Abstract
Objective:
Negative pressure wound therapy (NPWT) and oxidized regenerated cellulose (ORC)/collagen/silver-ORC (OCSO) dressings have individually demonstrated effectiveness in supporting wound healing, but few studies have examined their combined use. This retrospective data analysis compared wound outcomes following outpatient NPWT with and without OCSO dressings.
Approach:
A search of deidentified records from the U.S. Wound Registry resulted in 485 cases of wounds managed with NPWT with OCSO dressings. A matched cohort of patients who received NPWT without any collagen dressing (n = 485) was created using propensity scoring. For patients in the NPWT + OCSO group, OCSO was applied topically on or after the day of NPWT initiation and stopped on or before the day of NPWT termination.
Results:
Wounds managed with NPWT + OCSO were significantly more likely to improve and/or heal compared with wounds that received NPWT alone (p = 0.00029). The relative wound area reduction was 40% for patients receiving NPWT + OCSO, compared with 9% for patients receiving only NPWT (p = 0.0099). The median time to achieve 75–100% granulation coverage with no measurable wound depth was shorter by 8 days with NPWT + OCSO in all wound types (p = 0.00034), and by 14 days in surgical wounds (p = 0.0010), than with NPWT alone.
Innovation:
This is the first study examining the clinical outcomes associated with the integration of NPWT and OCSO dressings compared with the use of NPWT alone. These data support the novel practice of applying NPWT concurrently with OCSO dressings.
Conclusion:
This retrospective comparative analysis using real-world data demonstrated improved healing outcomes with integrated use of NPWT with OCSO dressings versus NPWT alone.

INTRODUCTION
Negative pressure wound therapy (NPWT) and oxidized regenerated cellulose (ORC)/collagen/silver-ORC (OCSO) dressings are two effective treatment modalities for improving wound outcomes. Studies have shown evidence that NPWT applied at −125 mmHg using foam dressings promotes wound healing by increasing perfusion, removing exudate, reducing edema, and stimulating granulation tissue formation. 1 –5 In addition, OCSO dressings provide an antimicrobial barrier in the dressing and maintain a moist wound environment conducive to healing. 6 –10 For wounds requiring multimodal support, OCSO dressings are frequently applied to facilitate wound closure after a duration of NPWT. 11 Despite their individual effectiveness, few studies have investigated the outcomes when these treatments are used concurrently. In this retrospective data analysis, wound outcomes following outpatient NPWT with and without OCSO dressings were compared.
INNOVATION
Previously, the simultaneous use of NPWT with OCSO dressings was not supported by the manufacturer’s instructions for use (IFU). Despite the lack of published evidence demonstrating the efficacy of these combined therapies, some wound care specialists hypothesized a synergistic benefit for wound healing. In 2020, a retrospective study reported that simultaneous use of NPWT with OCSO dressings supported the closure of 37 complex lower extremity wounds. 12 These wounds were difficult to heal by secondary intention, had critical structures that were hard to cover with granulation tissue, and could not be closed by flap reconstruction. Twenty-seven of the patients had unsuccessfully been managed with NPWT alone, suggesting that the combination therapy was critical to achieving closure. Recent updates to the IFU include guidelines for application of OCSO dressings with NPWT. The results of this study provide new evidence supporting this novel therapeutic approach.
CLINICAL PROBLEM ADDRESSED
Nonhealing and chronic wounds impose considerable physical, social, psychological, and financial burdens upon patients. These wounds are characterized by a disrupted healing cascade, rogue inflammatory response, and poor granulation. 13 Separately, NPWT and OCSO dressings were designed to support different mechanisms of wound healing. When applied sequentially, the use of OCSO dressings after discontinuation of NPWT has assisted the transition of stalled wounds into a state more conducive for healing, leading to eventual closure. 11,14 Research demonstrating the safety and efficacy of simultaneous NPWT with OCSO dressings could support a shorter time line for wound healing in some patients.
