Abstract
Significance:
Lower extremity traumatic wounds are associated with numerous perioperative challenges. Their etiologies determine the characteristics and extent of the injury. The timing of subsequent surgical intervention and wound healing optimization after lower extremity trauma are integral to successful perioperative lower extremity wound management.
Recent Advances:
Managing trauma to the lower extremities uses a multidisciplinary surgical approach. The objective of this review is to summarize lower limb trauma assessment, advancements in lower extremity trauma management, and the clinical applications of advanced wound care in lower limb traumatic wounds. The advent of lower limb reconstruction and the development of advanced wound care modalities have helped to improve the management of these complex injuries.
Critical Issues:
The extensive involvement of bone, soft tissues, nerves, and blood vessels of severe lower extremity trauma wounds presents a challenge for clinicians in both the acute care setting and during patient rehabilitation. If not properly managed, these injuries may be subject to a decline in limb function and may possibly result in limb loss. To reveal developing limb-threatening conditions, serial examinations should be performed.
Future Directions:
The majority of lower limb traumatic wound will benefit from the perioperative administration of an appropriate negative pressure wound therapy (NPWT)-based system, which can help to promote granulation tissue and remove wound exudate before definitive closure and/or reconstruction. NPWT should be included as an important adjunct in the surgical management of lower limb traumatic wounds.
SCOPE and SIGNIFICANCE
Lower extremity trauma exerts a significant burden on patients during the recovery phase. More severe lower extremity trauma injuries, such as open fractures, are prone to contamination and possible limb loss if not appropriately managed. Navigating the involvement of multiple tissue types in these defects can present numerous challenges. Where applicable, various negative pressure wound therapy (NPWT) systems can help to address some of the perioperative challenges encountered. Our purpose is to summarize the assessment of lower limb trauma, the advancements in managing trauma to the lower extremities, the importance of multidisciplinary surgical management, and the clinical applications of NPWT systems in this setting.
TRANSLATIONAL RELEVANCE
Many traumatic wounds of the lower extremities are not amenable to primary closure at presentation. After the initial debridement and washout, other modalities such as the NPWT can be used perioperatively to facilitate granulation tissue development and to assist with closure. NPWT helps to form a closed system against external contaminants and prepare the wound bed for eventual coverage or closure postreconstructive surgery. This review provides an overview on the use of a variety of NPWT modalities in traumatic lower extremity wounds. Each NPWT modality can be incorporated as a temporizing dressing between initial debridement and final reconstruction, a system to deliver topical wound solutions for wound cleansing, a bolster for grafts or flaps to help facilitate wound coverage, or a closed incisional management system of wound margins.
CLINICAL RELEVANCE
Traumatic wound management of the lower limbs is multidisciplinary, and treatment is often multimodal. The versatility of the perioperative applications of NPWT is a valuable adjunct in the management of lower extremity trauma wounds.
INTRODUCTION
Lower extremity trauma poses a substantial burden on patients throughout their recovery beyond the acute management phase. 1,2 Although such injuries can be successfully managed, the significant involvement of the lower extremities in activities of daily living constrains those affected and has been determined to result in high health care costs and functional impacts, which are associated with work absence, decreased productivity, and impaired quality of life. 2,3 The spectrum of injury patterns can range from a simple skin laceration to a mangled limb injury affecting all functional aspects, including bone, soft tissues, nerves, and blood vessels. 4 Extensive involvement of these tissues is a challenging problem for clinicians in both the acute setting and the rehabilitation phase of treatment. 5 Optimizing wound healing following lower extremity trauma and subsequent surgical intervention is crucial for long-term success and multiple treatment techniques have been developed to reflect this.
Literature review
In April 2023, a literature search was executed using EMBASE and PubMed. The search strategy used the following terms and keywords: ([“lower extremity” OR “lower limb” OR “leg”] AND [“trauma” OR “fracture” OR “degloving”] AND [“wound healing”]). Inclusion criteria encompassed articles that were published in peer-reviewed journals, studies that discussed risk factors that might impact traumatic wound healing in the lower extremity injuries, preclinical studies that discussed scientific evidence of wound healing, and prospective and retrospective cohorts (randomized and nonrandomized). Literature subject to exclusion criteria were conference abstracts, study protocols, letters to the editor, economic studies, veterinary studies, preclinical studies (in vivo and in vitro), individual case reports, and non-English publications.
The literature search returned 2,318 results with 569 and 1,749 items returned by PubMed and EMBASE, respectively. Duplicates (n = 1558) were removed. Seven hundred and sixty items pertinent to the topic were then evaluated. Following a review of the article titles and abstracts, 618 items were subsequently excluded, and 142 pieces of literature underwent a full-text review. A total of 104 were later excluded after full-text review. A total of 38 articles were included.
Definition, epidemiology, and etiology
Lower extremity trauma is defined as an injury to the anatomical structures below the hip, involving at least the bone and soft tissue that increases in severity as nerves and blood vessels are implicated. 4 Studies investigating the epidemiology of lower extremity trauma have made clear distinctions on the anatomical structures that constitute the lower limb; these are the pelvis, hip, femur, patella, tibia or fibula, the ankle, and foot bones where the ankle is not included. 6
The etiology of lower limb injuries determines the nature and characteristics of the injury. The major mechanisms of lower extremity trauma can be classified as penetrating or blunt force. 7 Penetrating trauma is characterized by foreign objects that breach the skin causing damage to the soft tissue of the extremities and the body cavity, 8 whereas blunt force trauma is characterized by a force striking the body, which can result in further injury. These may include a contusion, abrasion, laceration, and/or fracture. 9 Depending on the amount of force applied to the body, the severity of blunt force trauma can escalate certain trauma types into a penetrating type of trauma as seen in cases of open fractures.
