Abstract
Background: Trigeminal/neuronal hyperexcitability and spreading depression activating the trigemino-vascular system are discussed in migraine-pathophysiology. This study investigated trigeminal and olfactory event-related potentials in migraineurs. Methods: Nasal chemosensitivity was assessed in 19 female migraineurs with or without aura > 72 h before or after an attack and in 19 healthy females employing event-related cortical potentials (ERPs) after specific trigeminal stimulation of nasal nociceptors with short pulses of CO2, and specific olfactory stimulation with H2S. Odour thresholds and odour identification performance were also tested. Results: Migraineurs exhibited greater responses to trigeminal stimulation, indicated by significantly larger ERP amplitudes N1. In contrast, olfactory ERP amplitudes P1N1 were significantly smaller in migraineurs. A leave-one-out classification procedure on the basis of these two parameters assigned 76.3% cases correctly. The olfactory ERP amplitude discriminated better between groups than trigeminal ERPs (71.1 vs. 68.4% correct classification). Conclusions: Our data suggest trigeminal hyperexcitability in migraineurs. A general increase of nasal chemosensitivity is not supported because of smaller olfactory ERP amplitudes in migraineurs. Olfactory ERPs discriminate better than trigeminal ERPs between migraineurs and controls, emphasizing the significance of the olfactory system in migraine.
Current research on migraine pathophysiology focuses on neuronal hyperexcitability, i.e. increased amplitudes of visual evoked potentials (1, 2), deficient habituation (3–5) increased contingent negative variation (6), on alterations of the trigemino-vascular system (7), and on migraine as a cerebral channelopathy (5, 8–10). The degree of involvement of peripheral and/or central parts of the trigemino-nociceptive system in migraine is unclear. Some data suggest that cutaneous and mucosal trigeminal nerve fibers (11, 12) may be involved. Peripherally evoked trigeminal potentials in migraineurs showed significantly shorter latencies of early components than in controls. Other authors report involvement of central trigeminal structures (13–17).
There are also several reports of olfactory dysfunction in migraine, specifically about olfactory aura and olfactory hallucinations (18–20). Sensory hyperexcitability is frequently manifested by osmophobia (21, 22), and odours can be precipitants of a migraine attack (21, 23). In addition, a higher prevalence of anosmia or hyposmia in the interval between attacks has been suggested (24, 25).
This study investigated trigeminal and – for the first time – olfactory event-related potentials (ERP) elicited by specific trigeminal (26) and specific olfactory (27) stimulation of nasal receptors with CO2 and H2S, respectively. Psychophysical testing of olfactory function was assessed using validated tests for odour threshold and odour identification (28, 29).
Our aim was to investigate whether the hypothesis of neuronal hyperexcitability could be applied to both the trigeminal and olfactory modality. In addition, we wanted to identify electrophysiological parameters that might allow interictal discrimination between migraineurs and controls providing a more objective parameter than subjective reports. The latter was aimed at future investigations of pharmacological intervention, especially pharmacoprophylaxis.
Methods
Study population
Nineteen women suffering from migraine (age 28.8 ± 6.3 years) and 19 healthy women (age 27.2 ± 4.6 years) were included during June to August 1996. Thirteen women suffered from migraine without aura (international headache society (IHS) classification code 1.1) and six from migraine with aura (IHS code 1.2; in most cases visual auras) (30). Inclusion criteria were: (i) migraine with or without aura (IHS code 1.1 or 1.2); (ii) disease-duration of >2 years; (iii) frequency of 2–6 attacks per month within the last 3 months. Exclusion criteria were: (i) history of central nervous system or nasal pathology; (ii) chronic medication other than contraceptives; (iii) abnormal blood test results (full blood count, electrolytes, glucose, thyroid, renal and liver function). Both groups were also assessed for handedness (two left-handers in each group) and smoking behaviour (migraineurs, six smokers; controls, nine smokers). Taking into account reported differences in depression ratings (31), psychological tests for depression (Beck's depression inventory) and IQ (Raven's standard progressive matrices) were carried out, but groups were found to be comparable. The test hour and the stage of menstrual cycle were equally distributed between migraineurs and controls to prevent the results being confounded by these parameters.