MATERIALS AND METHODS
For this study, patient records between January 2005 and December 2016 from the U.S. Wound Registry 15 database were analyzed. The U.S. Wound Registry compiles information from more than 100 hospital-based outpatient wound centers across the United States and Puerto Rico since 2005, documenting details on patients undergoing treatment for chronic wounds and ulcers. Data are systematically collected at the point of care within the electronic health records of patients, including adherence to wound care quality measures established by the registry. The U.S. Wound Registry has received recognition from the Centers for Medicare & Medicaid Services as a qualified clinical data registry.
The study extracted measures from electronic health records, utilizing real-world observational data collected by wound care professionals. Records were restricted to patients who were treated with an outpatient NPWT unit (3M™ ActiV.A.C.™ Therapy System; Solventum Corporation, Maplewood, Minnesota) for various types of wounds, primarily including surgical wounds (SWs) (33.2%), pressure ulcers (PUs) (14.6%), traumatic wounds (TWs) (12.4%), chronic ulcers (CUs) (12.2%), and diabetic ulcers (12.2%). Additional selection criteria included the use of only one type of NPWT during the observation time frame, defined as the period between the initiation of NPWT and the last visit. The NPWT was applied to treat the target wound and terminated on or before the last visit date.
Two patient groups were identified, distinguished by the application of NPWT with (n = 485) or without (n = 19,487) an OCSO dressing (3M™ Promogran Prisma™ Collagen Matrix with ORC and Silver; Solventum Corporation). In the NPWT + OCSO group, OCSO dressing was applied topically on or after the day of NPWT initiation and stopped on or before the day of NPWT termination. OCSO was the only type of collagen dressing used during the treatment, applied solely in conjunction with NPWT. On average, the application of OCSO dressings overlapped with 64% of the NPWT duration in the NPWT + OCSO group. In the NPWT group, no collagen dressings of any type were used.
To control for preexisting group disparities, an overarching propensity score was computed for each case using logistic regression across 39 variables, including patient demographics, comorbidities, and wound characteristics. 16 Within each wound type, NPWT + OCSO cases were matched to NPWT cases using 1:1 nearest neighborhood propensity score matching within a ±0.01 range. The wound-specific cohorts were then compiled for NPWT and NPWT + OCSO groups, respectively, to facilitate subsequent comparisons. The propensity score was matched with an average distance of 0.000075.
The study endpoints included the rate of healing or improvement, rate of worsening, percentage reduction in wound area, and time to achieve optimal granulation. Wound status as “healing/improving” or “worsening” were categorical labels provided by the health clinician assessing the wound at each visit. The percentage reduction in wound area was calculated as the change in the percentage of the initial surface area between the start and end of NPWT use. A granulation score (values ranging 1 to 5), derived from the Bates-Jensen Wound Assessment Tool, 17 –19 was used to classify the percentage of granulation tissue coverage and wound depth. A granulation score of 1 indicated optimal progress with 75–100% coverage and 0 depth. Clinicians demonstrated an estimate accuracy of ±25%. The healing time to attain “optimal granulation” was defined as the number of days between the start of NPWT and the earliest date at which a granulation score of 1 was recorded. If the initial granulation score was 1 at the start of NPWT, the healing time was reported as 1 day assigned as 1, on the condition that the wound did not worsen during the observation period.
The standardized mean differences and χ 2 tests were used to compare the covariate balance between the two groups before and after the propensity score match. Endpoints were compared using two-sample t-tests for percent reduction in wound area and χ 2 or Fisher’s exact test for rates of healing/improvement and worsening. The time to achieve optimal granulation was compared using a Kaplan–Meier survival analysis to delineate wound healing trajectories. 20 –22 The Kaplan–Meier healing curves were compared for the entire study population as well as within each wound type.
