Abstract
Significance:
The laparotomy is a common surgical procedure with a wide range of indications. Ideally, once the goals of surgery were achieved, the incision edges could then be approximated and the abdomen primarily closed. However, in some circumstances, it may be impossible to achieve primary closure, and instead the abdomen is intentionally left open. This review discusses the indications and objectives for the open abdomen (OA), summarizes the most common techniques for temporary abdominal closure, and illustrates treatment algorithms grounded in the current recommendations from specialty experts.
Recent Advances:
Still a relatively young technique, multiple strategies, and technologies have emerged to manage the OA. So too have the recommendations evolved, based on updated classifications that take wound characteristics into account. Recent studies have also brought greater clarity on recommendations for managing infection and malnutrition to support improved clinical outcomes.
Critical Issues:
The status of the OA can change rapidly depending on the patient’s condition, the wound quality, and many other factors. Thus, there is a significant need for comprehensive treatment strategies that can be adapted to these developing circumstances.
Future Directions:
Treatment recommendations should be continuously updated as new technologies are introduced and old techniques fall out of use.

SCOPE AND SIGNIFICANCE
Abdominal injuries are often complex, requiring a multifaceted treatment plan and coordination across a multidisciplinary team. This review describes three key strategies for managing and resolving abdominal wounds: temporary abdominal closure, antimicrobial therapy, and nutritional support. Recommendations for treatment and decision-making algorithms based on the current literature are also summarized.
TRANSLATIONAL RELEVANCE
Injuries involving the abdomen and gastrointestinal structures ignite various damaging mechanisms that must be addressed in order to halt deterioration of the wound. Using therapeutic approaches that are based in the fundamentals of wound science, disease progression, microorganism colonization, and nutrition will provide a better understanding of how to improve the patient’s condition. This review summarizes the current understanding and approaches to abdominal wound care and reveals the gaps where further research into the underlying mechanisms is needed.
CLINICAL RELEVANCE
Abdominal injuries are frequently complicated and require surgical intervention to resolve. 1 In patients who are significantly physiologically compromised, minimal access surgery may not be an option. This necessitates the use of laparotomy, which is performed by making a large incision to access the peritoneal cavity. Doing so can not only help resolve the patient’s underlying condition but also create a surgical wound that must be subsequently managed. The key recommendations summarized in this review provide a resource for clinicians encountering abdominal wounds within their practice.
BACKGROUND/OVERVIEW
The open abdomen (OA), also known as a laparotomy, is a strategy in which the fascia is intentionally left open to prevent elevation of intra-abdominal pressure and allow easy and repeated entry into the cavity as part of a re-exploration strategy. In the modern age, the beginning of surgical management of diseases of the abdomen dates back to the last century. The first reference in the medical literature to OA appeared in 1897 when Andrew McCosh proposed managing generalized peritonitis as a large abscess, leaving the abdominal cavity open to allow its complete drainage. 2 Since then, an ongoing effort has been made to establish clear indications for this procedure, with special emphasis on early abdominal wall closure.
Currently, the strategic approach to laparotomy in severe cases involves multiphase damage control surgery (Fig. 1). 3 This staged approach, which uses initial abbreviated laparotomy, requires temporary abdominal closure, which we call the Deferred Closure Protocol. The recommendation according to many authors is that the abdomen should be closed definitively as soon as possible. 4 Based on this premise, early definitive closure of the abdominal wall should be the main objective of any strategy for OA management once the causes that originated it have been resolved. The consequences of not closing an abdomen in a timely manner can lead to frozen or hostile abdomens.
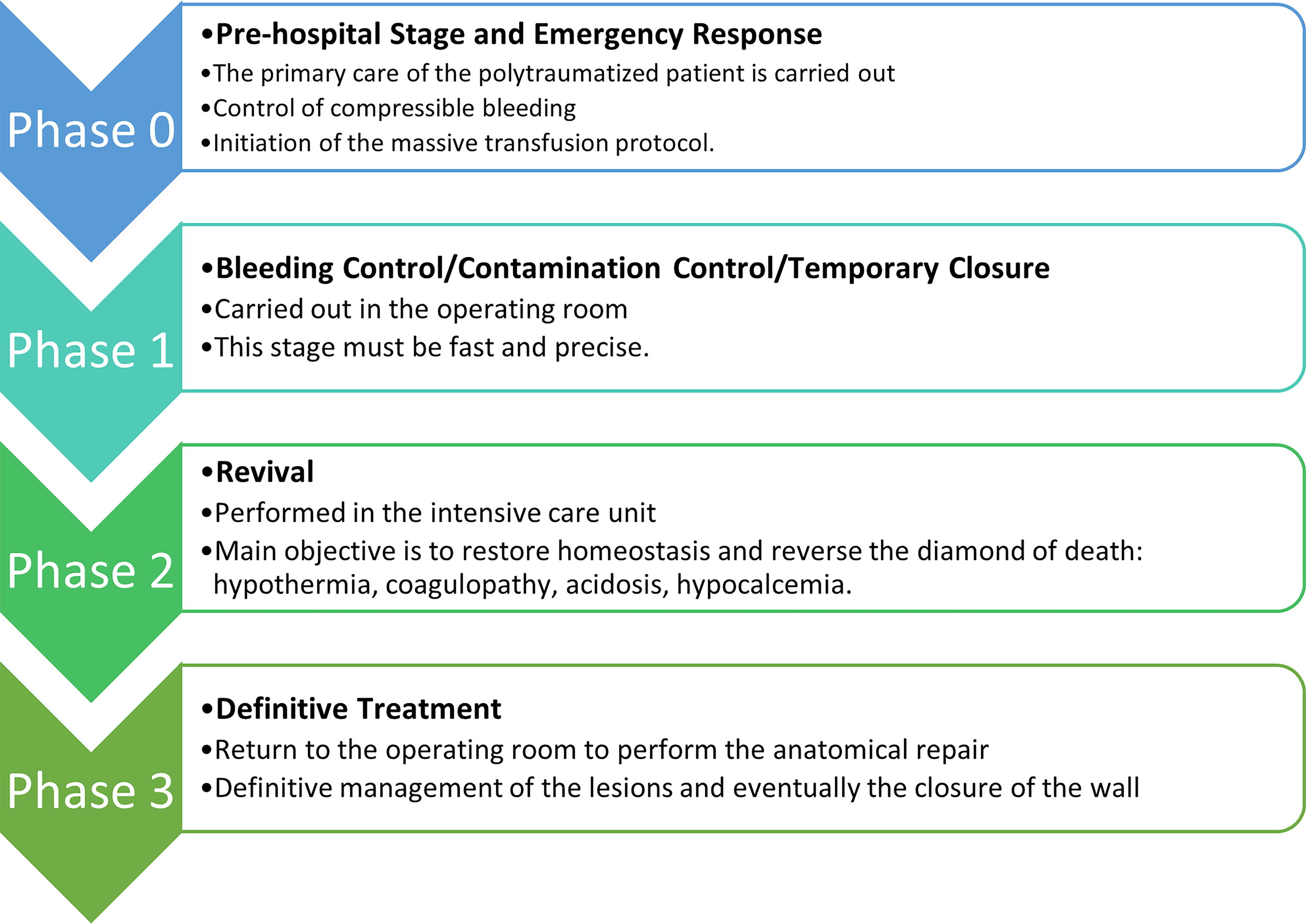
The phases of damage control surgery.
DISCUSSION
Candidates for an OA
In the treatment of various surgical conditions, such as peritonitis, intra-abdominal trauma, and mesenteric ischemia, early definitive closure of the abdominal wall (i.e., closure of the fascia and skin) may put the patient at risk of developing intra-abdominal hypertension (intra-abdominal pressure >12 mmHg) and/or abdominal compartment syndrome (ACS; intra-abdominal pressure >20 mmHg with new organ dysfunction). In these cases, a damage control approach may be preferable or necessary (Fig. 2).

Scenarios in which maintaining an open abdomen is indicated.
Other indications include increased lung pressure, hemodynamic instability before or after abdominal closure, massive intestinal edema, packing, and planned reoperation. The indication for OA in abdominal sepsis, whether due to postoperative peritonitis, intestinal perforation, infected pancreatic necrosis, or mesenteric ischemia, continues to be the most controversial of the indications, as it is associated with significantly higher mortality, especially in elderly patients and those with higher APACHE II scores. 5,6 Patients with peritonitis are often clinically complex and physiologically compromised. Thus, there is a need to shorten surgery time with a damage control strategy and undertake more refined procedures after adequate resuscitation. In these cases, delayed closure allows for stabilization while reducing the risk of ACS. 5 Keep in mind that OA is not always appropriate for patients with peritonitis, and in some cases, OA can increase the risk of major complications. 7 Some publications say that only 10–15% of patients with intra-abdominal infection (IAI) would qualify for this type of management. 8
In severe abdominal sepsis, OA and staged surgical procedures remain a validated strategy under these conditions:
8,9
Inability to control the source of contamination with a single surgery. The patient’s status makes it impossible to continue the operation. Visceral edema, increasing the risk of ACS. Massive loss of the abdominal wall due to necrosis or infection. Uncertainty about the viability of the remaining intestine. Uncontrollable bleeding due to coagulopathy secondary to septic shock.
Strategies for temporary abdominal closure
The mortality rates of OA patients are high, and delayed closure is associated with higher complication rates, emphasizing the need to aggressively pursue closure when it is safe to do so. 10 The need to leave the abdomen open forces us to think about the closing strategy, especially in those cases in which it is deferred. Here, we review the five most common techniques for temporary abdominal closure (Fig. 3).