Study design
All measurements were taken in the migraine-free interval, i.e. >72 h before or after an attack. Subjects were required to abstain from nicotine and alcohol for at least 12 h, and from food for 4 h prior to the experiments. The study was conducted according to the declaration of Helsinki on biomedical research involving human subjects (Somerset West amendment), and the protocol was approved by the University of Erlangen-Nürnberg Medical Ethics Review Committee. All subjects provided written informed consent.
Stimulation procedures
CO2 was used for specific stimulation of the trigeminal system. It produces stinging and burning pain (26). In contrast, H2S was used for olfactory stimulation, since it stimulates specifically the olfactory system, without simultaneous trigeminal co-stimulation (27). Monomodal stimulation was performed by the use of a special apparatus which allowed the application of chemical stimuli without causing concomitant stimulation of mechano- or thermoreceptors (32). This was achieved by embedding stimuli of 200 ms duration in a constantly flowing air stream (8 L/min) that was applied to the nasal mucosa. Temperature and humidity of this air stream were kept constant (36.5°C; 80% relative humidity). Since the rise time of the stimulus concentration pulse is <20 ms it is comparable to switching a checkerboard-reversal pattern on a TV screen (33). This allows eliciting late nearfield event-related potentials. During each session 20 stimuli of CO2 (200 ms; 70%v/v) or H2S (200 ms; 8ppm) were applied (interstimulus interval approximately 40 s), starting with CO2. During the experiment the subjects were comfortably seated in an air-conditioned room that was visually shielded. White noise via headphones (50 dB SPL) was used for acoustic shielding. All subjects underwent a training session prior to the experiment where they were familiarized with the experimental conditions and procedures as well as with a special breathing technique that avoids airflow in the nasal cavity (velopharyngeal closure) (32).
Event-related potentials (ERPs)
The EEG was recorded from five positions of the international 10/20 system (Cz, C3, C4, Fz and Pz) referenced to linked earlobes (A1 + A2). Possible blink artefacts were monitored from an additional site (Fp2/A1 + A2). Stimulus-linked EEG-segments of 2048 ms duration were recorded at a frequency of 250 Hz (hardware filter, band pass 0.2–70 Hz, pre-stimulus period 548 ms). ERPs were obtained by off-line averaging the digitized EEG-records, applying a digital band-pass filter (0.01–20 Hz). The ERP baseline was set at the mean of the digitized EEG recorded during the pre-trigger period. Records contaminated by eye-blinks (> 40 µV in the Fp2-lead, usually not more than 5 records per session, leaving at least 15 records for averaging) were automatically excluded from this process. Base-to-peak amplitudes P1, N1, P2, their latencies and peak-to-peak amplitude P1N1 and N1P2 of the ERPs were analysed using previously described criteria (34).
Psychophysical testing of the chemosensory function
Fifteen healthy volunteers had chemosensory function tested prior to the event-related potentials session by means of a set of psychophysical olfactory tests (28). Odours for the identification test were presented in felt-tip pens of approximately 14 cm length and 1.3 cm inner diameter. Instead of dye the pen's tampon was filled with 4 ml of liquid odourants or odourants dissolved in propylene glycol. For odour presentation the cap was removed by the experimenter and the pen's tip was placed approximately 2 cm in front of both nostrils for approximately 3 s. Subjects were free to sample each odour as often as necessary to select one out of four listed descriptors. Sixteen common odours were used, namely orange, shoe-leather, fish, cinnamon, peppermint, banana, apple, lemon, licorice, turpentine, garlic, coffee, rose, pineapple, clove and aniseed. Normal odour identification is indicated by 12.1±1.4 correctly identified odours. An interval of at least 30 s between different odours minimized olfactory desensitization. During the testing procedure subjects were blindfolded, and they received no immediate feedback regarding their decisions.
Odour thresholds were assessed using n-butanol as the odourant. A number of 16 dilution steps was established in a geometric series. Presentation of the odourants was similar to the identification task. Thresholds were determined by employing a multiple staircase method, using a triple forced choice paradigm, i.e. three pens were presented in randomized order, two containing the solvent and one the odourant at a certain dilution. Subjects had to indicate the pen containing the odourant. Each pen of a triplet was presented only once with an interval of at least 30 s between triplets. The subject had to correctly identify the odourant in two successive trials which then triggered a reversal of the staircase. The mean of the last four staircase reversal points of a total of seven reversals was used as the threshold estimate.