The epidemiology of lower extremity trauma provides further utility in establishing models of care for trauma patients. Wu et al. retrospectively reviewed all available disease and injury data from a range of data source types between 1990 and 2019 to identify the worldwide burden of fractures ranging from the skull to the bones of the foot. 10 In 2019, a total of 178 million new fractures were recorded globally, which resulted in a 33.4% increase in fracture prevalence and incidence since 1990. 10 Among the recorded cases, 78 million were noted as fractures of anatomical sites ranging from the hip to the tarsal bones with the most common anatomical area impacted being the patella, tibial shaft, or ankle group. 10
High-energy blunt force trauma results in open fractures and more severe lower extremity injury. However, the incidence of open fractures in general has experienced a downward trend. Court-Brown et al. calculated the incidence of open fractures as 30.7 cases per 10,000 in a year, based on 2386 cases over 15 years. 11 Epidemiological data also identified differences in the distribution of open fracture severity between anatomical regions. Data noted a greater percentage of open fracture severity, seen in the lower extremity (42.6%) compared with the upper extremity (18.6%). 6
In Australia, similar patterns of open fracture prevalence were observed where open fractures predominantly presented in the lower extremities. 12 In this population, motor vehicle accidents were the most common cause of injury (47.8%), followed by falls (39.7%) and other mechanisms (12.5%). 12 In addition to optimizing individualized management, understanding the epidemiology of trauma mechanisms provides additional information in developing management protocols and allocating resources for future service development planning. 12
History
In the pre-modern era, amputation was the mainstay of management for severe lower limb injuries such as those sustained on the battlefield. 13 The introduction and advancement of aseptic techniques and anesthesia resulted in safer and more successful operations. 13,14 Combined with advances in vascular reconstruction, antibiotics, and blood products, limb salvage has emerged as the new benchmark for lower limb trauma. 15
In the modern era, the most significant advancement in the management of lower limb trauma is the combined work of the British Association of Plastic, Reconstructive and Aesthetic Surgeons and the British Orthopedic Association. They have collaborated in recognition of the complexity of these injuries to provide guidelines and recommendations to minimize complications and optimize outcomes. 16 These guidelines emphasize the need for a specialist center capable of a multidisciplinary approach and appropriate surgical expertise when approaching significant lower limb trauma. 17 Management of lower extremity trauma is dependent on categorization of trauma and the extent of soft tissue involvement. 18 –20 Larger traumatic wounds may result in greater compromise of patient stability; however, this may also be dependent on patient demographics. 21 –23 As a rule, trauma management begins in accordance with the Advanced Trauma Life Support (ATLS) principles either in the prehospital or emergency department settings (Fig. 1). Timely referral and notification should be made to relevant surgical departments such as trauma, orthopedic, plastics, or vascular to ensure multidisciplinary management of the patient’s condition. 16,17
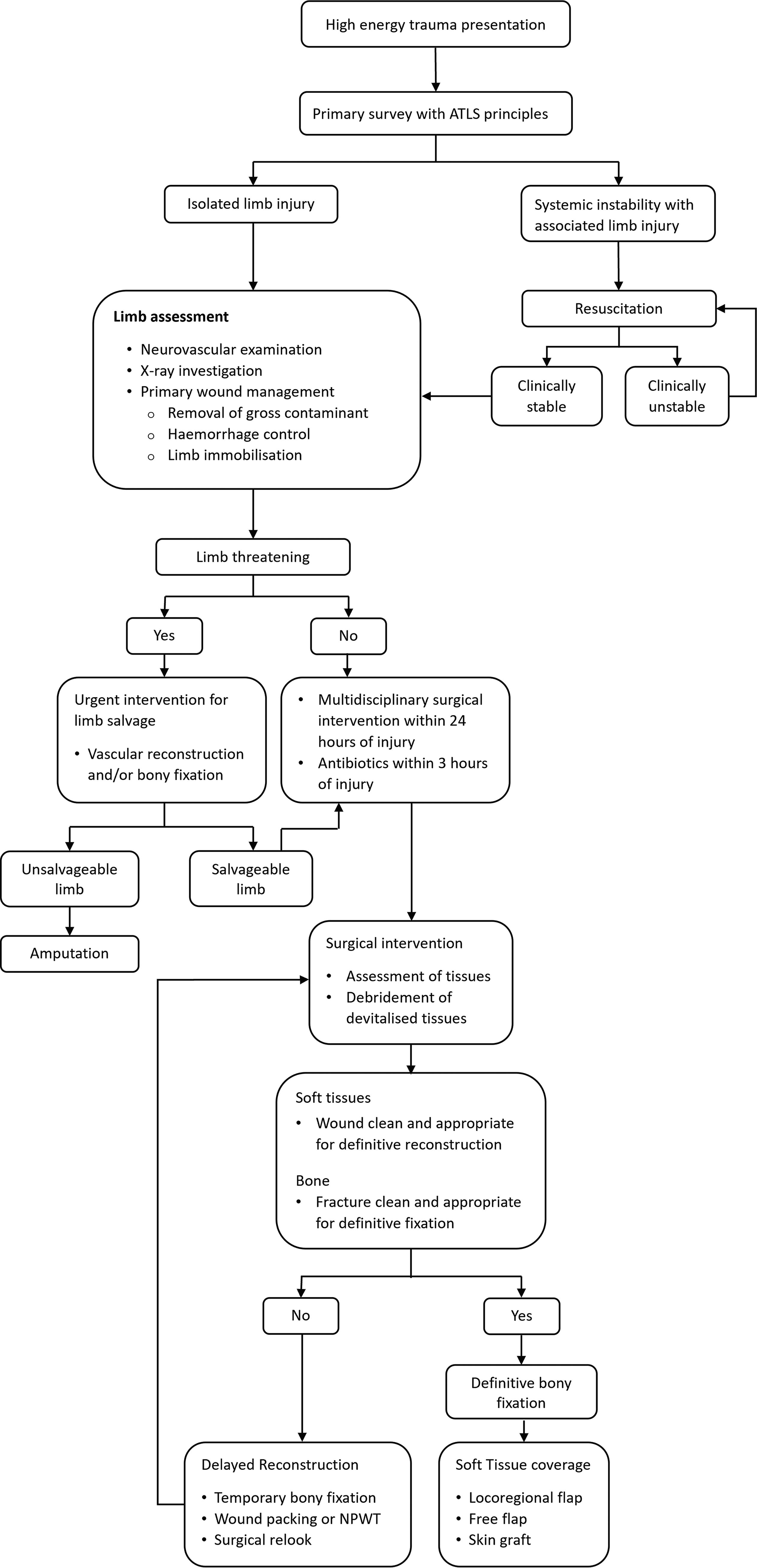
Management algorithm for high-energy trauma presentation.
LOWER LIMB RECONSTRUCTION
Lower limb injuries are often associated with soft tissue compromise requiring reconstruction. Different techniques are available for wound closure depending on the type of lower limb defect. 24 Following debridement, a small and superficial wound can be healed by secondary intention if there is no neurovascular bundle or bone exposure. This avoids an additional surgical intervention but can be a time-consuming process for the patient with recovery extending over weeks to months. Where possible, primary closure is ideal for the shortest recovery period, but it is only applicable to wounds where the wound edges can be approximated in a tension-free manner. 24
Most lower limb traumatic wounds are not amenable to primary closure either because of the extent of injury or the cylindrical nature of a limb and insufficient tissue laxity. In this setting, it is important to assess the wound bed and determine the structures that lie at its base. Wounds with a vascularized wound bed can support reconstruction with a skin graft from a distant donor site. 25 Lower limb defects with an ungraftable wound bed often consist of bare bone or tendon, and exposed vasculature or metalware, and require more robust reconstructive options. Locoregional flaps involve the transfer of tissue and inset into the injury site while retaining its vascular attachment. Careful consideration is required when utilizing this technique in lower limb trauma as the zone of injury may include adjacent tissues with subsequent degloving of superficial tissues from the deep fascia. 26,27 When this occurs, perforators to surrounding skin may be disrupted rendering locoregional flap options less reliable. Free flap surgery is often indicated for significant lower limb trauma involving large soft tissue deficits with exposure of bone, vessels, and metalware. Tissue is harvested from a site distant from the site of injury with a vascular pedicle, and the pedicle is anastomosed with one of the recipient vessels in the lower limb to provide soft tissue coverage. 28 Wounds that cannot be primarily closed have historically been packed with surgical gauze alone to provide initial wound coverage and continues to be used today because of its availability and surgeon preference. 29 However, removal and changing are often a painful challenge for the patient as the dressing becomes adherent to the wound bed and can mechanically debride the tissues if not appropriately soaked first. 30 It also does not provide appropriate sterile cover to the wound. 31
NEGATIVE PRESSURE WOUND THERAPY
Advancements in wound care have resulted in the development of a subatmospheric wound dressing system, known as NPWT, that can play a role in perioperative and postoperative wound care. NPWT was introduced in 1997 by Argenta and Morykwas who detailed techniques on the use of a reticulated open-cell polyurethane foam dressing attached to a canister and vacuum pump on acute, subacute, and chronic wounds. 32 NPWT has become an alternate surgical dressing particularly useful in large cavities with high exudate production and provides temporizing coverage between initial debridement and final reconstruction. It can help to prepare the wound bed through the promotion of granulation tissue development and removal of wound exudate while providing a sealed environment to minimize the risk of external contamination. 33 Orhan and Erdogan reported that NPWT use in high-energy extremity trauma can facilitate wound closure and temporization to ensure success of procedures lower on the reconstructive ladder. 34 Of the 48 lower extremity wounds included in this study, 91.6% of the wounds were able to be reconstructed with skin grafts alone. 34 Furthermore, the study found a moderate positive association between defect wound size and the required number of NPWT sessions to reach adequate wound healing.