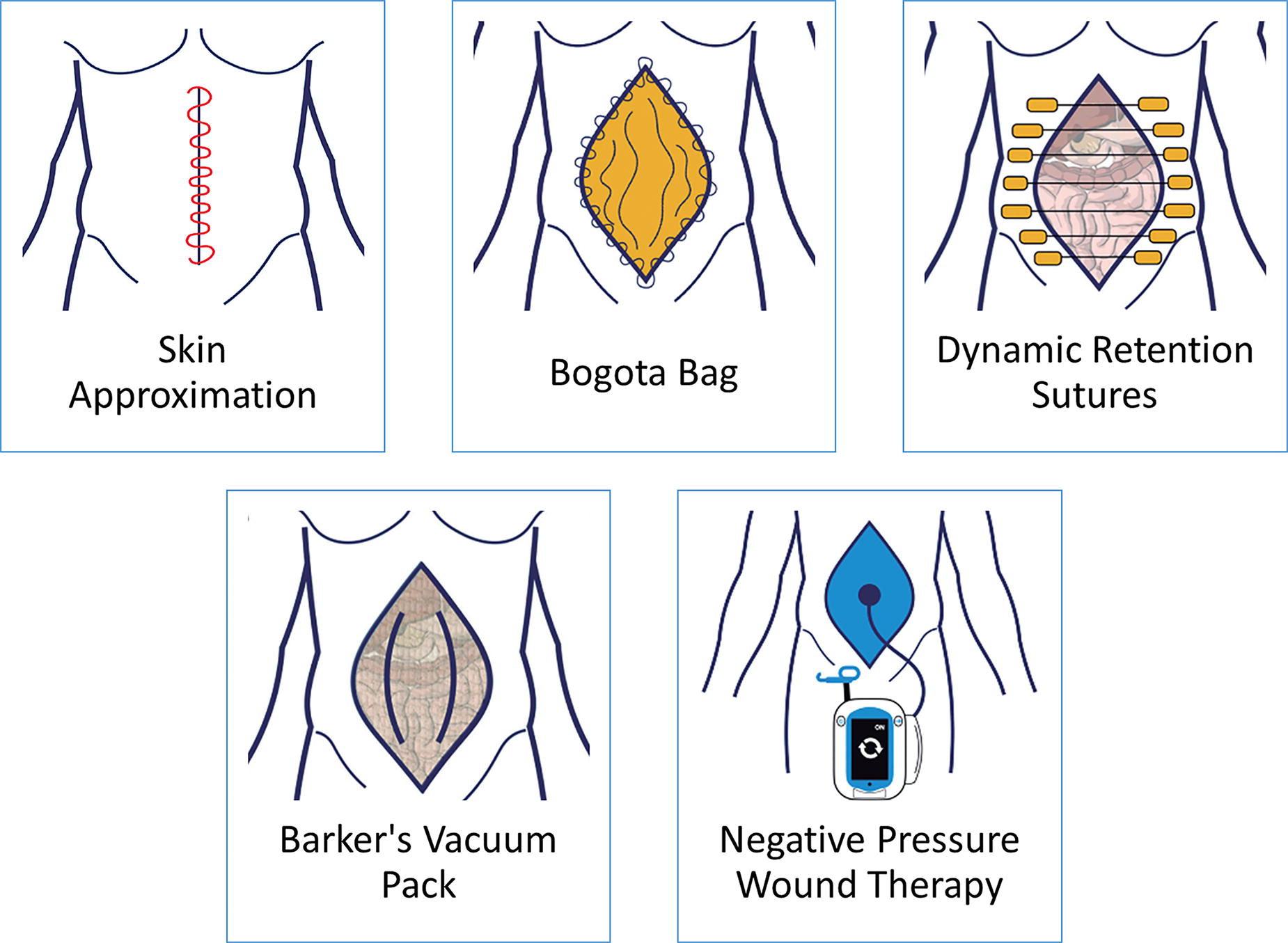
Common strategies for temporary abdominal closure.
Skin approximation
Approximation of the skin over the abdominal opening is a very easy, low-cost, and fast closure alternative (Fig. 4). This technique minimizes fluid and heat losses, though it does not prevent the establishment of ACS. 11 In addition, skin approximation lacks drainage support and can damage the skin.
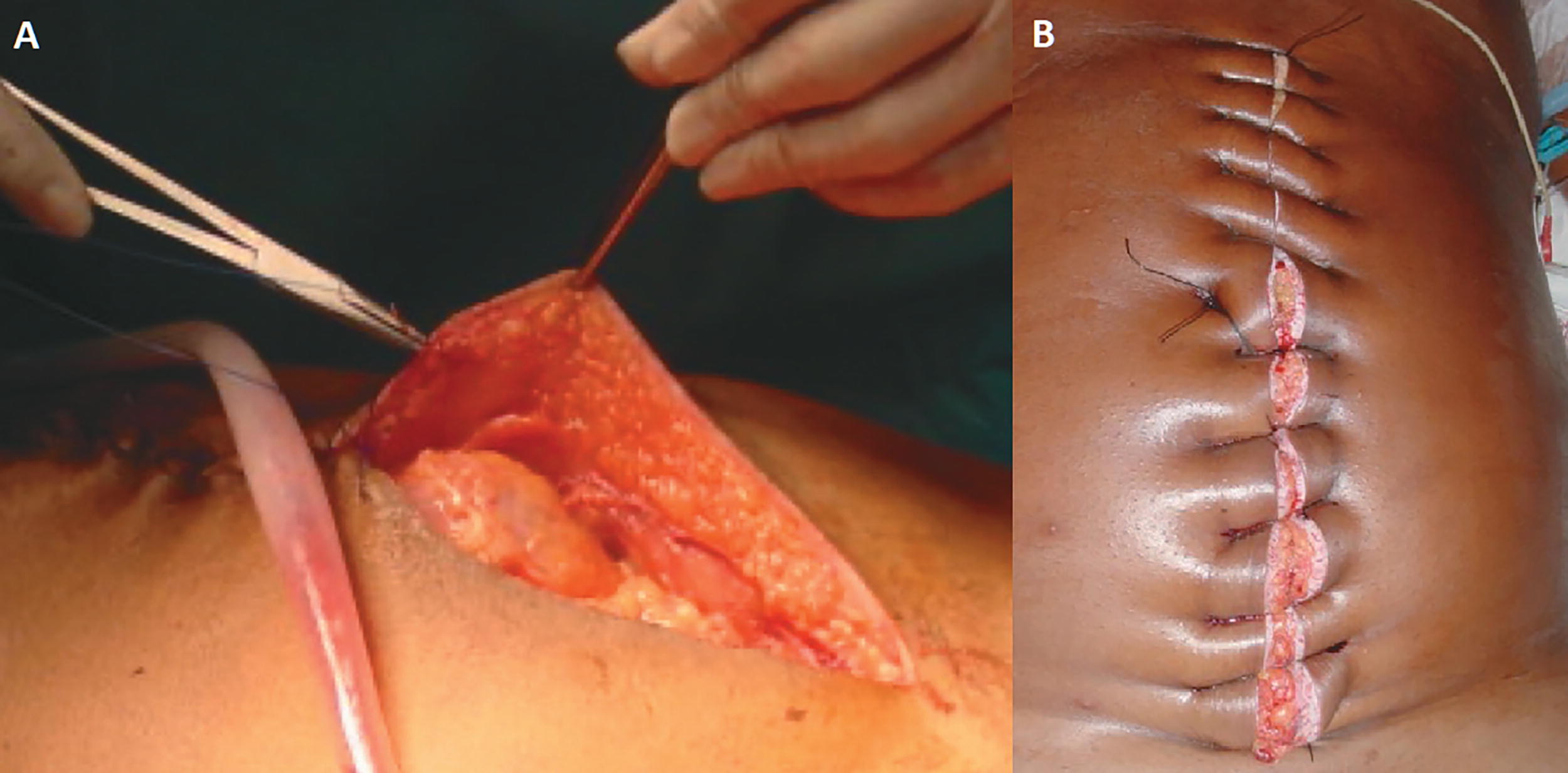
Temporary skin closure.
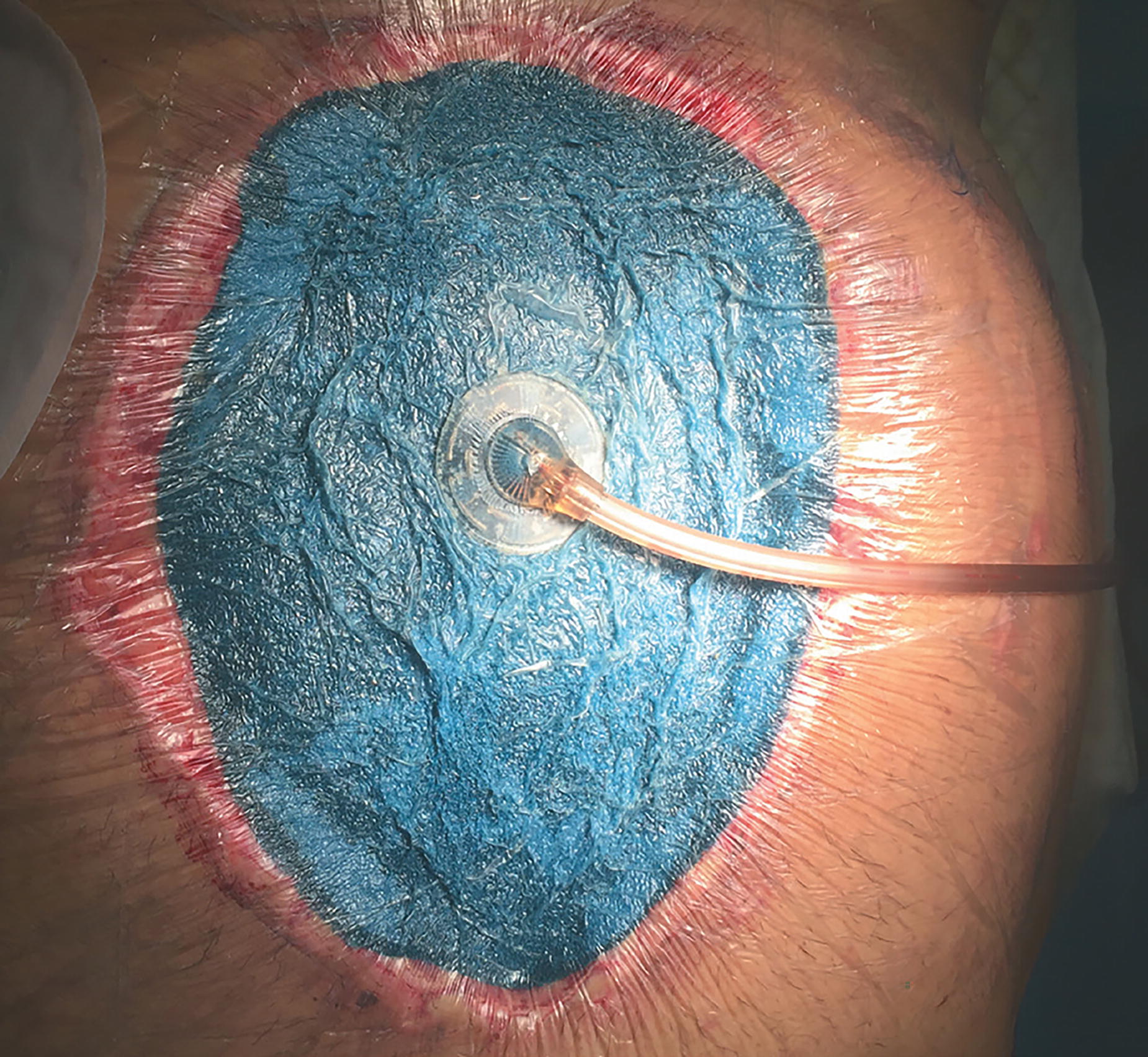
Bogota bag
The Bogota bag (also known as the Borráez bag) strategy involves the use of a sterile plastic bag attached to the skin or fascia (Fig. 5). The technique is easy, inexpensive, and fast. Given the establishment of a good container–content relationship, it could prevent ACS, especially if fixed to the skin. However, it is a passive system that does not actively promote closure. There is no exudate management or domain preservation. 11
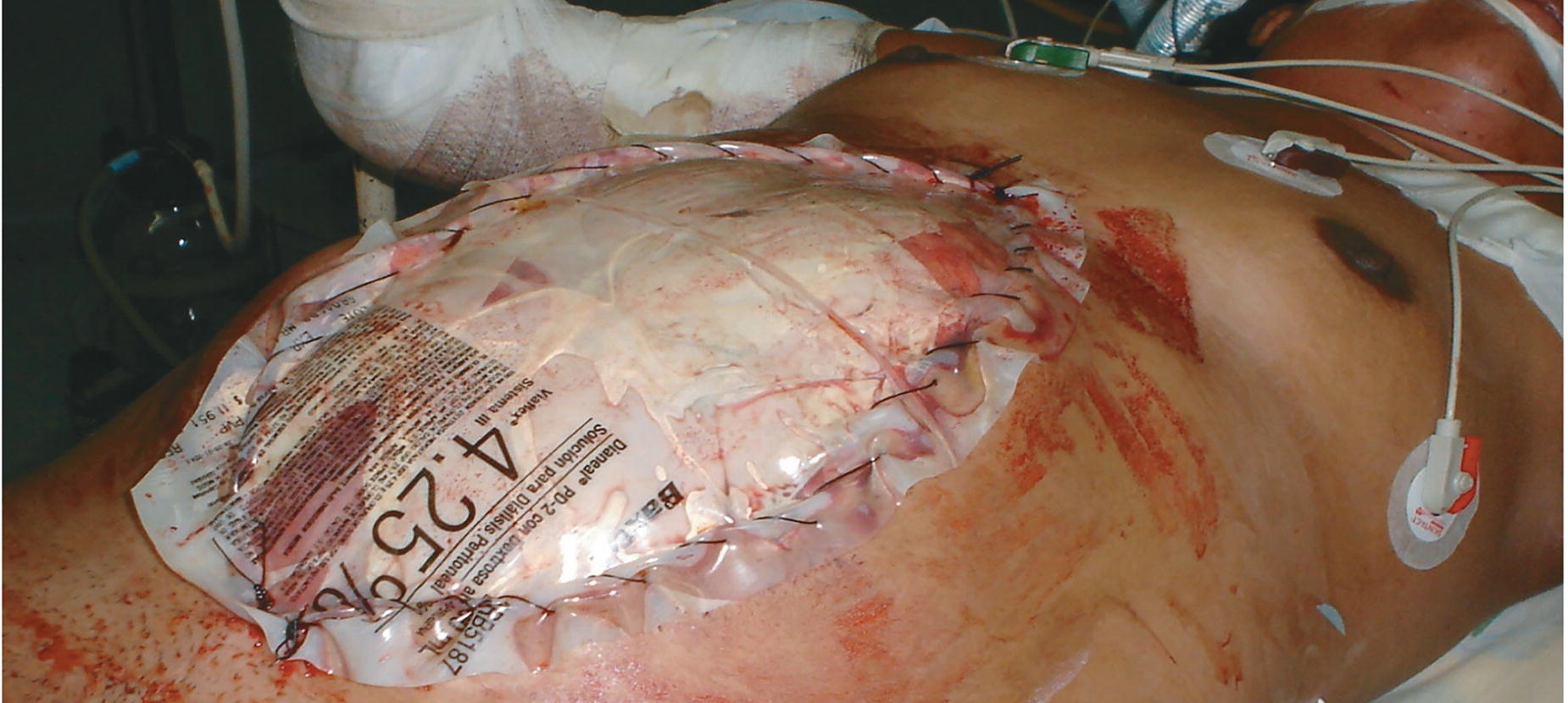
Attachment of a polyvinyl bag to the skin to temporarily close the abdomen. Note: in this case, a layer of latex is placed between the bag and the intestinal loops.
Dynamic retention sutures
Dynamic closure systems (Abdominal Re-Approximation Anchor System, ABRA™; Canica Design Inc., Almonte, Canada), also referred to as continuous fascial traction, use full-thickness elastomers in the abdominal wall to slowly pull the fascia under continuously variable tension (Fig. 6). It is easier to