This study applied a stringent critical value of 0.01 for significance tests in endpoint comparisons. For covariate balance tests between the two groups, a critical value of 0.05 was used because a false negative was deemed more costly than in the endpoint comparisons. To manage the issue of multiple comparisons in our study, conventional corrections for multitesting issues, such as Bonferroni or Benjamini–Hochberg, were intentionally not used. 23,24 This decision was grounded in the predesigned and complementary nature of these tests, rendering them nonindependent. 24 –26 While multitesting corrections are most effective for addressing random multiple comparisons in exploratory studies, our research followed a hypothesis-driven and theory-supported design, conducting comparisons in one cohesive direction. 25 Therefore, these corrections were avoided to prevent compromising statistical power. 27 All statistical analyses were conducted using SAS 9.4 (SAS Institute, Cary, NC). This study utilized deidentified registry data and was therefore exempt from institutional review board approval, in accordance with the U.S. Health and Human Services Code of Federal Regulations, title 45 CFR 46. 28
RESULTS
Before matching, the NPWT + OCSO and NPWT groups were comparable across most baseline patient demographics, comorbidities, and wound characteristics, although NPWT patients utilizing OCSO dressings showed a slightly higher risk profile compared with those without. Specifically, the NPWT + OCSO patients had a 4% higher arterial vascular disease (p = 0.029), 10% higher SWs (p < 0.0001), with 8% lower venous leg ulcers (VLUs) (p < 0.0001). In addition, they showed a 5% lower utilization of cellular- or tissue-based products (p < 0.0001), typically applied to VLSs, diabetic foot ulcers (DFUs), and burns.
Postmatching, there were no statistical differences across any of the examined patient demographics or comorbidities (Fig. 1). The mean ages at first treatment in the NPWT + OCSO group and NPWT group were 57.1 and 56.1 years, respectively (p = 0.34; Table 1). Both groups had equivalent distributions across sex (p = 0.48) and race (p = 0.49). More than 40% of patients in both groups were smokers (p = 0.85) with an average body mass index >30 kg/m2 (p = 0.25; Table 2). Both groups contained high-risk patients with an elevated risk of hypertension (>60%) and type 2 diabetes (>40%).