Foam dressings composed of either polyurethane or polyvinyl alcohol represent the most popular wound fillers used during commercially available NPWT. However, the use of gauze in NPWT as an alternative filler material to the more common reticulated open-cell foam (ROCF) dressing has also been well documented. 35 –37 Gauze-based NPWT may use either saline-moistened antimicrobial gauze or silicone-covered gauze as the wound interface material and has been characterized in the Chariker–Jeter application of subatmospheric pressure (−80 mmHg) in wound management. 38 Using gauze opposed to a foam dressing may have putative advantages such as the avoidance of granulation tissue ingrowth and reduced pain as has been observed during some foam dressing changes, reported ease of application, reduced force required upon removal, and the moldability of the gauze when filling complex irregularly shaped wounds. However, discomfort supposedly associated with tissue ingrowth within an ROCF dressing might be avoided with regular dressing changes (48–72 h) as per the instructions for use. Aside from a morphological difference in granulation tissue quality, with reports of thinner yet denser granulation tissue yielded from gauze-based NPWT, the clinical outcomes from using gauze-based NPWT are generally favorable and putatively similar to foam dressing use. 39
Although some literature has considered gauze dressing-based NPWT to be noninferior to NPWT using ROCF dressing, the putative advantages of NPWT using ROCF dressings over gauze-based NPWT may appear at the cellular level opposed to the outward clinical outcome. With ROCF dressing use during NPWT, there might be more than the eyeball test as evidenced by multiple bench studies. McNulty et al. conducted a series of in vitro wound studies investigating the effects NPWT paired with either ROCF or gauze-based dressings on human fibroblast cells grown in a three-dimensional (3D) porcine fibrin matrix. It was reported that relative to NPWT using ROCF dressing and static controls, human fibroblasts treated with gauze under suction exhibited less cellular migration resultant of chemotaxis and proliferation as well as statistically significant greater cell death. 40 In a subsequent investigation, it was noted that levels of bioenergetic markers in cellular metabolism such as cytochrome c oxidase, adenosine triphosphate/adenosine diphosphate, and energy charge were significantly elevated subsequent to NPWT using ROCF dressing. In addition, when using NPWT with ROCF dressing, levels of transforming growth factor-β (TGF-β) and isoforms of platelet-derived growth factor (80% vs. 53%, p < 0.05) were also increased over static control cultures but not after gauze under suction. 41
The more favorable effects of NPWT using ROCF dressing in cell culture led to an inquiry of global gene expression profiles in a TGF-β rat model. In this study, Derrick et al. used a whole-genome microarray survey for three different wound management modalities as follows: NPWT, moist wound healing, and gauze dressing under suction. In those animals treated with NPWT using ROCF, the pathways integral for inflammation, angiogenesis, and cytoskeletal regulation were associated with elevated gene expression. More specifically, 5,072 genes demonstrated greater than 1.6-fold change with NPWT relative to 3,601 genes and 3,952 genes for moist wound healing and gauze under suction, respectively. 42
The isolation of the hermetically sealed wound while under NPWT using ROCF dressing presented a challenge to assessing microdeformations at the interface of the ROCF dressing and the wound bed. However, a bench-test wound model for measuring the 3D strain field at submillimeter resolution for microcomputed tomography imaging of NPWT-induced microdeformations was developed. 43 Building on the earlier Wilkes study, another computational study measuring 3D microdeformations in tissue during the application of subatmospheric pressure was quantified using a 3D finite element model. As per the computational model, the ROCF dressing generated greater strain than gauze at all the subatmospheric pressure levels tested, which was statistically significant. Higher levels of microdeformation were noted in NPWT using ROCF dressing in contrast to gauze under suction. 44 The strain predictions reported were similar to the predicted findings from a prior study by Saxena et al., 45 which aimed to explain the mechanobiology of microdeformations via NPWT using ROCF dressings in granulation tissue regulation using a two-dimensional finite element model and computational studies.
It should be noted that the aforementioned results of bench investigations may help to inform or advance our understanding of how tissues may respond to NPWT using ROCF dressing, but they may not necessarily recapitulate what occurs in the context of human physiology. Further studies would be required to ascertain whether these findings at the bench or those that are derived in silico are actually representative, similar, or even disparate within human wounds.
NPWT with instillation and dwell time (NPWTi-d) is another development in NPWT and involves introduction of a topical wound solution to the wound bed while maintaining a closed vacuum system. 46 Prototypes of this technique can be attributed to Fleischmann et al. 47 who described a manual method of combining solution instillation (a syringe or an infusion system) with a vacuum suction system. Current NPWTi-d systems comprise the modernized NPWT devices that possess the technology to automatically administer and drain a preset volume of topical wound solution. This allows for the instillation of an appropriate topical wound solution to the wound bed, which varies depending on the clinical context. The type of instilled solutions may include a topical antiseptic solution to the wound bed or local anesthesia for pain relief particularly in the setting of an imminent dressing change. 46 In infected and chronic wounds, topical antiseptic solutions can be used and have produced satisfying results in the literature. 48,49 Saline instillations can also be applied to the wound with the goal to wash out the wound. Expert consensus recommends that the wound solution should be left to dwell in the wound bed for 10 min before draining. 46 Once the topical wound solution has attained the programmed dwell time within the wound bed, then the NPWT system facilitates aspiration of the topical wound solution to be from the wound bed environment and allowing subsequent cycles of the topical wound solution to be administered as needed. This process allows for the general cleansing of the wound bed.
Within the last decade, new advancements in NPWT have resulted in the development of battery-powered portable devices that have contributed to the breadth of available wound management options. Portable NPWT units allow the application of negative pressure therapy in a variety of patient settings and were pioneered with the aim of reducing inpatient wound care and early transition to home. 50 These devices have been designed to be smaller, lighter, and more user-friendly, allowing patients to independently continue their negative pressure therapy regimens in the outpatient setting. A consideration of the device’s smaller sizes is that they have a reduced ability to collect exudate, which may influence the type of wounds these devices can be used on. Another consideration of these devices is that they are often a single disposable unit that restricts them to a single use due to a preset lifespan determined by the manufacturers.