manage than mesh imbrication or sequential suturing and allows the abdominal wall to oscillate with the patient’s movement and breathing. Installation of the ABRA system unites the muscle and skin planes from their lateral retracted state with dynamic appositional traction, leaving the edge of the wound margins intact. This system can be used in combination with OA negative pressure therapy (OA-NPT), resulting in a comparatively high fascial closure rate. 11 –13
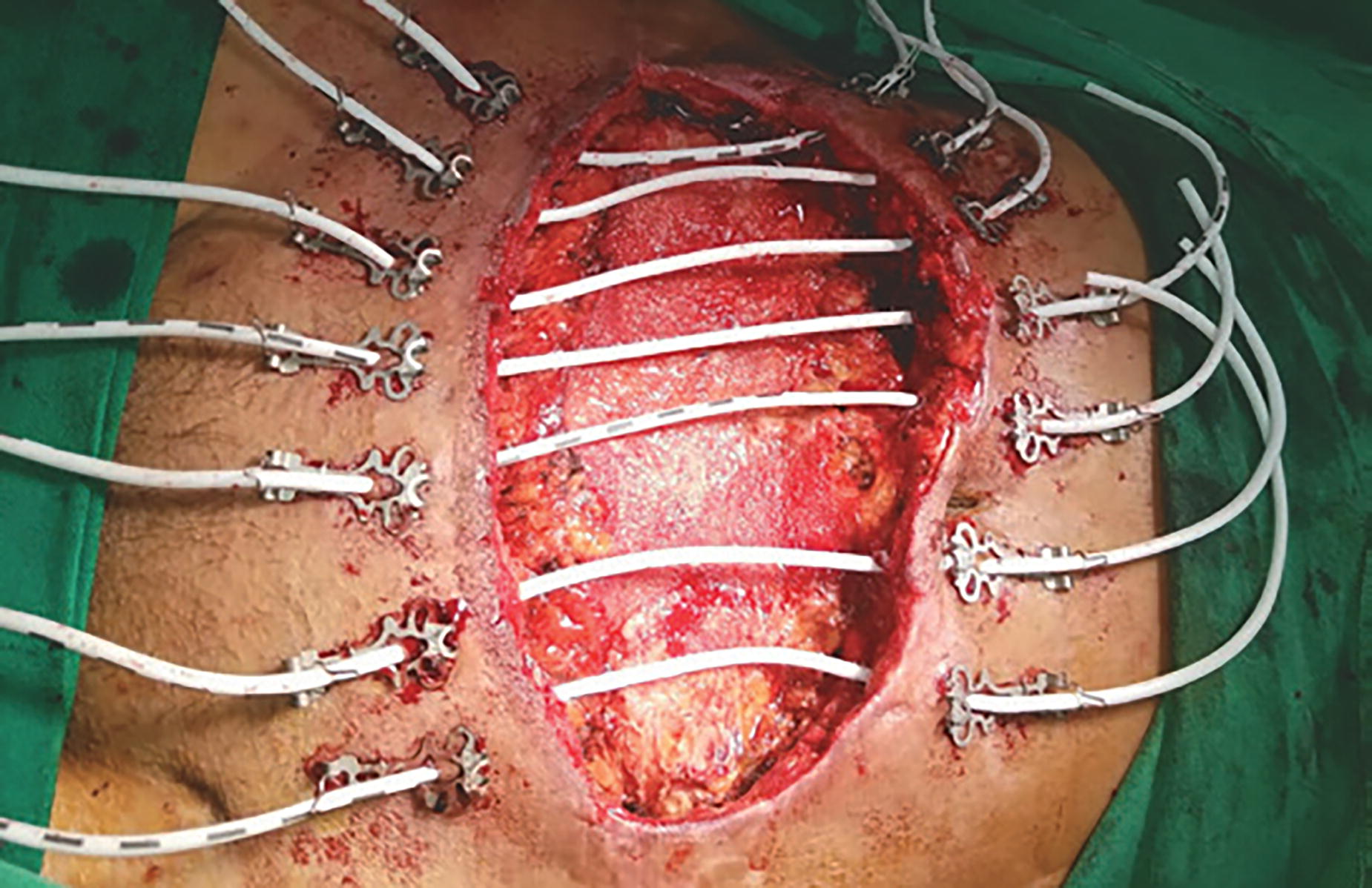
Application of the abdominal reapproximation anchor system.
Barker’s vacuum pack
The Barker’s vacuum pack strategy is carried out using an extended compress adhered to a surgical drape on which multiple holes are made. It is placed, extended into the intra-abdominal area with the vinyl polymer side on the intestinal loops. In the central area, adjusting to the abdominal wall defect, two to three wet compresses are added. Two Jackson–Pratt drains are placed on them, which are externalized about 5 cm by contraincision along the upper edge of the wound (Fig. 7). 14 The Barker technique for temporary closure of the abdomen is a simple, cheap, and reproducible method. However, because it is an artisanal technique, it lacks standardization and the development of materials with special characteristics studied for this purpose. Furthermore, it can be associated with high incidence of enterocutaneous fistula if the technique is not applied properly. 11
Application of the Barker system.
