Abstract
Objective:
While the use of negative pressure wound therapy (NPWT) with reticulated open cell foam (ROCF) is well established, the characteristics of ROCF do not allow for extended-wear use. There is the potential for dressing tissue ingrowth if left in place for greater than the recommended 2–3 days. An easy to use, novel peel and place dressing has been designed for extended wear with the wound management advantages of ROCF while alleviating the challenges of tissue ingrowth.
Approach:
Paraspinal, full-thickness or deep muscle excisional wounds were created in 11 and 2 swine, respectively, dressings applied with continuous negative pressure at −125 mmHg, and dressings changed weekly. Full-thickness excisional wounds were treated for 13 days and deep muscle wounds for 35 days. Wound dimensions were assessed. Granulation tissue thickness and re-epithelialization were measured via digital morphometry. Tissue quality, fibrinous material prevalence, and dressing removal peel force were analyzed.
Results:
The peel and place dressing substantially reduces dressing tissue ingrowth, is easy to remove with markedly low dressing peel force and promotes more granulation tissue at day 13 than ROCF with an interface layer. The extended-wear peel and place dressing, when applied to deep muscle wounds with weekly dressing changes, was applied for a total of 35 days. Successful wound closure was evident without any negative impact on wound healing.
Innovation:
This study assessed the wound management capabilities of an extended-wear peel and place NPWT dressing used until wound closure.
Conclusion:
The peel and place dressing is a suitable extended-wear NPWT dressing.

INTRODUCTION
Negative pressure wound therapy (NPWT), a technology that has revolutionized wound management, is indicated for wide-ranging wound types, including acute and traumatic wounds, 1 burns, 2 recalcitrant chronic ulcers, 3,4 dehisced wounds, and flaps and grafts. 5,6 The clinical outcomes of NPWT have been well documented through numerous publications over the past two decades. This large body of evidence has led to widely adopted NPWT usage across a spectrum of care settings worldwide, making NPWT an integral component of the wound care armamentarium.
Since the inception of NPWT over 25 years ago, reticulated open cell foam (ROCF) has been well-established as the dressing of choice for this wound management modality. 5,7 Much, if not most, of the literature elucidating the mechanisms of action of NPWT has utilized ROCF administered at −125 mmHg.
There is robust data indicating that the efficacy of NPWT, the physiological and biological mechanisms promoted by the therapy, is dependent upon the dressing material interfacing with the wound bed. 8 –12 McNulty et al., reported that NPWT with gauze stimulated less fibroblast migration and proliferation, key processes for the formation of granulation tissue to begin filling the wound deficit, than NPWT utilizing ROCF. 9 Parameters reflecting an improved energetic status, such as increased levels of cytochrome c oxidase, adenosine triphosphate/adenosine diphosphate, and energy charge, were also found following NPWT with ROCF treatment but not following NPWT with gauze. 12
In addition, genomic analysis indicated elevated expression of genes involved in angiogenesis and cytoskeletal regulation in rat tissues treated with NPWT plus ROCF versus NPWT plus gauze. 13 Additional cellular responses include significantly greater programmed cell death, or apoptosis, with NPWT with gauze. 9
While the use of ROCF with NPWT is efficacious in promoting wound healing, the inherent nature of ROCF limits the dressing wear time to the recommended 2–3 days to mitigate the potential risk of granulation tissue ingrowth. 14 Clinicians have been known to use nonadherent wound interface layers (IFLs) to mitigate this occurrence or to increase the dressing wear time. This practice has several drawbacks, including the shifting of the IFL between dressing changes. When this occurs, all or part of the wound bed is exposed to the ROCF which may lead to tissue ingrowth when the dressing is in place for longer than the recommended timeframe. Additional drawbacks are the increased dressing application and dressing change times required to trim the IFL to size and to secure it in place.
The aim of this study was to investigate whether a novel, easy to use, peel and place dressing would be as impactful as ROCF in promoting wound healing responses as an extended-wear NPWT dressing while mitigating the occurrence of tissue ingrowth in full-thickness and deep muscle excisional wounds utilizing a porcine model. The deep muscle wound study was allowed to progress to full wound closure to fully investigate the wound healing effects of the peel and place extended-wear dressing.
INNOVATION
The peel and place dressing is multilayered, including an ROCF polyurethane core for manifolding negative pressure. A silicone and acrylate adhesives hybrid drape for the periwound interface is incorporated for an all-in-one dressing (Fig. 1A). The peel and place dressing is a wound cover intended to provide less wound disruption than ROCF by reducing the potential of tissue ingrowth and foam adhesion to the wound bed (Fig. 1B). Ultimately, the utilization of the peel and place dressing may provide options to the current clinical treatment of wounds as an extended-wear 7-day NPWT dressing while promoting wound healing comparable or better than ROCF.
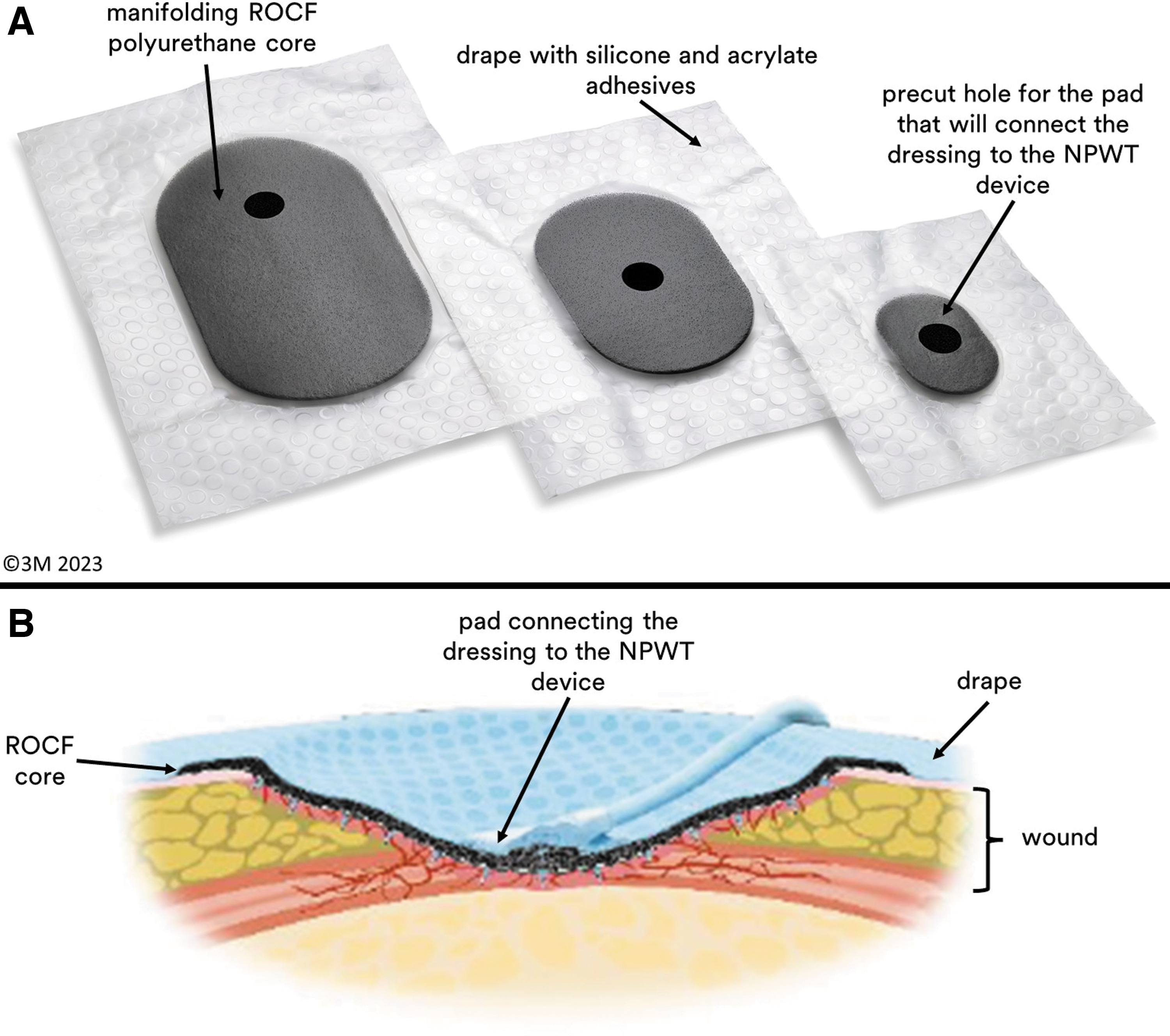
The Peel and Place Dressing.