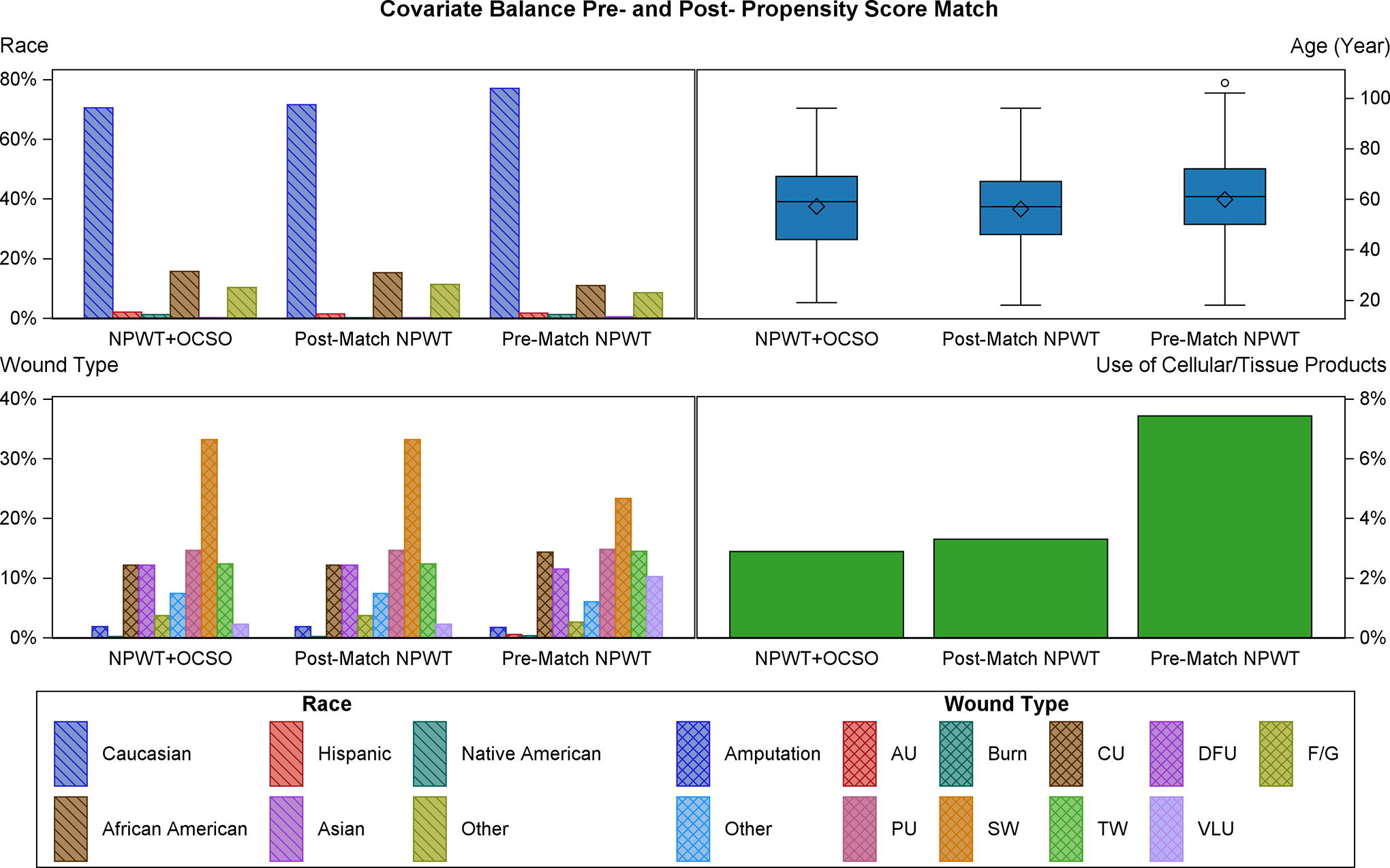
Covariate balance before and after the propensity score match. AU arterial ulcer; CU, chronic ulcer; DFU, diabetic foot ulcer; F/G, flap or graft; NPWT, negative pressure wound therapy; OCSO, oxidized regenerated cellulose (ORC)/collagen/silver-ORC dressings; PU, pressure ulcer; SW, surgical wound; TW, traumatic wound; VLU, venous leg ulcer.
Patient demographics before and after propensity score matching
NPWT, negative pressure wound therapy; NPWT + OCSO, negative pressure wound therapy + oxidized regenerated cellulose (ORC)/collagen/silver-ORC dressings; y, year; kg, kilogram; SD, standard deviation.
Patient comorbidities before and after propensity score matching
Wound characteristics and management therapies were statistically equivalent between the two groups. The average initial wound areas were 22.3 cm2 in the NPWT + OCSO group and 25.3 cm2 in the NPWT group (p = 0.37; Table 3). In both groups, 50% of wounds were present for more than 20 days upon the first encounter (p = 0.60), with over 85% displaying poor initial granulation. Similar wound therapies were administered in both groups, including NPWT active days (p = 0.54), hyperbaric oxygen therapy (p = 0.65), the use of cellular/tissue products (p = 0.71), and antibiotics (p = 0.61; Table 4).
Wound characteristics before and after propensity score matching
cm, centimeter; d, day; IQR, interquartile range.
Wound management therapies before and after propensity score matching
HBO, hyperbaric oxygen; IV, intravenous.
Wounds managed using NPWT integrated with OCSO dressings were significantly more likely to improve and/or heal compared with wounds that received NPWT alone (406 [83.7%] vs. 360 [74.2%]; p = 0.00029), and less likely to worsen (74 [15.3%] vs. 112 [23.1%]; p = 0.0019). The average wound area reduction between the start and end of NPWT was notably greater for patients receiving NPWT + OCSO at 40%, in contrast with 9% for patients receiving NPWT alone (p = 0.0099).
Across both cohorts, 78% of patients achieved optimal granulation tissue coverage during the observation period. The percentage of patients reaching this endpoint trended higher in the NPWT + OCSO group versus NPWT-only at all the examined time points (Fig. 2).