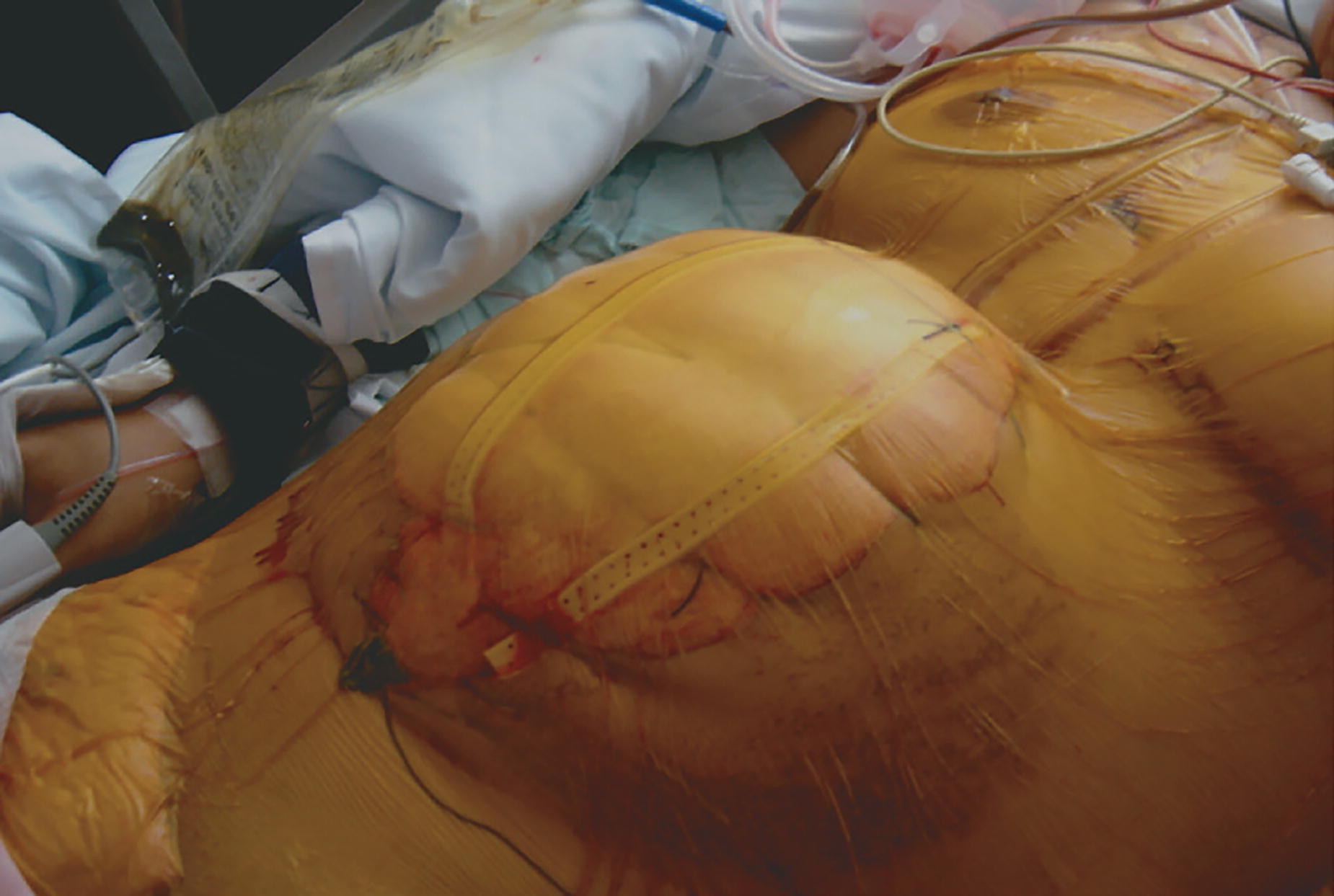
OA-NPT
OA-NPT (3M™ AbThera™ Open Abdomen Negative Pressure Therapy; 3M, St. Paul, MN, USA) is a commercially available temporary abdominal closure system designed to remove fluids from the abdominal cavity and provide medial tension. The goal is to help achieve primary fascial closure while protecting abdominal contents from external contamination. The reported fascial closure rate with OA-NPT ranges from 25% to 89.5% (Fig. 8). 13,15 –17 The application of OA-NPT consists of placing the fenestrated polyurethane drape (3M™ AbThera™ Fenestrated Visceral Protective Layer; 3M) over the viscera, under the fascia, to the retroperitoneum at the back, the diaphragm at the top, and the pelvis at the bottom. This prevents adherence of the viscera to the abdominal wall and allows mobilization of the fascia while avoiding injury to the viscera. A perforated foam dressing (3M™ AbThera™ Perforated Foam; 3M) is then placed on top, and the abdomen is sealed with a drape adhered to the skin. Tubing is attached from the foam to a negative pressure unit and −125 mmHg is applied. Dressings are changed every 24–72 h. Since the introduction of AbThera Therapy, other OA-NPT dressing kits have been introduced, including RENASYS® AB Abdominal Dressing Kit (Smith+Nephew, Watford, UK) and Suprasorb® CNP Wound Foam Abdomen (Lohmann & Rauscher, Neuwied, Germany), which are used at −80 mmHg negative pressure, and the Vivano® Med Abdominal Kit (Hartmann, Heidenheim, Germany), which can be applied using −125 mmHg.
Application of the open abdomen negative pressure wound therapy system (3M™ AbThera™ Open Abdomen Negative Pressure Therapy).
Classification of OA complexity
Patients with OA represent a heterogeneous group and are not typically encountered in daily practice. In this sense, many surgeons may be unfamiliar with their management. Hence, the importance of classifying the patient’s condition when establishing the OA strategy.
In Figure 9, we present the classification scheme that was developed during the consensus meetings on OA-NPT 18 that took place in November 2007 and January 2009 and is based on previous works of Banwell and Téot 19 and Swan and Banwell. 20 This classification system is based on the natural history of clinical improvement or deterioration in patients with an OA. 21 The key objective is to prevent its progression to a higher level of complexity.

Summary of the classification proposed by Bjork et al., which describes the grades or stages of OA, taking into account attachment, contamination, and the presence of fistula. OA, open abdomen.
Type 1
The classification begins with type 1, the simplest level referring to an abdomen without attachment or adhesions, which could have contamination and, in the worst case, the presence of a fistula. This situation often occurs after a decompressive laparotomy by ACS, from a ruptured abdominal aortic aneurysm, or after abdominal trauma unassociated with visceral perforation. As long as the patient does not have other risk factors for a poor outcome, the prognosis is good for an uncontaminated wound (type 1A; Fig. 10A). A type 1B scenario involves an inflammatory process or contamination, such as local or generalized perforation because of an infection, diverticulitis, or perforation because of ischemia, rupture of the anastomosis after colorectal surgery, or trauma affecting the gastrointestinal tract. In this case, the objective is to cleanse the OA and avoid rigidity or lateralization of the abdominal wall. A type 1B case may not close quickly, so it is important to consider additional strategies such as the use of mesh, elastic ties, or other adjuncts to help prevent fascial retraction and avoid progression to type 2. Finally, the presence of a fistula in these patients complicates the closure strategy because the priority becomes fistula treatment before attempting definitive closure.
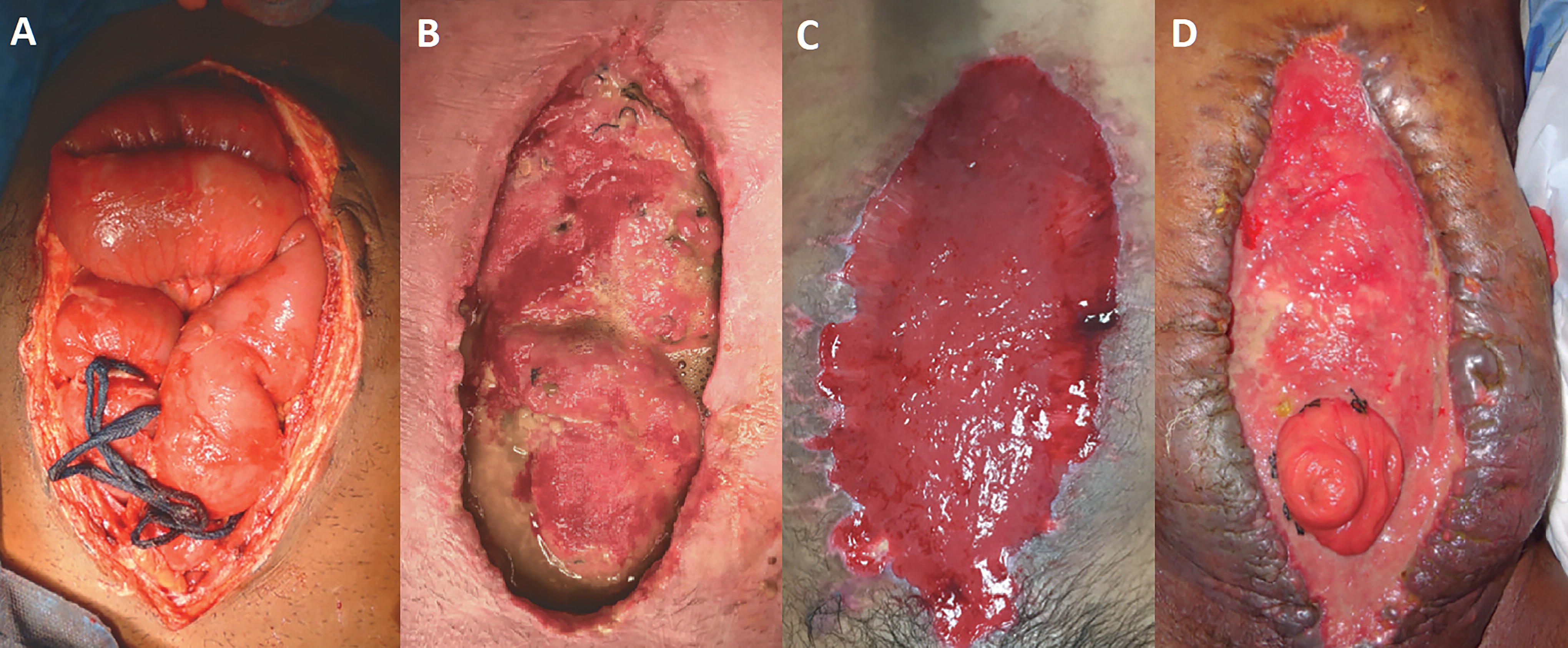
Examples of OA and their classifications according to Björk et al.
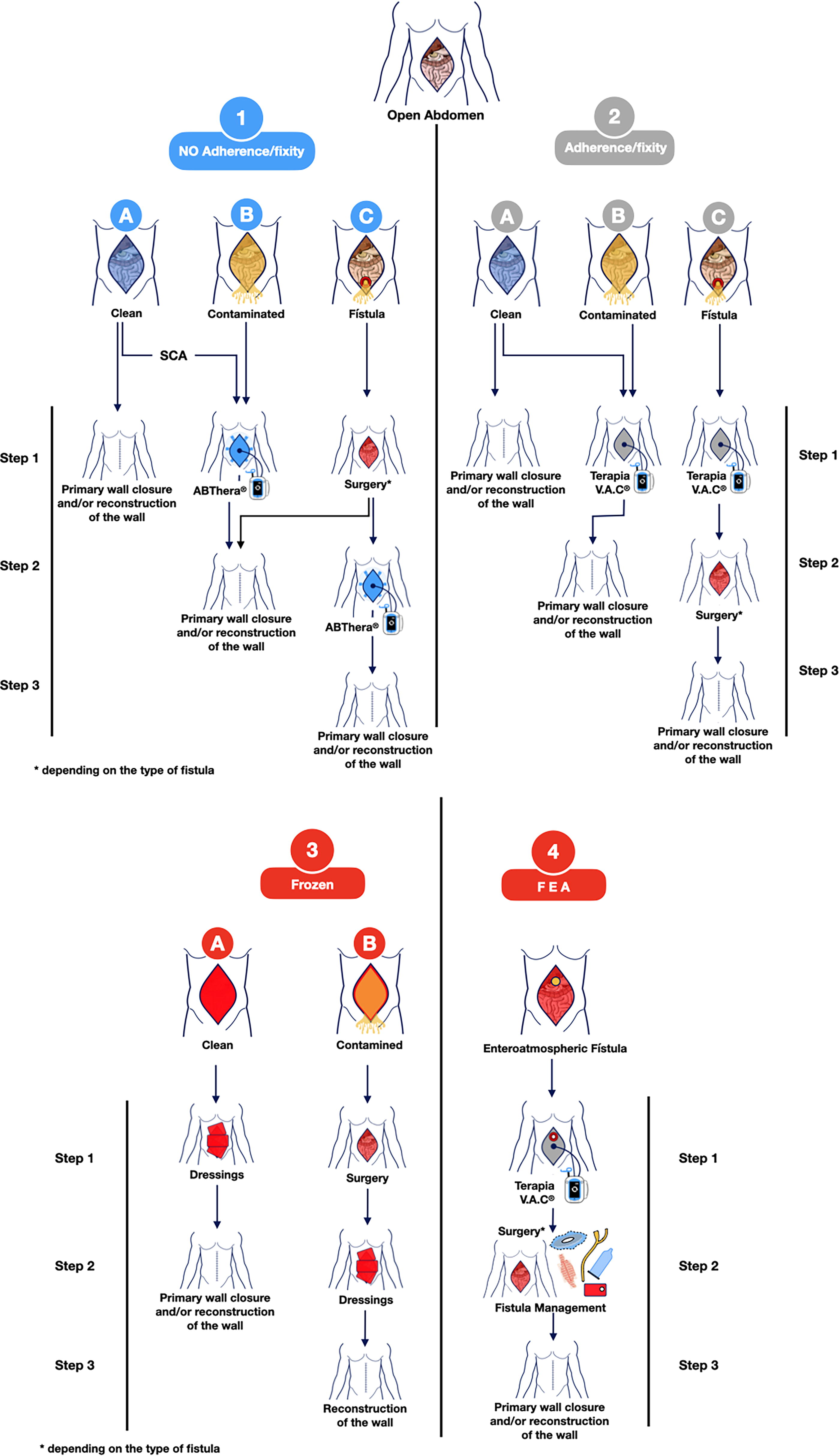
Proposed treatment strategy algorithm, according to the Björk classification.
