Wistar rats (250–350 g) were used. For immunohistochemistry, 48 animals were divided into three groups of 16. In the first group the rats did not receive any pretreatment. In the second group the rats received a subcutaneous (s.c.) injection of sumatriptan (Imitrex, Glaxo Smith Kline, Research Triangle Park, NC, USA) at a dose of 0.6 mg/kg. The animals of the third group were injected intramuscularly with lys-ASA (Aspegic, Sanofi-Synthelabo, Paris, France) at a dose of 50 mg/kg. Sumatriptan was diluted in physiological saline, lys-ASA in its commercially available solvent (distilled water). Ten minutes later in each group 8 animals received a s.c. injection of NTG (prepared from Nitrolingual pumpspray, Pohl-Boskamp GmbH, Hohenlockstedt, Germany) at a dose of 10 mg/kg and 8 animals received a s.c. injection of the vehicle (gift from Pohl-Boskamp).
Four hours after the NTG or placebo injections the rats were deeply anaesthetized by pentobarbital (Nembutal 80 mg/kg) and transcardially perfused with 100 ml physiological saline followed by 500 ml 4% paraformaldehyde in phosphate-buffered saline (PBS). The portions of cervical spinal cords comprised between −5 and −11 mm from the obex were removed and postfixed overnight for immunohistochemistry.
For Western blotting 30 rats were divided in three groups of 10 and received pretreatment and NTG or placebo injections as above. Four hours later, they were deeply anaesthetized and transcardially perfused with 200 ml of physiological saline. Cervical spinal cords between −5 and −11 mm from the obex were removed and frozen in liquid nitrogen for Western blotting.
Immunohistochemistry
After cryoprotection (30% sucrose overnight) 30 µm thick cryostat sections were cut and serially collected in 16 wells containing cold PBS. Each well received sections at a 0.5-mm distance throughout the rostrocaudal extent of the C1-C2 spinal cord. After pretreatment with 0.3% H2O2, the free-floating sections were rinsed several times in 0.1
Western blotting
The dorsal portions of spinal cord segments were homogenized in cold Tris-HCl buffer (50 m
Statistical analysis
nNOS-positive cells were counted by an observer blinded to the procedures in laminae I-III of the cervical spinal cord, in three different series of sections in each animal. The individual sections in these series were taken at 0.5 mm distances along the rostrocaudal axis. nNOS-positive neurones were scored, if they contained cytoplasmic and dendritic staining and a nucleus. The cell counts of nNOS and of relative Western blot optical densities were analysed with multiple variance analysis (
Results
Transverse sections of the cervical spinal cord demonstrated numerous nNOS-IR neurones in dorsal horn laminae I-III. These cells are small to medium sized neurones (8–15 µm diameter) with few dendrites were most common. There was no significant difference in the number of NOS positive cells at different levels of the C1-C2 region. NTG induced an increase of the nNOS-IR cells in the superficial layers of the caudal trigeminal nucleus in control, nontreated rats. A similar increase in the number of immunoreactive neurones was observed in the sumatriptan-pretreated group of animals. Lys-ASA pretreatment, on the contrary, abolished the NTG effect on nNOS-positive neurones (Figs 1 and 2).

nNOS-immunoreactivity on transverse sections of the upper cervical spinal cord, at −6 mm from the obex, in non pretreated (control) (a,b), sumatriptan-pretreated (c,d) and lysine-acetylsalicylate-pretreated (e,f) rats. Compared to vehicle (a,c,e), subcutaneous NTG administration (b,d,f) increases the number of nNOS-immunoreactive cells in controls (b) and after sumatriptan pretreatment (d) but not after lysine-acetylsalicylate-pretreatment (f). Scale bar = 50 µm.

Histogram showing the mean number of nNOS-immunoreactive cells in the superficial layers of the C1-C2 dorsal horns in the 3 animal groups 4 h after subcutaneous injection of vehicle (□) or NTG (▪) (mean + S.E.M, n= 8 per group). There is a significant increase of nNOS-immunoreactive cells after NTG injection in control and sumatriptan-pretreated rats (∗P < 0.05), but not in lysine-acetylsalicylate pretreated rats.
Western blot analysis of the C1-C2 region confirmed the results obtained by immunohistochemistry. We could identify a band at 155 kD characteristic for the nNOS protein. In animals, which had received NTG 4 h before, the density of the C1-C2 nNOS protein band was increased compared to vehicle-injected rats. This increase was similar in control and sumatriptan pretreated groups. After pretreatment with lys-ASA, the nNOS band was comparable after NTG or vehicle injection (Fig. 3). Densitometric analyses of the bands (corrected for protein loading) confirmed the results of the visual inspection of the Western blots: relative to vehicle injections, the optical density of the nNOS band was significantly enhanced after NTG administration with or without sumatriptan pretreatment, but not when the NTG injection was preceded by a pretreatment with lys-ASA (Fig. 4).
