Abstract
Significance:
The Wound Healing Foundation recognized the need for consensus-based unbiased recommendations for the treatment of wounds. As a first step, a consensus on the treatment of chronic wounds was developed and published in 2022. The current publication on acute wounds represents the second step in this process. Acute wounds may result from any number of conditions, including burns, military and combat operations, and trauma to specific areas of the body. The management of acute wounds requires timely and evidence-driven intervention to achieve optimal clinical outcomes. This consensus statement provides the clinician with the necessary foundational approaches to the causes, diagnosis, and therapeutic management of acute wounds. Presented in a structured format, this is a useful guide for clinicians and learners in all patient care settings.
Recent Advances:
Recent advances in the management of acute wounds have centered on stabilization and treatment in the military and combat environment. Specifically, advancements in hemostasis, resuscitation, and the mitigation of infection risk through timely initiation of antibiotics and avoidance of high-pressure irrigation in contaminated soft tissue injury.
Critical Issues:
Critical issues include infection control, pain management, and the unique considerations for the management of acute wounds in pediatric patients.
Future Directions:
Future directions include new approaches to preventing the progression and conversion of burns through the use of specific gel formulations. Additionally, the use of three-dimensional bioprinting and photo-modulation for reconstruction is a promising area for continued discovery.

SCOPE AND RELEVANCE
This body of work is intended to be a guide to clinicians in the treatment of acute wounds. Herein, we define an acute wound and focus on its management, including preparation of the wound bed, infection control, and pain control, with specific emphasis on the management of facial, torso, hand, foot, high velocity, and burn wounds. Lastly, we include techniques for optimizing long-term outcomes in the form of scar prevention.
TRANSLATIONAL RELEVANCE
This body of work does not focus on bench research or its translation to clinical practice.
CLINICAL RELEVANCE
The intended purpose of this expert consensus statement is to provide clinicians with practical guidance for the bedside treatment of acute wounds. As such, the focus is purely on clinical application. In serving as a guide for the practicing clinician, the included consensus statements were required to have at least an 80% agreement rate among the experts who participated in the panel. The content is broad and intended to be comprehensive to aid clinicians and learners caring for the broad spectrum of acute wounds encountered in any clinical care setting.
BACKGROUND
The Wound Healing Foundation recognized the need for consensus-based, unbiased recommendations for the treatment of wounds. As a first step, a consensus on the treatment of chronic wounds was developed and published in 2022. 1 The current publication on acute wounds represents the second step in this process.
Acute wounds can be created intentionally (surgical incisions), sustained accidentally by traumatic injury, or occur as a result of disease. Acute wounds are managed in a variety of clinical settings – the operating room, outpatient clinics, urgent care emergency centers, or even in combat environments – and by a diverse group of providers. This publication serves as a review of the current evidence on the care of acute wounds as presented by a diverse group of experts and practitioners. The publication is organized in treatment categories such that it is universally applicable to as many types of acute wounds as possible.
METHODS
The Consensus Panel members were invited by the two editors – Drs. Olutoye and Eriksson – both of whom are members of the Wound Healing Foundation. The members of the panel, all with extensive experience in clinical and research areas pertaining to acute wound healing, have summarized their conclusions in this article. The Consensus Panel on Acute Wounds was convened virtually on November 19, 2022. Consensus statements were based on 80% or higher concurrence among the panelists.
The editors defined the scope of work and outlined the various related sections. Participants were selected based on the following criteria: nationally recognized expertise in acute wound care and research; specialty diversity (ten clinical and two research specialties were represented, including dermatology, general surgery, vascular surgery, pediatric surgery, plastic surgery, orthopedic surgery, podiatry, pediatric emergency medicine, nursing and wound healing research); practice setting diversity (academic hospital, private hospital, wound care clinic, providing care in outpatient, inpatient, and emergency department [ED] settings); and geographic diversity.
Although 16 of the 17 Consensus Panel members were based in the United States, many foreign publications were reviewed, and most panel members participate frequently in international meetings. The conclusions of the panel should therefore be of interest to wound care practitioners worldwide. In addition, the panel included learners (fellow and resident physicians, medical students) to enhance applicability to providers at different levels of training.
Experts in each area were assigned to review and present the critical elements of each section. An extensive review was performed by experts assigned to each section, and a draft script of each section was prepared for and submitted ahead of the virtual meeting. This was followed by a formal slide presentation of each section by the assigned experts in a virtual meeting of all the participants. Key concepts were identified for each section, and discussion ensued, with consensus achieved through >80% concurrence among panelists. A formal vote was undertaken when there was dissent or lack of unanimous agreement. After cycles of iterative review, there was unanimous concurrence with the edited statements. All presentations were recorded, transcribed, and edited into this article.
DISCUSSION
Acute vs chronic wounds
A wound can be defined as a defect or disruption in the continuity of the epidermis and deeper structures of the skin or mucosa due to physical or thermal damage or because of an operation. 2 Less often, they are caused by reactions to medication or by inflammatory or blistering skin disease. Wounds are typically classified as acute or chronic, depending on whether the healing process proceeds in a timely or delayed fashion. An acute wound is damage to the skin that frequently heals completely within a predictable time range, depending on the wound depth, size, and the magnitude of the damage. 3 The primary causes of acute wounds include mechanical injuries (abrasions, tears, penetrating injuries, or surgical wounds), radiation, electricity, corrosive chemicals, thermal sources, or blistering skin diseases. On the other hand, a chronic wound results when skin injuries do not heal in a timely manner and may be at risk of recurring. 2,3 Frequently, patients with chronic wounds have underlying co-morbidities such as diabetes or vascular disease.
Determination of healing time and, as a result, whether a wound is designated acute or chronic, remains arbitrarily based on factors including anatomical location, shape, wound cause, patient age, and physical condition. For example, an elliptical wound on the face of a healthy child will likely heal faster than a circular burn wound on an elderly person with multiple comorbidities. Wounds are also categorized depending on the amount of skin layers affected. Very superficial wounds encompass those injuries affecting the epidermal surface only and are termed erosions. Ulcers that involve both the epidermis and part of dermal layers but retain adnexal structures (i.e., hair follicles, blood vessels, sweat glands) are called partial thickness (PT) wounds. A variety of terms have been used to describe the mechanisms or results of traumatic injury including avulsion (tearing away), abrasion (scraping or rubbing), laceration (rough, jagged tear), incision (cutting with a sharp object – knife or scalpel), crushing (press or squeeze with force) or shearing (cutting with two blades like scissors, or when the body moves in one direction and parts in contact with the surface do not move or move in a different direction) injuries are some examples.
Should deeper structures including the entire dermis or deeper, be involved, it is called a full-thickness (FT) wound. 2,4 With deeper wounds – involving part or all of the dermis and deeper layers – regeneration does not occur, but rather a repair process is initiated that usually results in scarring. Moreover, the adnexal structures are absent and are replaced with scar tissue, and keratinocytes epithelialize the wound only from its edges. FT wounds often heal in part by contraction, while there is minimal contraction in partial-thickness wounds. Contraction involves the movement of pre-existing tissue centripetally, and the wound area is decreased. This may result in cosmetically disfiguring contractures. Interestingly, regeneration can sometimes occur in specific situations even when the dermis is involved. One example of this is in early (first two trimesters) fetal skin wounds. Although not entirely understood, this is likely due to fibroblast heterogeneity, a reduced inflammatory response, and the presence of higher concentrations of glycosaminoglycans and type III collagen, as well as decreased levels of transforming growth factor b1, seen in fetal dermis. Overall, these factors are likely important in mediating regeneration rather than repair. 5,6
Primary, secondary and tertiary intention healing
Wounds heal by one of three general mechanisms. When an acute wound heals without intervention, it is termed secondary intention healing. Secondary intention healing is primarily by the contraction of myofibroblasts. 7 Primary intention healing arises when a surgeon directs closure of the wound by approximating the wound edges. Even if the wound edges are approximated and healing is by primary intention, the three phases of healing occur. Depending on the size, shape, and location of the wound, a surgeon chooses among three methods to repair a defect: direct closure, flap, or graft repair. A third type of wound healing, tertiary intention, is sometimes described. This refers either to delayed primary closure of a wound that is purposefully left open and/or when a primarily closed wound dehisces and is left to heal on its own. An example of healing by tertiary intention would be a patient who returns several days after an initial wound occurs, at which time debridement and wound cleansing are performed. This can be useful in the management of certain contaminated crush wounds or mammalian bites.
Methods of creating acute wounds
Acute wounds may be created utilizing a scalpel (steel), laser (heat), liquid nitrogen (cold), or chemicals (acid) to create a wound. 2 Alternatively, a patient may sustain an acute wound through accidental trauma, thermal, or chemical burns. Wounds heal differently and at different rates, depending on the inciting event. In general, wounds created by sharp steel, such as surgical incisions, heal faster than other methods of wounding. Healing of traumatic wounds may be slowed due to foreign substances inoculated into the wound, causing prolongation of the inflammatory phase. Acute wounds, especially crush injuries, can involve the skin and multiple other tissues and be at risk for wound infection, delayed healing, and other complications, and therefore benefit from a multidisciplinary approach to management. For complex wounds, the importance of patient participation and engagement in ensuring patient-centered care and optimum outcomes cannot be over-emphasized.
Preparation of the wound bed/infection control
Introduction – acute wounds at risk of infection
As indicated in the recent review of wound infection published by the International Wound Infection Institute (IWII) (Fig. 1), 8 all acute wounds are at risk of infection, with open wounds having the highest risk of infection, as essentially all open wounds are contaminated or colonized with microorganisms. The risk of acute wound infection is influenced by several key factors, including the characteristics of the individual patient, aspects of the patient’s acute wound, and the environment of the patient and wound. 9 –12 Major factors of individual patients include systemic diseases such as poorly controlled diabetes, peripheral neuropathy, conditions that produce hypoxia in wounded tissues, connective tissue disorders, malnutrition or obesity, alcoholism, illicit drug use, smoking, corticosteroid use, and poor adherence to treatment plans.