Assessment of vigilance
During the experiment, subjects performed a tracking task. On a computer monitor, using a joystick, they had to keep a small square inside a randomly moving larger one. Time of successful performance was automatically recorded (range 0–100% tracking performance) and served as a measure of vigilance.
Statistics
The analysis focused on differences between migraineurs and healthy volunteers. Implications of migraine-type, i.e. migraine with vs. without aura were studied only as an additional, secondary factor.
Unpaired t-tests were calculated for age, smoking behaviour, tracking performance, results of Beck's depression test, and those of Raven's intelligence test. Performance in odour identification and odour thresholds was subjected to univariate analysis of variance for repeated measures (rm-
Where appropriate analysis of variance was followed by univariate post-hoc comparisons to identify specific recording positions at which the groups differed significantly. For this purpose correlated unpaired t-tests with Bonferroni adjustment for multiple comparisons were performed (i.e. the α−level was set to 0.005). Finally, discriminant analysis was performed in order to identify parameters allowing for discrimination between groups and in order to estimate the discriminative performance of either pain or smell. Variables were chosen for entry into the analysis on the basis of how much they lowered Wilk's lambda. Group differences on the basis of the identified parameters were made by F-statistics, with adjustment for multiple comparisons, when appropriate. The analysis was processed by SPSS® 8.01 (SPSS Inc., Chicago, IL, USA). If not otherwise indicated, the α−level was set to 0.05.
Results
All enrolled subjects finished the study. Unpaired t-tests (smokers vs. non-smokers) for each parameter did not show any group differences. Smoking was therefore not further regarded. Migraineurs did not differ from controls in terms of age, handedness, intelligence or depression (P > 0.05, 95% confidence intervals (CI) for differences of −2 to 5.3 for age, −3.6 to 0.5 for IQ, and −1.4 to 4.9 for Beck's depression scale). None of these parameters was further regarded in the statistical analyses. In addition, no significant group differences for tracking performance were observed. Migraineurs scored 72.7 ± 3.2% (mean ± standard error) and controls 72.4 ± 5.0%.
Psychophysical tests of chemosensory function
All subjects had normal olfactory function according to published normative data of the ‘Sniff'n Sticks' test (28, 29). No differences were found between subjects with and without migraine as indicated by the absence of statistically significant main effect of ‘group’ or a significant interaction ‘threshold or identification’ by ‘group’ (Fig. 1).

Individual performance in the odour identification and the odour threshold tests. Higher values indicate better performance. No differences were found between subjects with and subjects without migraine. The data are plotted as vertical boxes with error bars showing the 10th, 25th, 50th, 75th and 90th percentiles (horizontal lines), and the 5th and 95th percentiles (horizontal dashed lines). The mean is marked with a horizontal dotted line. Individual data are presented as dots. As the identification tests provided only discrete numbers, some of the individual data are superimposed on each other.