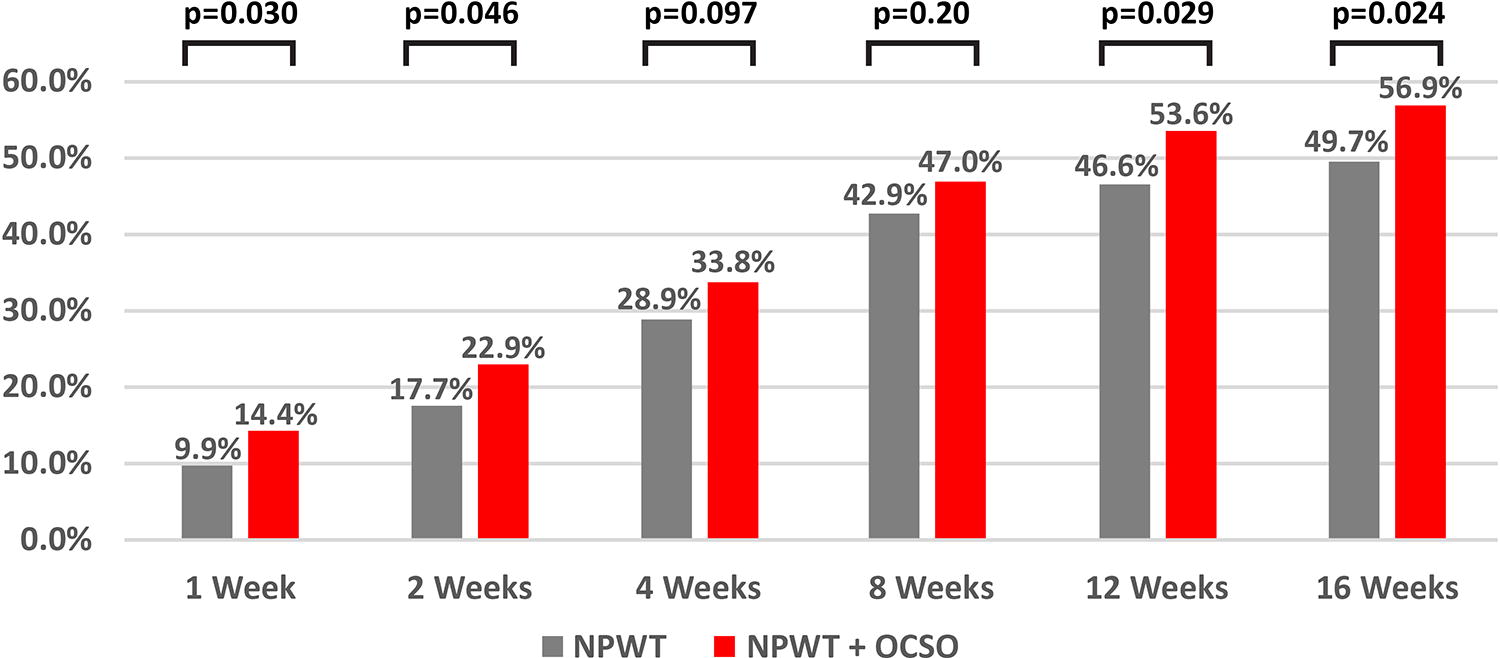
Percent of patients achieving 75–100% granulation tissue coverage with no measurable wound depth at six time points after initiation of NPWT with or without oxidized regenerated cellulose (ORC)/collagen/silver-ORC dressings.
The Kaplan–Meier healing curves by treatment groups across the entire observation period are shown in Fig. 3. For both groups, optimal granulation was achieved in 50% of cases within 40 days. An analysis of the weekly healing rates for the first 90 days showed that the NPWT + OCSO group reached optimal granulation faster than the NPWT alone (hazard ratio [HR] = 1.29; 95% CI 1.12-1.50; p = 0.00034). The median healing time was 21 days (interquartile range [IQR] 1–64) for NPWT + OCSO patients versus 29 days (IQR 5–93) for NPWT patients, and the mean time span was 34.3 ± 1.9 and 38.1 ± 2.1 days, respectively.