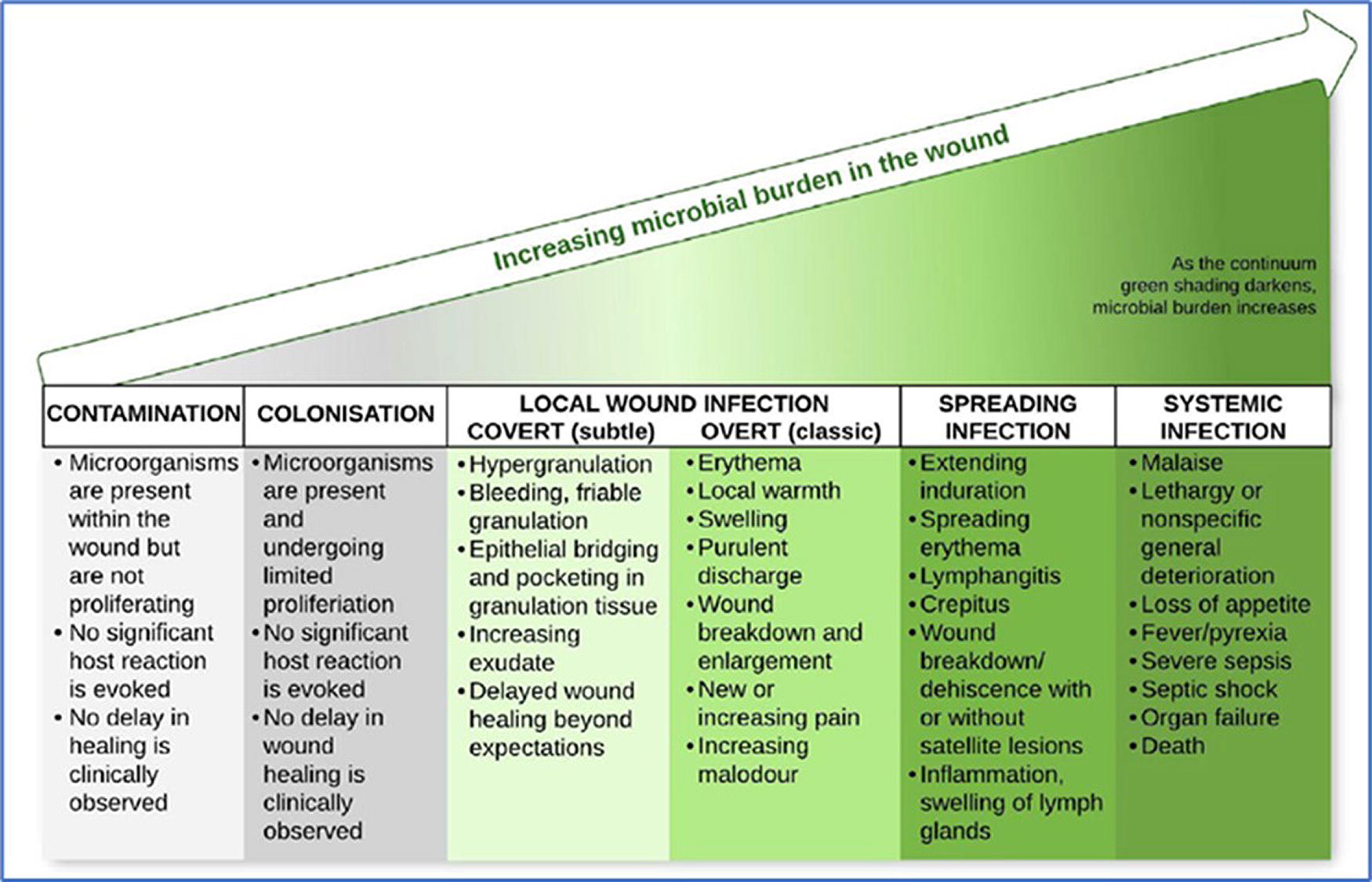
Wound infection continuum. The five major stages of wound infection with the local and systemic effects of increased microbial burden in wounds.
Risk factors based on the wound itself include if the wound is contaminated or dirty. This can occur due to acute trauma especially if a foreign body is present in the wound, if surgery is performed on contaminated or dirty tissues, if there is a hematoma or necrotic tissue, or if a prolonged surgical procedure is required. Increased duration of surgery increases the incidence of surgical site infections(SSI). Procedures complicated with SSI have a median duration of surgery at least 30 minutes more than those that do not develop a SSI. 9 For surgical incisions, adequate preparation before incision will mitigate the risk of infection. Finally, environmental factors for the wound and the patient can increase the risk or acute wound infection, including an unhygienic environment, inadequate management of wound moisture and exudate, and poor hand hygiene and aseptic techniques.
Identifying and assessing clinical signs and symptoms and stages of acute wound infection
In the broadest term, clinical infection of an acute wound is the invasion of a wound by proliferating microorganisms to a level that impairs normal healing. It is important to understand that acute wounds are essentially never sterile, so the goal of acute wound treatment is not to eliminate all microorganisms from an acute wound bed, but rather to mitigate the progression of the microbial “bioburden” to levels that would impede complete healing in a reasonable time. Thus, it is critically important to understand and recognize the clinical stages and signs of wound infection.
As shown in Figure 1, the Wound Infection Continuum presented during the IWII 2022 update of Wound Infection in Clinical Practice shows five major stages of wound infection. At the lowest end of the microbial burden scale, “contamination” refers to the stage in which microorganisms are present within the wound but are not rapidly proliferating, there is no significant patient reaction, and no delay in healing is clinically observed. In the “colonization” stage, microorganisms are present and undergoing limited proliferation, but the patient’s immune response can limit the expansion of the microorganism, and again, no clinical delay in healing is observed. In the third stage, the microbial burden has increased in the wound to produce subtle effects in the wound including friable hyper-granulation, increased exudate, epithelial bridging, and pocketing of granulation tissue, which combine to delay healing beyond expectations. Extensive tissue injury and necrosis (as in crush injuries) present a substrate for microbial proliferation if not debrided. As wound infection progresses, classical overt signs and symptoms become evident and may include erythema, local warmth, swelling, purulent discharge, increasing malodor, new or increasing pain, and wound breakdown and enlargement. Conversion of planktonic bacteria into biofilm bacteria phenotypes that develop high tolerance to a patient’s normal immune system, as well as treatment with antibiotics and antimicrobial agents may become a major factor at this stage of acute wound infection. If the wound microorganisms invade the tissue surrounding the acute wound, the wound infection progresses into the fourth stage of spreading infection that is characterized by extended induration, erythema, inflammation, swelling lymph nodes, and more extensive wound breakdown or dehiscence. In the fifth and final stage of wound infection, patients typically develop fever, malaise, severe sepsis, organ failure, and progressive decompensation, which can eventually lead to septic shock and death.
Multiple wound infection assessment tools have been developed that can aid in the evaluation of wound infection, with several that target specific wound types such as acute cardiac surgery wounds (ASEPSIS score), while other assessment tools target infection of acute or chronic skin wounds (IWGDF/IDSA, NERDS, STONEES, IWII) or all wound types (CSSC checklist, TILI). 8
Diagnosis of acute wound infection
Wound infection is a clinical diagnosis based on the presence of local and systemic signs and symptoms of infection. The clinical diagnosis can be confirmed by hematological, radiological, and microbiological investigations. These studies may uncover the systemic effects of infection, the presence of osteomyelitis, extension of the infection to deeper sites, or the identification of the microorganism responsible for infection.
Microbial analysis of the wound will inform the selection of appropriate antibiotic therapy for the specific infection. Such analysis should occur early in the patient’s presentation for clinical assessment. Indications for sending microbial studies include the following: (1) evidence of local spread or systemic infection; (2) failure to respond to antimicrobial intervention or clinical deterioration despite appropriate antimicrobial treatment; (3) surveillance of drug-resistant microbial species; and (4) identification of species that may be less amenable to surgical intervention.
Treatment
Wound bed preparation is the management of the wound to accelerate endogenous healing or to facilitate the effectiveness of other therapeutic measures for the wound to heal rapidly. Since acute wound infection is caused by the invasion of metabolically active planktonic microorganisms into viable tissue, local treatments and care to protect acute wounds from microorganism invasion is important. 13 Wound cleansing is a fundamental component of wound preparation, defined as the active removal of surface contaminants, loose debris, non-attached non-viable tissue, microorganisms, and/or remnants of previous dressings from the wound surface and its surrounding skin.
Debridement should be considered when non-viable or necrotic tissue, and/or foreign material is seen in acute wounds. There are various forms of debridement, each with varying degrees of complexity. 1 In acute wounds, mechanical and/or surgical debridement should be considered early when there is the presence of foreign material or contamination of the wound bed. Following debridement, maintenance of an optimal moist wound environment is important for driving wound healing potential. Since microbial load is directly related to infection risk, wound bed preparation with debridement and local and systematic antibiotics, as appropriate, will lead to effective control of the wound environment. 14
Prophylactic antibiotics are generally not recommended at the time of closure of acute wounds. Most acute open wounds do not require the use of antimicrobial therapy if there is no risk or active infection. That being said, there are exceptions, such as open fractures, contaminated wounds, bite wounds, and severe deep wounds for which antimicrobial prophylaxis may be indicated in accordance with published clinical practice guidelines (CPGs). 15
The signs and symptoms of local spread of infection may include worsening inflammation, erythema, or induration greater than 2 cm from the wound edge, streaking, crepitus, or wound breakdown. Wounds with a high risk of infection should be treated with antimicrobial therapy empirically. Factors that predispose wounds to infection include poorly controlled diabetes, prior surgery, radiation or chemotherapy, immune system disorders, protein-energy malnutrition, alcohol and drug abuse, poor tissue perfusion, and inappropriate antibiotic prophylaxis. 16 Other local wound factors are contaminated or dirty wounds, trauma with delayed treatment, pre-existing infection or sepsis, spillage from gastro-intestinal tract, penetrating wounds over four hours, inappropriate hair removal, and operative factors. 17,18 For hair removal, avoidance of razors in favor of hair clippers or depilatory cream has been shown to decrease the risk of infection. 19
Topical antimicrobial therapy should be administered at the lowest effective dose to minimize harm to the cells in the wound. Clinical efficacy of the therapy should be evaluated after no more than two weeks of usage. 20 Complete wound healing within 8–12 weeks, improvement in wound bed tissue type, reduction in clinical signs and symptoms of local infection, and reduction of laboratory data are signs of clinical efficacy of the topical antimicrobial treatments.
