Abstract
The zebrafish is becoming an increasingly popular research animal around the world. Its welfare is affected by an array of environmental factors, such as food access and water quality. Holding density is an important welfare aspect, not least due to its interaction with other housing conditions. Despite the extensive use of zebrafish in research, little is known of how densities affect its welfare. In this systematic review, we have performed a large literature search, compiled, and evaluated all publications regarding zebrafish holding density. We have analyzed how density effects growth, reproduction, and stress response, including behavior, water quality, and pathogenic outbreaks in young and adult fish. Our review shows that the holding densities tested vary largely depending on the research focus, for example, body growth or behavior. In fact, research indicates that future recommendations on holding density could depend on which welfare aspects are considered. Overall, there is a need for more studies investigating the interactive effects of density on welfare indicators, such as reproduction coupled with stress response. We stress the necessity of including holding density in universal housing guidelines and reporting information on holding conditions of larvae and adults when publishing zebrafish work.
Introduction
The zebrafish (Danio rerio) has become a common laboratory animal all around the world. It has been estimated that over 3250 institutes spread throughout 100 countries work with zebrafish. 1 Despite its popularity as a laboratory animal, there is relatively little research about zebrafish welfare conditions, and there are no general guidelines established from evidence-based systematic research and evaluation on different housing conditions. Apart from ensuring that laboratory zebrafish receive appropriate care, standardized guidelines also allow for reproducibility of experimental data, and the possibility of data sharing and meta-analysis, which in turn can reduce the number of animals used in testing.
The European Convention for the protection of vertebrate animals used for experimental and other scientific purposes states that “all experimental animals shall be provided with housing, an environment, at least some freedom of movement, food, water, and care, which are appropriate to their health and well-being.” 2 Zebrafish welfare thereby concerns both their physical and mental health. Our understanding of zebrafish welfare is currently limited to a few biomarkers that can be quantified, such as behavior, reproductive performance, and endocrine and metabolic functions. 3 However, the causation relationships between the welfare indicators and the welfare states are still not fully established. The welfare of zebrafish kept for research purposes can be affected by a number of factors, such as water quality, structural and social enrichment, tank size, and availability and type of feed, to name a few. Common to a number of these factors is that they are all affected by the holding density of the fish kept under these conditions, and vice versa (Fig. 1). Considering the potential importance of appropriate stocking densities, there are still few guidelines and legislations giving clear directions on housing densities.
The holding density does not only correspond to available space per fish but will also affect other factors relevant for fish welfare, such as the access to food and the resulting water quality, including oxygen levels and waste products.
The European Directive 2010/63/EU is dedicated to the protection of animals used for scientific purposes, including fish. 4 In section 11.4 of this directive, it is stated that “The stocking density of fish shall be based on the total needs of the fish in respect of environmental conditions, health, and welfare. Fish shall have sufficient water volume for normal swimming, taking account of their size, age, health, and feeding method. Fish shall be provided with an appropriate environmental enrichment, such as hiding places or bottom substrate, unless behavioral traits suggest none is required.” 4
In some countries, legislations are recommending specific numbers of fish, regardless of species, per volume of water. For example, in the Swedish Board of Agriculture's Regulations and General Advice of Laboratory Animals, it is stated that “Fish should have at least 1 liter of water per fish that is less than 5 cm long” (our translation). 5 However, this description includes many different fish species with different needs and social structures and the recommendation is not specific enough to allow for the appropriate welfare of all fish species.
Apart from national legislations, private nonprofit organizations, such as the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) International, work toward promoting the humane treatment of animals in science through voluntary accreditation and assessment programs. 6 More than 1,000 companies, universities, hospitals, governmental agencies, and other research institutions in 49 countries have earned AAALAC accreditation, demonstrating their commitment to responsible animal care and use. Moreover, the American Fisheries Society has published guidelines for the use of fishes in research, giving general directions regarding housing, husbandry, and experimental methods. 7
Wild Populations of Zebrafish
Zebrafish are naturally distributed in rivers and lakes in Pakistan, India, Nepal, and Bangladesh.8,9 Despite their large distribution area, descriptions of wild zebrafish populations and habitats are scarce in the literature. The zebrafish is known to occupy areas that are affected by the monsoon and thereby have a rainy season and a dry season every year. The monsoon season dramatically alters the biotic and abiotic factors in their habitats, such as water flow, water depth, food availability, water chemistry, turbidity, and temperature. 8 Zebrafish have been found in various surroundings, including fast surging rivers, slowly flowing streams, stagnant water pools, and paddy fields. 9 Their bottom substrate can vary between sand, silt, or boulders and the vegetation can be submerged, floating, abundant, dense, thin, or even absent. 10 The abundance and diversity of predatory fish fluctuate between different habitats. 11 The large variation in habitats and ecological contexts that different zebrafish populations have been exposed to has created interpopulation differences due to divergent selection in various traits.9,12
It has been found that the social behavior of zebrafish is learned from their early social environment and that the shoaling preference of the adult fish varies depending if they were raised individually or in groups as larvae. 13 This was also noted by Suriyampola et al. 8 when they investigated the behavior of wild zebrafish from different populations in eastern and central India. They observed an extraordinary variation in the social behavior of zebrafish at different sites, both in terms of group size, group cohesion, and aggressive behavior. In some areas, they observed zebrafish shoaling in groups of 4 individuals, in other sites up to 300 individuals. Shoal size is determined by a combination of genetics and behavioral plasticity to the local habitat features and complexity. 8 Another trait that varies in response to the ecological context is boldness. Roy and Bhat11,12 investigated wild zebrafish populations from various geographical areas in India. They found that individuals from high-predation and high-flow habitats were significantly bolder and more aggressive than zebrafish from low-predation and low-flow/stagnant water habitats. It is not only behavior that varies with ecological exposures, although. Shukla and Bhat 9 found significant morphological differences between zebrafish populations originating from various geographical and habitat regions in India. Using multiple regression models, the authors found the main predictors in the relationship between morphological differences and ecological context to be habitat differences in dissolved oxygen, stream velocity, and predator abundance. Altogether, it can be concluded that the environmental variations in various geographical areas shape the morphology and behavior of zebrafish, not least for shoaling density.