Type 2
When adhesions have developed between the intestines and abdominal wall and/or the fascia has retracted laterally, primary closure becomes difficult to achieve. The objective should be to minimize further retraction and avoid greater deterioration. If adhesion of the intestines to the abdominal wall is irreversible, it is preferable to avoid excessive manipulation, as this risks causing injury and creating a fistula (Fig. 10B). Temporary abdominal closures that cover the entire viscera, such as the Barker’s Vacuum Pack or OA-NPT, reduce contact between the intestines with the retracted abdominal wall, thereby reducing the risk of developing adhesions.
Type 3
When the abdomen is subjected to multiple surgical interventions or there is a history such as peritonitis or anastomotic leakage, “frozen abdomen” can be presented (Fig. 10C). This is defined as a loss of free natural spaces between intra-abdominal organs and compartmental structures (anterior abdominal wall, retroperitoneal space, pelvic cavity, etc.), caused by a severe adherent syndrome with fibrous and scar tissue. The prevention of adhesions and/or rigidity is of great importance to avoid progression to a frozen abdomen.
Type 4
The development of an entero-atmospheric fistula (EAF; Fig. 10D) or an enterocutaneous fistula represents an important deterioration that significantly impacts management. In most cases, the intestine requires specific repair since it will hardly revert or resolve spontaneously. At this stage, sepsis control, study of the fistula's anatomy, nutrition, and establishment of the proper moment of surgery are needed. The main objectives are to control the fistula and prevent loss of abdominal domain, lateralization of fascia, and skin damage. Sometimes, after controlling the fistula, simple skin closure over the granulated wound bed is possible. This strategy allows the early discharge of the patient but is associated with subsequent herniation and additional reconstructive surgery.
Suggestions for OA treatment strategies according to the Björck classification
Taking into account the description mentioned earlier, of the OA types based on the anatomical characteristics of the abdominal wall, we make the following suggestions:
Type 1A
When there is no risk of ACS, primary closure—an approximation and closure of the tension-free wall—should be attempted. In contrast, if ACS is confirmed or probable, the placement of an active closing system with OA-NPT is suggested, after which primary closure can be carried out.
Type 1B
Depending on the degree of abdominal wall contamination and considering the risk of ACS, we suggest the use of active closure with OA-NPT. This not only facilitates active closure but controls the infectious focus as well. In both cases, the use of passive closure systems such as the Bogota bag is becoming less frequent.
Type 1C
Although definitive closure of the abdominal wall is important, when a fistula is present, its treatment takes priority. The fistula management technique depends on many factors, including the anatomy of the fistula, amount of debris, and the condition of the patient. Recommended strategies include conservative treatment, surgical closure, and the use of OA-NPT. 22
Type 2A
If possible, detach the abdominal wall from the intestinal loops and seek the primary closure. If this is not possible, apply OA-NPT with the visceral layer pressed deep into the peritoneal recesses to separate the viscera from the fascia.
Type 2B
For this classification, the indications are the same as the previous level but with greater inclination to manage contamination and implement OA-NPT as described earlier.
Type 2C
The primary concern is initial isolation and handling of the fistula. Secondarily, seek closure as in types 2A and B.
Type 3A
When the abdomen is frozen in the absence of contamination, the closure for second intention is relevant, for which we recommend dressings that promote healing. In these cases, a ventral hernia will be created to be resolved later.
Type 3B
In the presence of contamination, it should first be controlled. Then, proceed in the same way as type 3A.
Type 4
The EAF must be isolated before OA-NPT can be applied. Resolution of the EAF usually requires surgical intervention. However, it is important to consider the technique and the timing. Many experts recommend it be done after 6 months from the start of the condition. The initial strategy is to convert an EAF into an enterocutaneous fistula and then surgically treat the latter with the aforementioned considerations.
Focus control and antimicrobial therapy
IAIs continue to be a frequent cause of admissions to critical care units worldwide. In some series, morbidity and mortality are still around 23% and can vary, reaching 50% in high-risk patients, and may even be affected by the diagnostic and therapeutic resources available in the different health systems. 23,24 There are three fundamental pillars of IAI treatment (Fig. 12). 25
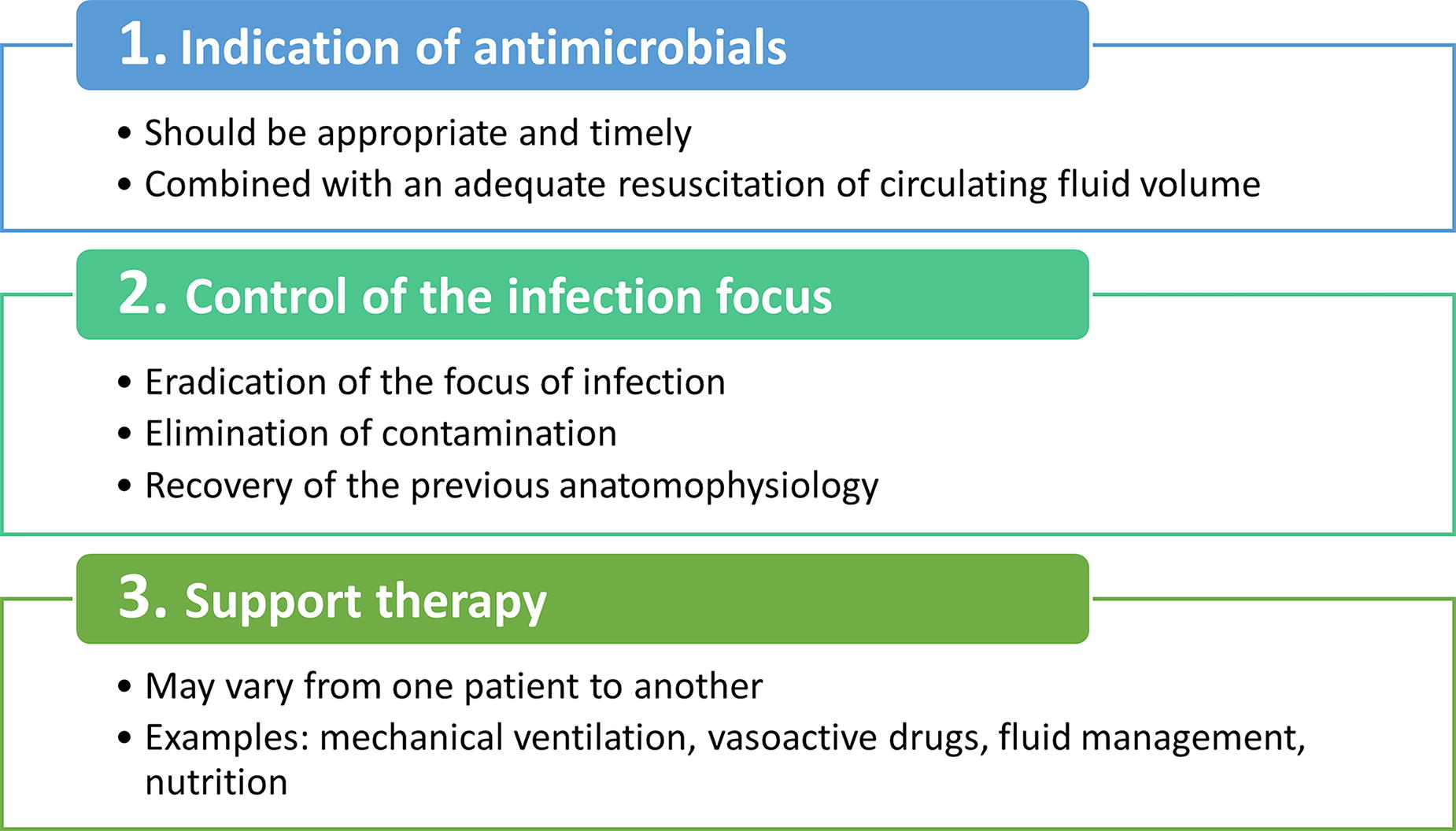
Summary of the three steps of intra-abdominal infection management.