Systemic infection results when the microorganisms spread throughout the body via the vascular or lymphatic systems, causing a host immune response affecting the whole body. Systemic antibiotics should be administered only when the degree of infection cannot be controlled with local intervention and has the risk of systemic dissemination. The choice of antimicrobial therapy is guided by microbial analysis; however, these data may not always provide conclusive information regarding the cause of infection. 21 Antibiotics should be chosen to avoid the development of multi-resistant organisms. 22 In general, systemic antibiotics are used for a seven to ten-day treatment course. When the wound is complicated by osteomyelitis or the wound involves an infected prosthetic implant or fracture of osteosynthetic material (i.e., plates, hardware), systemic antibiotic treatment is indicated for six or more weeks. Local and systemic antimicrobial therapy should be used along with local wound management and holistic patient care. 23
Acute pain management
The International Association for the Study of Pain defined pain as an “unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage.” 24 A discussion of the management of pain in acute wounds does not differ significantly from that of chronic hard-to-heal wounds, other than the expectation that pain associated with acute wounds should begin to diminish over time as healing progresses. This does not lessen the need to address pain with all patients in all situations. Inadequate management of acute pain is a risk factor for the development of chronic pain, with an incidence of up to 33% in intensive care unit patients. 25 Furthermore, the presence of pain increases stress and anxiety, which can, in turn, inhibit wound healing.
A review of the types and occurrences of wound pain can facilitate the delineation of the approach to management.
Pain type
Nociceptive
Begins with an injury involving pain receptors found in the skin, joints, or viscera. Signals are picked up by sensory receptors in nerve endings in the damaged tissue, which are then transmitted to the spinal cord and subsequently the brain. Such pain may be described as aching, throbbing, sharp, or stabbing.
Inflammatory
Inflammatory pain arises when tissue damage occurs due to surgery, trauma, or other inflammatory conditions. This pain usually resolves when the condition that provokes the inflammation is controlled. Inflammation is characterized by redness, heat, and swelling and is a normal healing response to injury. These symptoms may also indicate early signs of infection, and visual signs and symptoms should be monitored closely if the symptoms persist and visual signs increase.
Neuropathic
Arises from sensations processed by the central nervous system (as in phantom limb pain) or the peripheral system (as in neuropathy). Neuropathic pain is a major contributor to chronic pain and differs in character from nociceptive pain as it is often described as burning or tingling sensations.
Pain occurrence
Background pain
Pain in the wound/operative area that is present at rest.
Incident or cyclic pain
Pain that is caused by periodic and routine treatments, procedures, or activities such as turning and repositioning, coughing, ambulation, and scheduled physical therapy. This type of pain also triggers anticipatory pain which, due to past experiences, may cause a significant stress response.
Procedural or noncyclic
Provoked by sporadic occurrences such as debridement and dressing changes.
Assessing pain
Pain should be assessed at each patient encounter. If not assessed and documented, it may be ignored or not properly addressed. If the encounter is emergent, the pain may be presumed, depending on presenting findings, or obvious, depending on the patient’s response to examination or movement.
A patient’s pain should not be dismissed. The expression of pain is very individual and may seem to be exaggerated or minimized compared to the appearance of the wound. In addition to a pain scale (numeric, Wong-Baker [FACES], visual analog or verbal), additional questions should be asked related to the impact of the individual’s pain on their quality of life including impact on sleep, appetite, work, family, and social life, as well as a potential obstacle to mobility and exercise. The pain assessment should include location, duration, intensity, changes since the last assessment, and aggravating and relieving factors.
Managing pain
Overwhelming Procedural Pain: It is advisable that analgesia should be administered before painful procedures. For the hospitalized patient requiring significantly painful procedures such as large negative pressure wound therapy (NPWT) or large surface burn dressing changes, operative dressing changes with mild anesthesia or moderate/deep sedation at the bedside should be titrated to the lightest level to allow tolerance of the procedure. 25
Systemic pain management
The decision to prescribe systemic analgesics and opioids is individualized but also very regulated. As a result, many practitioners will not prescribe them; this may have a negative impact on the patient with a wound. The avoidance of pain medication may result in a patient who is not able to adhere to an optimal treatment plan but may also miss appointments, remove compression wraps, and not perform the requisite dressing procedures. Such patients may thus be labeled as “non-adherent,” when in fact their ability to adhere to the wound management plan is compromised by undermanaged pain.
Strategies for management of short-term pain
Over-the-counter acetaminophen or non-steroidal anti-inflammatory drugs (NSAIDs) are typically sufficient for inflammatory pain such as those experienced after cleansing or debridement. These can be utilized by the patient at the recommended doses on the packaging. Topical anesthetic gels, creams, or liquids can be utilized with supervision. With more severe pain refractory to other modalities, prescriptions of opioids in small quantities can be considered with limited refills only after re-evaluation by a provider.
Modifiable strategies related to wound pain
In addition to systemic strategies, pain in the patient with an acute wound may be mitigated by addressing modifiable factors. 26 Pain arising from an infection should improve as the infection is addressed with systemic antibiotics and/or management of the source of the pain, such as drainage of an abscess or collection of fluid. Using dressing materials that prevent adherence to the wound bed such as contact layers, choosing dressings after a thorough assessment of the environment to match the exudate levels of the wound, and altering the treatment plan as the wound evolves can minimize or eliminate much of the dressing-related pain. Reducing the frequency of dressing changes, leaving the wound undisturbed for longer periods of time through the use of newer higher absorbency dressings, and using topical anesthetics when dressing changes must occur will reduce the frequency of pain, as well as potentially reduce the stress associated with dressing changes.
Distraction strategies to reduce pain
Diverting attention away from pain does not remove it but rather turns the focus away from the effects of pain. For example, patients often comment that their pain is worse at night. This could potentially be attributable to overuse or increased activity during the day. In reality, many times the pain is likely about the same; however, when patients are more “distracted” during the day, it is easier to ignore or not to focus on the pain. In contrast, when trying to sleep, the pain becomes the primary focus, resulting in the perception of greater pain at night. Likewise, a patient in an outpatient setting may seem distracted or inattentive to questions related to their evaluation as they focus on their phone; however, this distraction may be a way to unknowingly reduce the stress and/or pain related to the visit. Similarly, children facing a potentially stressful situation such as a physician’s office or ED visit may be distracted with an electronic game device to help ease the stress that such an environment may induce. Situations such as these illustrate the importance of distraction strategies to reduce pain.
Once such diversion method is the use of virtual reality; an artificial computer-generated environment is created to replace real-world sensory input to take the patient to a place removed from the environment and focus on actual or potential pain. As distraction interventions work by competing for attention otherwise directed toward painful stimuli, pain tolerance, and pain thresholds have been shown to increase under virtual reality conditions. 27,28
Special considerations in the pediatric population
When managing acute painful procedures in children, some special considerations are necessary. In addition, children with special needs may require unique considerations. Pediatric patients are often fearful of procedures and, depending on their age, may have a limited understanding of terminology frequently used when healthcare providers describe the necessary steps for treatment. Taking some time before the procedure to explain in child-friendly terms what will occur can frequently alleviate stress in children old enough to comprehend this information. Avoiding the use of trigger words such as “needle,” “cut,” and “shot,” and words that children may not readily understand such as “suture” and “anesthetic,” may help to mitigate their stress and potential response to pain.
If a child life specialist is available, they should be involved before the procedure begins. It is best to set up the procedure tray out of view of the child. During the procedure, allow and provide appropriate distractions such as videos or music. Parents can be helpful at providing comfort and distraction for the patient during procedures. Many young children with minor wounds can be comfortably restrained in a bedsheet or on a papoose board with an assistant helping to immobilize the area of repair, whereas school-aged children may be able to cooperate. All children may benefit from a parent or assistant distracting them and/or holding their hand to prevent disruption of the sterile field. Parents or family should be encouraged to remain in the room for minor wound care.
Depending on the child’s age and level of anxiety, inherent pain of the wound repair/procedure, and the expected duration of the procedure, multiple approaches for pain management may be considered. 29
It is advisable to select the least painful wound care or repair procedure whenever possible. For example, many lacerations may not require sutures. In cases where adhesive tapes or tissue adhesives are sufficient for wound closure, those options may be preferable to sutures. In cases where sutures are required, consider utilizing a rapidly absorbable suture such as fast-absorbing gut. This prevents a subsequent suture removal procedure, which can cause significant anxiety for young children.
On arrival to the ED or treating unit, a pain evaluation should be performed. Oral NSAIDs or acetaminophen can be administered for mild to moderate pain. Systemic opioid pain medication should be considered for those in moderate to severe pain, based on provider discretion, with options for administration including oral, intranasal, or intravenous (if access is already available).
When possible, children should have local anesthesia provided before wound repair. In some cases, pain associated with injections can be mostly avoided by using topical analgesics such as lidocaine-epinephrine-tetracaine. Topical analgesics should not be used on mucus membranes, areas of poor vascularization, or genitalia. Toxicity related to topical analgesics is rare and mostly due to intravenous administration of the agent or overdosing. The ester family of local anesthetics consists of 2-chloroprocaine, tetracaine, and procaine, which are metabolized by plasma cholinesterase. Adverse effects from ester local anesthetics are typically from the potential for toxicity with pseudocholinesterase deficiency or absence and allergic reaction to the metabolite para-aminobenzoic acid. These ester local anesthetics are not typically used in children. The amide family of local anesthetics consists of lidocaine, prilocaine, bupivacaine, levobupivacaine, mepivacaine, etidocaine, and ropivacaine. These are typically metabolized in the liver by the cytochrome P-450 system. Hepatic enzymes needed to metabolize these amide local anesthetics agents are not as normally developed in infants, and they may therefore be prone to toxicity. 30,31 Caution should be used in younger infants with a predisposition to methemoglobinemia as well. 31 In some cases, additional injectable buffered lidocaine may provide further anesthesia if incomplete with topical. When topical analgesics are not optimal, then local infiltration with an anesthetic such as buffered lidocaine with or without epinephrine may be considered. Physicians should be aware of the possible complications of local anesthetics such as cardiotoxicity and central nervous system symptoms. Providers should be aware of pediatric weight-based dosing considerations for all types of topical and local anesthetics they would consider utilizing. 32 For example, the recommended dose of Lidocaine is 3-5mg/kg/dose, not to exceed the recommended adult maximum dose of 300 mg/dose and not to be repeated within 2 h. For lidocaine with epinephrine, the recommended dose is 5–7 mg/kg, not to exceed the adult maximum dose of 500 mg; and for longer-acting bupivacaine, the recommended dose is 0.2-0.3 mg/kg/dose not to be repeated within 16 h. 32 Minor overdose of local anesthetics may present initially with tinnitus, metallic taste, and numbness of the lips. At higher doses, convulsions may occur. Even higher doses may lead to ventricular dysrhythmias and cardiovascular collapse. Shorter-acting agents such as lidocaine are less toxic as the effect wears out quicker. 30 Dysrhythmias from the cardiac toxicity of the longer-acting agents like bupivacaine may be more difficult to treat. Ropivacaine has less cardiotoxicity to bupivacaine 33 although the CNS toxicity is similar. 33,34
When it is anticipated that a wound procedure will cause significant pain despite local anesthetic, or that the procedure may require a moderate amount of time to repair, or if the patient has high levels of anxiety, it is reasonable to consider adjunctive medications to reduce anxiety and/or sedate the patient. It is important to note that any sedated procedure should be performed by a healthcare provider trained and credentialed to provide sedation, including the necessary rescue for unanticipated anesthetic complications. For minimal sedation, intranasal or oral midazolam may be administered. For procedures that are moderately to severely painful or have a longer duration, moderate or dissociative sedation may provide better analgesia and control of the environment. For example, burn debridement for a larger body surface area may require moderate, dissociative, or even deep sedation. More extensive wound repair or debridement for which a longer or deeper anesthetic is needed should occur in consultation with an anesthesiologist and surgeon, as operative management may be necessary.