Olfactory and trigeminal ERPs
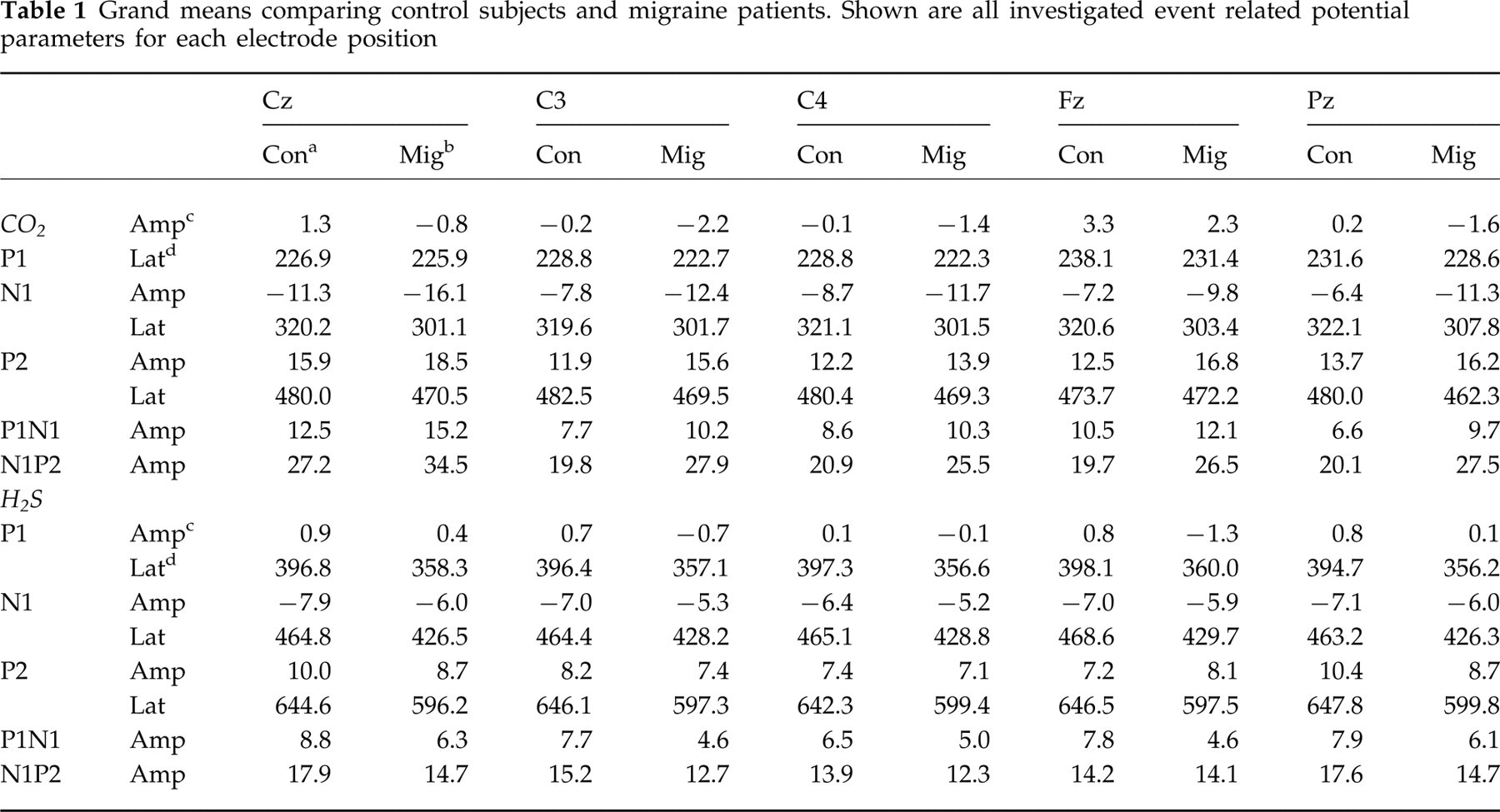
Amplitudes of olfactory event-related potentials were smaller in migraineurs. Trigeminal ERP amplitudes N1, P2 and peak-to-peak amplitudes P1N1 and N1P2 were greater in migraineurs; only trigeminal ERP amplitudes P1 were smaller in migraineurs. Latencies of both olfactory ERPs and trigeminal ERPs were shorter in migraineurs. Differences were consistent within each modality for each parameter at all recording positions (Table 1). Grand means, i.e, ERPs averaged among all subjects, separately for recording position, and for group are presented in Fig. 2. ERPs of individual subjects showing minimum, maximum and median amplitude N1 for each group and modality are shown in Fig. 3. Differences between migraineurs and controls were unequivocally spread among ERP parameters and among trigeminal and olfactory chemosensitivity (Fig. 4).