Initial experiences with the 3M™ V.A.C.® via Negative Pressure Wound Therapy System (Solventum Corporation, Maplewood, MN, USA) portable device allowed for early patient mobilization and early transition to home for further recovery. The reduction in the length of patient stay did not compromise the effectiveness of wound healing, suggested by the adequate granulation tissue growth and complete wound closure reported. 51 Similarly, Canonico et al. reported encouraging wound healing results with the application of a novel mobile NPWT device developed by Smith & Nephew Srl (Agrate Brianza, Milan, Italy) in 2012. 52 However, owing to the device’s portable nature and limited lifespan, clinical discretion needs to be made to ensure the device is used on appropriate wounds. Wounds that may benefit the most from portable NPWT are the ones that have been covered by a graft or a flap, which are also low exudative, which is defined as <80 mL/day, and are small-to-medium sized. 51 These wound characteristics are subject to change depending on individual specifications of the portable NPWT device in question.
Furthermore, the success of the electronic portable NPWT systems can also be attributed to its ease of operation. The 3M V.A.C. via Negative Pressure Wound Therapy System (Solventum Corporation) was designed with a one-discreet button operation that made operation easy for caregivers and patients. 51 Patients also reported that the application of the suction to the wound bed by the portable device was comfortable. Although patients reported ease in operation of the portable NPWT device, the researchers reported that some patients did require to return to clinics due to inexperience with dressing changes. Overall, the studies reported that portable NPWT devices were safe to use in the outpatient setting and resulted in positive wound outcomes.
The reported success in wound healing, the simple design, and the ease of operation of the portable NPWT devices resulted in an increase in the number of systems available on the market. Currently several battery-powered portable systems have been reported widely in the literature and have further dichotomized into either a canister-based or canister-free design. Smith+Nephew has developed a portable device called the PICO™ single-use negative pressure wound therapy that has a growing body of evidence suggesting that its use helps improve healing rates of chronic wound types and reduces the associated wound care burdens (cost and nursing time) when compared with standard care. 53,54 The Avelle™ NPWT Pump system (ConvaTec, Deeside, UK) is another portable system that utilizes a canister-free NPWT design. The canister-free design can be attributed to a proprietary dressing derived from 100% sodium carboxymethylcellulose (Hydrofiber®) that is able to autolyze debrided wound tissue and facilitate evaporation of wound exudate. 55 Another recent addition to the portable NPWT arsenal is the Avance® Solo NPWT System (Mölnlycke Health Care AB, Gothenburg, Sweden), a canister-based portable device that has been found to be suitable for use in acute and home care settings. 56 Table 1 summarizes the commercially available battery-powered portable NPWT products currently on the market.
Commercially available portable negative pressure wound therapy devices
NPWT, negative pressure wound therapy.
More accessible models of NPWT have been developed in the form of mechanically powered portable NPWT devices. 68,69 These devices consist of a cartridge with an activation/reset key and a dressing unit with an integrated tubing that covers the wound bed. The cartridge acts as the source of a preset, single level of negative pressure and doubles as a container to collect any exudate from the wound bed. It is a small and lightweight device that is mechanically powered to exert the negative pressure. As a result, there are clinical limitations to the utility of these mechanically powered portable devices. Tettelbach et al. recommend that these devices be suitable for smaller wounds and with little exudate. 68 The mechanical nature of the disposable NPWT (dNPWT) also restricts the ability to apply a range of negative pressures. Some devices readily on the market include the 3M™ Nanova™ Negative Pressure Wound Therapy and 3M™ Snap™ Negative Pressure Wound Therapy System, which are two mechanically powered dNPWT devices. 69,70 These systems comprise a pump and spring mechanism that generates a preset, continuous negative pressure environment to the wound bed.
There are several beneficial outcomes associated with NPWT use. NPWT has been demonstrated to promote granulation tissue formation and to help with wound healing. Morykwas et al. observed a significantly high increase in new granulation tissue following NPWT use. 71 One of the most strongly cited reasons for NPWT is the improvement in tissue perfusion. Morykwas et al. were the first to describe NPWT use in North America in 1997. 32 In a series of animal studies, Doppler flowmetry measured blood flow in subcutaneous tissue and muscle during periods of intermittent and continuous suction. Perfusion at different strengths of suction was also investigated. Morykwas’ article is the source of the recommended −125 mmHg for optimal tissue perfusion as a fourfold increase in perfusion was observed during periods of negative pressure compared with the baseline where negative pressure was not used. 71 This circulatory improvement has since been replicated on humans by Wackenfors et al. and Timmer et al. Laser–Doppler flowmetry was utilized to measure tissue perfusion during NPWT use. 72,73 An improvement in microcirculation blood flow was observed with maximal improvement during intermittent therapy.
These changes were further investigated on a histological level by Chen et al. who applied NPWT to Japanese rabbit ear wounds with the contralateral ear as a control. 74 Tissue from the wound edges were serially harvested. Following 30 min of NPWT, capillary bud formation was observed; and after 24 h, capillary caliber and density were near normal. In comparison, no capillary buds were observed in the control group at 24 h and capillary morphology was still irregular and stenosed. 74 More recently, Liu et al. concluded that NPWT use can help to improve microvessel density in chronic, refractory wounds by influencing the expression of regulation of angiogenic mediators. 75 On an immunohistochemical level, Walgenbach et al. investigated the degree of angiogenesis by analyzing endothelial cell component antibodies in tissue samples of patients after NPWT, which revealed an increased proliferation of endothelial cells in the newly formed granulation tissue. 76
Nonconventional NPWT systems have been developed in response to cost barriers associated with conventional NPWT devices. Gupta et al. reported that a nonconventional NPWT system comprised a nonproprietary sterile polyurethane foam, adhesive drape, and suction tube that was connected to a generic wall-mounted vacuum suction pump that delivered −125 mmHg of pressure. 77 Application of this nonconventional NPWT device resulted in statistically significant improvements in granulation tissue cover, reducing exudation and the necrotic tissue profile.
Despite the benefits of NPWT, adoption of its use should be judicious and targeted to appropriate patients and wound types. Bleeding is a significant complication of NPWT use and wounds should be assessed for any bleeding potential before foam application. An unrecognized vascular injury can lead to large-volume blood loss into the NPWT apparatus, which can be missed by clinical staff if the exudate canister is not directly visualized. In addition, anticoagulated patients can also experience hemorrhage from the wound bed, which may lead to life-threatening outcomes. 78
Staff training is required to ensure safe and effective utilization of NPWT at the time of intraoperative application and ongoing management. 79,80 This training is essential to mitigate potential problems such as retained foam as granulation ingrowth can become embedded into the foam and small pieces can remain in the wound bed when removing or changing the dressing. In deep and undermined wounds, this may be unrecognized due to poor visualization and these foam remnants can become foci for infection. 78
Technical proficiency is also necessary to troubleshoot any device malfunctions. Loss of power to the machine apparatus from battery depletion or disconnection from the primary power source can lead to loss of vacuum pressure. 81 A blockage within the foam or tubing can also result in loss of suction and unintentional interruption to therapy, whereas loss of a seal renders any negative pressure delivery futile. Loss of a seal often occurs because of inadequate application of the adhesive tape over the foam and surrounding tissues. Application of the seal can be particularly difficult if navigating around tricky topography such as limb flexures, soft tissue creases, and external fixation pin sites. 82,83 Securely sealed edges may need to be reinforced between formal dressing changes as patient movement may inadvertently cause the adhesive tape to lift.
