Abstract
The swim bladder functions to maintain the fish balance at a certain position under water. Although the motoneuron-dependent swim-up behavior is important for swim bladder inflation, the underlying molecular mechanism remains largely unknown. We generated a sox2 KO zebrafish using TALEN and found that the posterior chamber of the swim bladder was uninflated. The tail flick and the swim-up behavior were absent in the mutant zebrafish embryos and the behavior could not be accomplished. As the tail flick behavior is absent, the mutant larvae therefore cannot reach the water surface to gulp air, ultimately leading to the uninflation of the swim bladder. To understand the mechanism underlying the swim-up defects, we crossed the sox2 null allele in the background of Tg(huc:eGFP) and Tg(hb9:GFP). The deficiency of sox2 in zebrafish resulted in abnormal motoneuron axons in the regions of trunk, tail, and swim bladder. To identify the downstream target gene of sox2 to control the motor neuron development, we performed RNA sequencing on the transcriber of mutant embryos versus wild type embryos and found that the axon guidance pathway was abnormal in the mutant embryos. RT-PCR demonstrated that the expression of sema3bl, ntn1b, and robo2 were decreased in the mutants.
Introduction
Zebrafish exhibit fixed but coordinated swim-up behaviors, including spontaneous muscle contractions, touch-evoked coiling and swimming, both in embryo and adult. Motor neurons have specific subtypes and different morphological characteristics, and also they extend axons to different target muscles. 1 In the process of neural development, the direction of axon growth is guided by many molecules outside the axon in the microenvironment, which can guide the axon to choose the right path to reach the target.
The swim bladder is a unique organ in vertebrate evolution. The formation process of swim bladder is divided into three stages: budding phase, growth phase, and inflation phase. The completion of the air filling of the anterior and posterior chambers of the swim bladder indicates that it can perform a complex function. The inflation of the swim bladder is a landmark stage in the growth process of zebrafish larvae.2,3 Hh and Wnt signaling are involved in the development of zebrafish swim bladder. Shha and ihha are expressed in the epithelium of zebrafish swim bladder. The receptor genes ptc1 and ptc2 of Shh signaling are expressed in the fish swim bladder mesenchymal cells. Wnt5b began to express in mesenchymal cells from 36 hpf. The two wnt signaling receptor genes, including fz2 and fz7b and coactivator gene lef1, are expressed in mesenchyme and outer mesothelium.4,5
In zebrafish, sox2 gene belongs to SoxB1 subfamily, which includes six members: sox1a, sox1b, sox2, sox3, sox19a, and sox19b. 6 When a single knockdown of sox2, sox3, sox19a, and sox19b occurred, there was no obvious abnormalities except that the tail was slightly bending upward in the sox2 knockdown embryos at 30–33 h. It proved that SoxB1 family are functionally redundant in early embryonic development. 7 When the four genes sox2, sox3, sox19a, and sox19b were knocked down at the same time, obvious abnormalities appeared in embryonic development, including delayed development, shortened A-P axis, and abnormal development of central nervous system. 7 Similar to the MO phenotypes, Gou et al generated the sox2 knockout (KO) zebrafish by TALEN and found that the mutants also showed minor defects in the tail and the failure of the inflation of the swim bladder.8,9
Although it proved that KO of sox2 can lead to tail bending and swim bladder uninflation, the exact role and mechanism of sox2 in zebrafish embryonic development remain poorly defined. In the present study, we detected the expression of sox2 in swim bladder region, and then we examined the tail flick and swim-up behavior in sox2 mutant embryos by ZebraTower recording system and studied the underlying mechanism involved in this process.
Materials and Methods
Zebrafish breeding and embryo collection
The zebrafish were raised in the zebrafish facility of the Model Animal Research Center, Nanjing University as previously described 10 according to the routine procedures approved by international AAALAC. 11 The procedures were approved by the Institutional Animal Care and Use Committee at the Model Animal Research Center, Nanjing University.
Construction and synthesis of zebrafish sox2 TALEN
The TALEN targeted sites for zebrafish sox2 were designed by the TALENT software. 12 In brief, nucleotide sequence of the HMG domain of sox2 gene was put into the box to search the target sites. TALEN plasmids were assembled as previously described. 13 The in vitro synthesis of capped mRNA was performed with the mMessage mMachine SP6 Transcription Kit (Ambion). Poly (a) Tailing Kit (Ambion) was used to add poly(A) tail to the transcribed mRNA.