This interesting reflection on the plasticity of the zebrafish in response to their environment raises the question of how conditions in the wild relate to the housing conditions of laboratory strains. Over the past century, the zebrafish has become an important model organism in various biological research fields, including evolution, genetics, molecular biology, and biomedical research. 14 While wild zebrafish are continuously exposed to the seasonal variation associated with the monsoon, laboratory strains of zebrafish are exposed to a predictable and constant environment. The environmental variation should give rise to greater flexibility in plastic responses in wild zebrafish, since they are continuously exposed to unpredictable environmental changes compared to laboratory strains held in aquaria. Bhat et al. 10 investigated this by comparing behavioral traits between wild and laboratory zebrafish populations. They found that laboratory-raised zebrafish fed more readily after a disturbance compared to wild populations, but found no difference in aggression or shoaling behavior. The authors concluded that genetic and environmental factors interacted to establish the behavioral response.
Understanding the life cycle and behavior of wild zebrafish will enable us to improve the welfare in captivity by evaluating factors such as water quality, life stage-specific requirements, and reproductive behavior. Animals held under optimal conditions giving rise to good welfare can be used more effectively in research and increase the reliability of data, hence proving their welfare is a key to successful experiments. 15 Density is one of the important factors that affect the welfare of zebrafish and we need to identify the appropriate range of densities that ensure healthy fish. The large variability in shoal size and density in wild populations demonstrate the need to evaluate the effects of different holding densities in captivity. The aim of this review is therefore to summarize the current knowledge on effects of holding densities on zebrafish welfare.
Methods
In this systematic review, we conducted a literature search between December 10, 2020, and December 17, 2020, on the Web of Science (including records from 1945 to 2020), using the “Abstract” advanced search function. Additional articles were identified using the “allintitle” function in Google Scholar. The search terms and results from the literature search are summarized in Table 1. The search yielded 360 articles in total. Once duplicates were removed, the remaining abstracts were critically evaluated. Articles irrelevant to the field were eliminated, leaving 40 articles. Of these, four were excluded due to not being available in English. In total, 36 articles were included in the study according to the process schematized in Figure 2. Subsequent to the literature search, we evaluated the addressed topics of all articles and divided them into six larger categories that corresponded to different important aspects of zebrafish welfare. The welfare topics that we chose were growth, reproduction, stress response in terms of physiological responses, behavior, water quality, and pathogens, and rearing juveniles and larvae. We found that dividing the articles into these different subsections allowed for a thorough review on each welfare aspect and that all publications fit under at least one of these topics. The research under each subsection was united in this discussion to perform a wider summary and to suggest future research directions in this area.