Audiovisual alarms exist within NPWT machines as a safety feature to alert when negative pressure delivery has been interrupted. Despite this, it is important to educate patients where possible on device malfunctions, which can be escalated to clinical staff. 80 This can be achieved by recognizing system alarms or individual awareness of tactile suction loss. Checking appropriate NPWT machine function is integrated in protocolized use in the clinical setting. In both inpatient and outpatient environments, clinical staff are required to assess the NPWT dressing at regular intervals. This is a resource-intensive task, which may deter widespread adoption of this technology and the benefits of NPWT use must be weighed against resource limitations.
Clinical applications of NPWT in lower limb injuries
The role of NPWT in lower limb trauma reflects the nature of the injury and eventual reconstruction. In the acute traumatic setting, NPWT acts as a temporizing dressing following initial debridement. 33 In comparison with traditional dressing options, it provides a sealed environment against further contamination while facilitating granulation and local perfusion. Large defects and traumatized local tissue produce significant exudate as part of the inflammatory response and NPWT can help minimize retention of fluids within the wound bed. 84 Similarly, traumatic mechanisms can lead to significant degloving injuries with an accumulation of blood and extracellular fluid between the subcutaneous tissue and deep fascia. Following debridement of any devitalized overlying tissue, a large cavity can be revealed, and reduction of this dead space can be achieved with NPWT. 85 Care should be taken to adequately protect any vasculature exposed from the initial injury or debridement as direct contact with these structures by the foam can result in inadvertent injury.
NPWT has also become a main component of wound management following limb fasciotomy. Lower limb fasciotomy involves release of all compartments of the leg via a lateral and medial incision. This is either therapeutically or prophylactically performed to reduce pressure in the compartments, which can lead to muscle necrosis. By utilizing NPWT following fasciotomy, large open wounds can remain clean and deep tissues are protected from external contamination. 86 The negative pressure exerted on the wound edges can counteract the skin’s natural tendency to retract following an incision, which can be exacerbated by post-traumatic edema causing muscle to swell and separate the wound edges further. 87 The antiedema benefits of NPWT aid in reducing leg swelling and increase the likelihood of approximating the wound edges and directly closing the fasciotomy wounds. 88
Soft tissue loss of the lower limb can often be the only evidence of injury. The extent of integumental loss can range from extensive to minimal following debridement of devitalized tissues and may not be amenable to primary closure. In these settings, NPWT can optimize the wound bed and aid in reconstructive recovery. 89 A split-thickness skin graft is a commonly used reconstructive option for cutaneous defects. It involves harvesting the whole epidermis and variable dermis depth of uninjured skin to provide coverage of the defect. The graft can be expanded via a meshing process, which increases graft surface area at a variety of ratios depending on the meshed apertures of the graft. Graft loss can be attributed to loss of contact between the graft and wound bed, which can occur due to the accumulation of fluid or blood, infection, shearing forces, and inadequate wound bed vascularity. Granulation tissue formation and tissue perfusion are known outcomes of NPWT that optimize the wound bed for a graft. However, NPWT can be used to bolster a graft. 90 The negative pressure capacity of NPWT allows for continuous drainage of wound fluid and reduces the risk of a seroma or hematoma forming beneath the graft. 91 Application of the foam dressing should include a nonadherent interface between the foam skin grafts to provide a layer of protection. Even distribution of pressure through the foam dressing provides uniform contact of the dressing and the graft onto the wound bed. This is possible despite any contour irregularities of the wound bed due to either the nature of the wound bed itself or due to any challenging anatomical regions such as the foot or ankle. 92 The sealed nature of the NPWT system also provides a consistently moist environment that helps prevent graft desiccation and loss. 93 A recent adjunct to skin graft reconstruction is the advent of temporizing matrices. These are man-made synthetic polymers that form a bioabsorbable matrix and an overlying sealing membrane. 94 The matrix is designed to minimize scarring and contracture by providing a scaffold for cellular migration to form neo-dermis. Upon application of the product to the wound bed, a suitable outer dressing is required that will not adhere to the matrix, absorbs exudate, and encourages contact with the wound bed. NPWT is an ideal dressing option that fulfils the proposed criteria and has been utilized to help support the integration of dermal substitutes. 95
The use of NPWT following lower extremity free flap reconstruction for traumatic wounds has recently gained traction. Free flap venous congestion is a known cause of flap failure and the use of NPWT has been documented to optimize previously compromised or threatened flaps. Qiu et al. applied NPWT to 12 venously congested pedicled or free flap reconstructions with flap survival in all cases documented. 96 A similar resolution of venous compromise was observed by Vaienti et al. who applied NPWT to three congested pedicled flaps and one free flap. 97 This clinical improvement is thought to be attributable to NPWT’s ability to promote blood flow and venous return. These therapeutic properties of NPWT have resulted in its application in the immediate postoperative phase to help reduce complication rates. Chen et al. successfully described the application of NPWT dressings to a fasciocutaneous flap inset while leaving an opening to avoid compression of the pedicle. 98 This design allows for flap monitoring and minimizes the risk of pedicle compromise from the vacuum pressure. The use of NPWT in flap reconstructions is likely safe and reliable for both flap optimization and as a primary dressing at the time of flap inset; however, further investigation is required to understand the full implication of negative pressure therapy after flap reconstruction.
Application of a closed-incision negative pressure therapy (ciNPT) adopts the properties of traditional NPWT from an open wound bed to a closed surgical incision. While the clinical use of NPWT revolves around wound optimization following trauma or injury, ciNPT may prophylactically help in the reduction of some postsurgical complications. 99 Foam is placed directly onto an incision line and an adhesive drape is applied to seal the foam in place with delivery of negative pressure via the foam to the skin. Recognition that ciNPT provides a closed clean environment while allowing drainage of exudate through a sutured or stapled incision led to the development of the ciNPT as an incision management system. 100 This system was designed to provide continuous negative pressure to directly closed wound edges providing additional external support and reducing tension along the incision line. 101 The dressing includes a silver layer at the skin interface to counteract microbial colonization in the fabric, while the occlusive drape acts as a physical barrier to external contamination. 102 ciNPT has been shown to reduce the incidence of wound infections and seroma formation when compared with nonsuction postoperative dressings. 103 Use of ciNPT in lower limb wounds following orthopedic and vascular surgeries is associated with reduced incidence of incision complications and the need for revision procedures. 104 The antiedema properties of NPWT can also be adapted for prophylactic purposes. Recently, next-generation ciNPT that expands the area of foam coverage to include intact skin beyond the incision line has been developed. The larger surface area of negative pressure therapy bolsters incisions and the surrounding soft tissue. 105 The application of this dressing to the lower limb has wide-ranging possibilities for the management of soft tissues following lower limb surgery.
Representative cases
Case 1—NPWT use as an adjuvant modality in the management of a traumatic acute wound
One week after a mechanical fall, a 72-year-old female presented with an infected traumatic hematoma overlying her right ankle lateral malleolus which was unrecognized at the time of injury and subsequently developed an area of skin necrosis and surrounding cellulitis. This was on a background of peripheral vascular disease and atrial fibrillation for which she was prescribed anticoagulant medication. The patient underwent initial debridement of necrotic skin to evacuate the hematoma and for tissue sampling to guide antimicrobial management. The resulting defect measured 3 × 5 cm2 with intact periosteum at the wound base and was regularly dressed using Betadine-soaked gauze. On resolution of the infection and confirmation with repeated negative wound cultures, NPWT was applied to encourage wound margin contraction and promote granulation tissue. Hydrocolloid dressing was applied to the surrounding area as an added layer of protection for the patient’s mature fragile skin (Fig. 2A).