Microinjection and verification of zebrafish sox2 TALEN mRNA
One nL of the sox2 TALEN mRNA was injected into the zebrafish embryos at the 1–2 cell stage. The injected embryos were then grown in the incubator at 28.5°C. The genomic DNA was extracted to examine mutated sox2 when reaching 24 hpf. Sox2 TALEN F (sequence 5′-TTTCCCAGCAAAGTTACCTC-3′) and sox2 TALEN R (sequence 5′-CTCATGTCGTAGCGGTGCAT-3′) primers were used for cloning the 722 bp of sox2 containing target sites of the TALENs.
Screening and identification of zebrafish with sox2 mutant
After sexual maturity (denoted as Founder), the zebrafish were allowed to mate freely for F1. The genomic DNA was obtained for sequencing. Finally, one mutation carrying 4 bp deletion (deletion of 4 base pairs) was generated and identified by PCR with the forward primers of 5′-ATTCGGAAATAAGTAAGCG-3′ (wild type, F2wtest-F) and 5′-AATTCGGAAATAAGTAACTC-3′ (−4 bp type, F2mtest-F) and reverse primer of 5′-AGCTGCTCCTGCATCATGC-3′ (F2test-R).
Whole mount in situ hybridization and paraffin section and hematoxylin eosin staining
These methods are described in the Supplementary Materials.
Behavioral recording and observation
These methods are described in the Supplementary Materials.
RNA sequencing and data analysis and real-time fluorescent quantitative PCR
These methods are described in the Supplementary Materials.
Data statistics
The frequency of the swim-up behavior was compared between two groups using χ 2 test.
Results
KO of sox2 leads to abnormal development of the swim bladder of zebrafish larvae
A positive adult F1 carrying a 4 bp deletion was selected to establish the KO line for further experiment. The 4 bp deletion caused a frameshift and the truncated protein comprising of 69 amino acids lacked the HMG box domain and C-terminal region (Fig. 1A). The sox2 KO zebrafish embryos showed the phenotype of tail upward bending and shortened anterior posterior axis from the late stage of pharyngeal arch (32 hpf) to hatching stage (48–72 hpf).
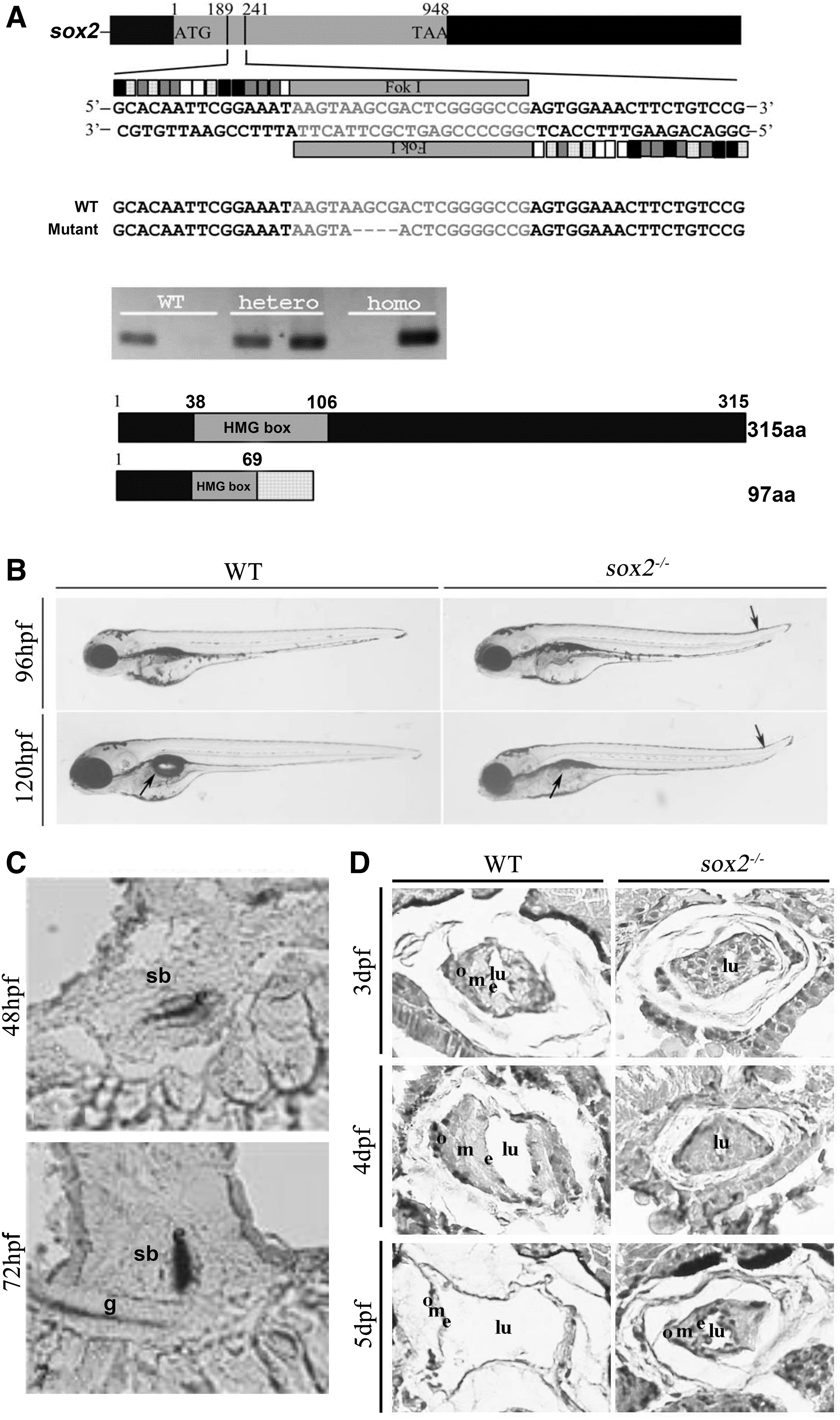
Knockout of sox2 in zebrafish by TALEN leads to abnormal inflation of the swim bladder.