CLINICAL PROBLEM ADDRESSED
Evidence indicates that patients living with chronic wounds find “dressing removal as the time of greatest pain”. 15 This pain exceeds that of the wound presence itself, with arterial and venous ulcers associated with more frequent pain. For at least half of the participants surveyed, the resolution of pain resulting from a dressing change took longer than 1 h for several different wound types ranging from ulcerations, surgical, trauma, and burn wounds. 15 The ideal clinical solution would be one that uses a nonstick surface to prevent pain upon removal and also extends the time between dressing changes, thereby reducing the number of painful events. This could greatly improve the dressing change experience for both the patient and caregiver.
To date, ROCF has not been utilized as an extended wear dressing due to the potential for granulation tissue ingrowth and embedded foam fragments remaining in the wound bed upon dressing removal. The accompanying wound bed disruption and the associated pain that may occur if left in place for greater than the recommended 72 h have deterred widespread longer use. The data presented herein of an easy-to-use, peel and place dressing demonstrates the dressing's capabilities of providing the advantages of ROCF while addressing tissue ingrowth as an extended-wear NPWT dressing.
MATERIALS AND METHODS
Surgical procedures and application of dressings and NPWT
All work was approved by the Institutional Animal Care and Use Committee (IACUC) and animal care complied with applicable national and local regulations. The animals were housed at a test facility accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care (AAALAC). The animals were fed twice daily with water ad libitum unless not feasible due to fasting the day before an anesthesia procedure or a scheduled sedation. Thirteen female (nulliparous and nonpregnant) swine (Sus scrofa domesticus) of Yorkshire cross were utilized weighing ∼80–95 kg at the time of the study. All animals were treated humanely, and the standards conformed to those of current ethical animal research practices.
The animals were sedated with a combination of Telazol® (Zoetis Services LLC, Parsippany, NJ) (100 mg/mL) and Xylazine® (Santa Cruz Biotechnology, Inc., Santa Cruz, CA) (100 mg/mL). General anesthesia was administered via isoflurane inhalation on the days of wound creation, dressing changes, and pretermination. For analgesia, buprenorphine (10 mg/mL) was administered as needed throughout the study. Excede® Sterile Suspension (Zoetis Services LLC) (100 mg/mL) was administered for antibiotics on wound creation day. The animals were prepared for surgical procedures by closely shaving the hair from catheter sites and surgical access sites (paraspinal sites/dorso-lateral back and sides). To remove gross debris, these areas were cleansed with mild soap and water followed by a chlorhexidine scrub. The surgical sites were prepared for aseptic surgery with a surgical scrub solution.
For 11 animals, 5 full-thickness 7.5 × 3 cm excisional wounds were created on each side of the spine for a total of 10 wounds per animal between the crest of the shoulders and the coccygeal tuberosity. The excisional wounds extended down to the subcutaneous fascial layer (just over the muscle). In 2 additional animals, 10 paraspinal deep muscle excisional wounds (approximate average of 2 cm deep) at 7.5 × 3 cm were created per animal. The distance between the wounds created was ∼3–6 cm. The day of wound creation was designated as day 0.
For the 11 animals with full-thickness wounds, the wounds were either dressed with 3M™ V.A.C.® Granufoam™ Dressing (3M Company, San Antonio, TX), an ROCF-based dressing, and Mepitel® One (Mōlnlycke, Gothenburg, Sweden), a perforated polyamide net with silicone adhesive, as a wound IFL or a novel peel and place dressing. The ROCF and IFL were cut to size to fill the wound, whereas the peel and place dressing was utilized as a wound cover with the dressing extending beyond the wound edges by at least 1 cm. The ROCF was secured in place in the wound with 3M™ V.A.C.® Drape (semiocclusive adhesive drape) (San Antonio, TX).
The peel and place dressing was secured over the wound and immediate periwound area with the incorporated 3M™ Dermatac™ Drape (silicone and acrylic hybrid adhesive drape) (San Antonio, TX). The dressings were connected to the NPWT device (3M™ ActiV.A.C.™ Therapy Unit, San Antonio, TX) via a 3M™ SensaT.R.A.C.™ Pad (San Antonio, TX).
A dressing change was performed for all wounds on day 7 (Fig. 2). The wounds were not debrided nor wiped clean after dressing removal. A new peel and place dressing was applied to wounds previously treated with the peel and place dressing and NPWT continued. At the 7-day dressing change, only IFL (with sterile gauze on top for exudate absorption, when required) was applied to wounds previously treated with ROCF + IFL. These wounds did not receive NPWT for the remainder of the study. The study was terminated at day 13 (13-Day Study Group) and the animals euthanized in an ethically acceptable method via heparinization (250–350 U/kg) and overdose of pentobarbital (390 mg/mL).
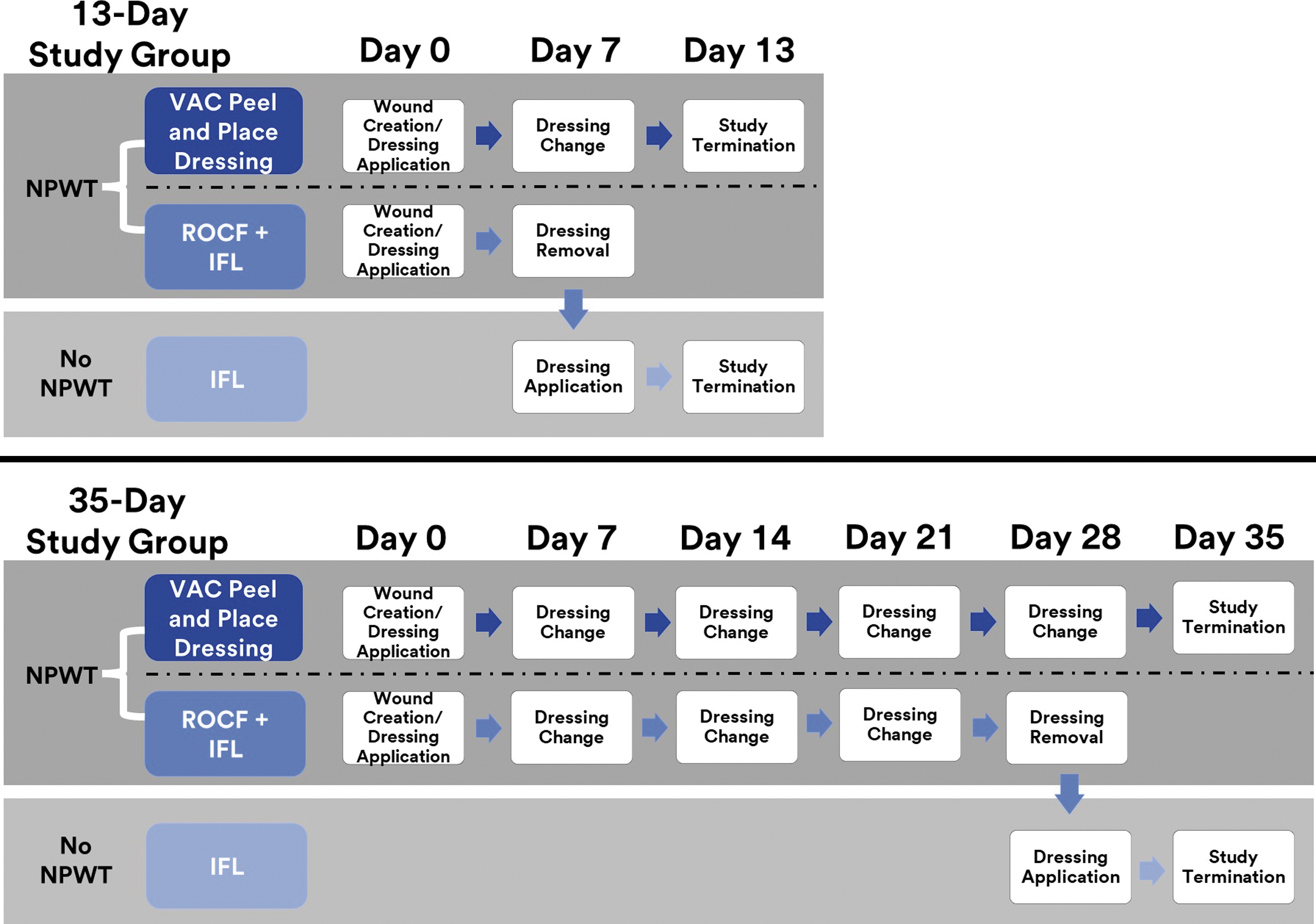
Study design, NPWT application, and dressing application for the 13-Day Study Group (n = 11 animals) and 35-Day Study Group (n = 2 animals). NPWT, negative pressure wound therapy; ROCF, reticulated open cell foam; IFL, interface layer.