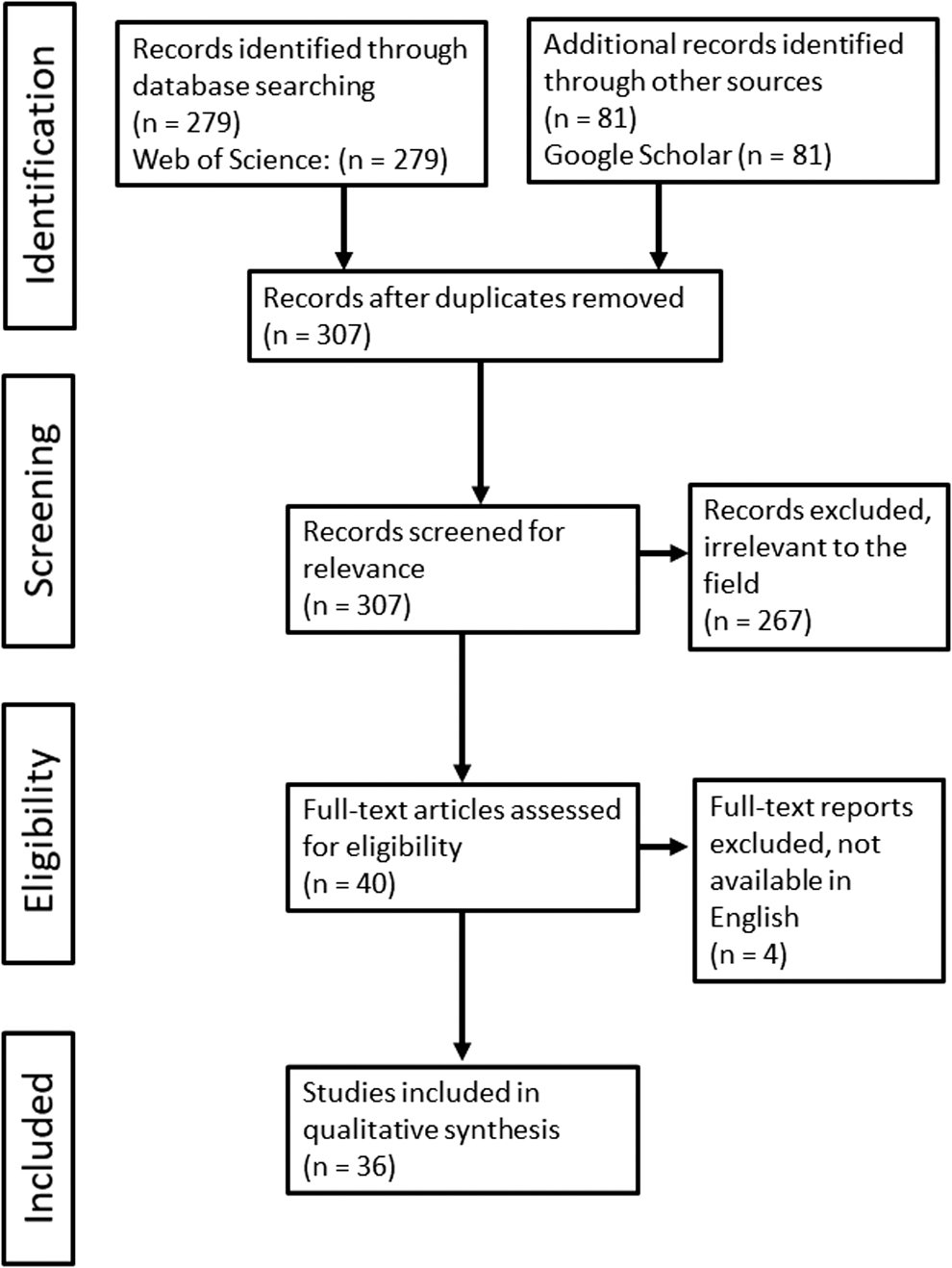
A PRISMA flow diagram illustrating the methodology of the literature search process. PRISMA, preferred reporting items for systematic reviews and meta-analyses.
An Overview of the Search Terms and Results Generated by the Database Search on Zebrafish Density and Welfare
Growth
Fast growth is an important criterion in a zebrafish research facility since many experiments are size dependent. In addition, the reproductive output of a large fish is better compared with a small fish. 16 There are, however, challenges associated with achieving fast growth, mainly because there are many parameters that influence the growth. The growth of zebrafish is affected by a multitude of environmental factors, including temperature, feed, water quality, and population density. 17 A balance of these parameters must be found to achieve optimal growth without implicating the welfare and health of the fish. This section aims to summarize the effects that holding density has on growth.
Hazlerigg et al. 18 found that the holding density of adult zebrafish had a strong effect on body growth. In their tested densities of 2.5, 7.5, and 30 fish/L, they found that the higher the density, the lower the body growth. Egge 19 found similar results in holding densities of 4.2, 8.3, and 16.6 fish/L, with significant reductions in body mass and length with increasing housing densities. The optimal holding density for growth in that study was therefore concluded to 4.2 fish/L. On the contrary, when Rabbane et al. 20 investigated growth in relation to density, they received a different result. They found the growth rate and length gain to not be significantly different between 2.5 and 7.5 fish/L, while both variations generated significantly higher results than holding densities of 12.5, 17.5, and 22.5 fish/L. This would suggest that zebrafish holding densities can be kept up to 7.5 fish/L without adversely affecting the growth.
An interesting article by Leibold and Hammerschmidt 21 studies hyperphagia and caloric restrictions by varying the holding density in combination with various feeding regimes. This is perhaps the most realistic experimental layout as it demonstrates how growth is simultaneously affected by both feeding strategy and holding density. The authors found that regardless of their holding densities, 2 and 20 fish/L, respectively, the standard variation of body mass growth was very small. This indicates that the food uptake was not affected by the different social factors associated with dominant or subordinate behavior. It raises the question of what causes the difference in growth at different holding densities, if it is not differences in food intake due to social differences. A possible explanation is that higher holding densities increased the stress levels, which reduce the fish's ability to absorb and assimilate the nutrients in the feed. It is also possible that the growth difference is due to other holding parameters, for example, elevated or reduced temperature levels, which affect the metabolism and growth. If the feeding regime and technique differ between the studies discussed above, that could also explain why studies with similar testing densities found contradictive results.
Reproduction
Since many, if not most, zebrafish laboratories have continuous breeding programs in place, reproductive output is an important factor when considering holding density. In fact, the main reasons that zebrafish are such popular laboratory animals is their short generation time of 2–4 months 22 and high number of offspring at each mating event. This section will summarize the articles on how reproductive output is affected by holding density.