In addition, we observed that all the sox2 mutant larvae failed to inflate the swim bladder at 4 dpf (96 hpf), while 31.1% (28/90) of the wild-type larvae had inflated the posterior lumen of the swim bladder. At 5 dpf (120 hpf), 93.6% (74/79) of the wild-type larvae were inflated, but all the mutant larvae were still not inflated (Fig. 1B). These data collectively suggest that disrupting sox2 may influence the swim bladder inflation.
As sox2 KO mutants displayed abnormal swim bladder inflation, we examined whether the sox2 expression exist in swim bladder or not. WISH results indicated that sox2 was highly expressed in swim bladder at 48 and 72 hpf (Fig. 1C). To rule out the possibility of the sox2 expression in swim bladder caused by probe trapping, WISH of sox2 with a sense probe was also performed (Supplementary Fig. S1). The three tissue layers of the swim bladder comprising epithelium, mesenchyme, and outer mesothelium had been formed and exhibited obvious differentiation in 3 dpf wild-type larvae.
We next examined the formation of the three tissue layers of the swim bladder in sox2 KO embryos. Compared with control at 3 dpf, the mutants showed no obvious abnormality in the three layers (Fig. 1D). At 4 dpf, the lumen of the swim bladder in wild-type zebrafish expanded dramatically and the three tissue layers proliferated obviously. Contrary to the control, the middle lumen of the swim bladder did not expand obviously in sox2 mutant larvae. At 5 dpf, both the area of the swim bladder and the posterior lumen of the wild-type larvae increased significantly. The three tissue layers became very thin and were surrounded by a large chamber in the middle, which was consistent with the phenotype of the inflatable swim bladder.
However, the three tissue layers in the sox2 mutant did not change, and the middle lumen still not expanded significantly, which was consistent with the phenotype of the noninflatable swim bladder.
We next analyzed several molecular markers of the three tissue layers and some markers related Wnt and Hh signaling involved in the swim bladder development by WISH. It revealed that the expression level and pattern of hb9 (epithelium), fgf10a and acta2 (mesenchyme), and anxa5b (outer mesothelium) in sox2 mutant embryos were not significantly changed compared to control, which is consistent with the histological result (Supplementary Fig. S2A).
In addition, the expression pattern of the shha, shhb, ptc1, ptc2, and wnt5b also showed no significant difference between mutant and wild-type embryos (Supplementary Fig. S2B). The pneumatic duct connects the esophagus to the swim bladder. The gas was swallowed and passed through the duct to inflate the swimbladder. 14 Compared with the wild larvae, the pneumatic duct formation was normal in 5 and 6 dpf sox2 mutant larvae, which indicated that the inability to inflate the swim bladder in sox2 KO zebrafish was not caused by the pneumatic duct abnormality (Supplementary Fig. S3).
Loss of sox2 leads to deficits in tail flick and swim-up behaviors in zebrafish larvae
When the embryos developed to 3 dpf, both wild-type and sox2 mutant embryos hatched from the shell membrane. The behavioral recording results showed that 100% of the wild-type larvae (5/5) had swimming behavior. The larvae would swam forward with their back up accompanied by tail flick. Compared with the wild-type larvae of the same batch, only 10% of the sox2 mutant larvae (1/10) moved forward, while 90% of the zebrafish larvae stayed with the side close to the bottom of the culture dish (Fig. 2A).