The same dressings were also evaluated for the 2 animals with deep muscle excisional wounds. The study was terminated at day 35 (35-Day Study Group) with dressing changes on days 7, 14, 21, and 28 for both treatment groups. This allowed evaluation of the peel and place dressing up to wound closure. These wounds were also not debrided nor wiped clean after dressing removal on dressing change days. NPWT was administered for all wounds treated with the peel and place dressing throughout the duration of the study. NPWT was also administered for wounds treated with ROCF + IFL up to day 28. On day 28, only IFL was applied and NPWT discontinued until study termination and euthanasia (Fig. 2).
For both animal groups, treatment dressings were applied to the wound sites as per a randomization scheme that differed per animal. Continuous negative pressure was administered at −125 mmHg. Wheras it is recommended that ROCF dressing changes are performed every 2–3 days, the objectives of this study required that dressing changes occurred every 7 days until study termination for all treatment groups to evaluate the dressings' long-wear capabilities (such as healing progress via gross observations, wound dimension measurements, histopathological analyses, and dressing peel force evaluation). Therefore, an IFL was used with ROCF treatments to mitigate granulation tissue ingrowth as practiced in the clinical setting to extend wear time.
Traditionally, in the clinical setting, NPWT is eventually replaced by an advanced wound care dressing since NPWT is not generally used once a wound has fully granulated. To mimic a standard clinical care pathway, wounds treated with ROCF + IFL received only IFL application, and NPWT was discontinued until study termination on the last dressing change (day 7 or day 28) for both animal groups. The peel and place dressing treatment was assessed with NPWT throughout the study duration to determine if there were any limitations and/or impediments to healing.
Gross observations and wound dimensions measurement
At each dressing change and at study termination, the wound beds were visually inspected and prevalence of retained dressing fragments, if present, were noted. The level of bleeding upon dressing removal was also assessed. The periwound was examined for signs of irritation and tissue breakdown. These parameters were assessed semiquantitatively according to the following scale: none, mild, moderate, or significant. Three-dimensional (3D) digital images (inSight® camera system; eKare, Inc., Fairfax, VA) were acquired to enable measurements of wound area and volume on each procedure day.
Histological assessments
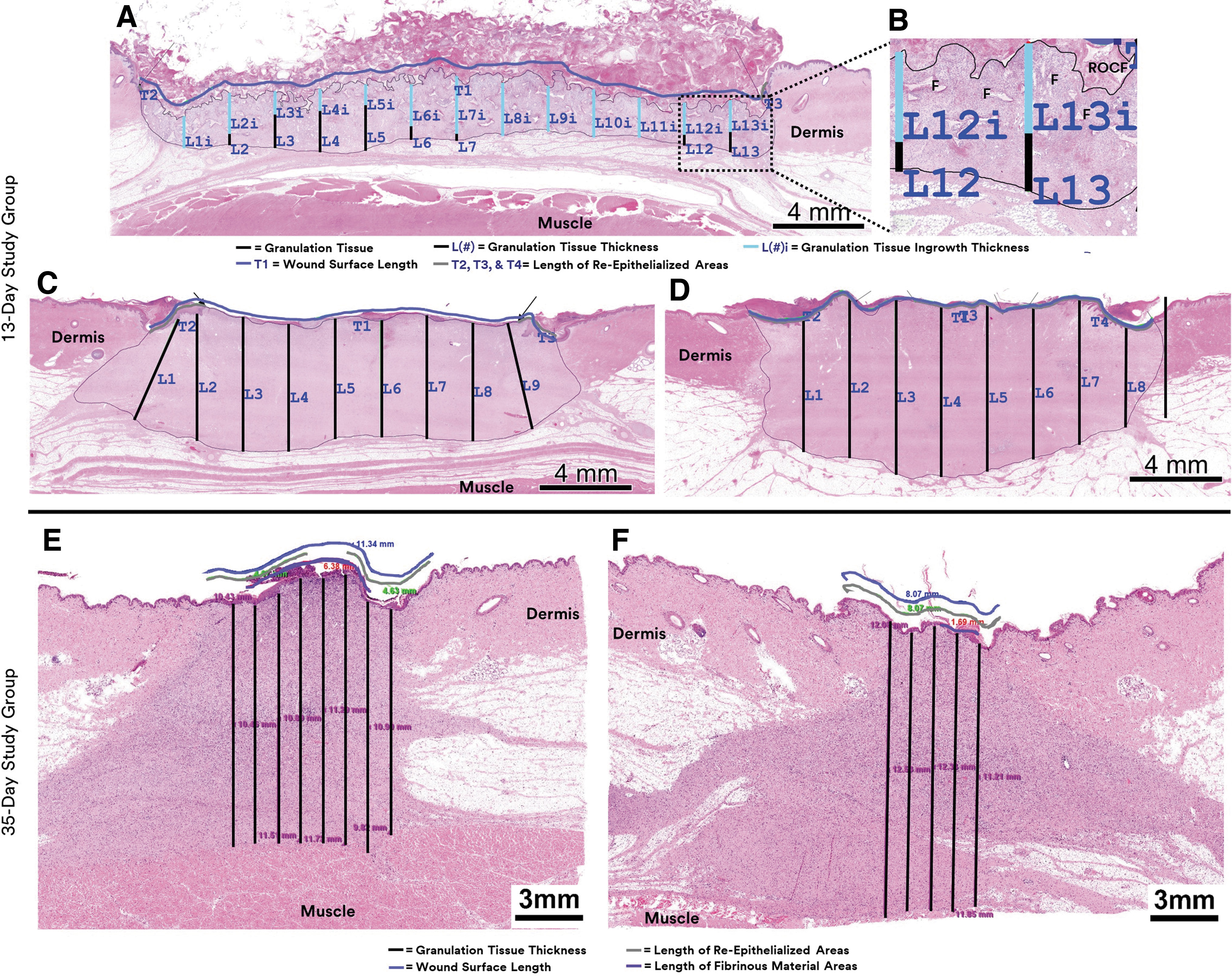
At study termination, all wounds in both study groups were excised en bloc with the excised portion extending to the underlying muscle and at least 1 cm beyond the wound edges. Before dressing removal for the 13-Day Study Group on day 13, ROCF + IFL/IFL dressings were bisected via a scalpel and the medial half of the dressing was removed from the wound bed according to the peel test methodology described below. The dressings were not removed from the lateral half of the wound bed to enable analysis of tissue ingrowth into the dressing material for that portion of the tissue sample (Fig. 3).
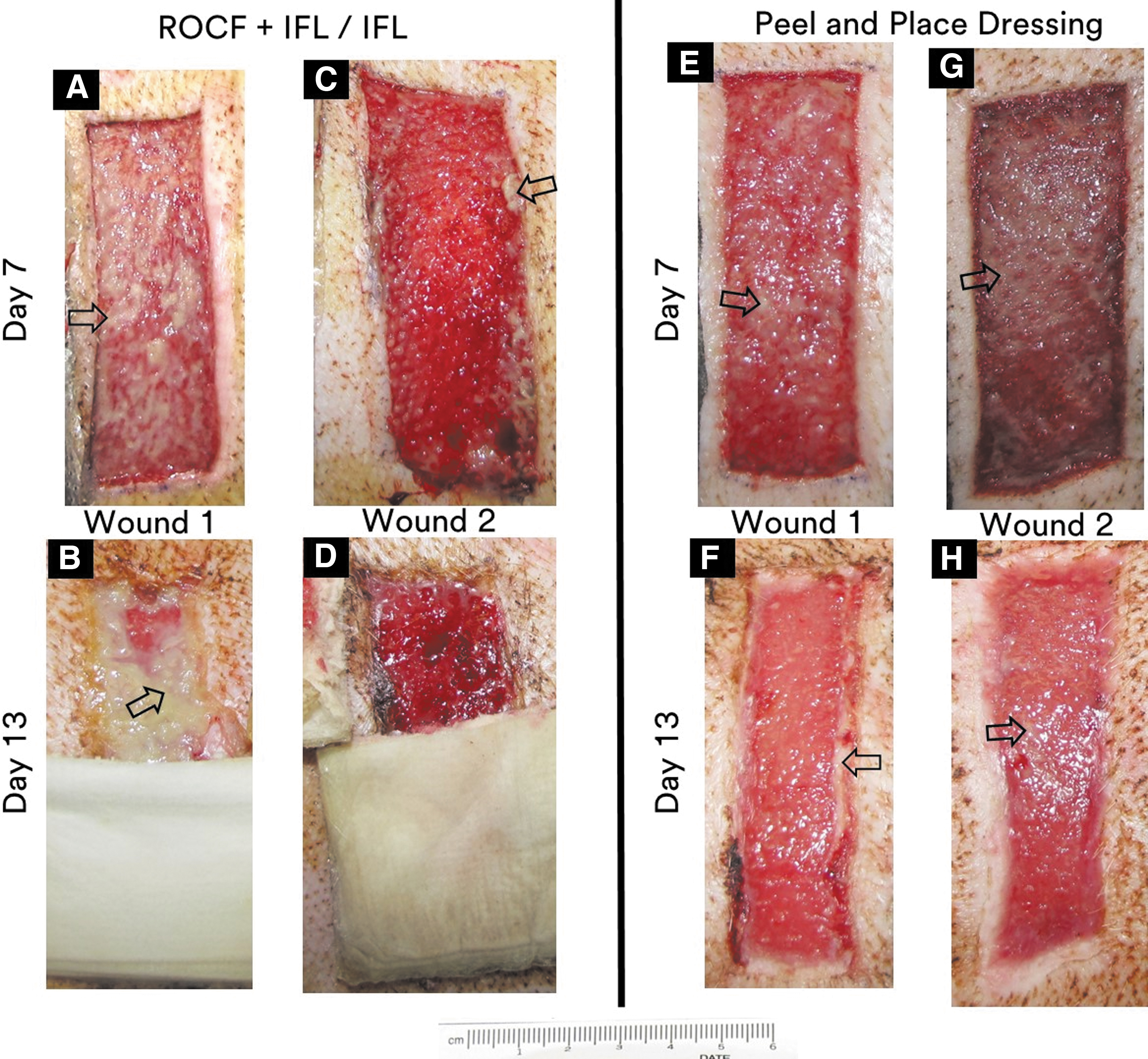
13-Day Study Group representative wounds.