Kolb et al. 23 tested how two different housing densities, 3.3 and 6.6 fish/L respectively, affected the reproductive outcome. They found that fish held at the higher density gave smaller embryos, but larger larvae. However, there was no difference in average clutch size or viability of the eggs. Castranova et al. 24 conducted a similar study by keeping fish in three different densities of 3, 6, and 12 fish/L for 1 month before setting up pairwise spawning. They did not find any significant difference between the treatments on average clutch size or percentage of spawning success, and viable eggs. Nor did Rabbane et al. 25 find any difference in fertilization rate, hatching rate, or viability of eggs from fish held at densities of 5, 15, or 25 fish/L for 2 months. At the very high testing densities of 35 and 45 fish/L, however, they did find a significant decline in reproductive success compared to the lower densities tested.
In addition, shorter experiments of holding fish in larger tanks with the access to spawning sites have been performed. For example, Spence and Smith 26 designed an experiment to test the effect of two relatively low holding densities, 0.05 fish and 0.25 fish/L, on reproduction. They found that the total number of eggs produced at an assigned spawning site during 4 days was significantly greater in the higher holding density. The authors suggested that the difference was due to increased male territorial behavior at the higher density. Spence et al. 27 studied how the exact same holding densities affected male reproductive success. Their results showed that density treatment of 0.05 fish/L led to increased mating success for territorial males, while holding fish in 0.25 fish/L diffused this difference and territorial and nonterritorial males were equally successful.
A study by Hutter et al. 28 reached a similar conclusion by studying the reproductive behavior of wild-caught zebrafish, although the amount or quality of eggs laid was not quantified. They found that pair spawning occurred more frequently in large tanks with low densities of 0.007 fish/L, while group spawning events were more common in smaller tanks with higher densities of 1.2 fish/L. However, territorial behavior was not observed at the lower density of 0.007 fish/L, indicating that the larger tanks provided enough spawning sites for the fish as they were not restricted to one assigned spawning site. This setup with such large tanks was unusual in the articles reviewed in this section, and while it might better correspond to wild environment, it was not directly comparable to other studies in the field.
In studies where breeding sites were limited, as in Spence et al., 27 territorial males were more successful at very low densities and large arenas. At higher densities, territorial males were unable to monopolize the spawning and group spawning occurred in its place, thus leading to equal mating opportunities for territorial and nonterritorial males. This would explain why there were no differences found in the studies that investigated holding densities of 3 fish/L and higher since the tested densities were above the threshold for allowing equal success regardless of the territoriality of the males. The overall conclusion from this section is therefore that holding densities could be kept between 0.05 and 25 fish/L to avoid adverse effects on reproduction.
Stress Response
The stress level is an important consideration when discussing the welfare of research animals, since one of the main ethical premises of using research animals is to minimize unnecessary suffering and stress. 15 There are two main methods to evaluate stress in zebrafish, physiological responses and behavior. We will start by discussing the physiological stress responses to various holding densities in this section and then proceed to the behavioral stress responses in the next.
The physiological stress response in zebrafish is controlled by the hypothalamic-pituitary-interrenal axis through the release of a series of hormones, with cortisol being the primary. 29 Over time, cortisol has become one of the main markers for evaluating stress in zebrafish. Despite that, few studies have been conducted to evaluate the effects of various holding densities on the cortisol response of zebrafish.
Ramsay et al. 30 crowded zebrafish at a very high density of 40 fish/L for two different exposure times, 3 h and 5 days, respectively. They found that the crowded zebrafish from both the 3-h and 5-day treatments had significantly higher cortisol levels than the control zebrafish housed at 0.2 fish/L. Thus, the study confirms that exposure to high densities will influence the physiological stress response of the fish. Gronquist and Berges 31 also conducted a study to evaluate the effect of three different stocking densities (0.13, 0.30, and 1.2 fish/L) on the cortisol response in zebrafish by measuring cortisol dissolved in tank water. They did not find any significant difference on cortisol secretion between the treatments. Pavlidis et al. 32 exposed zebrafish to four different holding densities of 4, 10, 20, and 40 fish/L, respectively, and found no difference in trunk cortisol between the treatments. In addition, they conducted experiments in tanks where the number of fish was kept constant at 10 fish, but varied the water volume inside between 0.25, 0.5, 1, and 2 L. Interestingly, they now found the stress response in 10 fish at 2 L (5 fish/L) to be similar to controls kept at 0.2 fish/L, while treatments with 10, 20, and 40 fish/L had again significantly higher trunk cortisol levels than control conditions. Ramsay et al. 30 also found that tank size affected the response and that crowding fish at 40 fish/L caused a significant increase in cortisol in large tanks (76 L), but not in small tanks (4 L).
Both Ramsay et al. 30 and Pavlidis et al. 32 found that the effects of crowding zebrafish at 40 fish/L generated different cortisol responses depending on the tank size and water volume. However, the articles reached opposite conclusions where Ramsay et al. 30 concluded that fish were more stressed during crowding in larger water volumes, while Pavlidis et al. 32 found fish to be more stressed in smaller water volumes. There were considerable differences between the two studies that could explain the results. The main difference is that Ramsay et al. 30 used tanks of different sizes, while Pavlidis et al. 32 maintained a constant tank size and varied the water volume.