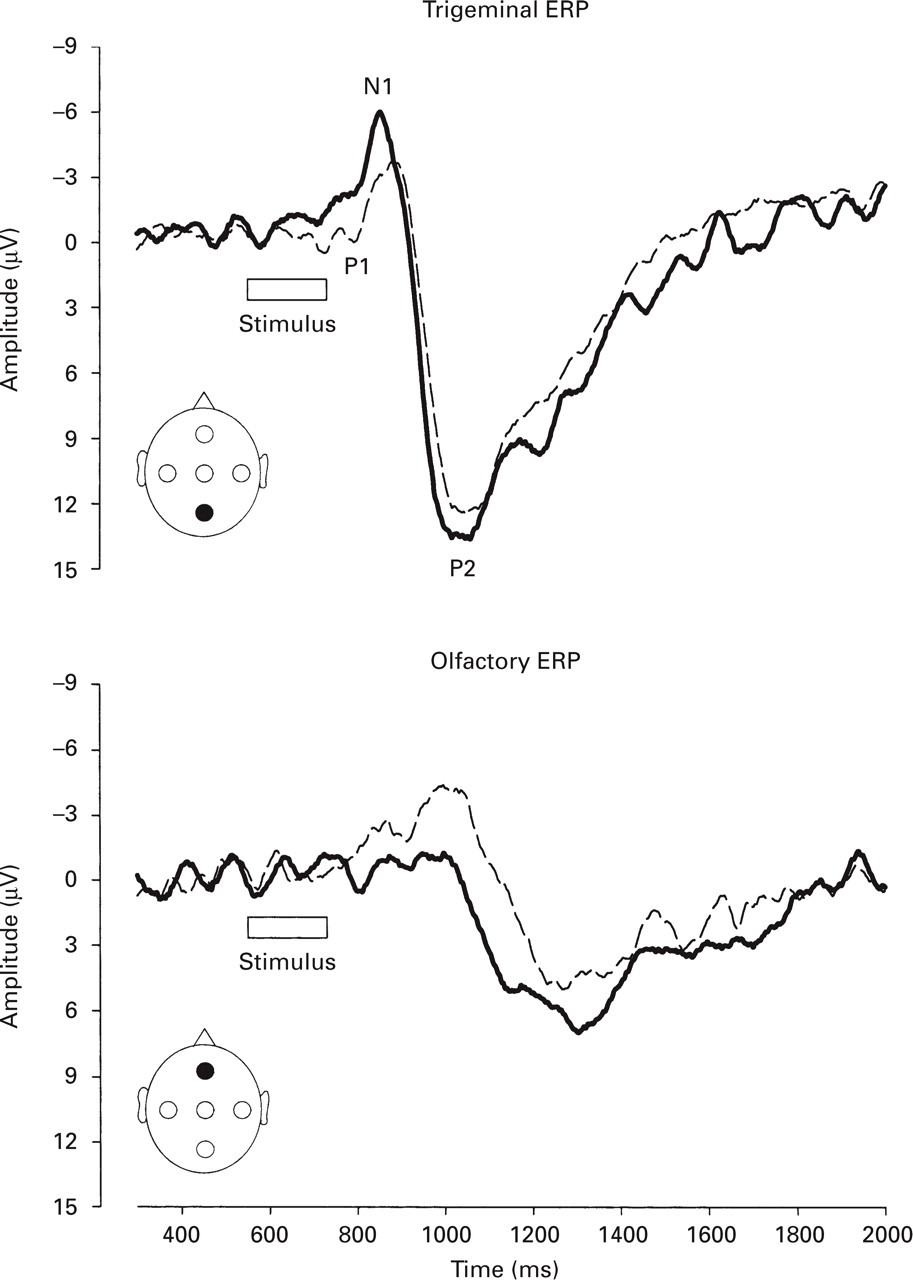
Grand means (i.e. averages across all subjects per group) of trigeminal (top) and olfactory (bottom) event-related potentials at recording positions Fz and Pz, respectively. Amplitudes P1, N1 and P2 are annotated. Note that the Y-axis indicates positive values drawn downwards and negative values drawn upwards. Migraineurs (bold lines) are compared with control subjects (thin dashed lines).