The first step in choosing an appropriate antimicrobial agent is to understand which microorganisms are commonly involved in the infectious abdomen. The initial treatment is usually empirical and should be broad spectrum. It should guarantee coverage of Gram-negative pathogens, although they are less common. These types of patients require immediate treatment, while culture results and susceptibility tests are obtained in 48–72 h to guide specific treatment, especially in critically ill patients. 26
Rate and timing of microbial colonization of the OA are unknown. 27 However, complicated IAIs managed with OA are typically polymicrobial. Evidence indicates that the vast majority of these patients are in a state of sepsis, and they require basic coverage against Gram-positive, Gram-negative, aerobic, and anaerobic bacteria. 28 In addition, there is frequent need for coverage against Enterococcus spp., P. aeruginosa, Candida spp., and resistant pathogens, such as extended-spectrum β-lactamase-producing Enterobacteriaceae. This will largely depend on microbiological aspects, epidemiological patterns, and factors related to the patient. Intravenous administration is the only route recommended for early antimicrobial treatment in IAI. Oral administration should be avoided at this stage, as absorption will generally be unreliable and effect may be delayed. 29 The source of secondary peritonitis must always be taken into account, since it directly influences the spectrum of pathogens involved. Specifically, the stomach, upper small intestine, lower small intestine, and large intestine have distinct flora in terms of microbial species and density (Fig. 13). 30 It is important to note that source control is recommended within 24 h of IAI diagnosis, using the least invasive approach possible. In critically ill patients, abbreviated laparotomy and temporary abdominal closure are recommended if closure would create excessive hypertension, the patient’s physiological reserves are severely compromised, or if there is a need for further procedures for source control or second-look laparotomy. 26
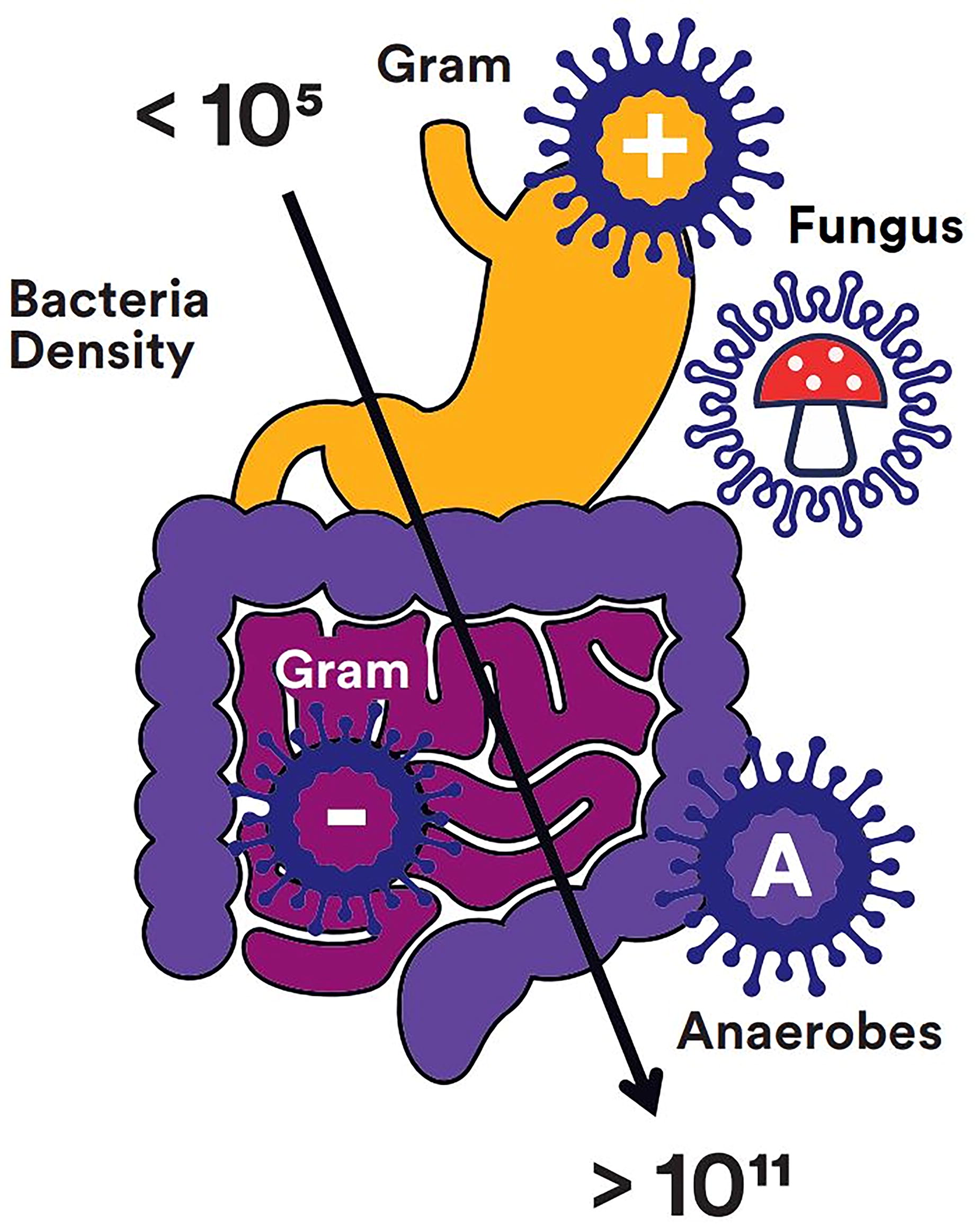
Distribution of pathogen density across the digestive system. 30
There is a predominance of Gram-negative and anaerobic bacteria in IAI of colorectal origin or appendicitis. Examples of intestinal injuries with complex IAI presentations are shown in Figure 14. In gastroduodenal perforation, Gram-positive bacteria and yeasts are more frequently isolated. 30 In IAI of the small intestine, there is a relative balance between the four different groups of pathogenic microorganisms. However, in subsequent weeks, there is a change in culture results, showing an increase in Gram-positive bacteria, whereas the prevalence of Gram-negative bacteria decreases. 30 The choice of empirical antibiotic regimens for IAI should be based on the clinical condition of the patients, the individual risk of infection by resistant pathogens, and the epidemiology of local resistance.
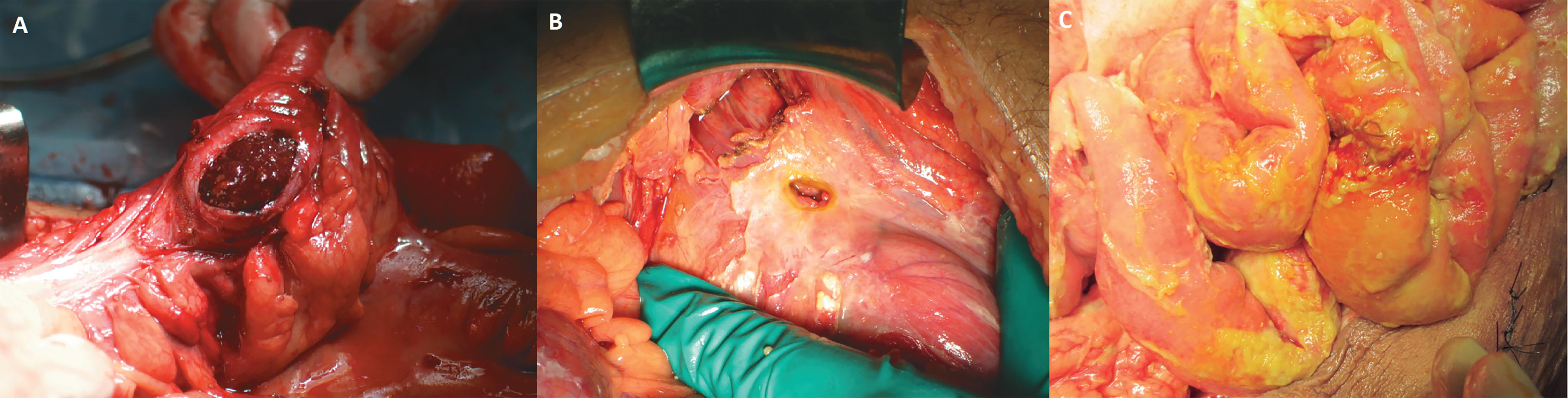
Examples of complicated intra-abdominal infections.
Intra-abdominal candidiasis
Intra-abdominal candidiasis is a serious infection for which adequate early treatment is important, but antifungal prophylaxis for all patients with peritonitis does not appear to be beneficial. Fungal involvement in IAI is common, particularly in nosocomial infections and in patients who have been exposed to prior antibiotic therapy. 31 Cultures are not always reliable for diagnosing candidiasis in a timely manner; thus, it is important to take into account risk factors and where appropriate use nonculture biomarkers to guide therapy. 32 Intra-abdominal candidiasis has been associated with high mortality, and its late treatment is a risk factor for mortality. Candida albicans remains the most common pathogen, and its incidence is highest in gastrointestinal perforations above the angle of Treitz. Other risk factors for its appearance include prolonged hospital stay before surgery, preoperative cardiovascular failure, and generalized peritonitis. 33 –35 One should consider the use of fluconazole for preventive and pathogen-directed therapy of susceptible strains of C. albicans in adults and children who are not critically ill. Also consider the use of voriconazole for empirical or pathogen-directed therapy of fluconazole-nonsusceptible Candida strains in noncritically ill adults and children older than 1 month of age. Do not routinely use amphotericin B or its lipid formulations for empirical or pathogen-directed management of intraabdominal candidiasis in adults or children. 26 In the confirmed presence of Candida spp., adequate therapy is imperative, and an echinocandin may be an appropriate, especially for critically ill patients. 36 When possible, de-escalation to azoles may be considered in patients who are clinically improving. 37
Taking cultures of IAIs
In order to use antibiotics responsibly, it is critical to appropriately take and interpret cultures from the initial surgery. From this, we can administer a targeted therapy, eliminating the empirical concept at this point of infectious treatment. With the result of the culture, we can do de-escalation therapy as a safe strategy and thus prevent contributing to microbial multiresistance. 38,39 Similarly, it is important that empirical antibiotic treatment should be guided by sensitivity patterns of each institution.
Cultures often reveal a mixed flora in which it is difficult to distinguish contaminants from true pathogens. In cases of IAI acquired in the community, with secondary peritonitis, cultures rarely influence patient management as the flora found is generally suspected. 40 In patients with complicated IAI, it is always recommended to perform intraoperative cultures (Fig. 15). These allow the antimicrobial regimen to be broadened if the initial choice is too narrow, the resistance pattern to be known, and de-escalation if the empirical regimen is too broad.
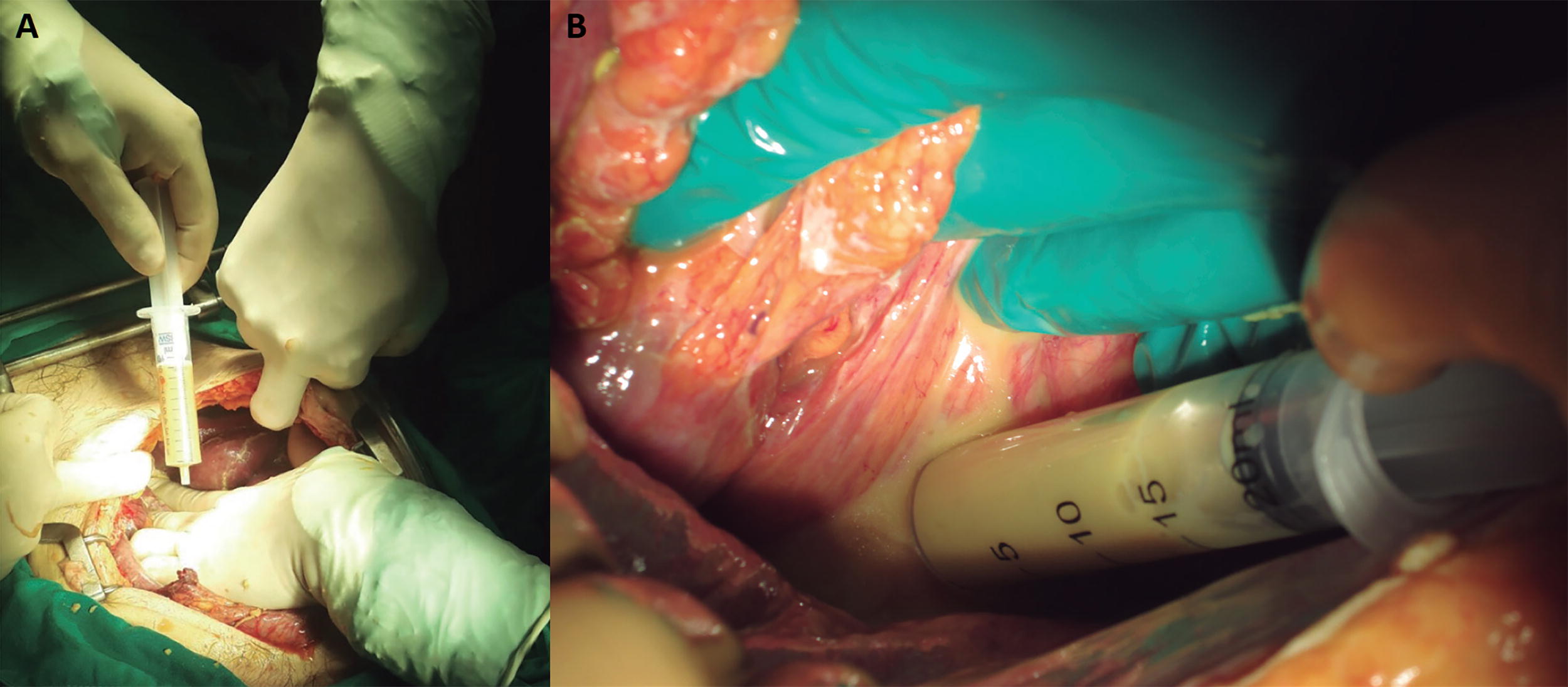
Taking cultures using a sterile syringe.