It is difficult to propose clear housing guidelines based on how density affects the physiological stress response of zebrafish from these few articles. The available evidence indicates that zebrafish should be held at a density of 5 fish/L to minimize the physiological stress levels. There is, once again, a need for more experiments in this field, particularly to evaluate the stress response to long-term exposures at different densities.
Behavior
The social behavior of zebrafish changes depending on their housing conditions, such as holding environment and density, and certain behaviors indicate anxiety and stress.33,34 Behavioral changes that indicate anxiety and stress include increased swimming speed, tighter school cohesion, freezing bouts, and fish remaining close to the bottom of the tank. 29 Kleinhappel et al. 35 studied the behavior of laboratory zebrafish in response to acute stressors. They found that stress exposure caused a change in social group dynamics, with higher shoal densities and reduced distance between individuals in the shoal. Shelton et al. 36 analyzed how the physical and social environment influenced the spatial distribution of zebrafish and found density to be the main defining factor. Their results indicated that the distance between zebrafish in a group depended mainly on the holding density and that zebrafish at a higher density of 1.4 L water per fish swam closer than zebrafish held at a lower density of 2.8 L water per fish. It can thereby be concluded that the holding density of zebrafish has a strong influence on their behavior. Studying the behavior of fish is a popular research method since it is unobtrusive, low cost, and relatively simple to perform. Despite the benefits, there are few studies on how various holding densities affect the behavior of zebrafish.
Gronquist and Berges 31 exposed zebrafish to three different holding densities of 0.13, 0.30, and 1.2 fish/L, respectively, and studied differences in their behavior. They found no significant difference between treatments on fin display, flutter, aggression, gape, or chatter. Spence and Smith, 26 on the other hand, found a difference in male territorial behavior when housing zebrafish at two different densities of 0.05 and 0.25 fish/L. They observed that males in the higher density displayed significantly more aggressive behaviors than males in the lower density. On the contrary, a study by Paull et al. 37 found that aggressive behavior decreased as density increased. The found aggression to occur most frequently in a density treatment of 0.3 fish/L, less frequently in the 0.8 fish/L treatment, and even less frequently in the 1.6 fish/L treatment. Thereby, the three different studies that have investigated density effects on aggressive behavior have all found different results.
The reason that Spence and Smith 26 found increased aggressive behavior at higher densities could be due to them having a shorter exposure period for the fish at 4 days, compared to 8 days in the study by Paull et al. 37 and 10 days in the study by Gronquist and Berges. 31 It is possible that social behavior changes with exposure time, and that the hierarchies at 4 days of exposure differ from those of 8 days or longer. Another explanation for the conflicting results between the studies could be that the authors tested different spans of holding densities. For example, the highest tested density by Spence and Smith, 26 0.25 fish/L, was lower than the lowest tested density by Paull et al., 37 0.3 fish/L. Since the social behavior of zebrafish seems to change with different holding densities, the trend in aggression level could diverge depending on which density span was tested.
The presumption that higher holding densities cause aggression has led to some interesting follow-up experiments on how to reduce the behavior. Carfagnini et al. 38 investigated the effects of increased spatial complexity on aggressive behavior by adding artificial plants to zebrafish aquaria with fish held at 2.4 fish/L. They found that spatial complexity reduced aggression and suggested that providing covers could increase the welfare of zebrafish housed at high densities, although this was not tested in their study. Wilkes et al. 39 on the contrary, did not find any reduction in stress levels or aggressive behavior after the addition of glass rod structures into zebrafish tanks. They did, however, mention that the lack of effect could be due to the short exposure time of the enrichment, since the experiment lasted only 1 week. It is also possible that the type of spatial complexity is important and that certain objects have a greater effect on reducing stress than others.
Since zebrafish are found in shoals in the wild, 8 laboratory zebrafish are typically housed in groups. There are, however, some studies that indicate this may not be the optimum way to house zebrafish. Parker et al. 33 found that the basal cortisol levels of individually housed fish were lower compared with group-housed fish. Shams et al.40,41 found that socially raised fish placed in chronic social isolation had reduced anxiety behavior and lower serotonin levels compared to control fish. However, in a third study, the same research team found no difference in stress response between life-long developmentally isolated fish and socially raised fish, 42 indicating that the exposure time of social isolation affects the response. This indicates that housing zebrafish in groups, as is done in most zebrafish laboratories, may not provide optimal welfare of the fish. However, keeping fish isolated in tanks has several drawbacks, including higher costs and increased space requirements. It is also more difficult to study the social behavior of zebrafish that have been chronically isolated as there is no baseline for their control behavior in response to conspecifics. Therefore, although it is important to not disregard that isolated fish may be less stressed, it is unrealistic in practice to house the fish this way. We will therefore focus this review on the premises that zebrafish are housed in groups.