The ratio of control to treatment sites was 1:2 in the 13-Day Study Group; therefore, dressing bisection was not required for the peel and place treatment group. Only half of the wounds in the peel and place treated group were utilized for tissue ingrowth assessment (n = 11); the remaining 11 wounds were utilized for peel force testing as described below. All dressings were removed at study termination for the 35-Day Study Group.
Tissue samples were fixed in 10% neutral buffered formalin for at least 72 h to allow for proper fixation of large tissue samples, then embedded in paraffin. Paraffin-embedded tissue sections were mounted on slides and stained with hematoxylin and eosin. The stained slides were digitally scanned and evaluated by a board-certified pathologist blinded to the treatment groups. Histomorphometry analyses were performed to provide quantitative measurements of granulation tissue thickness and percent re-epithelialization. Granulation tissue thickness included measurement of the wound bed and, for the 13-Day Study Group only, the tissue ingrowth into the dressing material. Histopathology evaluations provided semiquantitative assessment of other parameters of healing for both studies, such as the maturity of collagen, the level of vascularization, the extent of inflammation, and the presence of edema/seroma.
The inflammatory response was scored according to the presence of neutrophils, eosinophils, lymphocytes, plasma cells, macrophages, and multinucleated giant cells. Irritancy ranking analysis, based on the inflammatory response, was performed for the 35-Day Study Group, as per Annex E, the International Organization for Standardization (ISO) 10993-6:2016, Biological evaluation of medical devices—Part 6: Tests for local effects after implantation.
Prevalence of fibrinous material analysis
Digital wound images were acquired and used to quantify the prevalence of fibrinous material present on the wound beds for both animal groups. The 35-Day Study Group further investigated the impact of the fibrinous material on wound healing by allowing the study to progress to wound closure. The fibrinous material area coverage of each wound for each time point was determined via pixel count analysis of the digital wound images using ImageJ, version 1.53o (National Institutes of Health, Bethesda, MD). ImageJ enabled scaling, wound perimeter delineation, and color thresholding to capture total wound area and fibrinous material prevalence. The 2D wound images were imported into ImageJ and the wound isolated by selecting the area of analysis (the wound edges delineated).
Color thresholding was applied to exclude the reflected light captured on the wound image as this could be falsely discerned by the software as fibrinous material. The resulting image was then analyzed to provide the total area of the wound. The red-white and color spectrum was then adjusted to only include the areas of fibrinous material of the wound image. In essence, the image was converted to binary data, or a mask of the wound bed was created in black and white with the black areas designated as the fibrinous material. The black areas were then quantified to determine the prevalence of the fibrinous material compared to the total area of the wound.
Dressing peel force testing
Dressing peel force assessments were performed at dressing change (day 7) and pre-euthanasia for the 13-Day Study Group only. All wounds were subject to dressing peel force assessments on day 7 to measure the amount of force required to remove the dressing from the wound bed. At pre-euthanasia, peel force assessments were performed on half of wound sites treated with the peel and place dressing (n = 11 of 22). Only the bisected medial dressing halves of the ROCF treatments were evaluated.
The animal was placed on a tilting table to allow for a bespoke peel tester device to peel the dressing from the wound at ∼180˚ relative to the peel tester. Exact angles were measured via a digital protractor for each wound evaluated. The dressings were peeled from the medial to the lateral direction at a constant rate and peel force readings recorded through NI™ LabView (NI, Austin, TX) as measured via a Shimpo digital force gauge (Electromatic Equipment Co., Lynbrook, NY).
Statistical analysis
All data are reported as means with standard error of the means (SEM) or medians with interquartile ranges (IQR). For the 13-Day Study Group, the number of wounds per treatment group were 9–11 except for wound dimensions analysis and dressing peel force, which were 21 or 22 wounds. For the 35-Day Study Group, the number of wounds were 8 and 12 for the ROCF and peel and place dressing treatment groups, respectively. Statistical analysis was performed using JMP 13.0 software (Cary, NC). Shapiro–Wilk goodness-of-fit test was used to assess normality. Analysis of variance (ANOVA) was performed for measures that were normally distributed, such as granulation tissue thickness and percent re-epithelialization. The Tukey method for pairwise comparisons was then used if differences were detected via ANOVA at the alpha level of 0.05.
For nonnormally distributed measures, such as collagen maturation, vascularization, inflammation, and prevalence of edema/seroma, the nonparametric ANOVA (Kruskal–Wallis) was utilized to test for differences. The Steel-Dwass (Tukey's nonparametric) test was then performed for pairwise comparisons if differences were detected via Kruskal–Wallis. For the 35-Day Study Group repeated measures, such as wound volume, wound area, and fibrinous material prevalence, the multivariate analysis of variance (MANOVA) was utilized to determine the impact of treatment over time. Alpha was set at 0.05. All data are recorded in the electronic laboratory notebook BIOVIA I Workbook, version 2018.1.0.375 (Dassault Systemes, Waltham, MA).
RESULTS
Improved wound healing quality with peel and place dressing and NPWT
Wound and periwound visual evaluation
13-Day Study Group
The full-thickness excisional wounds treated with either dressing indicated healing progress throughout the study duration (Fig. 3). The wound fill was discernable on day 7 for all wounds with a corresponding decrease in wound area by day 13. In addition, no visible dressing fragments were found in any of the wound beds. Fibrinous material presence, at varying degrees, was visible on the surface of most wounds on day 7. Healthy tissue was present underneath the fibrinous material, as illustrated by the healing progress observed on day 13. The prevalence of this fibrinous material noticeably decreased on day 13. There were no visual adverse signs to indicate that healing was hampered, regardless of fibrinous material prevalence. In addition, there was no aberrant smell during dressing removal. Finally, the periwound showed no signs of tissue breakdown nor irritation, regardless of treatment group, at the time points assessed.
35-Day Study Group
All deep muscle excisional wounds also showed healing progress throughout the study duration (Fig. 4). There was mild to moderate bleeding associated with the dressing removal of ROCF + IFL/IFL on day 7. In some instances, the IFL shifted and certain parts of the wound interfaced with the ROCF directly, thereby inducing bleeding upon ROCF removal in those areas. At times, the shifting was considerable causing a substantial portion of the IFL to fold, which also caused wound bed disruption upon removal. The IFL folding was witnessed at the earlier time points, days 7 and 14. Bleeding was also observed in wounds where the IFL shifting did not occur. No dressing fragments were found in any of the wound beds. Greater wound fill was visually observed on day 7 with the peel and place dressing-treated wounds compared to ROCF + IFL/IFL-treated wounds.