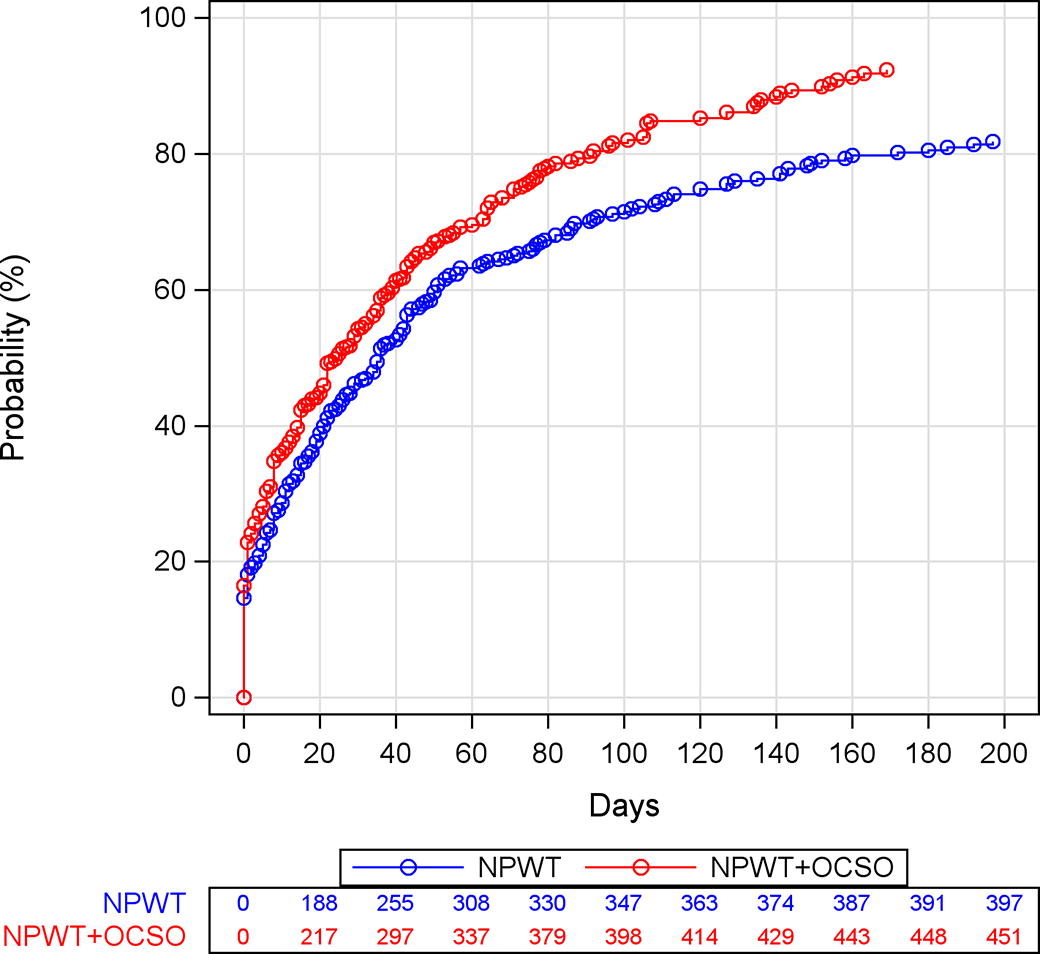
The Kaplan–Meier healing curves showing the probability of achieving 75–100% granulation tissue coverage with no measurable wound depth for the entirety of the observation periods.
A further investigation of treatment effect by wound type was conducted. The category with the largest sample size, SWs (n = 322), exhibited a significantly faster rate of achieving optimal granulation for NPWT + OCSO versus NPWT alone, with a median healing time of 29 (IQR 8–65) and 43 (IQR 13–143) days, respectively (HR = 1.52; 95%CI 1.18-1.97; p = 0.0010). Seven other types of wound had small sample sizes and their differences did not reach statistical significance: amputations (n = 18, p = 0.34), CUs (n = 11, p = 0.52), DFUs (n = 118, 0.051), flaps or grafts (n = 36, p = 0.059), PUs (n = 142, p = 0.66), TWs (n = 120, p = 0.080), and VLSs (n = 22, p = 0.14).