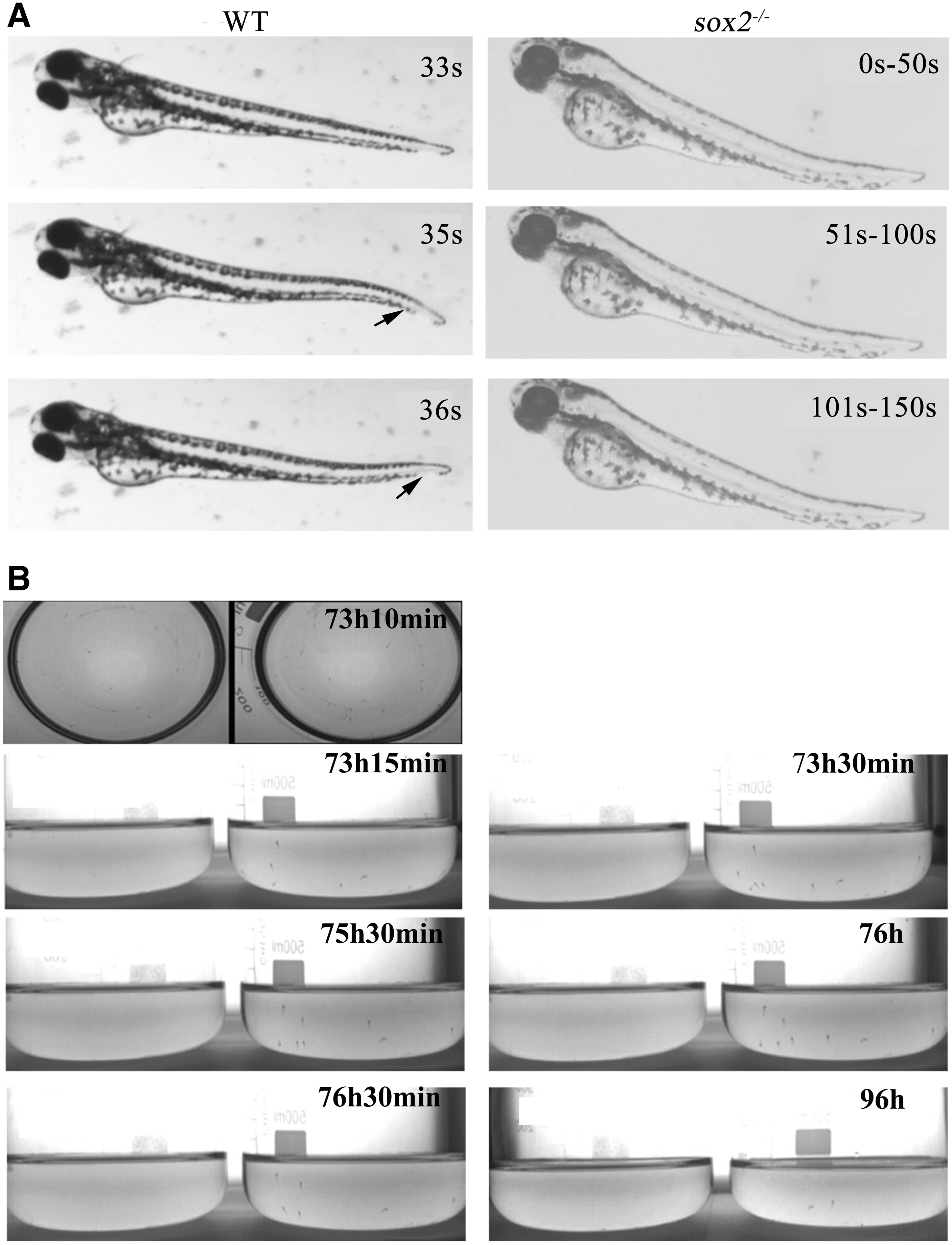
Abnormal inflation of the swim bladder upon sox2 mutant might be caused by its swim-up behavior defect.
Existing literature has shown that swim-up behavior is propelled by a short series of tail flicks, including larval attachment to and released from the tank wall repeatedly. 15 With the absence of tail flick in sox2 mutant embryos, we examined whether the subsequent swim-up behavior was affected. The swim-up behavior was recorded with a camera connected to Zebra Tower recording system. At 73 h 15 min, a part of wild-type larvae were observed to slowly attach to the tank wall, and then the larvae moved along the tank wall with a distance of 0.5–1 cm from the bottom of the tank.
After that, the larvae would keep swimming up until they reached the air-water interface between 73 h 30 min and 76 h 30 min, which was indispensable for the inflation of the swim bladder. 16 At 96 hpf, the wild-type larvae could swim freely in any depth of water, while the sox2 KO mutant larvae showed obvious abnormal movement behavior. From 72 hpf, it was found that the mutant embryos were close to the bottom of the tank in a side-up posture, and their free movement were also in a side-up posture. From 73 h 15 min to 76 h 30 min, the sox2 mutant larvae did not exhibit the swim-up behavior of wild-type larvae that initially adhered to the tank wall and then slowly moved to the water surface.