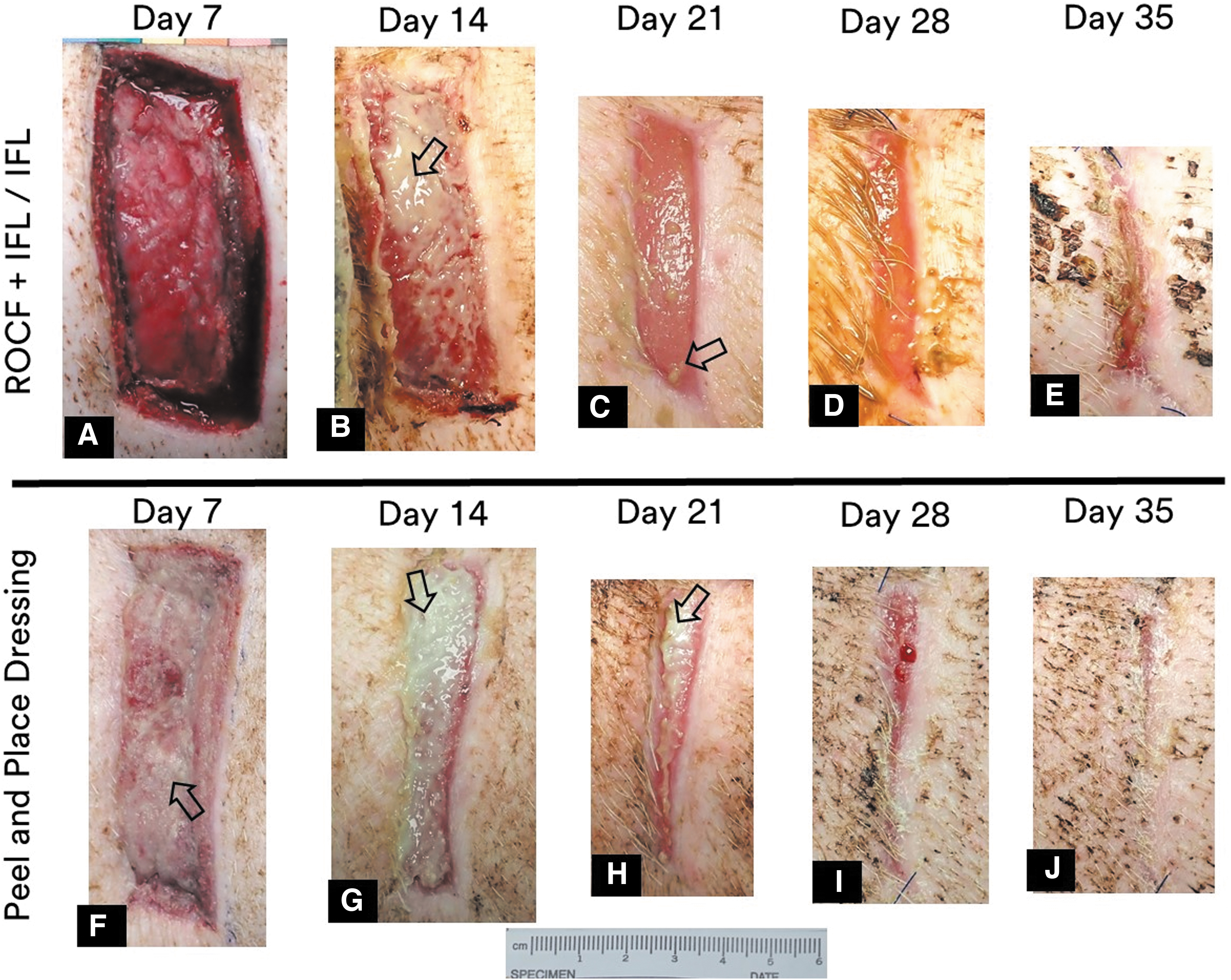
35-Day Study Group representative wounds from each treatment group on each procedure day. Wound treated with ROCF + IFL/IFL at
Differences in wound fill between treatment groups at other time points were not as readily obvious by eye. The decrease in wound area for all wounds as the study progressed was readily obvious (Figs. 4 and 5).
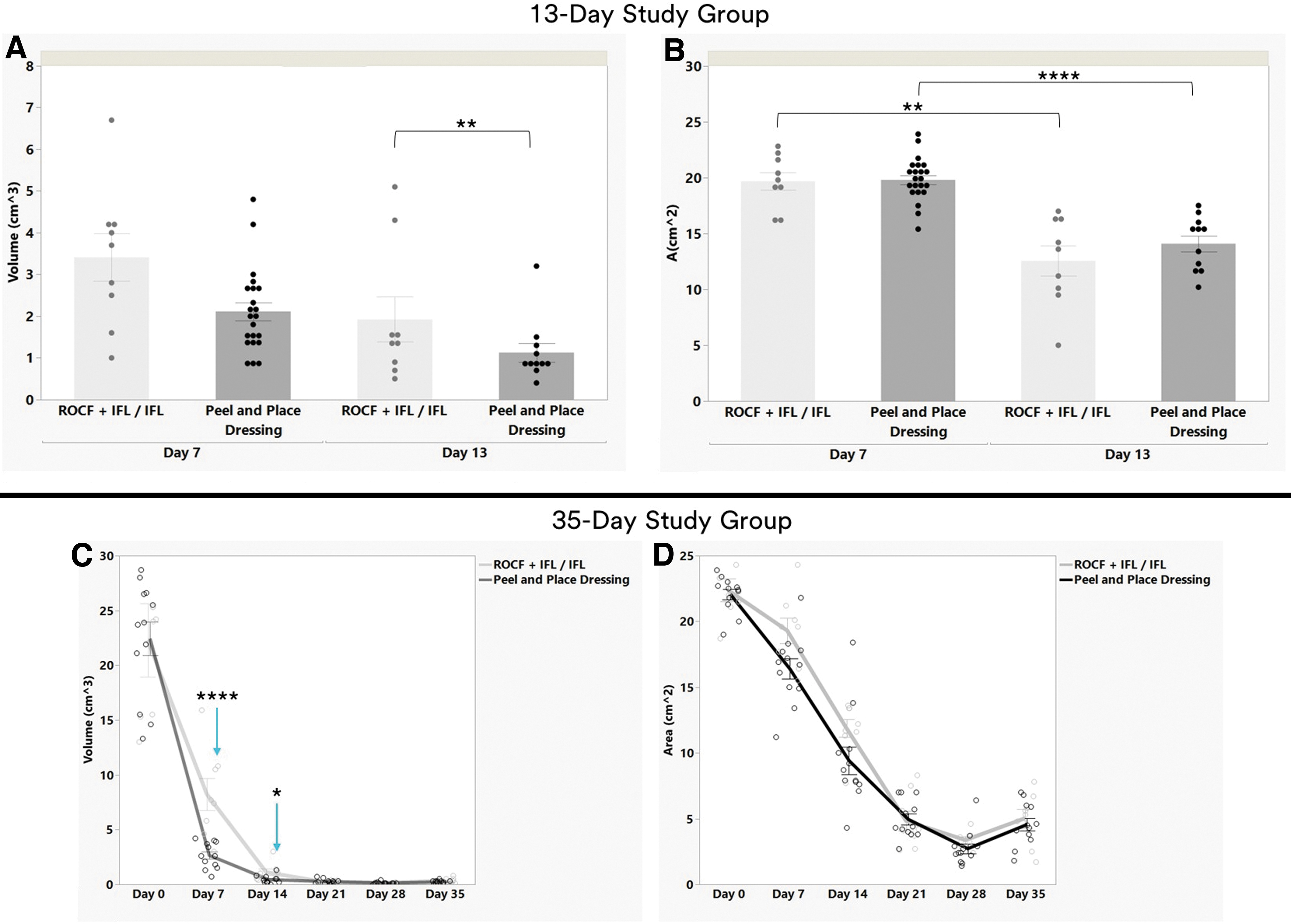
Wound dimensions analysis (mean ± SEM).
The deep muscle excisional wounds in the 35-Day Study Group also exhibited the presence of fibrinous material. The fibrinous material was present on the wound beds of both treatment groups and was in greater prevalence on days 7 and 14. As with the 13-Day Study Group, healthy tissue was present underneath the fibrinous material, as illustrated by the continued healing observed throughout the study duration. The amount of fibrinous material evident on the wounds decreased with time. By day 21, most of the wound did not have fibrinous material present. As seen in the 13-Day Study Group, there were no visual adverse signs, no aberrant smell, and the periwound showed no signs of tissue breakdown nor irritation.
Wound volume (wound deficit) and wound area analysis
13-Day Study Group
There was no difference in the average volume between ROCF + IFL/IFL-treated wounds and peel and place-treated wounds, mean ± SEM were 3.4 ± 0.6 cm3 and 2.1 ± 0.2 cm3, respectively, on day 7 (Fig. 5A). Although on day 13, the mean volume of ROCF + IFL/IFL-treated wounds was 1.7 times greater at 1.9 ± 0.5 cm3 versus 1.1 ± 0.2 cm3 for peel and place dressing-treated wounds (p < 0.01). No significant differences were found in the wound area evaluations between treatment groups (Fig. 5B). The wound areas within a treatment group did show significant decreases (p < 0.05) between day 7 and 13, mean ± SEM were 19.7 ± 0.8 cm2 versus 12.6 ± 1.3 cm2 for ROCF + IFL/IFL and 19.8 ± 0.4 cm2 versus 14.1 ± 0.7 cm2 for the peel and place dressing.