DISCUSSION
NPWT and OCSO dressings have been used separately to manage chronic or difficult-to-heal wounds. For wounds with stalled healing, implementation of a multimodal treatment plan may be appropriate to resolve the various dysfunctional repair mechanisms. NPWT can be applied to clean wound surfaces to create a barrier against external contaminants, remove exudate, promote perfusion, and support granulation. OCSO dressings can be placed directly onto the wound bed, and when moistened, transform into a biodegradable gel that maintains a balanced wound environment. After wound bed preparation with NPWT, OCSO dressings are frequently used to bridge the wound to closure. 11,14,29 Health care providers have postulated that OCSO dressings can be used simultaneously with NPWT, delivering the beneficial effects of both therapies and potentially shortening the duration of wound care. Recently published data support the successful integration of these therapies, particularly in fostering granulation tissue growth for complex and nonhealing wounds, even those with exposed critical structures. 12 Consequently, the manufacturer’s IFU were amended to include guidance for the use of OCSO dressings with a portable, outpatient NPWT unit for the management of certain wound types. In light of these changes, we conducted a retrospective analysis of registry data to assess the potential of OCSO dressings to bolster NPWT clinical outcomes.
This is the first study to use data from the U.S. Wound Registry to report on the outcomes of outpatient NPWT integrated with OCSO dressings, in comparison with the use of NPWT alone. We identified 485 patients who underwent the combined therapies, and used a matched cohort to control for confounding variables. There were no significant differences in the baseline demographics, comorbidities, or wound characteristics. Despite the initial similarities, substantial differences were seen in the reduction of wound area pre- versus post-NPWT between the groups, with the NPWT + OCSO group exhibiting a 40% reduction, compared with the 9% seen in the NPWT group. The combination therapy group also showcased an approximate 30% reduction in time to achieve optimal granulation tissue coverage within 90 days of starting NPWT.
A deeper analysis separating wounds by type revealed that SWs benefited from combination therapy over NPWT alone. Although granulation rates diverged for all other wound types, favoring NPWT + OCSO over NPWT only, these differences were not statistically significant, possibly owing to their small sample sizes. However, these early data support a more detailed investigation of combination therapy and its potential benefits to specific wound types or patient populations. Notably, the 2020 study by Loh et al. reported that NPWT + OCSO facilitated closure in treatment-resistant chronic wounds, including DFUs, venous ulcers, TWs, and PUs. 12
The findings in our study population suggest that neither therapy interfered with the efficacy of the other and there could be a synergistic effect owing to their simultaneous use. While both treatment groups benefited from NPWT, the components of OCSO dressings may have conferred a healing advantage over NPWT alone. This is supported by prior clinical reports that wounds managed with OCSO dressings exhibited lower rates of infection and shorter time to re-epithelialization compared with standard-of-care dressings. 6,10 Ionically bound silver, as found in OCSO dressings, is a well-known antimicrobial agent, and may help suppress the growth of common wound pathogens. 30 Bourdillon et al. examined the effect of OCSO upon in vitro biofilms of P. aeruginosa, reporting that OCSO was associated with reduced biofilm population without inhibition of human dermal fibroblast proliferation. 31 These data need to be confirmed with clinical studies showing in vivo efficacy, but suggest utilizing multiple mechanisms for supporting a wound environment conducive to wound healing. In human studies, the use of ORC/collagen components of OCSO dressings for the treatment of lower extremity ulcers has been associated with decreased activities of elastase, plasmin, and gelatinase in wound exudate and lower concentrations of matrix metalloproteinase-2. 32,33 These proteases are frequently elevated during delayed wound healing, and modifying their activity may be a therapeutic strategy for wound care. 34,35
Because OCSO dressing forms a permeable gel on the wound surface, it is unlikely to impair the effects of negative pressure to the wound bed. However, it is recommended by the manufacturer’s IFU that before application the dressing should be trimmed to fit the wound shape and fenestrated to ensure pass-through of exudate away from the wound surface. Standard foam dressings can be placed over the OCSO dressing and NPWT applied as usual, with dressing changes performed every 72 h or as needed.