At 96 hpf, the mutant larvae were still at the bottom of the tank (Fig. 2B). The video record of 52 randomly selected wild-type larvae revealed the swim-up behavior frequency of 80.7% (42/52) as evidenced by the inflation of the swim bladder, while the swim-up behavior frequency is 0 (0/20) in the sox2 mutants (Supplementary Video S1). The frequency of the swim-up behavior in wild-type larvae was significantly larger than the sox2 mutants (χ 2 = 38.769, p = 0). These results indicated that the tail flick and swim-up behavior were absent in the sox2 mutant larvae.
Abnormal axon development of caudal neurons in sox2 mutant embryos
To verify, the tail flick behavior defection was associated with the abnormal caudal neurons development, we crossed the sox2+/− zebrafish with the Tg(huc:egfp) to generate the Tg(huc:egfp)/sox2−/− on the Tg(huc:egfp) background. The tail fluorescence intensity of Tg(huc:egfp)/sox2−/− embryos was a little weaker than that of wild-type embryos under the fluorescent dissecting microscope (Fig. 3A). To clearly visualize the development of the caudal neurons, we scanned the two embryos under the confocal microscopy. It demonstrated that the caudal neurons would stretch out many axons, which were dendrimer crossed at 48 hpf in the wild-type embryos (7/7). However, the axon development of mutant embryos in caudal neurons is abnormal and its number decreased significantly (2/10) or even disappeared (8/10) compared to control, reflecting the faulty regulation of neural-mesodermal progenitors in mutants.17,18
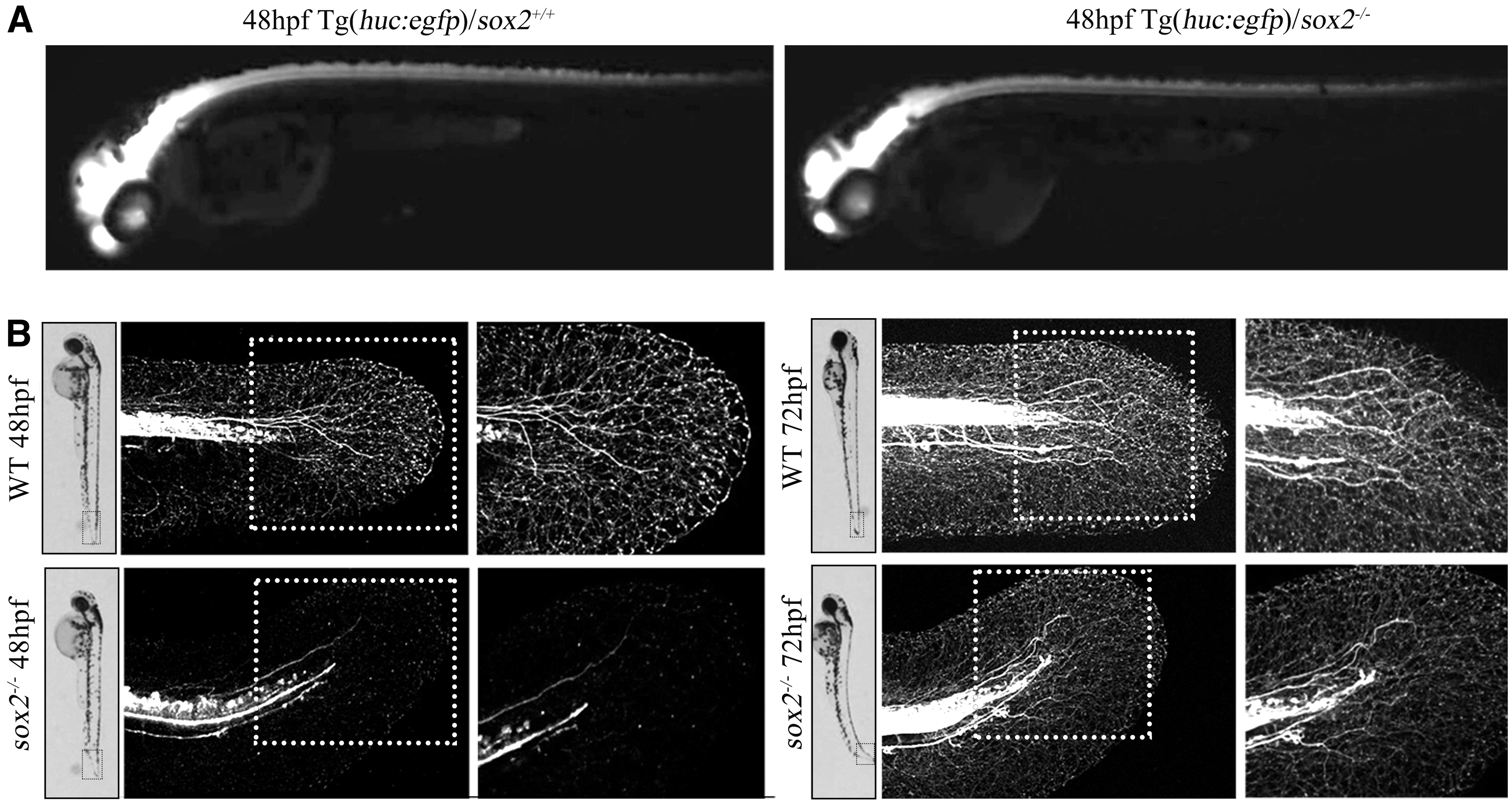
Abnormal axon development in sox2 mutant zebrafish caudal neurons.