35-Day Study Group
For both treatment groups, the wound volumes of the deep muscle wounds showed a downward trend from day 0 to 14 with the volumes nearing 0 cm3 as the study progressed (Fig. 5C). There were significant differences in the wound volumes between treatment groups at certain time points. The average volumes at day 0 were not different between the treatment groups, 22.3 ± 3.3 cm3 for ROCF + IFL/IFL-treated wounds and 22.4 ± 1.5 cm3 for peel and place dressing-treated wounds. At day 7, the average volume of ROCF + IFL/IFL-treated wounds was three times greater at 8.2 ± 1.5 cm3 versus 2.7 ± 0.3 cm3 for peel and place dressing-treated wounds (p < 0.0001).
At day 14, it was 2.8 times greater with ROCF + IFL/IFL at 1.1 ± 0.3 cm3 and 0.4 ± 0.1 cm3 for peel and place dressing wounds (p < 0.05). No differences were found at days 21, 28, and 35 between treatment groups. Analysis of the rate of change in wound volume indicate a significant difference between ROCF + IFL/IFL and peel and place dressing treatments (p < 0.0001). The peel and place dressing promoted a significantly faster rate of granulation tissue fill compared to ROCF + IFL/IFL.
The wound areas showed a downward trend from day 0 to day 28 for both treatment groups (Fig. 5D). The wound areas decreased approximately six-fold for the ROCF + IFL/IFL group and approximately seven-fold for the peel and place dressing group from wound creation to study termination. The average area at day 0 for ROCF + IFL/IFL-treated wounds was 22.4 ± 0.9 cm2 and 22.1 ± 0.4 cm2 for peel and place dressing-treated wounds. By study termination, the average wound areas were 5.0 ± 0.7 cm2 and 4.6 ± 0.5 cm2, for ROCF + IFL/IFL-treated wounds and peel and place dressing-treated wounds, respectively.
Unlike wound volume, there were no differences found between treatment groups at any of the time points evaluated. Also, unlike wound volume, no significant difference was found in the rate of change in wound area between ROCF + IFL/IFL and peel and place dressing treatments. Although the average wound area increased from day 28 to 35 for both treatment groups, the difference is not statistically significant.
Histomorphometry analysis of granulation tissue thickness and percent re-epithelialization
13-Day Study Group
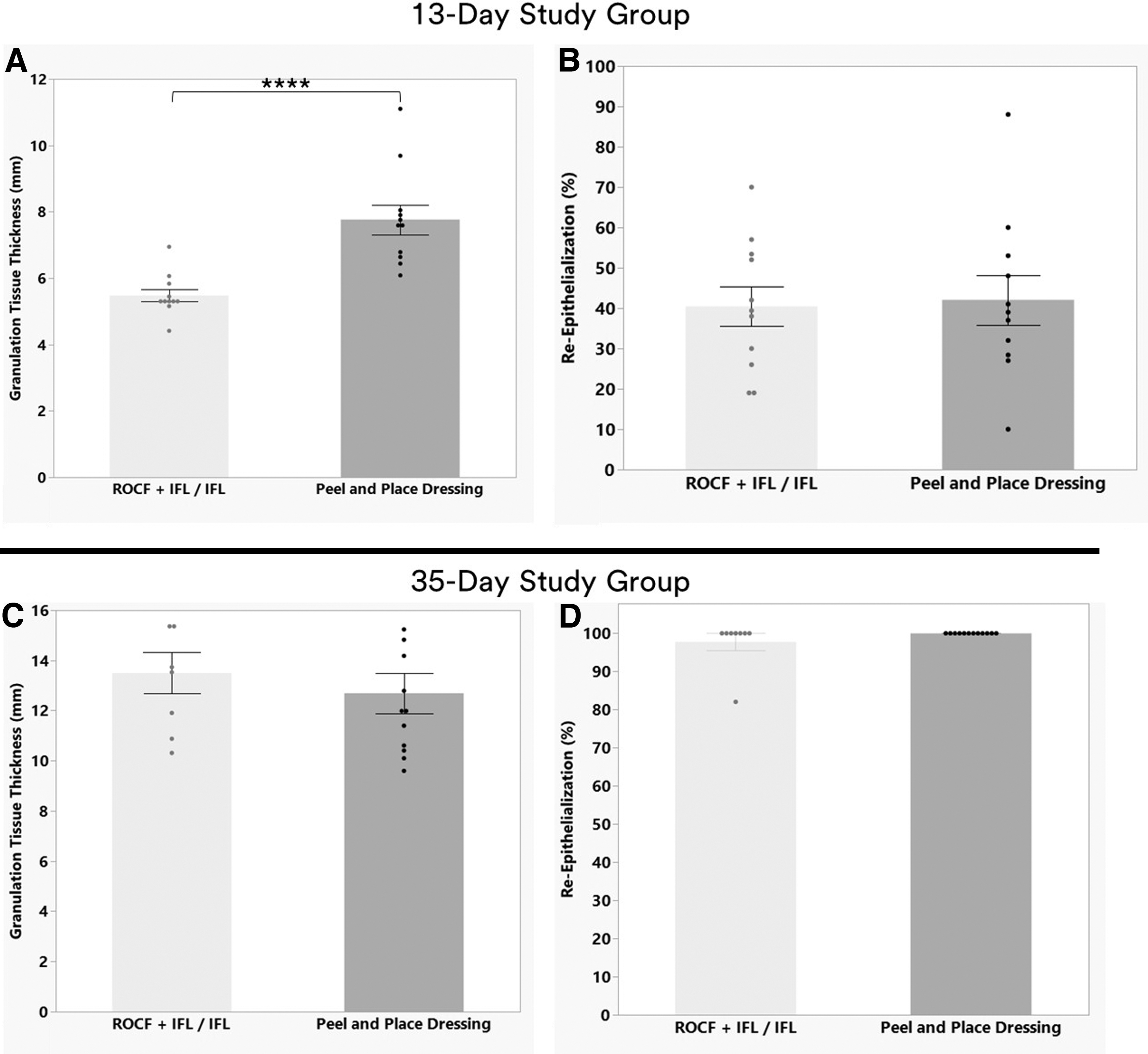
For the full-thickness excisional wounds study, the peel and place dressing induced significantly greater (1.4 times) average granulation tissue thickness (Fig. 6). Mean ± SEM were 7.8 ± 0.4 mm versus 5.53 ± 0.2 mm for ROCF + IFL/IFL (p < 0.0001) at day 13 (Figs. 6B, C, and 7A). There were no instances of granulation tissue ingrowth into the dressing for either treatment group. No differences were found between treatment groups in relationship to percent re-epithelialization (Figs. 6B, C and 7B). Both treatment groups promoted an average of ∼41% re-epithelialization.
Representative histology images.
Histomorphometry analysis (mean ± SEM). 13-Day Study Group for
35-Day Study Group
The treatment groups induced similar levels of average granulation tissue thickness in the deep muscle tissue excisional wounds on day 35 at 13.5 ± 0.8 mm and 12.7 ± 0.8 mm for ROCF + IFL/IFL and peel and place dressing, respectively (Figs. 6E, F, and 7C). The average re-epithelialization levels at day 35 were also comparable with mean ± SEM of 97.8% ± 2.2% for ROFC + IFL/IFL wounds and 100% ± 0.0% for peel and place dressing wounds (Figs. 6E, F, and 7D).
Granulation tissue quality at day 13 and irritancy ranking at day 35 via histopathology
13-Day Study Group
The deepest 30% of the wound bed (deep wound bed) was assessed for granulation tissue quality on day 13 via histopathology. No differences were found between treatment groups. The full-thickness excisional study wounds scored (median ± IQR) a “3” (3 ± 0) regarding collagen maturation (Fig. 8A). The wounds exhibited notable collagen deposition with dense stroma. The stroma was organized with collagen fibers oriented parallel to the skin surface and the vasculature of all the wounds was “moderate” (3 ± 0.2) (Fig. 8B). The inflammatory levels promoted were “rare” to “minimal” (1 ± 1 and 1 ± 0, for ROCF + IFL/IFL and peel and place dressing, respectively) and edema/seroma levels ranged from “absent” to “minimal” (Figs. 8C D).

Box plots of 13-Day Study Group histological assessments of granulation tissue quality at day 13. Semiquantitative scoring of
35-Day Study Group
The local responses of the deep muscle excisional study wounds were evaluated as per Annex E, ISO 10993-6 for irritancy ranking. Total irritancy score = (neutrophils + eosinophils + lymphocytes + plasma cells + macrophages + multinucleated giant cells + necrosis) × 2 + (vascularization + fibrosis + entrapped adipocytes). A ranked irritancy score was determined for the peel and place dressing by subtracting the average ROCF + IFL/IFL (clinically relevant control) irritancy score from the average peel and place dressing irritancy score. Since the mean total irritancy score for the ROCF + IFL/IFL was greater than that of the peel and place dressing, thereby resulting in a negative rank irritancy/reactivity score, the score was considered to be 0. The peel and place dressing was a nonirritant.