This study builds upon prior research on the combined therapies of NPWT and OCSO dressings for complex lower extremity wounds, expanding the scope to include a broader spectrum of wound types. The study population reflects a diverse range of chronic wounds at different complexities and stages. The combined therapeutic approach yielded significant clinical advantages, including expedited coverage with granulation tissues, ultimately contributing to enhanced patient safety and improved outcomes.
Wounds managed with outpatient negative pressure wound therapy (NPWT) integrated with oxidized regenerated cellulose (ORC)/collagen/silver-ORC (OCSO) dressings were significantly more likely to improve and/or heal compared with wounds that received NPWT alone, and less likely to worsen. Patients undergoing NPWT + OCSO exhibited a higher relative wound area reduction compared with those receiving NPWT alone. NPWT + OCSO patients achieved 75 − 100% granulation tissue coverage with no measurable wound depth faster than NPWT-only patients. These findings suggest that utilization of OCSO dressings can be commenced alongside outpatient NPWT, effectively harnessing the synergic efficacy of both therapies.KEY FINDINGS
LIMITATIONS
This study is subject to certain limitations due to its retrospective nature involving the analysis of preexisting data. The observational design, while valuable, inherently imposes constraints on causal inferences. Despite conscientious efforts to mitigate baseline discrepancies among groups with regard to crucial wound healing and infection risk factors such as smoking, obesity, diabetes, autoimmune diseases, and vasculopathies, it is important to acknowledge that the study was limited by the lack of information regarding the complete patient pathology and health care providers. Incorporating these factors would have facilitated a more nuanced assessment of patient risk profiles, concurrently managing potential clustering effects attributed to different health care providers.
Furthermore, the study did not have data on specifics concerning wound locations and surgical history. This lack of information potentially compromises the accuracy of patient risk categorization, as different body parts and prior injuries could impact wound healing. In addition, the study had little data on the operational duration and frequency of OCSO dressing application. Not accounting for the “dosage” of intervention raises the possibility of an underestimation of the intervention’s true effectiveness.
There exists high interobserver variability and inaccuracy in estimating wound size within clinical practice. 36 Although clinicians achieved a ±25% accuracy in estimating wound size and granulation tissue coverage in this study, measurement errors could potentially introduce systematic bias into the data. The assumption made was that the clinicians responsible for measuring wounds were not correlated with the therapies administered to patients.
While the study endeavored to incorporate 39 distinct patient and wound characteristics for propensity score matching and risk adjustment, it is imperative to recognize that the assessment of preexisting confounders is predominantly contingent upon the available registry data. This reliance on data availability underscores the potential influence of unaccounted factors on health care providers’ decisions regarding the application of NPWT with or without OCSO dressings, introducing the possibility of uncontrollable selection bias. Nevertheless, the findings reported in this study may catalyze initiatives for randomized controlled trials to further investigate this topic and enforce a causal inference regarding the clinical advantages attributed to the integration of NPWT and OCSO.
CONCLUSION
These study findings introduce novel evidence that OCSO dressings have the potential to augment the effectiveness of outpatient NPWT. This integrated therapeutic approach emerges as a useful option for clinicians seeking additional strategies to facilitate the healing of intricate wounds.
Footnotes
ACKNOWLEDGMENTS AND FUNDING SOURCES
The authors thank Mikaela M. Sifuentes, PhD (Solventum), for article preparation support. Funding for this study was provided by Solventum (Maplewood, MN).
AUTHOR DISCLOSURE AND GHOSTWRITING
Y.H. and L.G. are employees of Solventum (Maplewood, MN).
AUTHORS’ CONTRIBUTIONS
Y.H. contributed to the conceptualization, data curation, formal analysis, investigation, methodology, project administration, supervision, writing, and editing of the article. L.G. contributed to the conceptualization, data curation, methodology, supervision, and review and editing of the article.
ABOUT THE AUTHORS