Recordings of individual subjects demonstrating group maximum (panel A), median (panel B) and minimum (panel C) of amplitudes N1 (or N1P2 where graphically indicated) of trigeminal (top) and olfactory (bottom) event-related potentials, respectively, at recording position Pz. Amplitudes P1, N1 and P2 are annotated. Note that the Y-axis indicates positive values drawn downwards and negative values drawn upwards and a change of scale of the Y-axis for group minima (panel C). Migraineurs (thick lines) are compared with control subjects (thin dashed lines). The group of migraineurs showed consistently shorter latencies. An exception is shown in panel C.

Means and standard errors of differences of parameters of trigeminal (top) and olfactory (bottom) ERPs after specific stimulation of the nasal mucosa with either CO2 (CN V) or H2S (CN I). Positive differences of amplitudes indicate higher amplitudes in migraineurs than in controls except for amplitudes N1, where because of their negative sign, the same difference is indicated by negative differences. Negative differences of latencies indicate shorter latencies in migraineurs than in controls. Significant group differences were found for amplitudes N1 of trigeminal ERPs at recording position Pz, and amplitudes P1N1 of olfactory ERPs at recording positions Cz, C3, and Fz (∗P < 0.005, ∗∗P < 0.001). The respective amplitudes were greater in migraineurs than in controls with trigeminal ERPs but smaller with olfactory ERPs.
Grand means comparing control subjects and migraine patients. Shown are all investigated event related potential parameters for each electrode position
Rm-
Discriminant analysis identified the trigeminal ERP amplitude N1 at recording position Pz together with olfactory ERP amplitude P1N1 at Fz to distinguish best between migraineurs and controls. The olfactory parameter discriminated better between groups (Wilk's lambda 0.654 for olfactory ERPs vs. 0.785 for trigeminal ERPs; significance of the probability that discrimination between groups is preserved if the variable is removed: P < 0.001 for olfactory ERPs and P = 0.027 for trigeminal ERPs). Pairwise group comparisons were highly significant for the olfactory parameter alone (d.f. = 1,36, F = 17.39, P < 0.001) and for both parameters together (d.f. = 2,35, F = 12.39, P < 0.001). Classification made on this basis assigned 78.9% of the cases correctly. In 76.3% cross-validation still made a correct group assignment. Group centroids were −0.819 and +0.819 for migraineurs and controls, respectively.
Discriminant analysis to distinguish between controls, migraineurs with aura and migraineurs without aura identified amplitudes N1 at Pz and P1N1 at Fz for trigeminal, and amplitudes P1N1 at Fz for olfactory ERPs (Figs 5 and 6). The trigeminal parameters discriminated migraine with aura from migraine without aura (F = 3.89, P = 0.03), and migraine with aura from controls (F = 4.62, P = 0.017), as well as migraine without aura from controls (F = 4.56, P = 0.018). Olfactory ERP amplitude P1N1 at recording position Fz was found to distinguish best between migraineurs with vs. without aura vs. control (Wilk's lambda: 0.618; significance of the probability that discrimination between groups is preserved if the variable is removed: P < 0.001). It clearly discriminated migraineurs with aura from controls (F = 18.19, P < 0.001), migraineurs without aura from controls (F = 9.59, P = 0.004), but not the two types of migraine (F = 3.2, P = 0.083). Cross-validated correct classification into the three groups was only 60.5%. When only migraineurs with aura and controls were included in this analysis olfactory ERP amplitude P1N1 at Fz was identified again to discriminate best. The cross-validated correct classification then was 96%.