Significant decrease in prevalence of fibrinous material on the wound beds of peel and place dressing as the study progressed
13-Day Study Group
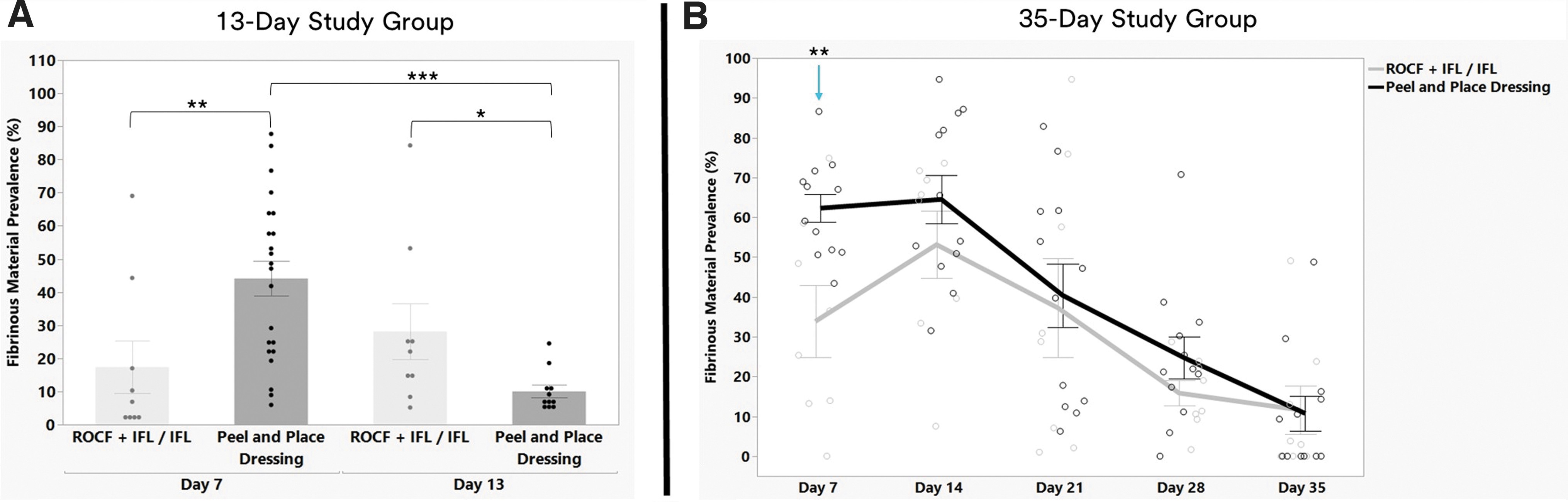
The presence of fibrinous material on the full-thickness excisional wound bed surfaces was notable, specifically at earlier time points, regardless of treatment group. Although there was greater prevalence of the fibrinous material on the peel and place dressing wound beds on day 7, mean ± SEM at 44.1% ± 5.3%, versus the ROCF + IFL/IFL wound beds, 17.4% ± 7.9% (p < 0.01), this trend was reversed at study termination (Fig. 9A). By day 13, the fibrinous material presence was 10.0% ± 1.9% and 28.1% ± 8.4% for the peel and place dressing and ROCF + IFL/IFL, respectively (p < 0.05). The fibrinous material presence was similar for ROCF + IFL/IFL on days 7 and 13 but was significantly decreased as the study progressed for the peel and place dressing group, 44.1% ± 5.3% and 10.0% ± 1.9%, respectively (p < 0.001).
Percent prevalence of fibrinous material at different time points.
35-Day Study Group
Fibrinous material presence was also observed on the wound bed surfaces of the deep muscle wounds. The fibrinous material was also most prevalent at the earlier time points for both treatment groups, with the most prevalence on day 14 (Fig. 9B). There was greater fibrinous material for the peel and place dressing wounds versus the ROCF + IFL/IFL wounds at 62.3% ± 3.6% and 33.9% ± 9.0%, respectively, on day 7 (p < 0.01). Except for day 7, there were no differences regarding the prevalence of fibrinous material between the treatment groups at other time points. From day 14 onward, the amount of fibrinous material decreased for both treatment groups to 13.3% ± 6.6% and 9.6% ± 4.0%, by day 35 for ROCF + IFL/IFL-treated wounds and peel and place dressing-treated wounds, respectively.
At the later time points, such as days 21, 28, and 35, the presence of long hairs laying on the little that is left of the wound beds can be erroneously discerned as fibrinous material by the software despite all efforts to prevent such false positives. There was no significant difference in the rate of change in the prevalence of fibrinous material between ROCF + IFL/IFL and peel and place dressing treatments.
Low force required to remove the peel and place dressing from the wound bed
The force required to remove the dressing was measured at every time point except for wound creation (day 0) for the study assessing full-thickness excisional wounds in the 13-Day Study Group. There were no differences regarding average dressing peel force between treatment groups at dressing change day (day 7) or study termination (day 13) (Fig. 10A). The dressing peel force ranged from mean ± SEM of 0.3 ± 0.03 N (peel and place dressing at day 7) to 0.5 ± 0.08 N (ROCF + IFL/IFL at day 13). For maximum dressing peel force, the peel and place dressing was significantly lower at day 7 compared to ROCF + IFL/IFL, 1.1 ± 0.2 N and 1.7 ± 0.2 N, respectively, (p < 0.05) (Fig. 10B). There were no differences between time points within a treatment group in relationship to maximum dressing peel force.
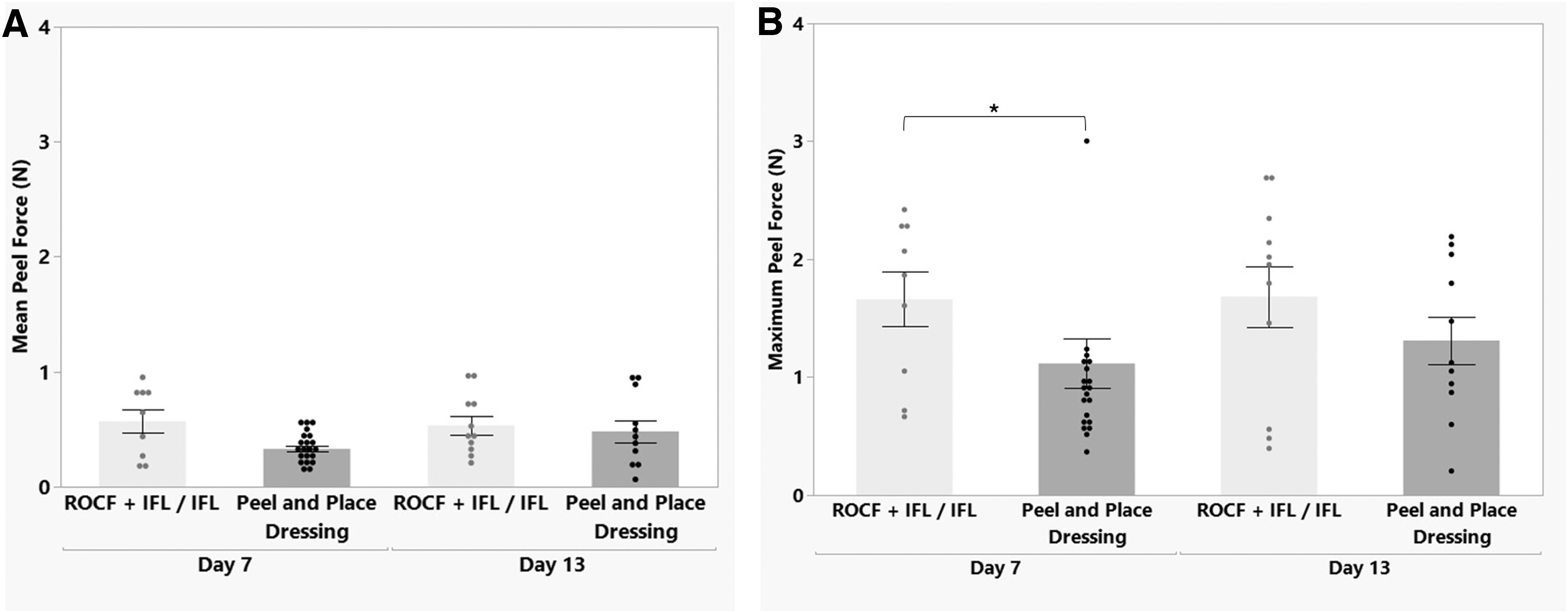
13-Day Study Group dressing removal peel force at days 7 and 13 (mean ± SEM).