Water Quality and Pathogens
Holding fish in laboratories at high densities comes with challenges, not least for maintaining good water quality. The largest challenge is to prevent the accumulation of the toxic waste product ammonium by converting it into nitrate, which is much less toxic. 43 Accumulation of waste products and other metabolites, such as secreted cortisol, 32 can be problematic in both stationary and recirculating aquaculture systems affecting both development and growth of zebrafish43,44 and adult zebrafish behavior. 45 The problem is mainly during the start-up period of the aquaria since it takes time to establish an appropriate bacterial culture of nitrifying bacteria. Although few systematic studies have been performed to investigate how water quality is affected by fish density, Pavlidis et al. 32 showed a reduction in water quality and dissolved oxygen in their static tanks during the crowding experiment at 40 fish/L. However, when cortisol release was measured from static tanks and compared to crowding experiments with recirculating water, there was no difference in trunk cortisol responses following the crowding experiment, indicating that the crowding had a larger influence than water quality when the latter was adjusted. Once again, we express a need for more projects of how holding densities affect important parameters.
A slightly more explored and related avenue is whether high holding densities provide an increased risk of bacterial infections in the fish. Murray et al. 46 tested the effects of two different holding densities, 3.3 and 4.9 fish/L, respectively, on the development of subclinical mycobacteriosis over a period of 1 year. They found a greater proportion of fish infected by Mycobacterium in the higher holding density. A similar study by Ramsay et al. 47 found a higher cumulative mortality from Mycobacterium infections for fish exposed to crowding and handling stressors compared to control fish. The same authors also evaluated the effects of crowding and handling zebrafish on infectious outbreaks of Pseudoloma neurophilia and once again found significantly higher mortality in response to crowding and handling. 48 The elevated mortality was accounted to chronic stress resulting in elevated cortisol levels and increased immunodeficiency. Pullium et al. 49 described their experiences when housing zebrafish at a density of 5.4 fish/L and discovering a 15% mortality due to the infectious outbreak of motile Aeromonas septicemia (MAS), although they had no other holding density to compare with. The authors discussed that crowding fish could have caused increased stress and thereby elevated the risk of an MAS outbreak, although also suggesting that poor water quality could have caused it. In fact, Murray et al. 46 found that the cleaning method of the tank had a large impact on the infection rate, which indicates that infection outbreaks may be prevented by applying efficient cleaning techniques.
For these studies, it can be concluded that holding density plays a critical role in disease control of zebrafish. It is, however, difficult to determine with certainty at what densities the risk starts increasing. Only in one of the articles, Murray et al. 46 designed their study to investigate the direct impact of holding density on disease outbreaks, while the other studies used crowding stress in combination with other stressors. Regardless, it is clear that minimizing husbandry stress is an effective preventive method to reduce pathological outbreaks. There is a need for more studies investigating the direct impact that holding density has on water quality and pathogens.
Rearing Larvae and Juveniles
Although the main focus of this review is density effects on adult zebrafish, we will focus this section on the impact of holding density when rearing zebrafish larvae and juveniles. There is a multitude of parameters that influence the survival, growth, and sex determination of zebrafish larvae, including water quality, light cycle, feed, and stocking density. 50 The sex determination system in laboratory strains of zebrafish is controlled by the same external factors, such as temperature and rearing density, during the gonadal development period at 15–45 days postfertilization. 51 Ribas et al. 51 tested the effects on masculinization on zebrafish larvae raised at four different densities of 9, 19, 37, and 74 fish/L. Their findings demonstrated that as the rearing density increased, there was a larger proportion of males in the brood. The authors recommended a rearing density of 13–20 fish/L to avoid overmasculinization of the larvae. The skewed sex ratio, however, might be simultaneously affected by other influencing factors as well. For example, Krug 52 evaluated the relationship between masculinization at a rearing density of 40 fish/L and food availability. The results indicated that when food rations were adjusted to the number of fish, there were no skewed sex ratios at high rearing densities of 40 fish/L compared to 10 fish/L.
In addition to sex determination, there are other density-affected processes during zebrafish rearing. For example, higher holding densities can cause increased mortality during the early juvenile phase, 18 decreased growth during the first 3 weeks to 3 months,51,53 and increased occurrence of skeletal deformities during the first 3 months. 54 To optimize the growth, fish larvae should be held at densities between 12 and 22 fish/L according to Delomas and Dabrowski, 53 and at 9 fish/L according to Ribas et al. 51 To reduce the risk of skeletal deformities, fish larvae should be held below 8 fish/L. 54 Although there are some contradictory evidence, the overall recommendation taken from these publications is that the rearing density of larvae should be kept below 12 fish/L.
Discussion
The effect of density on zebrafish welfare is a largely understudied field. As illustrated in Figure 1, multiple parameters influence the welfare of the fish, and density is one of them. The aim of this review was to summarize publications focused on how various holding densities affect an array of welfare indicators of zebrafish (Fig. 3).