Evaluation of a patient’s suitability to receive sedation includes assessing the patient’s age, clinical condition, risk factors, past medical history, co-morbidities, and institutional criteria for anesthetic administration. If the patient does not meet eligibility or is deemed high-risk, alternative options should be discussed, and the patient may need to be transferred to a higher level of care and/or consult with an anesthesiologist and surgeon for operative management. If the patient is a good candidate for moderate sedation, it should be ensured that appropriate monitoring and safety measures are in place.
There are various medications that can be used for procedural sedation. The choice of medication for procedural sedation should be determined with the goal of maximizing patient comfort and safety. Additional factors include institutional preference and resources, provider experience with a particular regimen, expected pain level for the procedure, anticipated duration of the procedure, availability of intravenous access, and any patient-specific contraindications. For example, with the use of nitrous oxide, it would be possible to perform wound repair without the need for intravenous access.
Many children can achieve adequate pain control with acetaminophen and/or NSAIDs after the repair of acute wounds. In cases of moderate to severe pain refractory to other interventions, an oral opioid analgesic prescription may be considered. If pain is expected to remain severe even after repair, admission for pain management and further wound care should be considered.
In general, the pain related to acute wounds peaks in the first 1–3 days following injury and lessens as the wounds heal. Persistence or recurrence of pain may be an early indication of complications (such as infection) and warrants re-evaluation of the wounds.
Burns
Burns are an injury to the skin or other organic tissue primarily caused by heat, either wet or dry, or due to radiation, radioactivity, electricity, friction, or contact with chemicals. Burns are classified, based on their depth, into categories of superficial thickness (ST), partial thickness (PT), and full thickness (FT) and their size is calculated as a percentage of total body surface area (%TBSA). The identification of minor burns according to the American Burn Association is described in Table 1. To be considered minor, a burn must be an isolated injury that does not involve the face, hands, perineum, or feet, does not cross major joints, or involve the entire circumference of a body region.
The identification of minor burns according to the American burn association
To be considered minor, a burn must be an isolated injury that does not involve the face, hands, perineum, or feet, does not cross major joints, or involve the entire circumference of a body region.
TBSA - total body surface area.
Diagnosis
Depth determination is mostly made by clinical assessment. Split-thickness, first-degree burns affect only the epidermis; their site is red, painful, dry, and has no blisters, e.g., sunburns. PT), second-degree burns involve the epidermis and part of the dermal layer. Depending on the extent of the dermis affected, PT burns can be superficial or deep. In superficial PT burns, the site appears red (the erythema blanches with pressure), blistered, swollen, and painful. Deep PT burns appear partially yellow or white, are dry, and do not blanch with pressure. FT, third-degree burns damage the epidermis and entire dermis. The burn site appears white or charred and has no sensation.
Burn wounds are usually non-uniform in-depth, and many have a mixture of deep and superficial components. They are dynamic and can progress or convert to deeper wounds over a period of several days. It is often difficult to initially distinguish between superficial PT and deep PT burns; these non-uniform burn wounds must be reevaluated frequently (at least daily) during the first 72 h. The methods to determine %TBSA include the “rule of palm” (where a size of the patient’s palm and digits represents 1%TBSA), “rule of 9” in adults, and Lund and Browder chart with various modifications. 35 ST (first-degree) burns do not get included in the %TBSA.
Initial care
A comprehensive care plan for burn injuries is a complex, multi-staged process. It starts with first aid and continues through close assessment to identify the need for specialized care to scar prevention and treatment, with ongoing psychological support to ensure maximal recovery.
First aid is performed by cooling the burn for 20 minutes with lukewarm (∼12–15°C/55–60°F) running water applied within 3 h post-burn. This has been shown to reduce the depth of tissue damage, enhance the speed of wound healing, and reduce the need for grafting. 36 Hypothermia should be avoided and closely monitored, especially in young children. Alternatively, a small burn wound may be covered with clean, cool wet fabric reapplied every 2 minutes for a maximum of 20 minutes. Burnt/overlying clothing, contact lenses, and any jewelry should be removed. Chemical burns require immediate irrigation. Electrical burns assessment should include cardiac monitoring.
Disposition
Most patients who meet criteria for minor burns can be treated in an outpatient setting. Inpatient hospital care, preferably at a burn center, is mandated for any patient who has at least one of the following: 1) comorbid illness; 2) questionable patient reliability/social circumstances; 3) risk of noncompliance; or 4) suspected or known abuse. Minor burns can be treated outside of a burn center as long as the treating team is familiar with burn care, is able to utilize telemedicine for expert consultation, and refer to a burn center (outpatient or inpatient) in accordance with the American Burn Association recommendations. 37 FT burns >5%, pediatric patients, older adult patients, and those with chemical, electrical, and inhalational injuries benefit from the multidisciplinary team offered by burn centers.
Treatment
Initial management includes ensuring an up-to-date tetanus immunization status and treatment of pain and/or anxiety. Systemic antibiotics are not indicated for minor burns unless there is an obvious infection. ST burns only need skin moisturization and pain control. Open burn wound surfaces are prone to rapid bacterial colonization with the potential for invasive infection. The first step of partial-thickness and FT burn wound care is cleansing and superficial debridement, which removes loose, devitalized, and necrotic tissue or epithelium. Soap, water, or non-irritating skin disinfectants should be used. That, in combination with a topical antimicrobial dressing, is sufficient in the majority of cases for the treatment of superficial PT burns.
Ruptured blisters should be debrided, while small intact blisters <2 cm in diameter may be left alone if they are neither expanding nor impairing the range of motion. There is no consensus on which topical agent or dressing is best suited for such minor burn wounds. 38 For minor burn wounds on the face, ointments/gels alone might suffice, but all other anatomical locations generally should be covered. Antimicrobials, either in the form of ointments applied directly to the wound and covered with a primary dressing, or sustained-released products (usually silver), are first-line therapy. Silver-sustained release dressings provide active antimicrobial coverage for an average of seven days. 39 Synthetic polymers have the advantage of one-time application and reported pain reduction for a PT burn. Nonadherent materials such as films, foams, nylons, silks, or hydrogels can serve as primary dressings. A secondary dressing can be used to secure the primary dressing in place, to provide padding, compression, and additional absorption of exudates. The frequency of dressing changes ranges from twice daily to weekly depending upon the amount of exudate and choice of dressing material.
For deep PT burns, specialized care is required. Such burns tend to form pseudo-eschar, thus delaying wound healing. The wound care may involve enzymatic or mechanical-surgical debridement. Enzymatic debriding products include proteolytic enzymes derived from honey, bacteria, and plants. The latter has shown effectiveness in fast eschar removal, less need for escharotomies in circumferential burns, as well as a reduction of the need for autografting due to selective debridement. 40,41
Surgical sharp excision is indicated for FT burns and is often necessary for deep PT burns. Mechanically debrided wounds need definitive coverage, generally with autologous skin. The development of skin substitutes has provided an alternative when autologous skin is not available in the supportive treatment for deeper PT burns and acceleration of FT burn healing. A variety of dermal repair scaffold products are available in the form of dermal matrices and bioengineered skin substitutes. 42 Use of biological dressings (e.g., amnion, dermal substitutes, etc.) may reduce the need for frequent dressing changes and the associated pain and stress. 43 –45 Non-uniform wounds with various level of PT/FT burns treated by skin substrates may heal without grafting or with a reduced graft size, which decreases donor site morbidity. FT burn wounds (unless very small) necessitate skin grafting. Definite closure is accomplished with an autograft; FT skin graft (for functionally important areas i.e., hands), split-thickness skin graft, and/or spray-on skin cells. The utilization of the spray-on cell (a suspension of the skin’s basal cells) has significantly reduced donor site size. In superficial partial burns, especially in cosmetically sensitive areas, it can prompt the pigment return. 46
Scarring
Formation of hypertrophic scar (HTS) is directly proportional to the time of wound healing, with 14–21 days considered to yield lower rates of scarring. 47 An association has been demonstrated between burn healing time and the formation of HTS in pediatric burns indicating that every day matters. Each additional day that it takes for a burn wound to heal is correlated with an odds ratio of 1.138 for developing an HTS; however, patients with fair skin (Fitzpatrick type 1–3) had rates of HTS of < 15% when healed before 21 days, while patients with darker skin (Fitzpatrick type 4–6) showed higher rates of HTS (up to 50% higher) even when the burn wound was healed before 21 days. 48 Patients should be referred to a burn center promptly at the first sign of hypertrophic scarring or if the wound fails to meet usual epithelialization milestones (Table 2):
Epithelialization milestones
Described are the expected epithelialization milestones as stratified by skin tone (Fitzpatrick type) and age. If a would fails to meet the described timeline, patients should be referred to a burn center. For example, a patient with Fitzpatrick type 4 skin and/or who is aged 2 years should be referred to a burn center if the wound has not epithelialized by days 10–14. Similarly, an adult patient with Fitzpatrick 3 skin should be referred to a burn center between days 14–21 if the wound has not sufficiently epithelialized.