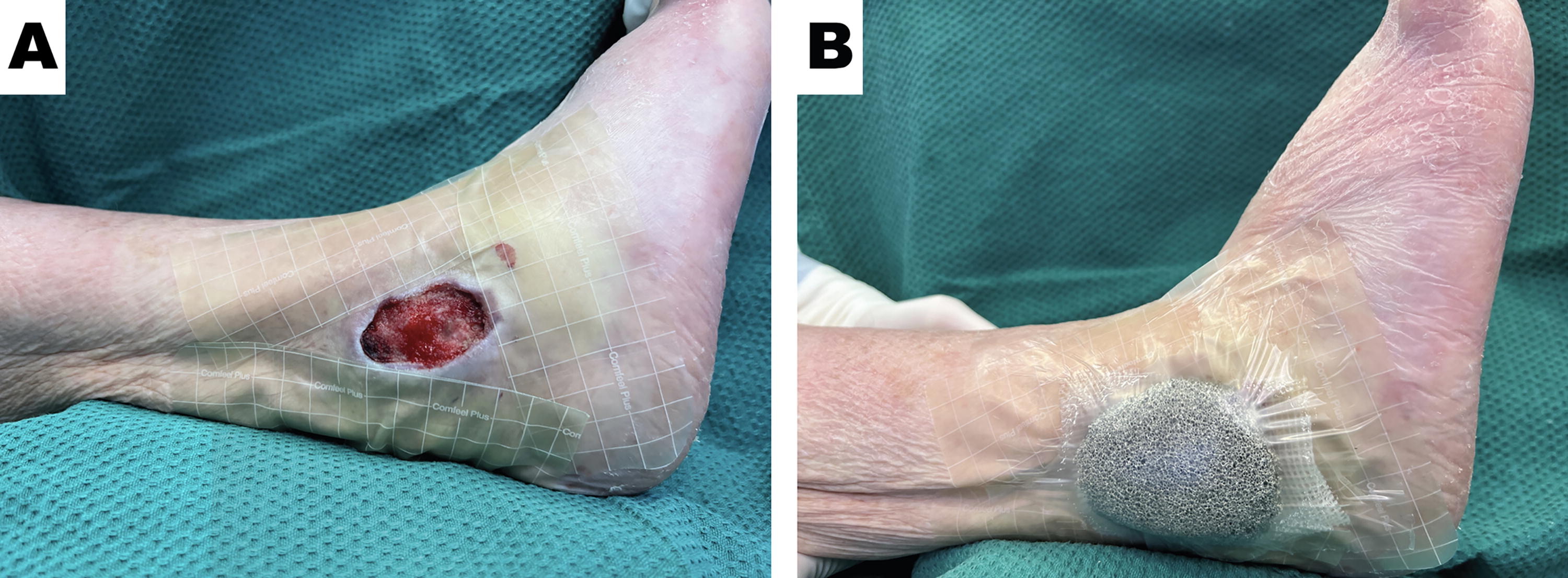
NPWT use as an adjuvant modality in the management of a traumatic acute wound. (
Care was taken to ensure that the occlusive drape for NPWT was in contact with the hydrocolloid dressing rather than the patient’s skin (Fig. 2B). The patient tolerated regular NPWT dressing changes without anesthesia and was discharged home with nursing support to facilitate regular dressing changes. Despite poor limb vascularity, the wound dimensions were reduced and the wound bed was occupied with healthy granulation tissue following NPWT. The patient ultimately underwent reconstruction with a protected skin graft that was bolstered using NPWT. Completed graft take was achieved and the patient’s wound fully healed.
Case 2—NPWT use as a temporizing dressing to manage a high-energy open fracture
A 42-year-old male sustained an open left tibia fracture following a high-speed motor vehicular accident. Clinically, the limb was perfused distally; however, sensory loss to the dorsum of the foot and limited ankle dorsiflexion were noted. This injury was associated with rib fractures and a traumatic pneumothorax, which was addressed in the acute setting as per ATLS procedures. The patient was brought to the operating theater as a combined ortho-plastic case for wound debridement and tissue assessment. Devitalized, contaminated, soft tissue was debrided intraoperatively, and the fracture site was exposed. Surgical exploration confirmed an intact vascular supply to the foot, a contused but intact tibial nerve, and injury to the anterior compartment of the leg muscles. The patient underwent temporary bony external fixation. NPWT was applied as a temporizing dressing to the soft tissue defect. When applying the occlusive adhesive drape, pin site location added a layer of complexity. However, re-enforcement of the drape edges was required to form an appropriate seal. The patient returned to the operating theater after 48 h for further review of the soft tissues. An area of previously identified degloved tissue was further debrided for an integumental loss totaling 2% of the total body surface area (Fig. 3A).
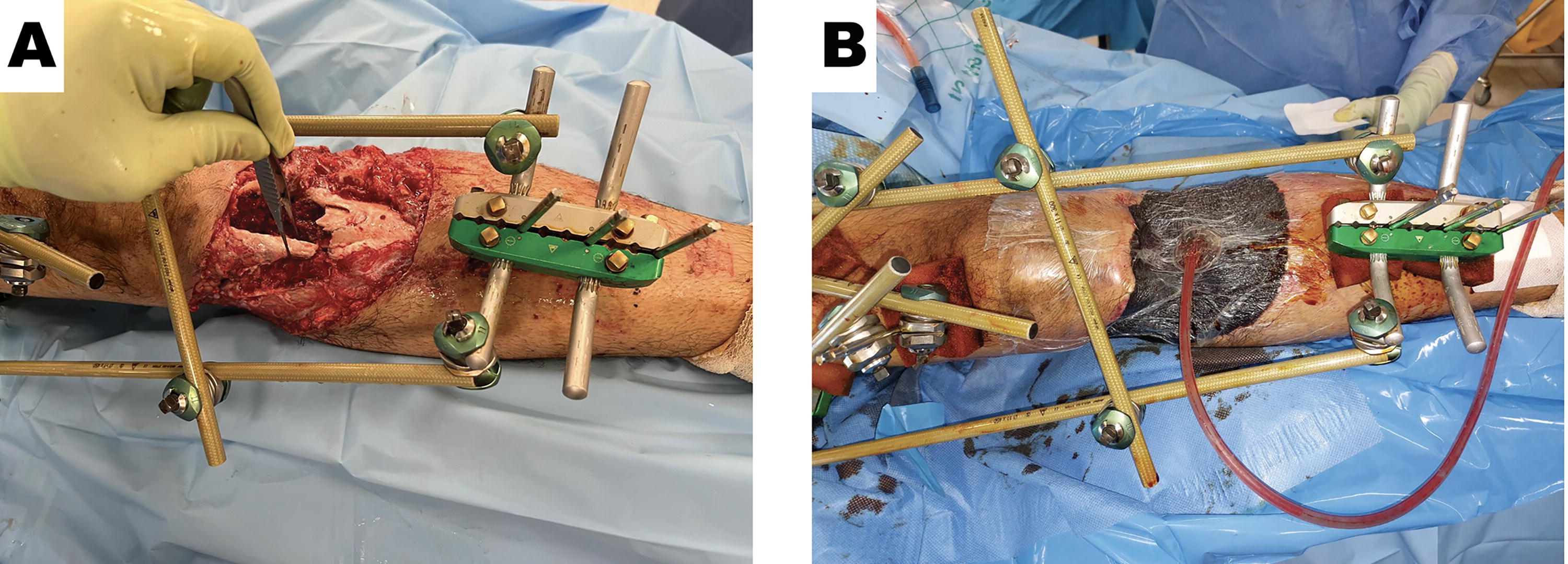
NPWT use as a temporizing dressing to manage a high-energy open fracture. (
Again, NPWT was applied as a temporizing dressing and helped to minimize the risk of exposure against external environmental contaminants and remove wound exudate (Fig. 3B).