When a microorganism is identified in clinical cultures, antimicrobial susceptibility testing should always be performed and reported to guide antibiotic therapy. Consideration should be given to changing antimicrobial therapy based on culture results in selected patients.
Specific recommendations include the following (Fig. 16):
Proposed treatment algorithm for managing the infectious open abdomen.
Do not change antimicrobial therapy based on culture results in low-risk patients who have had a satisfactory clinical response to source control and empiric therapy.
Consider modification of antimicrobial therapy in higher-risk patients if culture results identify organisms resistant to the initial empiric regimen, and additional antimicrobial therapy is planned.
Routinely reduce or optimize antimicrobial therapy in higher-risk patients to the narrower-spectrum agent or agents that have activity against the isolates once definitive culture results are available.
Nutritional support
If a patient develops a systemic inflammatory response to injury or trauma that is exaggerated or exceeds the host’s responsiveness, a state of hypermetabolism and catabolism develops. This hypercatabolic state is associated with muscle proteolysis, acute protein malnutrition, impaired immune function, and the subclinical development of multiorgan dysfunction. 41
The presence of inflammation often limits the effectiveness of nutritional interventions, and associated malnutrition may compromise the clinical response to medical therapy. 42 Patients with an OA represent one of the most dramatic, inflamed, and consequently hypermetabolic groups of surgical patients. The acute phase inflammatory response to the patient’s underlying disease or injury affects nutritional status by increasing resting energy expenditure and nitrogen excretion. In addition, the large open wound associated with an OA is a major catabolic drain, and exposed bowel is vulnerable to injury and fistula formation. Meeting nutritional requirements in these patients is extremely difficult, and nutritional supplementation alone partially reverses or prevents muscle protein loss in active inflammatory states.
Although achieving positive nitrogen balance is widely considered the primary goal of nutritional therapy, multiple studies show that negative nitrogen balance is a common event in patients with an OA. 43,44 Maintaining an OA leads to a significant loss of fluid, electrolytes, and protein from the exposed viscera. This value ranges from 15 to 30 g of nitrogen loss per liter of abdominal fluid, which represents a loss of 94 to 188 g of protein. 44,45 Not including this source of protein loss in the nitrogen balance equations leads to an underestimation of the total nitrogen loss and indirectly leads to decreased wound healing, increased infections, and decreased survival. In general, most patients with an OA will require 25–35 kcal/kg/day of nonprotein calories and 1.5–2.5 g of protein/kg/day. 4,46
The issue of whether we should feed critically ill patients with their full estimated caloric needs remains an ongoing debate in many intensive care units (ICUs). Questions surrounding this conundrum include whether to feed, the optimal feeding route and time to start nutrient delivery, the volume delivered with each administration, and/or the type of formula to use. Options for nutritional therapy in critically ill patients include providing only parenteral nutrition (PN), only enteral nutrition (EN), or combination therapy. Unfortunately, owing to the complexity of intensive care together with the heterogeneity of the patients and the underlying pathology, optimal nutritional therapy remains a difficult topic to study, 41 such that the self-care of critically ill patients with OA, including the best manner to optimize nutritional therapy is challenging. 41,47
Enteral nutrition
Multiple studies have shown a benefit of early administration of EN. These recommendations are supported by meta-analyses and recommended by experts from societies such as the American Society for Parenteral and Enteral Nutrition (ASPEN), European Society for Clinical Nutrition and Metabolism (ESPEN), and European Society of Intensive Care Medicine. 45,48,49 Despite this, there is still reluctance and uncertainty about the use, safety, and timing of EN in those patients seriously ill, much less with an OA. Compared with other ICU treatments, feeding continues to be considered a lower priority, probably because of fear of potential complications and lack of nutrition-focused or specific education.
Reasons cited for withholding enteral feedings in patients with an OA include fear of intolerance, development of ileus and intestinal edema, possibility of aspiration, risk of small bowel necrosis, and malabsorption with vasopressor use, fear of difficulties with glucose control, and contraindication to enteral feeding with the presence of intestinal discontinuity after damage control surgery. These concerns about the potential adverse effects of diet have led to heterogeneous nutritional support practices among physicians providing care to ICU patients and patients with OAs. Although several studies now support EN as the preferred method of providing protein and calories, this method often does not adequately meet nutritional needs. The frequency with which undernutrition occurs has been demonstrated in multiple comparative clinical studies with a supply ranging from 25% to 80% of required calories in different intensive care populations in multiple ICUs. 50 –52
Parenteral nutrition
ASPEN experts suggest that, in the critically ill patient with low nutritional risk, exclusive PN should be withheld for the first 7 days after ICU admission if the patient cannot maintain voluntary intake and if early EN is not adequate or feasible. 53 In contrast, in critically ill patients at high risk of malnutrition or severe malnutrition, when EN is not feasible, ASPEN experts suggest starting exclusive PN as soon as possible after admission to the ICU.
Clinical judgment on a given patient’s metabolic tolerance to dextrose (monitoring of glycemic control), lipid emulsions (monitoring of serum triglyceride concentrations), and amino acid dose is key to the administration of appropriate PN. 53 On its part, ESPEN recommends individually evaluating the safety and benefits of starting PN in patients who do not tolerate the full dose of EN during the first week in the ICU; the safety and benefits of initiating PN must be weighed on a case-by-case basis. It also suggests not starting PN until all strategies have been tried to maximize tolerance to EN. 49 Where feasible and appropriate, PN and EN can be used simultaneously, allowing for a gradual transition between the two methods.
Starting supplemental PN before this period of 7–10 days, in critically ill patients, does not improve outcomes and may be detrimental to the patient. 45,53 Patient-specific clinical judgment should be used regarding initiation of supplemental PN in the first 7 days for special cases (Fig. 17).
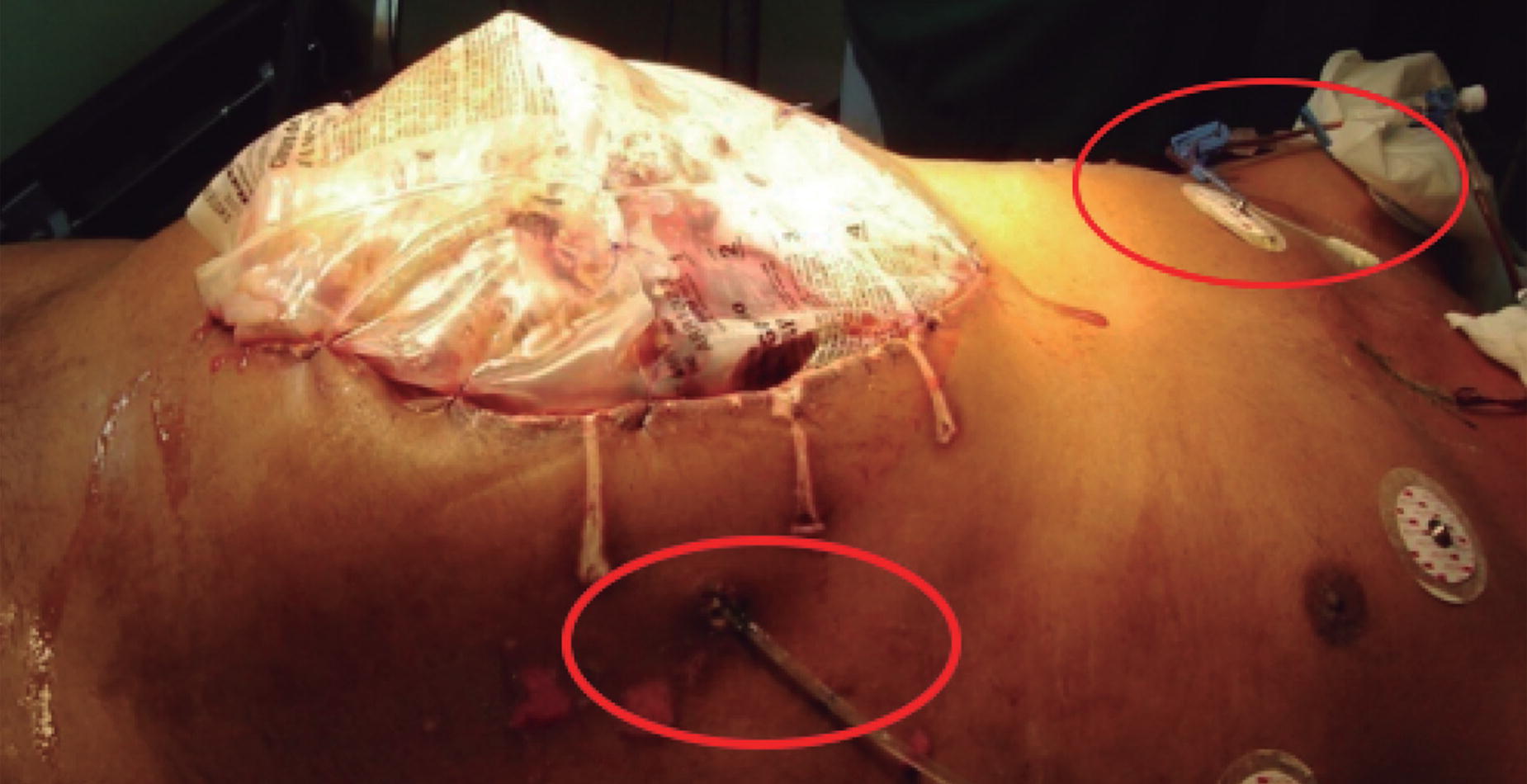
Example of a patient with an open abdomen with a Bogota bag and mixed nutritional support. (Bottom center) Enteral support via jejunostomy. (Top right) Parenteral support via central venous line.
Nutritional support for patients with EAFs
Patients with EAF present a unique challenge for the nutritional support team. Individual nutritional plans should aim to replenish fluid and electrolytes lost through the EAF and provide adequate calories and protein. Fistula acidity can be controlled using proton pump inhibitors or H2 channel blockers, which are recommended as part of a standard treatment regimen for high-output fistulas. Antidiarrheals such as loperamide or codeine are also useful tools for decreasing fistula output. 54 If enteral feeding is insufficient to compensate for the high output or if nutritional requirements are not met, then PN may be indicated. The ASPEN–FELANPE (Latin American Nutrition Federation of Enteral and Parenteral Nutrition) clinical guidelines indicate that patients with high-output fistulas (>500 mL/d) may require PN to meet fluid and electrolyte requirements and nutritional needs. 55 ESPEN recommends mixed nutrition when it is not possible to provide more than 60% of the requirements via enteral route (e.g., patients with high-output fistulas). 56 Care should be taken to ensure that there is no evidence that the fistula is feeding an abscess cavity or otherwise complicating the patient’s fluid and electrolyte balance.