Scar prevention and treatment comprises techniques to accelerate healing, post-healing massaging/lotion use, and compression therapy by garments and silicone gels. Treatment of the formed scars includes laser therapy, topical steroids, and surgical scar revision. 49
Physiotherapy
Physical and occupational therapy is essential for preventing stiffness, scarring, and contractures and includes anti-contracture positioning, range of motion stretching exercises, and splinting. Fingers and toes should be wrapped individually to facilitate range of motion. Early mobilization and restoration of daily activities are essential for the best functional outcomes. 50
Pain/anxiety and pruritus control
When planning treatment, psychological and emotional factors should be considered as burn debridement and dressing changes are painful procedures. Adequate pain control is essential, and it is important to note that the experience with the first dressing change can influence the consequent experiences. 51 In an acute hospital setting, the administration of opioids, and possibly sedative or dissociative agents, may be required. For outpatients, non-narcotic pain control is preferred and should be optimized using scheduled, alternating doses.
Itching is a common problem during the healing process, and its cause is multifactorial, including environmental factors (especially heat), physical activity, and stress. Systemic antihistamines and topical agents, including moisturizing lotions, are utilized for control. Pruritus usually stops after the wound is healed but can persist when HTS form. Fractional laser therapy can be helpful in refractory HTS pruritis. 49
Factors contributing to healing and outcomes after burns
Nutrition, specifically a diet rich in protein, vitamins, and minerals, plays an important role in burn wound healing. Similarly, prevention of infection, timely management of poorly healing wounds, optimal control of pain, anxiety and pruritis, and proper physiotherapy optimize healing. 51 The outcome-influencing factors include %TBSA involved, depth of injury, anatomical location, age of the patient, co-morbid conditions, and associated injuries.
Future directions
A potential new approach to preventing the progression and conversion of burns is the microcapillary gel, a topical gel embedded with the anti-inflammatory drug infliximab. The gel has shown effectiveness in reducing inflammation within partially burned skin and reducing progression to FT burns. 52 The use of three-dimensional bioprinting for skin reconstruction following burns is also promising in achieving faster wound closure, with the potential to enable accurate placement of skin cell types and constructs to replace injured skin. 53 Photo-biomodulation therapy can speed up recovery and reduce inflammation. 54 Lastly, the ability to accurately assess an individual's potential genetic susceptibility to raised scarring may lead to a more personalized approach to burn management in the future.
Torso wounds
Acute torso wounds can arise from sequelae of trauma, infection, or surgical intervention. These wounds can be in any part of the body, and the management is similar for the most part.
Traumatic wounds
They can result from penetrating or blunt trauma. The first step in management is to ensure that the patient is stable and that organ space injuries underlying the skin and soft tissue injury are fully evaluated and addressed.
Penetrating torso trauma wounds from stab wounds can appear small at the skin level but may have internal injury depending on the length of the object. Once this has been ruled out or addressed, these wounds can be closed primarily if clean, closed over a passive drain such as a vessel loop or Penrose drain, or packed with gauze to heal by secondary intention with dressing changes as needed. Penetrating wounds from firearms should be managed along similar principles. Once the internal injuries have been addressed, the skin wounds can be packed and will heal by secondary intention. Some firearms such as shotguns or high-velocity assault-type rifles can cause large FT skin and fascial defects. These wounds will need to be closed in layers, possibly requiring a fascial synthetic patch or negative pressure therapy to hasten the granulation and contraction of a large soft tissue cavity. Firearm skin wounds can lead to superficial and deep infections as they transport microbes from external or internal sources, especially in the case of bowel or bladder injuries, and prophylactic antibiotics should be considered, particularly for high-velocity bullets. 55 Knife wounds are often dirty and require tetanus prophylaxis and precautions for the development of wound infection.
Blunt torso trauma wounds can present in various forms. Bruising is most common from crush injuries, seat belts, or handlebars, or physical assault. This will heal without intervention. “Road rash” or skin abrasions that result from ejection during a motor vehicle crash or falling onto a pavement may involve a large portion of skin. These abrasions are typically ST skin wounds that will heal spontaneously, but there is a small chance of infection, thus an antibiotic ointment such as bacitracin should be applied to keep the skin breaks covered and for infection prophylaxis. Skin degloving can occur in severe cases and requires assessment of skin viability and an attempt to save as much skin as possible. Skin grafts or skin and soft tissue flaps may be necessary depending on the degree of the injury. 56
Open wounds are less common with blunt trauma, but when present, they are often dirty and should be thoroughly irrigated, debrided, and left open with or without negative pressure therapy, or closed over a passive drain. Open FT chest wall wounds, which could result from blunt or penetrating trauma, can cause a sucking chest wound. They should initially be covered on three sides to avoid accumulation of tension pneumothorax, and chest tubes should be placed. Repair in the operating room may require rib plating or chest wall replacement using synthetic, biological or composite meshes with or without titanium plate to restore chest rigidity, followed by soft tissue or flap coverage, depending on the extent of the injury. 57 Defects > 5 cm or > 4 ribs are generally reconstructed given the risk of paradoxical chest wall motion and lung herniation, especially for anterolateral defects, while this is not required for large apico-posterior defects given the support of the scapula and shoulder girdle. 57
Infectious wounds
Necrotizing fasciitis can occur anywhere on the body and requires wide surgical debridement that leaves large skin and soft tissue defects, which can be challenging to manage. The primary goal is to treat the infection with multiple debridements. During this period, negative pressure therapy is very useful as a temporary dressing. 58 Once the infection has been managed, wound closure is required, for which the approach will vary based on the amount of skin and soft tissue missing. Large defects are often managed with negative pressure therapy until the wounds granulate to skin level, after which they are covered by skin grafting (Fig. 2a–c). Smaller defects may be primarily closed, at least partially. In some cases, flaps may be required. Perineal infections due to Fournier’s gangrene can be tough to manage, as obtaining a seal around the anus for negative pressure therapy can be problematic. It is prudent to consider a diverting ileostomy or colostomy for these cases while the complex wound is being managed.
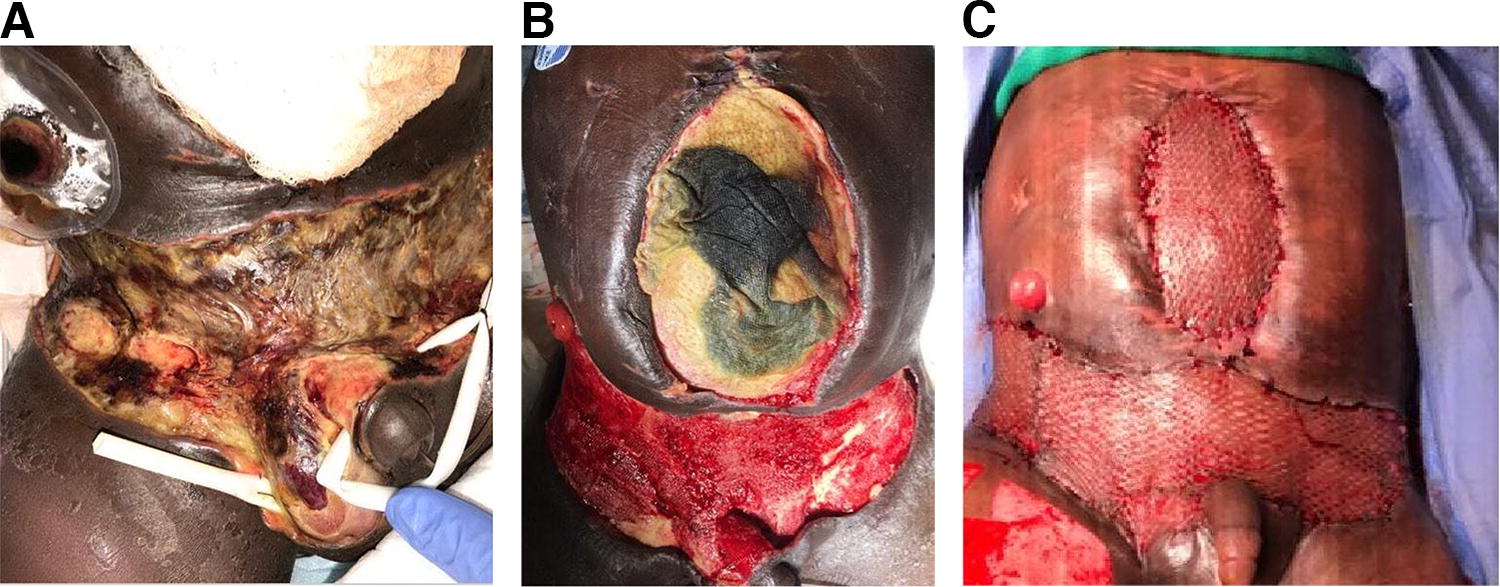
Open wounds of torso (Courtesy of Bindi Naik-Mathuria, MD);
Infected wounds may benefit from NPWT with instillation, which includes intermittent instillation of an antimicrobial (such as bacitracin/neomycin, polymyxin B/bacitracin or vancomycin/gentamycin/tobramycin) or antiseptic solution (such as polyhexanide, acetic acid, betadine) through the sponge device. Effectiveness has been shown with less need for surgical debridements and decreased time to wound closure. 59 An international consensus guideline recommends this use for a wide variety of acute and chronic wounds. 60
Surgical intervention
Abdominal, pelvic, chest, and back wounds can result from surgical incisions. In some cases, the skin of contaminated wounds may be left open to heal by secondary intention. Such wounds are generally managed by wet-to-dry packing with gauze bandage rolls or negative pressure therapy and contract and close over the course of a few weeks, or longer if infection develops. Wound or fascial dehiscence may occur following primary closure of torso wounds and can be managed similarly, or by re-operation and closure, depending on the situation and cleanliness of the wounds.
Trauma or severely medically ill patients undergoing abdominal operations may require an “open abdomen” approach in which the fascia is not closed initially, but closure is attempted after the initial physiological insult has improved. In some cases, this is not possible as the fascia retracts, and the abdomen expands. In these cases, closure can be complex. Biological and synthetic mesh (“patch closure”) followed by granulation and skin grafting and delayed ventral hernia repair (Fig. 2c) was common practice before the last decade. More recently, there has been a strong focus on primary fascial closure using adjunctive fascial traction techniques such as sequential fascial closure with tension sutures, primary fascial release through relaxing fascial incisions of the oblique muscles, or commercial fascial closure systems. 61,62 Negative pressure therapy is helpful, but high-quality data demonstrating improved outcomes is lacking. 63 Early fascial closure (within one week) is recommended for the best functional long-term outcome.