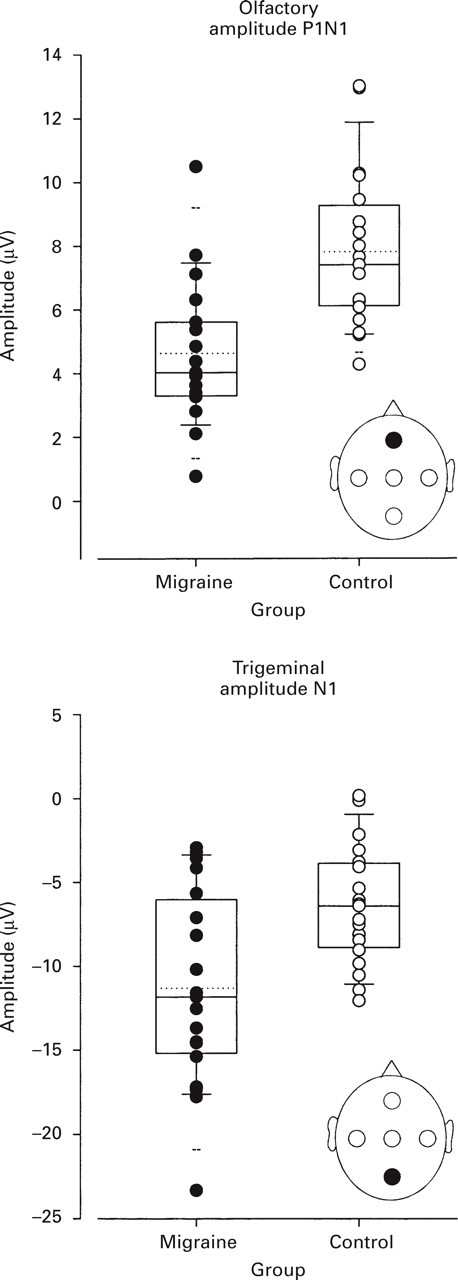
Parameters identified by discriminant analysis to distinguish best between migraineurs and controls: amplitudes N1 of trigeminal ERPs at recording position Pz, and amplitudes P1N1 of olfactory ERPs at recording position Fz. The data are plotted as vertical boxes with error bars showing the 10th, 25th, 50th, 75th and 90th percentiles (horizontal lines), and the 5th and 95th percentiles (horizontal dashed lines). The mean is marked with a horizontal dotted line. Individual data are presented as dots. Cross-validated classification assigned 71.1% of the cases to the right group. However, the olfactory ERP discriminated better than the trigeminal ERP (71.1 vs. 68.4% cross-validated correct classifications, when parameters taken alone). Note the different signs of amplitudes N1 (negative) and P1N1 (positive).

Migraine aura: Parameters identified by discriminant analysis to distinguish best between migraineurs without aura, migraineurs with aura, and controls: amplitudes N1 and N1P2 of trigeminal ERPs at recording position Pz and Fz, respectively, and amplitudes P1N1 of olfactory ERPs at recording position Fz. The data are plotted as vertical boxes with error bars showing the 10th, 25th, 50th, 75th and 90th percentiles (horizontal lines), and the 5th and 95th percentiles (horizontal dashed lines). The mean is marked with a horizontal dotted line. Individual data are presented as dots. Overall discrimination between groups was poor. Cross-validated classification assigned only 60.5% of the cases to the right group. Though the olfactory ERP discriminated better than the trigeminal ERP between migraineurs and controls, it was the trigeminal amplitude P1N1 at Fz that discriminated best between migraineurs without and those with aura.
Discussion
Olfactory ERPs discriminated better than trigeminal ERPs between migraineurs and controls. The only exception from this rule was the better discrimination between migraineurs with and without aura with trigeminal parameters. However, this explorative study focused on differences between migraine and control. Aura was introduced into the analysis only as an additional parameter. Therefore, the result of a better discriminative performance of trigeminal ERPs for ‘migraine type’ has to be considered as less reliable than the performance in the overall discrimination between migraineurs and controls.
The hypothesis of neuronal hyperexcitability in migraine is based on observations of higher amplitudes of visual (1, 2, 35, 36) and somatosensory (37, 38) evoked potentials. Recent findings based on magnetic resonance spectroscopy and evoked magnetic fields further emphasized interictal cortical hyperexcitability (39). Decreased habituation has also been proposed (40–43) as a feature of migraine, explaining higher amplitudes of evoked potentials as a result of the averaging process of single EEG records. Specifically, since the signal-to-noise ratio of single EEG records usually does not allow for reliable identification of ERPs, stimuli must be applied repeatedly to obtain single EEG records that subsequently are averaged to obtain event-related potentials. Habituation manifests itself as decreasing amplitudes from the first to the last single EEG record. When averaged, this results in smaller evoked potential amplitudes. Higher amplitudes in the averaged EEG can result from either or both, i.e. from consistently higher amplitudes in all single records or diminished habituation throughout the stimulation period.