DISCUSSION
The mechanisms by which NPWT with ROCF affects the wound healing process has been investigated for over 20 years. In addition to numerous reports from in vitro, preclinical, and clinical studies, finite element modeling has also been applied to predict tissue displacement and stress distributions based on the foam-wound bed interaction under negative pressure. 11,16,17 Many of the physiological responses promoted by NPWT with ROCF, as published by investigators of laboratory and theoretical studies, contribute to the proliferation stage of wound healing. 18,19 NPWT with ROCF has been demonstrated to impart macrostrain at the tissue level by drawing the wound edges together, thereby facilitating wound shrinkage. 10 At the cellular level, the mechanical forces of microstrain cause cell stretch that stimulates cell proliferation and chemotaxis, 8,9 consequently stimulating wound bed vascularization 20 and granulation tissue formation. 7
The molecular mechanisms leading to these biological effects include increased gene expression of wound healing biomarkers such as vascular endothelial growth factor (VEGF) 21 and fibroblast growth factor 2 (FGF2), 22,23 as key stimulators of angiogenesis, and platelet-derived growth factor (PDGF) 12 and transforming growth factor beta (TGFβ), 12,23 as crucial factors in granulation tissue production. 24 Adequate blood flow via neovascularization provides oxygen to the wound bed, is required to prevent the progression of the injury, 25,26 and is critical for proceeding through the healing continuum. Removal of exudate is also an important mechanism contributing to the proliferation stage. Exudate/edema removal allows for a stable wound site to promote the fragile process of re-epithelialization in burn patients receiving a skin graft or for split-thickness skin graft donor sites. 2,27
The authors report upon a promising easy to use, peel and place, extended wear NPWT dressing that demonstrates much of the described physiological and biological responses contributing to the proliferation phase of the wound healing continuum seen with ROCF. The peel and place dressing, with a single dressing change at day 7 during a 13-day study, promoted 1.4 times greater granulation in full-thickness excisional wounds than those treated with ROCF with an IFL for 7 days followed by treatment with only an IFL for the following 6 days (no NPWT for the last 6 days). This difference could be due to the dual benefits of the continuation of NPWT and using the peel and place dressing longer for the wounds treated with the peel and place dressing.
The tissue quality of the peel and place dressing-treated wounds was comparable to wounds treated with ROCF + IFL/IFL for per key parameters, such as collagen maturation and level of vasculature via histology. The extent of re-epithelialization was also comparable between the two groups. These finding are similar to results from a previous 22 swine 7-day study conducted assessing the peel and place dressing versus ROCF (no IFL). 28
Deep muscle excisional wounds were also treated with −125 mmHg negative pressure and either the peel and place dressing or the ROCF + IFL/IFL treatment for 35 days with weekly dressing changes. Analysis of wound dimensions acquired via 3D images confirmed the visibly apparent reduction in wound volume as the study progressed. At earlier time points, days 7 and 14, the wounds treated with the peel and place dressing were smaller in volume compared to ROCF + IFL/IFL. From day 21 on, ROCF + IFL/IFL-treated wounds became more similar to the wounds treated with the peel and place dressing. By day 35, the granulation tissue thickness and percent re-epithelialization promoted by both treatment groups were equivalent, although it is noteworthy that the peel and place treatment did promote a faster rate of wound fill compared to ROCF + IFL/IFL treatment.
These results illustrated that the peel and place dressing, in conjunction with NPWT, was successful in promoting granulation as well as in supporting re-epithelization. Numerous studies have shown that ROCF successfully promotes wound filling, but application of ROCF under NPWT until wound closure, or full re-epithelialization, is not a common clinical practice outside of bolstering split thickness skin grafts, flaps, and burn wounds. The success of the peel and place dressing, with very minimal dressing changes, suggests impactful positive biological outcomes.
There were no adverse effects noted with the peel and place dressing. Inflammatory levels were not elevated, and presence of edema/seroma was not detected. This study also demonstrated that there was no observed tissue ingrowth or retained dressing fragments on the wound beds with either the peel and place dressing or ROCF + IFL/IFL. The lack of both tissue ingrowth and wound bed disruption observed in the peel and place dressing treatment have been consistent in all studies conducted to date and can be directly correlated to the markedly low peel forces, also consistently demonstrated, for dressing removal. This, in turn, is a predictor of the ease of removal by the peel and place dressing.
This is in contrast to some of the surprising experiences concerning ease or removal with the IFL in the ROCF + IFL/IFL-treated wounds when there was considerable shifting, and consequently, folding of the IFL upon itself. In these instances, the IFL was not so readily visible at dressing removal. The clinical concern would be the possibility of inadvertently leaving the IFL at dressing removal, and the potential ingrowth and wound bed disruption in the areas in direct contact with the ROCF during extended wear time.
In all studies performed to date with this dressing, some fibrinous material has been present on the surface of the wounds at the initial dressing change (day 7). Fibrinous material was also present on the wound beds of the ROCF + IFL/IFL. Although on day 7 in the 13-Day Study Group, there was 2.6 times more area covered by the fibrinous material on the peel and place dressing-treated wounds than those treated with ROCF + IFL/IFL, this trend was reversed by day 13. By day 13, the wounds treated with the peel and place dressing contained almost three times less area covered by the fibrinous material than the wounds treated with ROCF + IFL/IFL.
To better understand if the presence of the fibrinous material negatively impacted wound healing, a study was performed that allowed progression of the wound to closure via the deep muscle excisional wounds. As with the 13-Day Study Group, there was more fibrinous material prevalence on the wound beds of the peel and place dressing at earlier time points. In the 35-Day Study of deep muscle wounds, the level of fibrinous material present on the peel and place-treated wounds was 1.8 times greater on day 7 than the ROCF + IFL/IFL-treated wound beds. No differences, however, were evident in the occurrence of the fibrinous material between treatment groups at the remaining timing points. It was also visibly evident that the presence of fibrinous material lessened over time for both treatment groups.
There were no signs of adverse effects visually, and pathology indicated that there were no impediments to healing induced by the presence of this material on either the wound bed or adjacent periwound. These are consistent findings with all studies conducted thus far. It is worth noting that while the ROCF wounds in the recently published article, Allen et al., 28 lacked the occurrence of fibrinous material, this was not the case for the ROCF + IFL/IFL-treated wounds in the current study. The presence of the fibrinous material with ROCF with an IFL was surprising as this has not been historically reported in the literature. Anecdotal evidence, however, has indicated the presence of similar-looking fibrinous debris/material on wound bed surfaces when skin substitutes are utilized. Clinicians have left the fibrinous material in place if the wound is progressing along its healing trajectory, and the wound continues to heal.
To complement the demonstrated extended-wear duration and the ease-of removal qualities, user studies were conducted to examine the ease-of-use aspects of the peel and place dressing. The user studies indicated several important points: the health care practitioners noted that the peel and place dressing was easier and simpler to use, and that application of the peel and place dressing was 6 times faster compared to ROCF (no accompanying IFL). 29
The findings reported herein indicate that the biological response promoted by the peel and place dressing is either comparable to or more robust than biological response promoted by ROCF (with or without an IFL). Future studies are anticipated to further elucidate the mechanisms of action by which the peel and place dressing promoted such wound responses and to determine how the mechanisms could differ from those of ROCF.
KEY FINDINGS
A newly designed, easy to use, peel and place dressing, when assessed as an extended 7-day wear NPWT dressing, promoted 1.4 times greater granulation tissue formation in full-thickness excisional wounds on day 13 than ROCF with an IFL/IFL only.
The peel and place dressing was successful in closing deep muscle excisional wounds with NPWT application for 35 days as an extended-wear dressing with only 4 weekly dressing changes.
No tissue ingrowth was observed with the peel and place dressing; this correlates directly to the markedly low peel force required for dressing removal.
Footnotes
ACKNOWLEDGMENTS
The authors thank Pedro Maldonado and Deepanjan Ghosh, PhD, for their efforts in data quality control. The authors thank Meridith Peratikos for statistical analysis guidance. Finally, the authors acknowledge Ben Stokes, PhD, and Rosella Kendrick for their efforts in the usability studies.
AUTHOR CONFIRMATION
The study was conceptualized by Diwi Allen, Dr. Kristine Kieswetter, and Dr. Timothy Robinson. The study investigation was executed by Diwi Allen, Marisa Schmidt, Samantha Mann, and Dr. Kristine Kieswetter. Formal analyses were performed by Diwi Allen and Samantha Mann. Visualization was conducted by Diwi Allen and Samantha Mann. Project supervision was conducted by Dr. Kristine Kieswetter. The original draft of the article was prepared by Diwi Allen.
AUTHOR DISCLOSURE AND GHOSTWRITING
This article was expressly written by the listed authors. No ghostwriters were used to write this article.
ABOUT THE AUTHORS
The authors and acknowledged contributors of this article are affiliated with the Medical Solutions Division at 3M Health Care.
Funding Information
Financial support was provided by 3M Health Care.