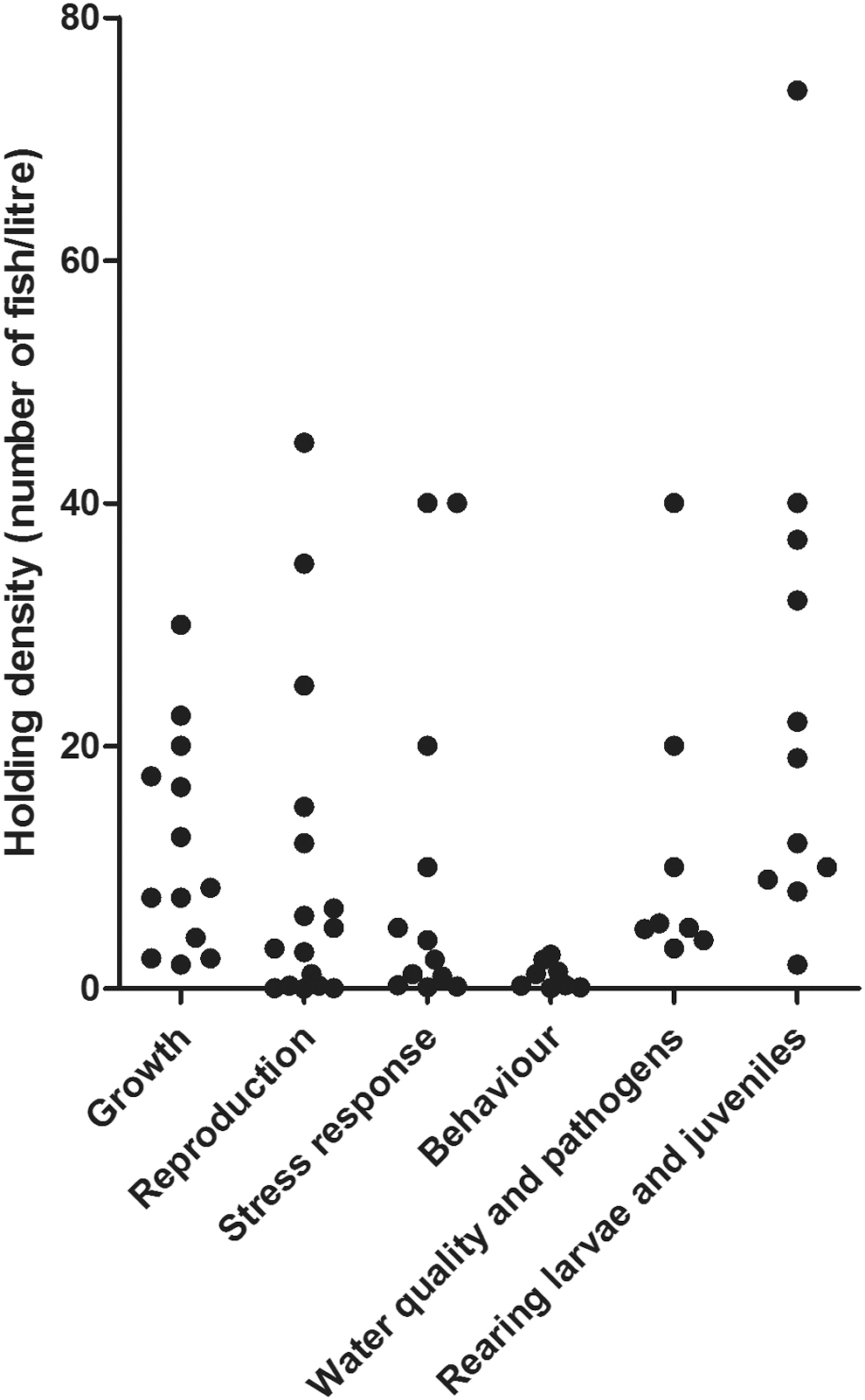
Holding densities of fish in the experiments reviewed in this article, divided into categories corresponding to research focus.
The conclusion from our analyses of published research is that the appropriate density levels depend on what welfare indicators are focused on. To optimize the growth of zebrafish, research indicates that densities should be kept below 7.5 fish/L, although it has been suggested that improved feeding regimes and techniques may compensate for the adverse effects of higher densities. For maintaining an effective reproductive output of the fish, studies suggest that densities can be kept anywhere between 0.05 and 35 fish/L. Studies on stress response in relation to density suggest a holding density of 5 fish/L to minimize the physiological stress response, although only two research articles were available on the subject. Behavioral studies have only been conducted at densities below 1.2 fish/L, which makes it difficult to pronounce any recommendation on the subject. Similarly, there is a complete lack of studies on the connection between density and water quality, although our own experience suggests that once a bacterial flora has been established, densities as high as 16 fish/L can be held without adverse effects on welfare. Multiple publications have shown that higher densities are associated with a higher risk of disease outbreak, although it can be moderated by using effective cleaning methods.
As outlined throughout the review, there is a lack of consistency in the experimental design of the studies. Figure 3 summarizes the tested densities in the articles reviewed in this study. It illustrates that, while growth and reproduction have been tested over a larger scale of densities, stress response has mainly been tested in densities below 1.2 fish/L or above 40 fish/L. Studies on behavior and pathogenic outbreaks have only been done at densities of 5 fish/L or lower. The relativity of what is considered a “high density” or a “low density” in the various studies complicates comparing their results. Referring to a “high density” can mean any value between 0.25 and 30 fish/L depending on the study. Similarly, a “low density” can be anywhere between 0.05 and 5 fish/L. These inconsistencies provide a challenge when creating guidelines to improve holding and welfare for zebrafish. There is a need for more studies within all welfare parameters on a more even scale of tested densities, especially where the few existing publications show contradicting results. In particular, there is a lack of behavioral studies on densities above 1.2 fish/L and how any holding density affects water quality.