Migraineurs showed larger responses to trigeminal stimuli. The smaller amplitudes of olfactory ERPs indicate a decreased olfactory sensitivity in migraineurs. A single explanation for these different findings might be found in the involvement of the serotonergic system in migraine pathophysiology (44–46). This theory postulates that serotonergic neurones in the brainstem play a modulatory role in cortical information processing (47). Low activity in this system leads to high cortical reagibility (48, 49) and may also diminish habituation. In pain processing, serotonin appears to have an inhibitory effect (50–52). The higher amplitudes of trigeminal ERPs might reflect an increased excitability of the pain-processing system in migraineurs. The low activity of the serotonergic system could have caused glomerular atrophy in the olfactory bulb, as known from studies in experimental animals (53). A smaller number of receptors might have been activated by the olfactory stimuli, producing smaller amplitudes of olfactory ERPs in migraineurs. In addition to serotonin, the dopaminergic system may have contributed as well to the present findings. Dopamine is a major neurotransmitter in the first relay station of the olfactory system, the olfactory bulb (for review see 21, 23, 54). Until to date, however, the role of dopamine in olfaction is poorly understood. For example, application of dopaminergic agonists does not lead to consistent changes of olfactory function (55–57).
Latency-differences did not pass the conservative statistical procedures and interpretation is speculative, but it is noteworthy that consistently shorter latencies were observed in migraineurs for both the trigeminal and olfactory modality. A recent study reported significantly shorter latencies of early components of trigeminal evoked potentials in migraineurs (58). We therefore consider our results in line with a hypothesis of neuronal, possibly trigemino-nociceptive hyperexcitability.
All participants of the present study had normal olfactory function as established by means of elaborate psychophysical testing. This contrasts with a previous report of deficits in olfactory thresholds found in migraineurs (25) where 12 out of 67 migraineurs were found to be anosmic or hyposmic. Reasons for the different findings may relate to different degrees of disease progression, different olfactory hazards in the past, concomitant intake of antimigraine drugs, or simply to differences of test procedures. Determination of odour thresholds was done by Hirsch et al. by testing the subjects once with a total of three dilutions of an odourant and a blank. The subjects' task was to tell which dilution still evoked an odourous sensation (25). In contrast, olfactory performance in this study was assessed applying 16 dilution steps in a triple forced choice–multiple staircase paradigm for threshold measurements in combination with an odour identification test consisting of 16 common odours (28, 29).
Smokers and non-smokers were equally distributed among groups in the present investigation. It is therefore unlikely that smoking has biased the results. In addition, it is known that smoking has a limited effect on olfactory function (59). For example, a dose of 30 cigarette pack-years leads to the additional misidentification of approximately one item in a 40-item odour identification test (60).
During enrolment only one man reported to the laboratory. Since women have been found to have larger amplitudes of olfactory ERPs (61), we decided to study females only, to avoid confounding of the results with gender differences. Since migraine prevalence in women is approximately three times higher than in men, the clinical relevance of the present study is preserved. With respect to the menstrual cycle, no influence on olfactory function was seen in the presently tested women, although it is known that the phase of the menstrual cycle may influence olfactory function (62, 63). However, fluctuations in olfactory sensitivity are so small that they are not observed by many authors (64–66).
This study identified parameters that discriminate between migraine and controls in the headache-free interval. These parameters could be challenged for their potential to assess interictal effects of migraine treatment. In comparison with the assessment of severity and frequency of migraine attacks these parameters may provide more information on whether the effects of pharmacological intervention were symptomatic or due to a re-normalization of underlying pathophysiology.
In summary, the present study investigated nasal chemosensitivity in migraine for the first time by concomitant assessment of event-related cortical potentials and psychophysical olfactory tests. Higher amplitudes of trigeminal event-related potentials support increased trigeminal nociceptive excitability in migraine. It is remarkable that olfactory event-related potentials discriminated better than trigeminal event-related potentials between migraineurs and controls.
Footnotes
Acknowledgements
This work was supported by BMBF(01EC9403) and by DFG (Lo 612/2–1). The authors would like to thank Dr E. Lang, Department of Neurology, University of Erlangen-Nürnberg for performing the diagnoses of migraine.