Based on our observations, the abnormal axon in sox2 mutants are more likely axons of sensory neurons and are not likely to be directly involved in the swimming defects in sox2 mutants (Fig. 3B).
Abnormal axonal guidance in trunk and tail motor neurons of sox2 mutant embryos
As huc marks all types of neurons in the Tg(huc:egfp), we crossed Tg(hb9:gfp) with sox2+/− zebrafish for monitoring the motoneuron development on the Tg(hb9:gfp) background. In line with previous data, 19 compared with wild-type embryos, the axon guidance of CaP in trunk and tail of Tg(hb9:gfp)/sox2−/− embryos was abnormal (Fig. 4A). Further morphological observation showed that the axons of CaP in wild-type embryos had many branches and innervated to the ventral musculature at 48 hpf. However, in sox2 mutant embryos, the axons of CaP stopped in the process of innervating to the ventral musculature, which made the length shorter, while other axons were accompanied by abnormal branches. At 72 hpf, although the axons of CaP in mutants significantly grew, their branches were obviously abnormal (Fig. 4B–C).
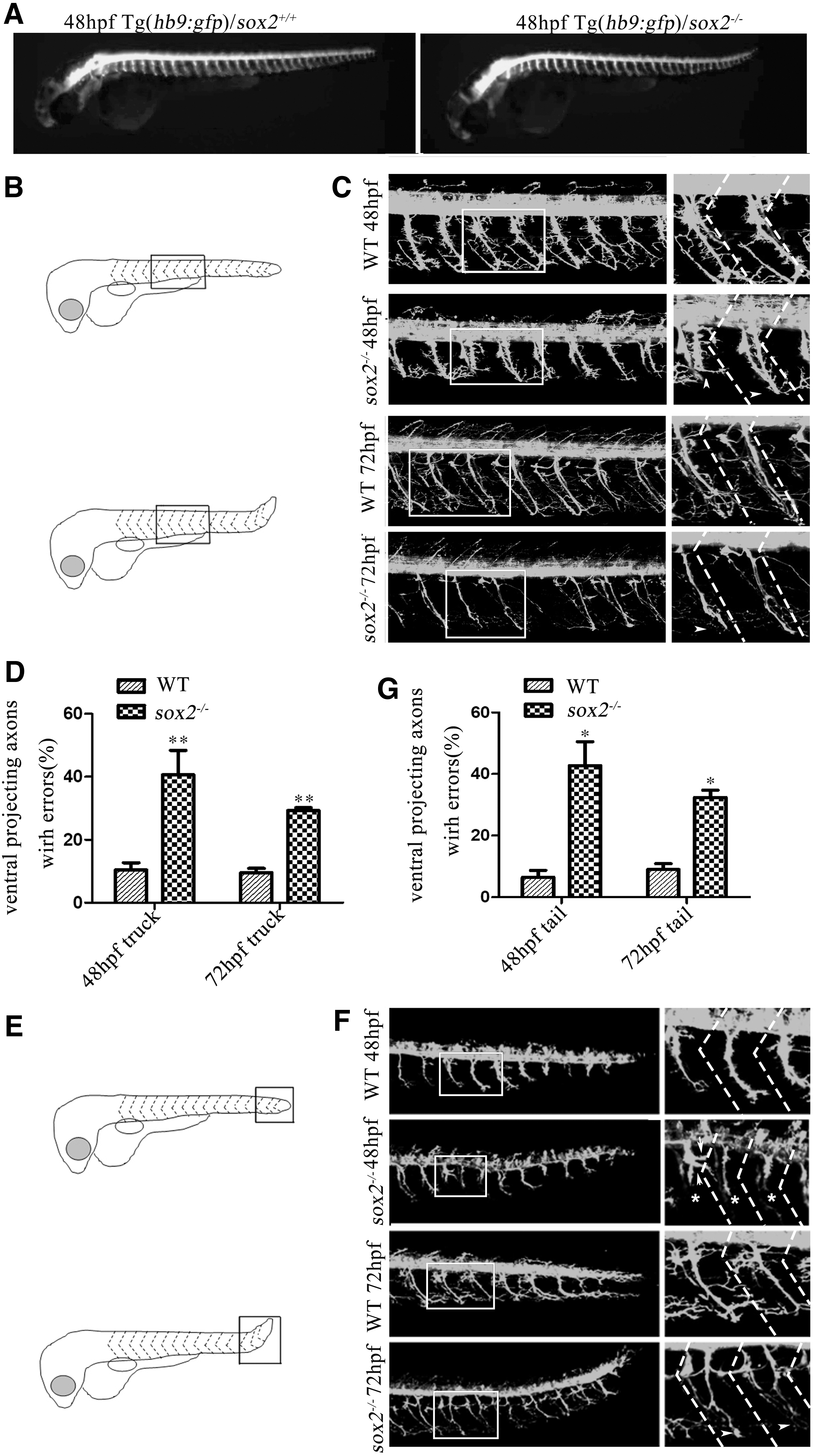
Abnormal distribution and axonal guidance of motoneurons in trunk and tail of sox2 mutant zebrafish embryos.
Among the 39 hemisegments in the trunk of 5 wild-type zebrafish embryos at 48 hpf, 10.4% of the CaP showed abnormal axonal extension to the ventral musculature or branching; However, 40.6% of the CaP from 32 hemisegments in the trunk of 4 mutant embryos showed deficits of axonal extension to the ventral musculature or branching (p < 0.01). Among the 52 hemisegments in the trunk of 8 wild-type zebrafish embryos at 72 hpf, 9.5% of the CaP showed abnormal axonal extension to the ventral musculature or branching; However, 29.3% of the CaP from 113 hemisegments in the trunk of 13 mutant embryos showed deficits of axonal extension to the ventral musculature or branching (p < 0.01) (Fig. 4D and Table 1).
Percentage of Abnormal Motoneuron Axons Extending from Trunk to Ventral Musculature in the Two Genotypes of Zebrafish Embryos at 48 and 72 hpf