Facial wounds
Face and scalp injuries are very common, and together with hand injuries, are the most frequent injuries seen in the ED. 64
Diagnostic considerations
A careful diagnosis is requisite for all facial injuries. The skin or scalp injury may sometimes be so severe that the evaluating provider may be distracted from ruling out fractures and nerve injuries. Most facial wounds can be managed by ED clinicians. When the lips, nostrils, eyelids, or ears are involved, consultation with a plastic surgeon is often required. If the teeth or jaws are injured, the expertise of an oral-maxillofacial surgeon is typically needed. Injuries to the eye require evaluation by an ophthalmologist. When anesthesia or special instruments are needed to treat facial injuries, the patient should be managed in the operating room.
Crush injuries, abrasions, lacerations, and avulsions
Abrasions are usually caused by the face sliding over a rough surface. Sometimes gravel or asphalt particles become imbedded in the skin – “traumatic tattoos” – which must be acutely removed under local or general anesthesia. The abraded area can be treated with a moist dressing, moisturizing cream, or ointment until healed.
Lacerations are the most common facial injuries, with crush injuries being a close second. 64 The initial evaluation is critical so that deeper injuries to nerves, muscles, ducts, teeth, and bone are recognized and treated early. Sometimes there are injuries to the eyebrows, eyelids, or the eye itself. After irrigation with saline and conservative debridement, these lacerations can usually be closed. Old teaching dictated that lacerations more than 8 h old had to be left open, but often they can be closed after 18–24 h (or even later if the edges are excised) if the edges are aligned and well-irrigated (more than 250 mL) with saline under low pressure. Irrigation is particularly important for contaminated wounds, especially animal and human bites. The patient should be advised that there remains a high risk of infection. 65 Avulsions (defined as tissue loss) may require reconstruction with a flap or skin graft, typically by a plastic surgeon.
Compound injuries
Compound injuries are injuries that include, in addition to skin, muscle, nerve, a duct such as the lacrimal or parotid duct, bone, and/or teeth. Such injuries are best treated acutely. They often require access to an operating room, anesthesia, and more than one specialist, which may defer treatment for a day or two. Observation or treatment of a head injury may also delay facial treatment.
Facial burns
Epidermal burns without blistering (first degree) can usually be evaluated and initially treated in the ED, with management at home consisting of a moist dressing or a moisturizing cream or ointment. Deeper burns with blistering (second-degree) and blackened or whitened skin from a full-thickness injury (third-degree) need to be referred to a burn center for evaluation and treatment. In deep facial burns, the treatment is often more conservative, with the goal of preserving delicate facial skin. Debridement is frequently performed using a specialized saline jet or ultrasound to better preserve viable tissue. If the facial burn wounds have not healed in 2–3 weeks, skin grafting may be indicated. 66
Wound closure and suture removal
When a laceration can be optimally closed with surgical tape (Fig. 3) or glue, this should be done. This usually eliminates the need for local anesthesia and sutures for subsequent removal. This is a particularly attractive option for pediatric patients. When sutures are needed, a two-layer closure is recommended. The deep layer is approximated with an absorbable, monofilament suture such as poliglecaprone in 5.0 or 6.0 size. The superficial layer can usually be closed with 6.0 or 7.0 nylon or polypropylene. Everting, vertical mattress sutures (Fig. 4) are desirable and are mandatory at the lacerated edges of the lip, nostril, eyelid, and ear. The eyelids often only require one layer of sutures except when the orbicularis or levator muscle has lacerated, in which case sutures are required in the muscle. 67
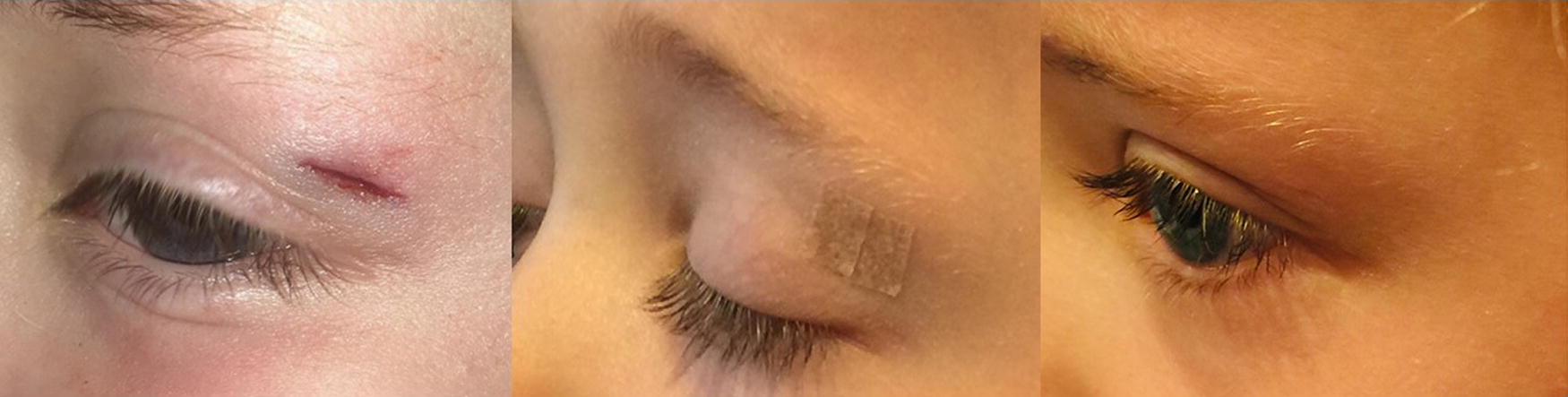
Eyebrow laceration (Courtesy of Elof Eriksson, MD, PhD). Eyebrow laceration in a child approximated with surgical tape that resulted in an excellent cosmetic result.
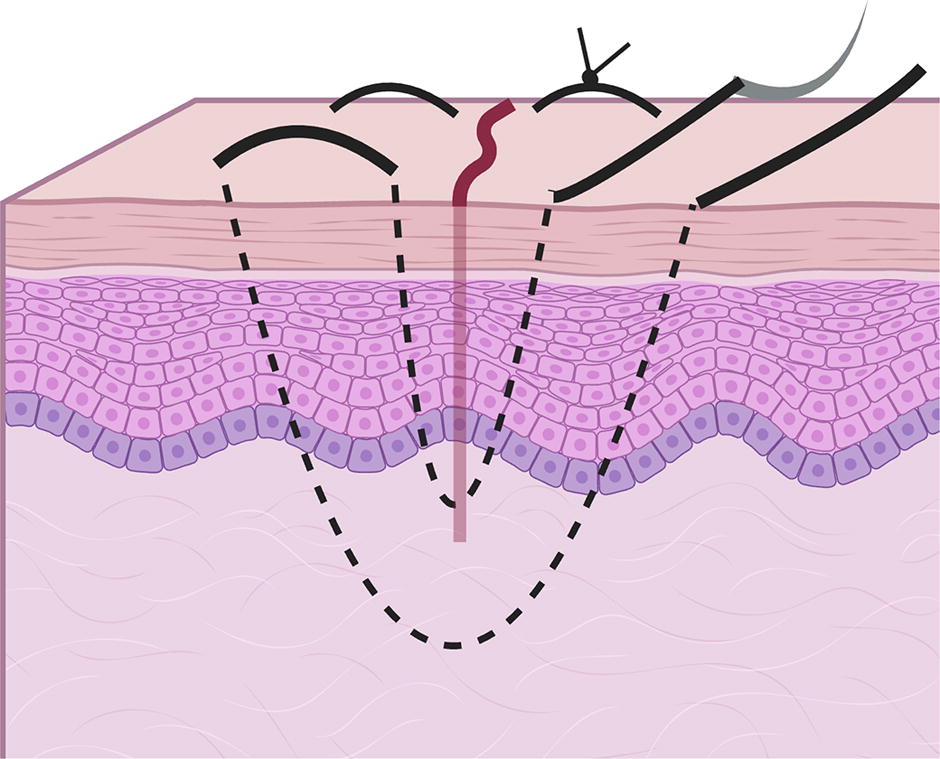
Vertical mattress sutures. (Created with Biorender.com) Sutures are placed deep in one direction and then more superficially in the reverse direction along the same vertical plane to allow the skin edges to evert.
Most facial sutures can be removed after five to eight days with the exception of areas under tension and in children where the risk of reinjury is high. In pediatric patients, it is often preferable to use absorbable (chromic) sutures to avoid the often painful and often traumatic process of suture removal. 68,69 The recommended choice of suture and size based on the location of the wound is summarized in Table 3.
Choice of suture and duration of use
General guidelines for the use of sutures and how long they can be left in place for different parts of the body. Appropriately sized cutting (or reverse cutting) needles would ensure easier passage of the sutures through the skin. In addition, skin staples can be used especially for hair-bearing areas where identification of the sutures for removal may be challenging.
Care should be exercised in the application of surgical glue to avoid the practitioner being stuck to the glue or the eyelids being stuck together. Acetone can be used to remove objects (gloves) stuck to the skin, but acetone should be avoided around the eyes. Erythromycin ointment and moist gauze can help release fused eyelashes. Cutting the eyelashes may be necessary. Consultation with an ophthalmologist may be required.
Skin grafts
If a facial wound cannot be closed because of too much tissue loss, a skin graft is often more attractive than a flap because, contrary to a flap, the donor site wound is more easily hidden. When skin grafts are required, usually a FT graft is preferred, and whenever possible, ideally ‘like’ skin—for example, from eyelid to eyelid or pre- or post-auricular skin to the rest of face—should be used. For larger areas, the upper inner arm skin usually provides a good color and texture match.
Hand wounds
Hands are the means through which the tactile world is explored. As such, they are frequently injured. A recent review found that 10% of all traumas seen in EDs in the United States involved hands or upper extremities. 70 Of these, 36.5% are lacerations, 19.9% fractures, 12.3% sprains/strains, and 12.1% contusions or abrasions. 71 This section will not serve as a definitive compendium on how to treat such injuries but rather outline elements of the diagnosis and treatment of common hand injuries that attained concordance among a panel of wound healing experts.