Moreover, we are critical to the lack of studies that are using the setups that zebrafish today are most commonly being held in, that is, rack systems consisting of 3–11 L tanks. The rack systems limit the available tank sizes, possible enrichment, and light conditions, and maintain the water parameters, such as temperature, salinity, and pH, at stable levels. In addition, some studies have indicated that tank size can affect the experimental effects, such as stress response30,32 and breeding behavior. 28 It would therefore be more relevant for understanding zebrafish housing to use standard sizes as they are becoming the most commonly used setting that zebrafish are held in worldwide. We support future studies making use of established housing conditions.
Similarly, there is a large variation in exposure time at the various densities before the testing was performed. Certain studies have set the exposure time to 5 days while others have used exposures of 1 year or even longer. Intuitively, this would create large differences between the studies and complicate comparisons between them. The rationale behind why different studies have chosen specific testing densities or exposure times remains unknown. However, when it comes to understanding welfare as a result of housing fish at a specific density, a study would benefit from the animals being held at the investigated conditions as long as possible, to capture the less acute changes in welfare and to unravel possible acclimatization to the environment. This is supported by data from studies such as Pavlidis et al., 32 who showed that trunk cortisol levels were elevated after 2 h in their setup regardless of density, while these levels normalized after 5 days in the tanks. Moreover, for future research, it would be preferable if all baseline studies on zebrafish welfare (i.e., not concentrating on crowding) focused on a certain span of densities, for example, between 1 and 20 fish/L. This would improve the comparability between studies. Looking at previously studied densities within the field and aiming to fill in gaps would also improve the knowledge within the field.
There is a need to improve the connection between the research and the experience from laboratories around the world. Multiple studies refer to knowledge and recommendations for holding densities that obviously exist within the zebrafish community, but remain unpublished. Within the welfare field, it is crucial that holding guidelines for zebrafish are well anchored in scientific, peer-reviewed publications. This would improve the precision and relatability of publications within any field that use zebrafish as a laboratory animal, as well as creating a foundation on which to base guidelines and laws.
A related topic is that details on all welfare parameters of the studies must be published in the Methods section, such as feeding regime, water quality, and structural enrichment. Feeding regime and feeding amount in particular must be adjusted to the number of fish held at each density. Universal guidelines need to be developed and adapted for all these factors to ensure that the study of any one factor is not affected by a change in another. In addition, the initial holding environment and welfare parameters of the zebrafish may also affect how the adult fish responds to various situations and surroundings. There is therefore a need to standardize how all zebrafish are held in different laboratories. The ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines provide specific guidelines for zebrafish and should be adapted and used in all laboratories around the world. 55 This would improve the quality of science within the welfare field. To enable this in all laboratories, however, the costs must be reduced for tools and test kits used in evaluating water parameters. The availability of cheap, yet reliable, testing equipment is vital to improve zebrafish welfare worldwide.
Conclusion
Reviewing this literature has clearly demonstrated how crucial density is for the welfare of zebrafish. It affects a wide array of parameters, including growth, reproduction, stress response, behavior, water quality, and pathogenic outbreaks. Density should be included when creating universal holding guidelines for laboratory fish and must be kept constant between experiments when varying other parameters. Despite its importance, there is a lack of peer-reviewed publications within the field of how holding density effects zebrafish welfare and a dire need for more studies to be conducted in the future. It has been suggested that zebrafish welfare can be improved as a result of increased funding of resources and research in this area to create a scientific evidence base, greater education of staff, new technologies, and spread of information between facilities within the community. 56 However, there is a gap between the political demand to improve animal welfare and the lack of research and research funding focusing on identifying scientifically sound biomedical indicators for animal welfare, being the base for housing guidelines.
Moreover, we recommend zebrafish researchers publishing their work to use the ARRIVE guidelines and include information on tank/housing system (type and dimensions), food (type, composition, amount, supplier, and feeding regime), water temperature, sanitation (water quality and frequency of tank cleaning and water changes), social environment (group size and composition/stocking density), and lighting (type, schedule, and intensity), as well as environmental enrichment. Information regarding conditions during the raising of larvae and juveniles are of importance; therefore, if conditions differ from the housing of the adult fish, such as holding densities during raising of juveniles, it should be stated.
Footnotes
Authors' Contributions
M.A. and P.K. participated in the conception and design of the article, as well as data collection, data analysis, and interpretation. Both authors also worked together to draft the article and performed critical revision of the article, and have both approved the final version of the article to be published.
Acknowledgment
We thank Tad Heppner for feedback on the article.
Disclosure Statement
No competing financial interests exist.
Funding Information
This work was funded by grants from Formas (2019–2096), a Swedish government research council for sustainable development and The Carl Trygger Foundation for Scientific Research (CTS 20:220).