In addition, we observed the development of CaP in the tail. It showed that the axonal extension and branching of the mutant embryo tail CaP were defective, which is similar to the abnormal axonal development in the trunk CaP (Fig. 4F–G and Table 2).
Percentage of Abnormal Motoneuron Axons Extending from Tail to Ventral Musculature in the Two Genotypes of Zebrafish Embryos at 48 and 72 hpf
Transcriptome sequencing revealed that sox2 mutation in zebrafish leads to changes in axon guidance signaling pathway
To further characterize the mechanism underlying the abnormal axon guidance in trunk and tail motor neurons, we performed transcriptome sequencing of caudal tissues posterior to the yolk sac isolated from wild type and sox2 mutant zebrafish. A total of 1432 differentially expressed genes (DEGs) were screened, of which 576 were upregulated and 856 were downregulated (Supplementary Fig. S4A).
The pathway enrichment showed that the DEGs were enriched in the axon guidance pathway, including nine DEGs (Supplementary Fig. S4B and Table 3). ntn1b, ntng2a, and unc5db are members of Netrin family; sema4g and sema3bl belong to semaphorins family; robo2 is a member of the Robo/slit family; epha4l is a member of Eph-ephrins family; ppp3cca is a member of protein phosphokinase; and nadl1.2 is a member of neurotrophic factor family. qRT-PCR showed that the expression levels of sema3bl, ntn1b, and robo2 were significantly downregulated in sox2 mutants at 72 hpf (Supplementary Fig. S4C).
The Results of Differentially Expressed Genes in Cluster Analysis Showed the Cluster of Genes in Axon Guidance Pathway
Discussion
Sox2 has been reported to play an important role in numerous biological processes, including neuronal development, pluripotent properties of stem cells maintenance, and so on.20,21 The role and mechanism of sox2 in zebrafish embryonic development are not fully understood. In the present study, we established a sox2 KO homozygous mutant zebrafish model by TALEN. Similar to the phenotype of MO and previous reported KO mutants,6,8 we also observed that the sox2 mutant zebrafish exhibited an upturned tail phenotype and abnormal swim bladder inflation. The behavioral recording and observation showed that the tail flick and swim-up behavior were absent.