Diagnosis of acute hand wounds
Accurate diagnosis begins with a definitive history of how the wound was acquired, and some specific aspects of the patient’s history, including age. Was the wound acquired as a work-related injury? What is the dominant hand? What significant hobbies does the patient have? Was the environment particularly dirty or did the injury involve fresh or saltwater? What is the tetanus immunization status of the patient? Does the patient smoke, have diabetes, or other co-morbidities that portend poor wound healing? What prior upper extremity problems, if any, existed? Information on positioning of the hand/arm when injured is also important (e.g., grasping or opened hand). These factors all have significant implications for how to eventually treat the injury.
Physical examination of the hands needs to progress in a systematic and thorough fashion. Most injuries can be elucidated by this diagnostic modality alone. The examination should progress through the following sequence: circulation, sensibility, soft tissue deficits, bones, joints, and active muscle functioning.
Circulation
Circulation is assessed by the color of the skin and nail beds, and by measuring capillary refill after indenting the skin and blanching it. The appearance of the skin from the affected area should be compared with that of uninjured areas. Lack of or compromised arterial inflow will be marked by a pale, cool extremity or digit with prolonged capillary refill. Venous congestion is marked by a purplish turgid extremity or digit with faster than normal capillary refill. Brachial, radial, and ulnar pulses should be palpated, and an Allen test 72 may be done to assess patency of the radial and ulnar arteries. The test is performed as follows: 1) Ask the patient to elevate the hand and clench a fist to exsanguinate it while compressing both radial and ulnar arteries at the wrist. 2) When the patient opens the hand, the examiner releases the radial artery, and reperfusion of the hand from the radial side should be observed. 3) Repeat the exam, but this time releasing only the ulnar artery, and reperfusion of the hand should likewise occur. Incomplete refilling of the hand may occur in 10–15% of patients, which may indicate an incomplete superficial palmar arch or arterial occlusion. Ischemia may result from amputations, direct vascular injury, electrical shock, crushing, or compartment syndrome. Irreversible tissue death (myonecrosis) occurs within six hours of circulation deprivation, whatever the etiology.
Sensibility
Sensory deficits may be observed due to direct injury to a nerve caused by laceration, compression, or a degenerative process. Simple instruments such as a bent article clip or pencil tip may be used to assess light touch sensibility, static, or moving two-point discrimination. Again, comparison to uninjured parts provides a useful frame of reference.
A >6 mm static and >3 mm moving two-point discrimination at the fingertips indicates sensory nerve impairment. 73 Evaluation of the median nerve is best assessed by sensibility in the index fingertip, the ulnar nerve by the little fingertip, and for the radial nerve, assessing the dorsal side of the first webspace.
Soft tissue deficits
Soft tissue injuries to the hand should be documented, but the main issue to be addressed is whether or not definitive or even temporizing treatment in the operating room is required. Blind probing of open areas should be avoided, and debridement of devitalized soft tissue areas should typically be performed in the operating room, since there is rarely enough laxity or redundant skin to close after debridement, especially in the glabrous skin of the palm.
Bone injuries
Plain radiographs are an integral part of the workup for bone injuries. Tenderness elicited on gentle palpation over bones is a sufficient indication to obtain radiographs (anterior-posterior and oblique views). Both hands should be imaged so that anatomical discrepancies may be compared to uninjured appendages. Wrist injuries mandate true lateral views. It is beyond the scope of this work to delve into the management of fractures; however, significant bone injuries with malalignment will likely require pinning or plating techniques in the operating room.
Lacerations
All lacerations should be cleansed/irrigated to determine the severity and involvement of other structures. Bleeding may be controlled by elevation of the injured extremity and direct pressure. Indiscriminate attempts to clamp a vessel should not be done. If a tetanus booster has not been administered within the past ten years, consider administration of a booster, and for heavily contaminated wounds, tetanus toxoid may be administered. 74
When only skin is involved, careful reapproximation to align the skin and evert the edges is the goal. The dorsum of pediatric hands may be closed with absorbable suture material to avoid the trauma associated with suture removal; however, most adult wounds should be closed with nylon in a non-layered fashion for removal in one to two weeks.
Special mention is noted for lacerations of the nail bed since this is the structure most commonly injured in the hand and improper healing may result in deformity. 75 When required, debridement of the nailbed should be limited. Repair of injury to the nailbed matrix is facilitated by removal of the nail and repair of the nailbed matrix with absorbable sutures or cyanoacrylate. 76 Many hand surgeons no longer stent the nail fold when the nail is removed. Fingertip amputations <1 cm may usually be treated with dressing changes and allowed to heal by secondary intention.
Bite wounds
Human bite wounds are frequently clenched fist injuries, where a tooth penetrates the skin and the wound extends to the bone (the metacarpal head). Bite wounds are responsible for 20–30% of all hand infections. 65,77 All layers, including the joint capsule, are inoculated with oral flora (Staphylococcus aureus, Streptococcus viridians, anaerobic Bacteroides, and Eikenella corrodens). The wounds should therefore be copiously irrigated, not closed, and treated at a minimum with oral antibiotics. Repeat washout should be contemplated in 48–72 h, and at times operative debridement is warranted to prevent deep space infection or tracking.
Dogs are responsible for 40–50% of bite wounds to hands and forearms. 77 There may be a crush component since jaws are capable of 150–450 pounds per square inch, which can devitalize tissue. These wounds may require debridement and excision of wound edges. Some may be loosely closed. Cat bites are puncture injuries that account for only 5% of animal bites to the hand. Consider Pasteurella multocida and cat scratch fever when considering the choice of antibiotics. These puncture wounds are “cored-out” with a punch biopsy tool to keep the wound open a bit longer.
Acute hand infection may develop from any puncture wound to the hand. They can be classified as superficial infections that involve the skin and subcutaneous tissue or deep hand infections that can involve the deep fascial planes, tendon sheaths, joints, and bones. Hand infections can result in significant morbidity, hence early recognition, prompt management with elevation, irrigation, drainage, empirical antibiotic treatment, and prompt surgical consultation, when indicated, are important. 78
Burn wounds
The upper extremities and hands are common sites of burn wounds, including those sustained in occupational accidents. Flames, contact, tar/asphalt, scalds, chemicals, steam, and grease are common offenders. It has been estimated that 80% of burn patients have burns involving the hands and upper extremities. 79 Evaluating the depth of injury is important, as is careful attention to initial burn care so as not to allow desiccation and deepening of the initial injury. The management of burn wounds is covered in detail elsewhere in this consensus statement.
Deep wounds may require early operative intervention with fasciotomies (dorsal skin) or escharotomies (digits) to preserve vascular supply or prevent nerve compression or intrinsic muscle compromise. Less severe burns may be treated with burn ointment and dressing changes or dressing gloves which have been developed to mitigate moisture loss and pain in the early post-burn period.
Splinting the hand in a protected position of metacarpophalangeal (MP) joint flexion, interphalangeal(IP) joint extension, thumb abduction, and wrist extension during healing is of paramount importance. 80,81 This may require percutaneous Kirschner wire pinning for MP flexion or IP full extension to prevent extensor clawing. Early excision and grafting should be considered, especially to cover exposed tendons or joints. Palmar skin is less frequently in need of excision and grafting since victims tend to clench their fists, protecting volar skin from more serious injury. Flap surgery may be needed if vascularized coverage is required.
Mangled hands/amputations
Injuries involving tools, machinery, or motor vehicles moving at high speeds may have multiple levels of involvement including soft tissue, vascular structures, tendons, nerves, and bones. These wounds need operative exploration, usually emergently.
Soft tissue coverage not locally available, such as regional or free flaps, may be needed. Skin substitutes made of cadaveric tissue or silicone-covered glycosaminoglycan substrate may be used for temporary coverage.
Amputated parts may be preserved for replantation beyond the six-hour ischemic window if kept in sterile gauze/saline on ice. There are relative contraindications to reimplantation surgery, such as older patients, comorbidities increasing risks of long surgeries, avulsion injuries, prolonged warm ischemia time (digits survive longer than hands), massive contamination, psychological problems such as self-injurious tendencies, or single digit amputations (except thumb). 82
Foot wounds
Acute foot wounds may result from a variety of pathologies such as diabetic foot infections, foot burns, trauma (i.e., lacerations), and puncture wounds, among others. The assessment and treatment of acute foot wounds should incorporate a comprehensive approach that includes mechanism of injury, the extent of the wound, presence of a foreign body, neurovascular or tendon injury, as well as tetanus status and allergies (e.g., local anesthetics, antibiotics, or latex) (Fig. 5). Consultation with the appropriate surgical specialist may also be warranted.
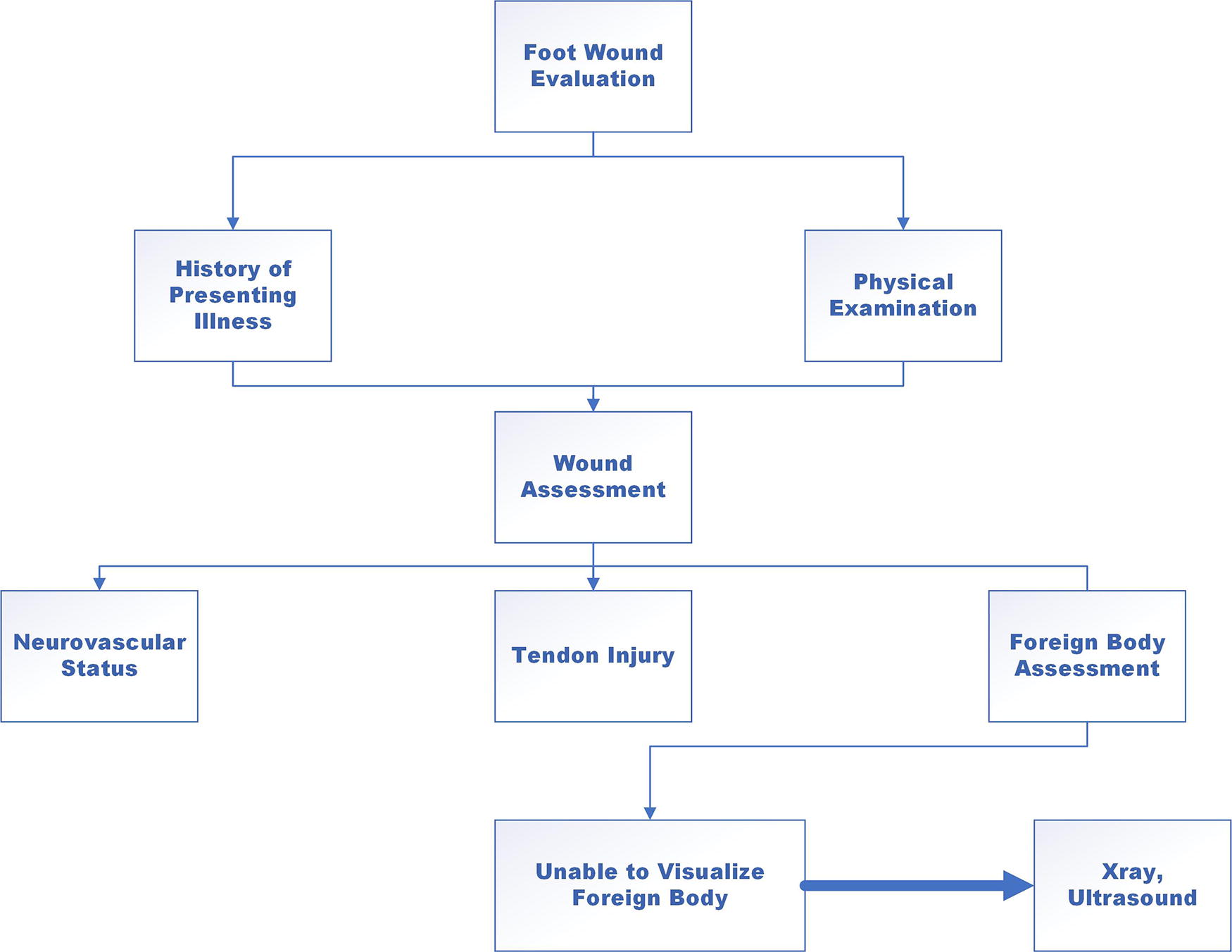
Comprehensive approach to foreign body evaluation. Assessment of a foot wound with particular attention to neurovascular status and tendon injuries. The suspicion of foreign bodies my require additional evaluation.