Fistuloclysis, or “distal feeding,” is an option that can provide nutritional support to those fistulas that cannot close spontaneously. 53,57 This strategy should only be initiated after a contrast study has demonstrated a minimum of 75 cm of unobstructed, non-fistulated distal small intestine. 58 Fistuloclysis pursues two main objectives: (1) nourishing the patient through the healthy distal intestine and (2) performing enterocyte or trophic nutrition to avoid atrophy of the remaining intestine for future transit reconstruction. Fistuloclysis is a cost-effective alternative to PN and can also stimulate the intestinal mucosa. The use of polymeric formulas and elemental formulas can provide effective nutritional support for patients with EAF (Fig. 18). 59
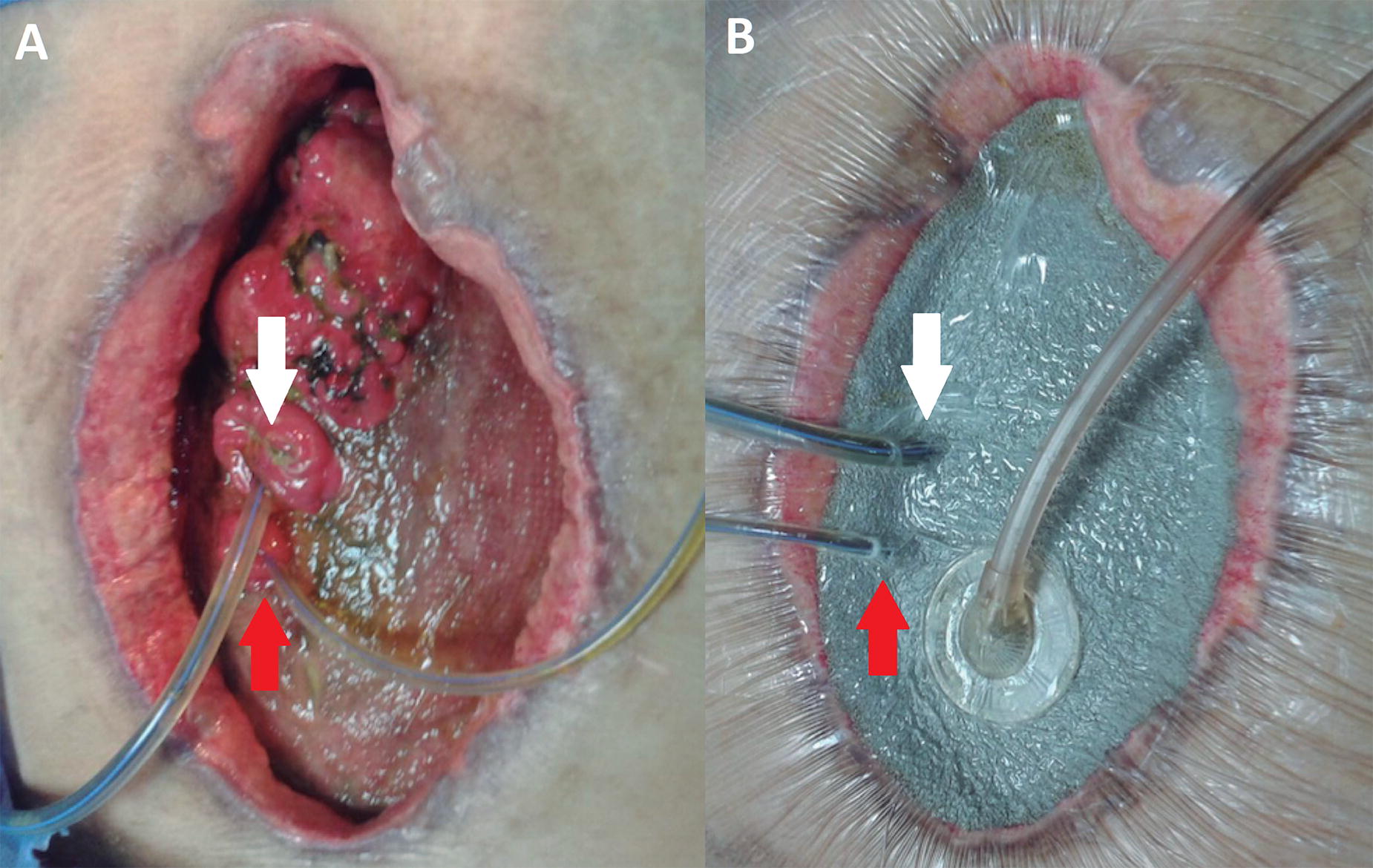
Example of a patient with open abdomen fistuloclysis (white arrow) and enteroatmospheric drainage catheter (red arrow).
The OA has become an important tool for the management of physiologically unstable patients requiring emergent abdominal surgical procedures. These patients present unique challenges for intensive care and nutritional support teams. Detailed attention to fluid and electrolyte management, meticulous wound care, prevention of EAF, and individualized nutritional supportive therapy are essential for successful recovery in this patient population (Fig. 19).
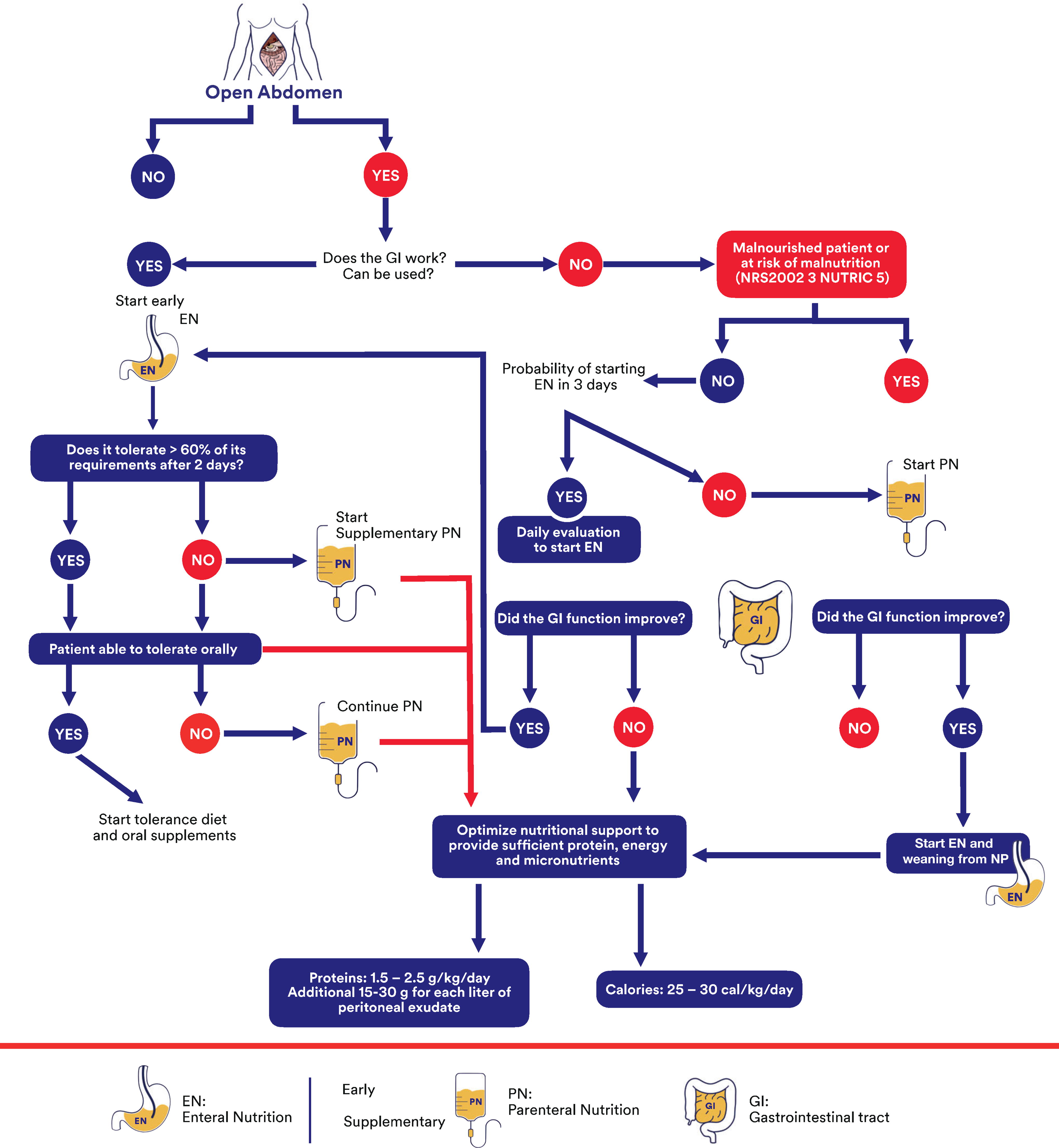
Algorithm for decision-making regarding the nutrition strategy in the patient with an open abdomen.
SUMMARY
Severe abdominal injuries require comprehensive treatment plans to stabilize the patient and prevent further deterioration. Staged surgical interventions and multimodal therapies can help improve patient status and ultimately lead to healing and resolution. These strategies include temporary abdominal closure, infection control, and nutritional support. The severity of the abdominal wound can be classified according to attachment, contamination, and fistulation. These classifications can be used to determine the correct treatment approach. Initial antimicrobial control should be empirical and broad spectrum. When possible, cultures should be taken to enable targeted therapy. In addition, patients with severe gastrointestinal dysfunction or open abdominal wounds frequently suffer from malnutrition, which should be rectified with adequate nutritional therapy. In this review, treatment recommendations are summarized and illustrated to support clinician decision-making for managing the abdominal wound.
TAKE-HOME MESSAGES
The OA is a strategy in which the fascia is intentionally left open, frequently as a result of damage control surgery. Candidates for temporary abdominal closure include patients at risk for ACS, peritonitis, and hemodynamic instability. Strategies for temporary abdominal closure include both active and passive techniques, and some are available as commercial products. Classification of the abdomen according to attachment, contamination, and fistulation can assist with selecting the appropriate treatment strategy. When appropriate, managing infection and malnutrition are strategies to support better healing outcomes.
Footnotes
ACKNOWLEDGMENTS AND FUNDING SOURCES
The author thanks Mikaela Sifuentes, PhD (Solventum), for article preparation support. No external funding was provided to conduct research for this article.
AUTHOR CONFIRMATION
P.O. was responsible for the conceptualization, investigation, writing, and review and editing of this article.
AUTHOR DISCLOSURE AND GHOSTWRITING
P.O. is a paid consultant to Solventum (St. Paul, MN).
ABOUT THE AUTHOR