NPWT use allowed time for combined discussions between the orthopedic and plastic teams to formulate a reconstructive plan for the limb. Any definitive bony fixation would result in exposure of both bone and metalware, necessitating free flap coverage of the defect. These complex and highly skilled operations required appropriate planning and resource allocation. In this patient, NPWT use was beneficial not just for local wound management, but it also allowed the surgical teams to formulate and successfully perform reconstructive surgery.
Case 3—NPWT use to encourage healing by secondary intention in an adversarial field
A 56-year-old female was previously diagnosed with a soft tissue sarcoma of her right leg following investigation of a rapidly growing subcutaneous growth. She underwent 5 weeks of neoadjuvant radiotherapy totaling 50 Gray (Gy) delivered in 2 Gy fractions daily. Four weeks after the completion of radiotherapy treatment, oncological resection was performed. Wide surgical margins were required to help minimize the risk of local recurrence while balancing the possibility of facilitating limb salvage. The tumor was excised with the tibia periosteum margin to provide adequate local control but resulted in exposed cortical bone at the base of the surgical site. A free, anterolateral thigh (ALT) flap was raised on the contralateral limb to provide a large area of soft tissue coverage. Soft tissue transfer was initially successful; however, tissue necrosis of native skin at the ALT inset was observed 2 weeks after surgery (Fig. 4A).
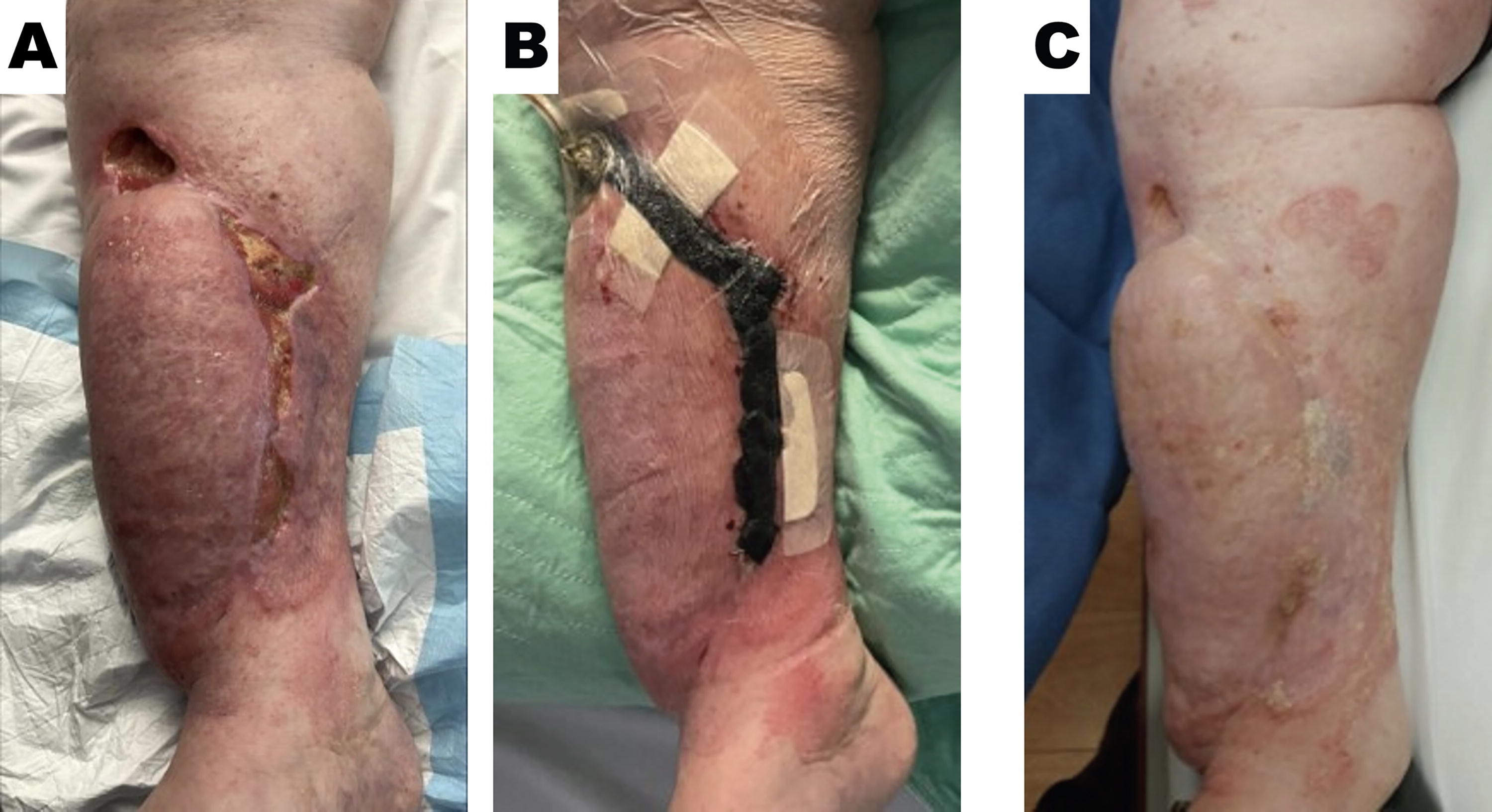
NPWT use to encourage healing by secondary intention in an adversarial field. (
Previously irradiated tissues had an increased risk of wound complications and poor healing following induced ischemia and fibrosis. NPWT use was recruited in this setting and applied to the area of skin loss to promote perfusion and granulation tissue generation (Fig. 4B).
Continued use of NPWT helped to enable complete wound healing by secondary intention despite poor tissue perfusion. In this patient, there was no need to generate a donor wound for skin grafting (Fig. 4C).
Case 4—NPWT use in traumatic wound management and wound bed optimization before grafting
A 25-year-old male sustained a degloving injury of his right plantar foot following a low-speed, motor bike accident. The plantar surface of his foot was completely undermined with longitudinal lacerations to the medial and lateral aspect of the sole resulting in a through-and-through defect. Plantar skin remained perfused via its proximal and distal attachments (Fig. 5A).