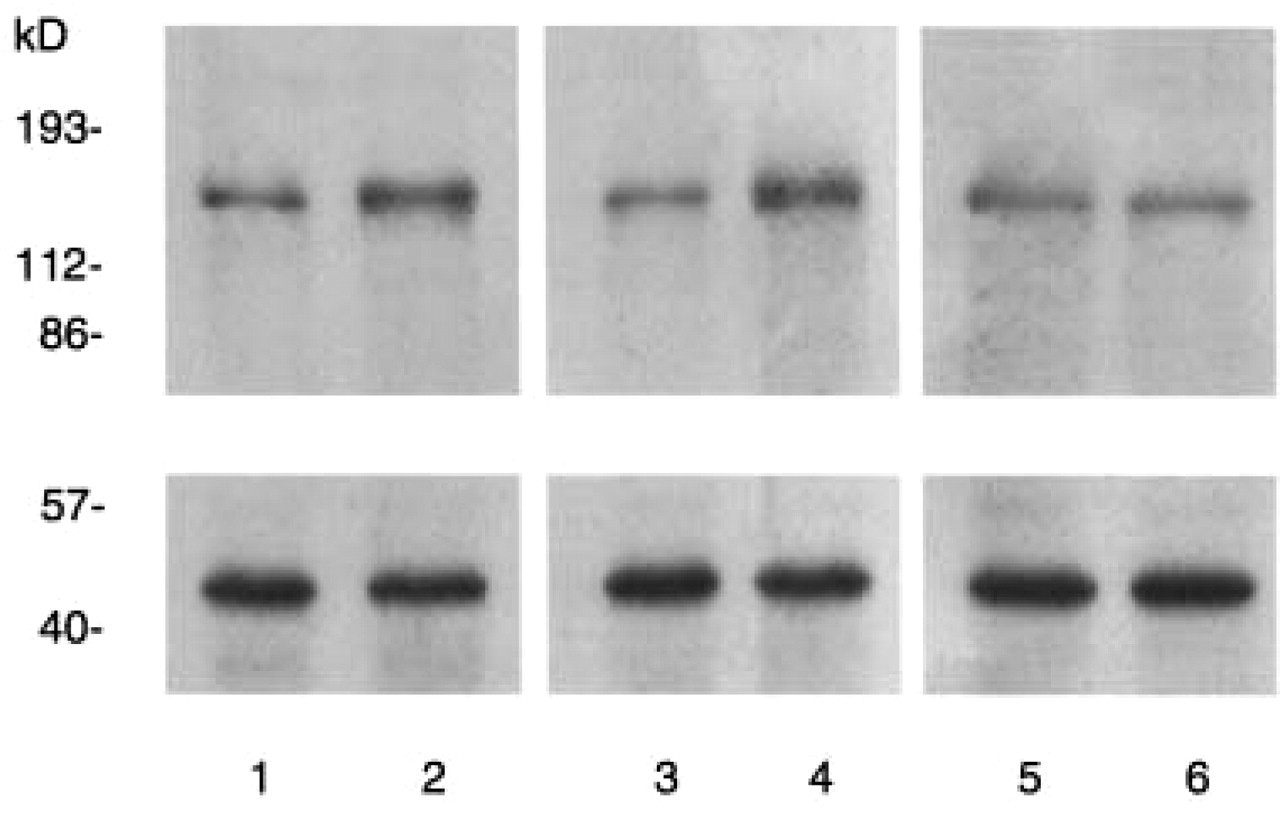
Western blotting of nNOS in C1-C2 spinal cord in control (lanes 1,2), sumatriptan- (lanes 3,4) and lysine-acetylsalicylate pretreated (lanes 5,6) animals. Compared to the vehicle (1, 3, 5), NTG administration (2, 4, 6) enhances the nNOS band (155 kD) in the control (2) and sumatriptan-pretreated group (4), but not in the lysine-acetylsalicylate-pretreated group (6). The corresponding β-actin bands are shown below for each animal group.

Histogram showing the optical densities of nNOS Western blots in the C1-C2 segments of the 3 animal groups 4 h after subcutaneous injection of vehicle (□) or NTG (▪) (mean + S.E.M, n= 5 per group). Data are expressed as proportion of β-actin. In controls and after sumatriptan pretreatment there is a significant increase of optical density in the 155 kD nNOS band (∗P < 0.05), while this increase is absent after lysine-acetylsalicylate pretreatment.
Discussion
To the best of our knowledge, this is the first study, which examines the effect of antimigraine drugs on the previously described NTG-induced increase of nNOS expression in the superficial layers of trigeminal nucleus caudalis (14). Pretreatment with sumatriptan failed to influence the increase in nNOS expression induced by the NO donor NTG, while the NSAID lys-ASA attenuated the NTG-induced nNOS activation.