Our findings show that abnormal motoneuron development, including trunk and tail CaP outgrowth and branches, are compromised in sox2 mutant zebrafish. The proper assembly of neural circuits during development requires the precise control of axon outgrowth, guidance, and arborization. Although the specific function and mechanism of the caudal neural arborization is not fully understood, we hypothesized that there are some axon extension or guidance molecules participated in neural circuit assembly. Moreover, RNA-seq showed that the DEGs were enriched in the axon guidance pathway. qRT-PCR showed that the expression levels of sema3bl, ntn1b, and robo2 were significantly downregulated in sox2 mutants.
Swim bladder is an important organ in fish, which can maintain buoyancy in water. The developmental process and molecular mechanism of zebrafish swim bladder are rarely known. Previous studies have demonstrated that hedgehog, 5 wnt, 4 and pbx122 signaling pathway-related proteins are expressed in all three tissue layers of swim bladder and involved in its development. For example, Morpholino-mediated gene knockdown of pbx1 caused abnormal inflation of the swim bladder, and lethality of zebrafish at 8 dpf. 22 In the present study, we found that the swim bladder formed normally in the sox2 mutant zebrafish with the presence of all three layers, but it failed to inflate. According to our results, the failure of the inflation of the swim bladder may be due to the abnormal development and guidance of axons in motoneuron, which affected the swim-up behavior in zebrafish, and thus hindered the process of swallowing air.
Moreover, Gou et al revealed that sox2 is required for the development of otic and epibranchial placodes, and its deficiency may lead to vestibular defects.8,9 Riley et al demonstrated that the vestibular function is to maintain fish balance and help to coordinate motor activity. 16 These data suggest that vestibular dysfunction caused by sox2 mutation also may be a possible contributing factor for the uninflation of the swim bladder.
It is well known that the development of motor neurons is regulated by a series of genes. In the present study, RNA sequencing revealed that the expression of DEGs was related to axon guidance, P53 signaling pathway, MAPK signaling pathway, and so on. Coinciding with our results, Gong et al also revealed that the DEGs between wild-type embryos and sox2 mutants were significantly enriched in axon guidance. 19 The results showed that the DEGs in axon guidance pathway comprised ntn1a, robo1, and sema5ba. In our study, qRT-PCR of the axon guidance molecules showed that the three genes, including sema3bl, ntn1b, and robo2, were significantly downregulated in the mutant embryos. Sema5ba and sema3bl belong to the semaphorins family, which are secretory or membrane-binding proteins. 23
All of them have a Sema domain with about 500 amino acid residues, and among of them 12–16 cysteine residues represent the specificity of each Semaphorin. 24 Semaphorins are considered as repellent factors of growth cones. 25 Sema3a is one of the most studied genes, which has been found to be involved in axonal repellent. The receptors of Semaphorin3 protein family are neurophilins. The transmembrane protein Npn1 forms a homodimer complex and binds to Sema3A. 26 The decreased expression of sema3bl in the mutants might be responsible for the length and branching of the axons in the swim bladder of mutant embryos. In addition, Netrin is a laminin-related diffusible molecule. In vertebrates, there are four members in netrin family, Netrin1-4 comprising of ntn1a and ntn1b. Netrin interacts attractively or repulsively with several receptors, including DCC. 27
The Netrin-1 plays an important role in axonal pathfinding, an evolutionarily conserved process demonstrated in several species.28,29 Robo1 and robo2, orthologs of mammalian Robo1 and Robo2, encode a transmembrane receptor that is essential for axon guidance in zebrafish. 30 Collectively, our work suggests that the identified three axon guidance molecules belong to the same family with the axon guidance pathway reported by Gong et al. 19
Nonetheless, there are limitations to this study. Although qRT-PCR was used to verify and confirm the expression of the three axon guidance molecules in sox2 mutants, external rescue of the defects of the motor neuron experiments are still not included.
Ethics Approval
The procedures were approved by the Institutional Animal Care and Use Committee at the Model Animal Research Center, Nanjing University.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Footnotes
Authors' Contributions
Conceptualization: S.C., Z.D., X.D., W.J., F.Z., and Q.Z.; Data curation: S.C. and Q.Z.; Formal analysis: S.C., Z.D., and Q.Z.; Funding acquisition: Q.Z.; Investigation: S.C. and Q.Z.; Methodology: S.C., Z.D., X.D., and W.J.; Project administration: S.C. and Q.Z.; Resources: F.Z. and Q.Z.; Software: Z.D., X.D., and W.J.; Supervision: Q.Z.; Validation: S.C., F.Z., and Q.Z.; Visualization: F.Z. and Q.Z.; Writing-Original Draft: S.C.; Writing-Review & Editing: F.Z. and Q.Z.
Disclosure Statement
The authors declare that they have no conflict of interest.
Funding Information
This work was supported by the National Natural Science Foundation of China (grant number: 31171434).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