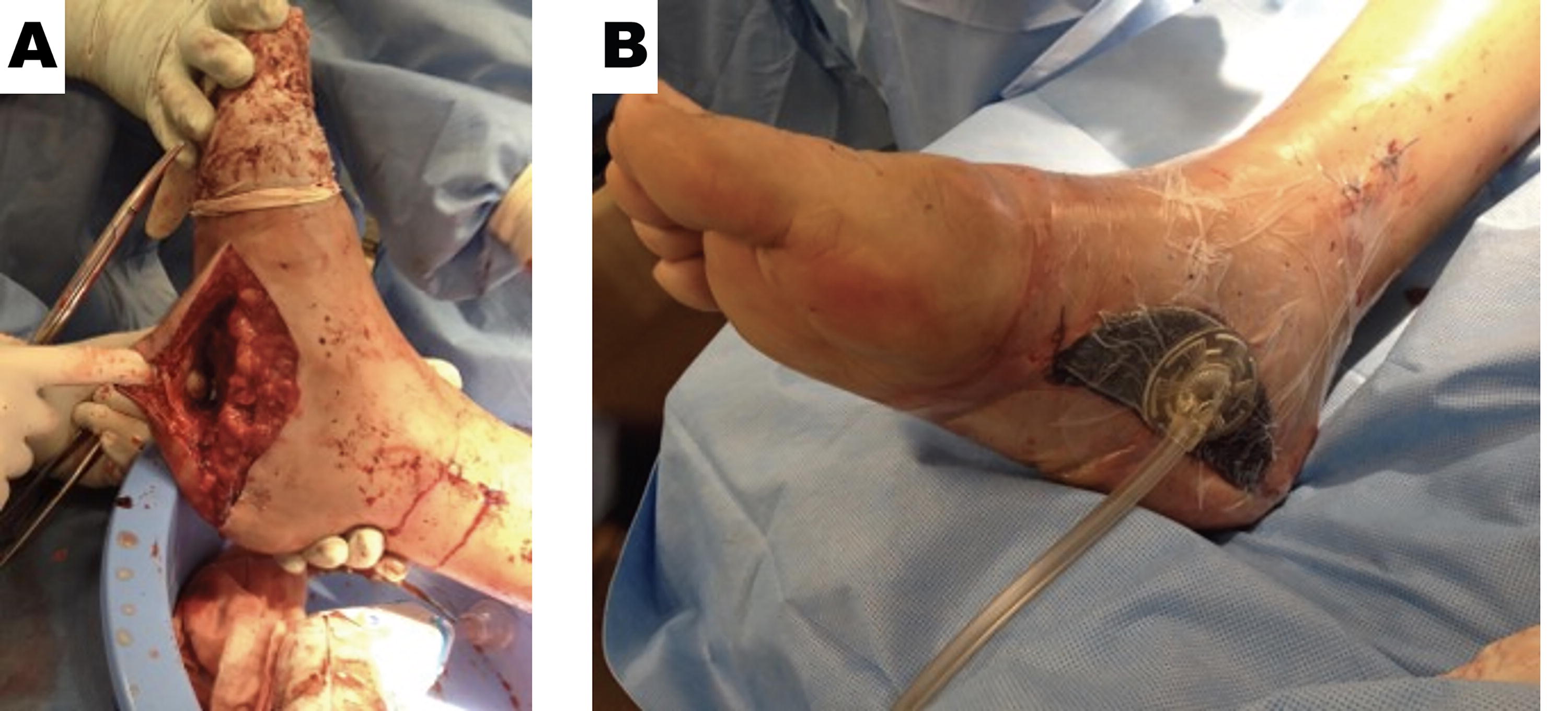
NPWT use in traumatic wound management and wound bed optimization prior to grafting. (
NPWT was used to first close the dead space formed by the overlying degloved tissue and to remove any exudate that could accumulate in the area. The lateral laceration was closed directly. Debridement of devitalized tissue resulted in a soft tissue defect on the medial aspect of the foot. NPWT use on this wound helped to promote granulation tissue formation (Fig. 5B).
Following NPWT, plantar subcutaneous tissue was able to adhere to the sole of the foot and the wound was optimized for a skin graft. This patient was able to achieve a full recovery from a wound healing and stable weight bearing perspective.
LIMITATIONS
There are some considerations surrounding the methodology and current state of negative pressure therapy research that contribute toward knowledge gaps in this critical review. The conclusions made regarding NPWT device use have been based on articles published and investigated in developed nations, which overall have less complex lower limb trauma presentations and easier access to advanced NPWT devices when compared with developing regions. Therefore, the conclusions of the review are subjected to a geographical bias, which influences insights and reported effectiveness of NPWT devices that have been collated. There is also a lack of comparative studies investigating the use of traditional NPWT devices to other nonconventional modalities of negative pressure therapy. Specific insights and advances into the nonconventional modalities of negative pressure therapy for wound management may have not been considered due the exclusion of non-English published articles and nonpeer-reviewed individual case reports/articles. As a result, these considerations contribute toward a gap in the NPWT literature. Pioneering cross-geographical comparative studies or considering a wider inclusion criterion may contribute to producing a more robust critical review of NPWT in the clinical context of managing lower limb trauma wounds.
The methodology of the critical review poses a limitation on the future relevance of our conclusions. The scope of the review can only cover the available evidence of the development of NPWT in wound management from its conception to its use in the clinical context of lower limb trauma in this point of time. Future developments in trauma wound care may render the techniques addressed in this review to be obsolete or potentially inferior to new adjuvant wound therapies. A final consideration is the large number of NPWT devices available on the market posing a difficulty in producing a comprehensive review on the nuances each device provides. Each iteration of the NPWT device has its own variation of dressing material, vacuum suctioning ability, and overall design, which can contribute to the effectiveness of the device in question. There are a multitude of studies that have been conducted to display the viability of each NPWT device and ensuring all the literature was covered in this review would be difficult, if not impossible. This large pool of NPWT devices also contributes to developing bias of the conclusions within this critical review. This is due to the lack of distinction in the NPWT devices used within the studies included.
CONCLUSION
Lower limb trauma consists of a broad spectrum of mechanisms and resulting injury pattern. Patients with varying degrees of bony, soft tissue and vascular compromise require an assessment and management tailored to their extent of injury. Challenges arise depending on the complexity of the wound and every effort is made to optimize wound healing. Particularly as amputation is no longer the primary form of lower limb surgical management, successful limb salvage entails a clean wound, bony stabilization, and adequate soft tissue coverage. Modern surgical principles call for the debridement of devitalized tissues and thorough exploration of the injured leg. The introduction of NPWT has revolutionized lower limb wound management. Its properties of enhancing wound healing by promoting tissue perfusion, reducing edema, and stimulating neovascularization while providing a clean moist environment have wide-ranging applications. From providing a temporizing dressing before definite bony fixation and soft tissue reconstruction to enhancing skin graft take and acting as a bolster for free flaps, NPWT continues to be a reliable and effective adjunct in the management of lower limb injuries.
TAKE-HOME MESSAGES
Bone fractures, especially those affecting the lower extremities and among the aged, represent a global health concern. Wound healing optimization using NPWT after lower extremity trauma and surgical intervention may help to achieve positive clinical outcomes. NPWT modalities are versatile advanced wound care interventions that have multiple applications within orthopedic traumatology of the lower extremities.
Footnotes
ACKNOWLEDGMENTS AND FUNDING SOURCES
The authors thank Willie M. Heard III, PhD (Solventum), for the assistance with article preparation and editing. This work received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
AUTHORS’ CONTRIBUTIONS
All the authors contributed to the design, development, writing, and reviewing of this work and gave final approval for publication. All the authors assume responsibility and accountability for the article.
AUTHOR DISCLOSURE AND GHOSTWRITING
S.N. is a consultant for Solventum. All authors contributed to the writing of this article, and no ghostwriters were used.
ABOUT THE AUTHORS