It is not known how NTG modifies nNOS in TNC. There are several possible ways for NO to induce nNOS expression in secondary trigeminal nociceptors:
a direct effect on these neurones;
via local laminae I-II interneurones;
via peripheral nociceptive afferents to TNC;
via descending central pathways.
Although in our previous study (14) the NTG effect on nNOS seemed to be specific for TNC as it was not found in upper thoracic segments, we cannot rule out the involvement of other neuronal subpopulations. It has been shown indeed that NO can exert a dual effect on nociception depending on the dose used (25, 26). Moreover, the exact extent of nNOS enhancement in TNC has not been determined; it may not be exclusively related to nociception and may vary between species. We hypothesized nonetheless from our first study that the most likely explanation was a secondary activation of second order nociceptive neurones and/or interneurones because of excitation by NO of their peripheral afferents (14).
If the latter hypothesis is correct, one would expect that an agonist of presynaptic 5-HT1B/D receptors which inhibit the release of transmitters in primary nociceptive afferents (27), is able to attenuate the NO effect on nNOS expression. Sumatriptan was indeed shown to reduce the CGRP release from meningeal trigeminal fibres after electric stimulation of the Gasserian ganglion in the rat, while lys-ASA was not (28). It was also reported that sumatriptan prevents the increase of dural perivascular CGRP-IR nerve endings after electric Gasserian ganglion stimulation (29). The lack of effect of sumatriptan in our study may therefore suggest that activation of nociceptive afferents has no role in the NTG-induced nNOS increase or that 5-HT1B/D receptors are not capable to oppose such a chemical activation. Earlier studies on migraine headache have emphasized a possible role for 5-HT2B/C (30), or more likely for 5-HT2B receptors, which mediate the release of NO from the endothelium (31). Recent experiments showed that 5-HT2A receptor activation was able to enhance nNOS expression in trigeminovascular neurones (32) and up-regulation of platelet 5-HT2 receptors was reported by the same group in transformed migraine (33). Taken together these findings underline that, besides 5-HT1B/D, other 5-HT receptor subtypes play a role in trigeminovascular pain, which could be an explanation for the lack of effect of sumatriptan in our model.
An alternative explanation for the ineffectiveness of sumatriptan must be taken into account: its hydrophilicity and reduced ability to cross the blood–brain barrier (34). For instance, in cat systemic administration of sumatriptan is able to inhibit the firing of central trigeminal neurones induced by sagittal sinus stimulation only after disruption of the blood–brain barrier by mannitol (35), which contrasts with more lipophilic triptans like zolmitriptan and naratriptan (36, 37). Interestingly, in acute migraine treatment sumatriptan has little efficacy, if it is taken at the time of the aura, i.e. before the headache phase (38), whereas aspirin will not lose efficacy on the headache if given during the aura (39).
There is no doubt that sumatriptan is effective in NTG-induced attacks in migraineurs (3), but such attacks are triggered by microgram-range doses of NTG administration, which seems to produce sensitization, but not c-Fos activation of trigeminal neurones (40). It may thus be possible that higher doses of NTG, such as those used here, induce a more robust activation of central trigeminal nociceptors.
Inducible cyclo-oxygenase 2 (COX-2) is expressed in the superficial dorsal horn of the rat spinal cord (41) and plays a role in central sensitization (42). It mediates most of the analgesic effects of NSAIDs (20). After hindpaw inflammation in mice some of the COX-2 expressing cells in laminae I-II are also positive for nNOS (43) suggesting an interaction between the two enzymes. Lys-ASA, like all NSAIDs, inhibits COX-1 and COX-2. It is effective in migraine, penetrates easily the blood–brain barrier and has a long–lasting effect as well as a long half-life (44). Lys-ASA may also exert an antinociceptive effect by acting directly upon the periaqueductal grey matter (45). Taken together, these data and our present results suggest that the stimulating effect of NTG, and thus of NO, on nNOS expression in secondary trigeminal nociceptors might be mediated by prostanoids and in particular by COX-2 expressing interneurones in TNC superficial laminae. To prove this hypothesis, further studies exploring the NTG effect on spinal COX-2 and the relative time course of nNOS and COX-2 expressions in the trigeminal sensory complex, are necessary.
Footnotes
Acknowledgements
This study was supported by Concerted Action 99/04–241 of the French Community of Belgium, by grant 3.4523.00 from the National Fund for Scientific Research (Belgium) and from the Hungarian Research Fund (OTKA T-043436 and M-036252). A Pardutz was a recipient of a Marie Curie Research Fellowship of the Headache Research Unit-Faculty of Medicine-University of Liège.