Puncture wounds
Puncture wounds of the foot are serious injuries, occurring more than 50% of the time on the plantar surface of the foot, with more than 90% of these involving penetration by a nail. Puncture wounds can lead to serious complications by penetrating into deep tissue spaces and structures of the foot, (including bones, joints, tendons, and deep fascia). It should be noted that the depth of penetration of the foreign object is a key factor in determining whether a wound will resolve without complex intervention. Susceptibility to infection is contingent upon several factors including location, type of penetrating object, retained foreign bodies, and penetration through shoes and socks. The most common organisms inoculating penetrating wounds are Staphylococcus aureus, beta-hemolytic streptococci, and various other anaerobic bacteria. 83 Pasteurella multocida is typical in cat and dog bite/claw scratch puncture wounds. 83 Pseudomonas aeruginosa is usually responsible for infection when the injury is the result of object penetration through shoes and socks. 84
The classical signs of infection typically include inflammation (redness, warmth, swelling, tenderness, or pain) or purulent secretions. It should be noted that the most common presenting symptom in the ED is persistent pain. 85 Pain out of proportion to the injury should lead clinicians to suspect that infection has developed. Puncture wounds involving the joint spaces lead to an increased risk of septic arthritis, which causes the rapid destruction of the articular structures. 86 Foreign-body penetration that extends to bone could potentially lead to direct extension osteomyelitis. 87
If the foreign object is readily visible or accessible through the wound, an attempt can be made to retrieve it in the ED (Fig. 6). In the event a foreign body becomes dislodged into the foot (a deep foot compartment space), surgical exploration and removal should be initiated under fluoroscopic guidance. It is recommended that children with wounds in which the top of the object cannot be completely visualized, wounds caused by broken glass, or those associated with other loose foreign bodies, receive plain radiographs. Ultrasound technology, when used by a properly trained clinician, can also be useful for detecting nonradiopaque foreign bodies.
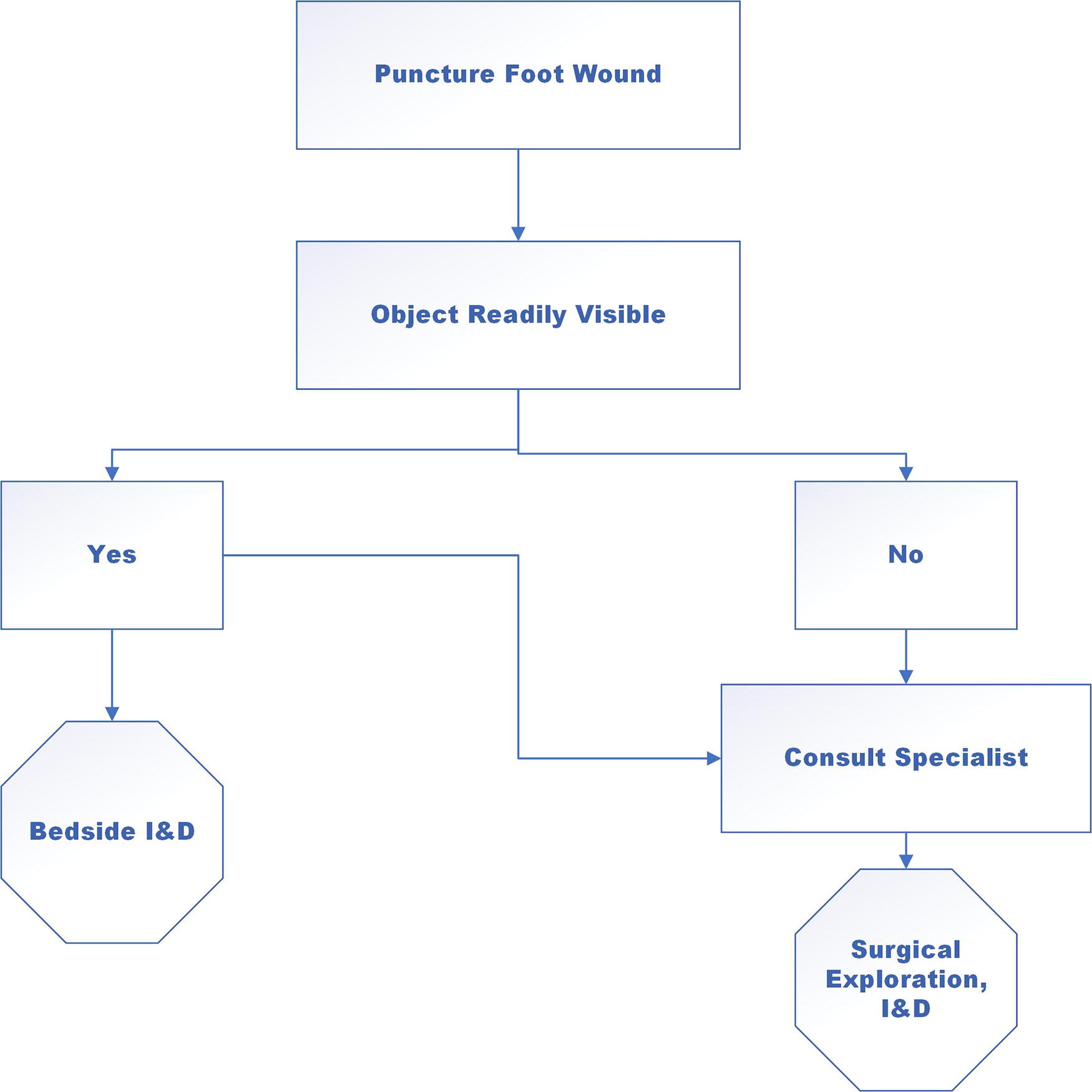
Puncture wound evaluation algorithm. A readily visible foreign body can be extracted at the bedside by an appropriately trained clinician. If the object is not readily visible or there is concern for associated injuries, a consult with the appropriate specialist may be warranted for surgical exploration.
All bedside procedures should be performed in a sterile fashion, including incision, drainage, and debridement with copious irrigation using sterile saline solution. Empirical antibiotic therapy should be initiated to target the suspected pathogens at hand, keeping in mind that Staphylococcus aureus is the most common bacterial contaminant. 83 Puncture wounds that occur in contaminated environments (e.g., farms and industrial areas) can result in polymicrobial infections, which include gram-negative and anaerobic bacteria. Superficial infections usually can be treated adequately with oral antibiotics. Deep wounds or those with suspected bone and joint involvement should include parenteral therapy and appropriate surgical debridement.
Special considerations for diabetic foot wounds
Individuals with diabetes are at risk for more complex courses following an acute puncture wound. This is due primarily to neuropathy, which can lead to a longer incubation time and a higher risk for delay in seeking treatment. 88 In addition, impairment of blood flow to the foot, a common feature in severe diabetes or peripheral vascular disease, also influences the management of foot wounds. 89 The Society of Vascular Surgery Wound, Ischemia, and Foot Infection (WIfI) classification system is a standardized method for classifying the severity of peripheral arterial disease in patients with diabetes (Fig. 7). 90 The use of this system has resulted in a paradigm shift in evaluating the risks of amputation and the potential benefits of vascular intervention. 90 The WIfI classification system takes into account the severity of limb-threatening ischemia (reduced blood flow), the presence of foot wounds or ulcers, and the extent of foot infection. Patients with higher WIfI scores generally have a worse prognosis and may require more aggressive treatment to prevent limb loss. 90
Wound, ischemia and foot infection classification of foot wounds. The nature of the wound and associated ischemia and/or infection may impact the outcome of acute foot wounds, and are useful in counseling. This figure illustrates the objective assessment of each of these components of a foot wound. SIRS, systemic inflammatory response syndrome; TCPO2, transcutaneous oximetry.
Management of diabetic foot ulcers and offloading
The management of new onset diabetic foot ulcers generally begins with a thorough analysis of patient-centered factors and a comprehensive assessment of the ulcer. The presence of any local or systemic infection, underlying neuropathy, malnutrition, peripheral artery disease, and any structural osseous deformities should be actively treated and addressed systematically. Patients with diabetes who present with neuropathic plantar forefoot ulcers devoid of poor circulation or infection should heal in six to eight weeks with adequate offloading. Foot ulcers that are subjected to frequent or sustained pressure and stress vectors (i.e., pressure-related heel ulcers or forefoot sub-metatarsophalangeal joint ulcers) or repetitive moderate pressure (plantar foot ulcers) will benefit from pressure reduction. 91 Conservative measures include the incorporation of mechanical offloading devices. Several shoe gear devices are available to achieve mechanical offloading, including those described below:
Total contact cast (Courtesy of David Armstrong, DPM). Full cast with a heal for off-loading.
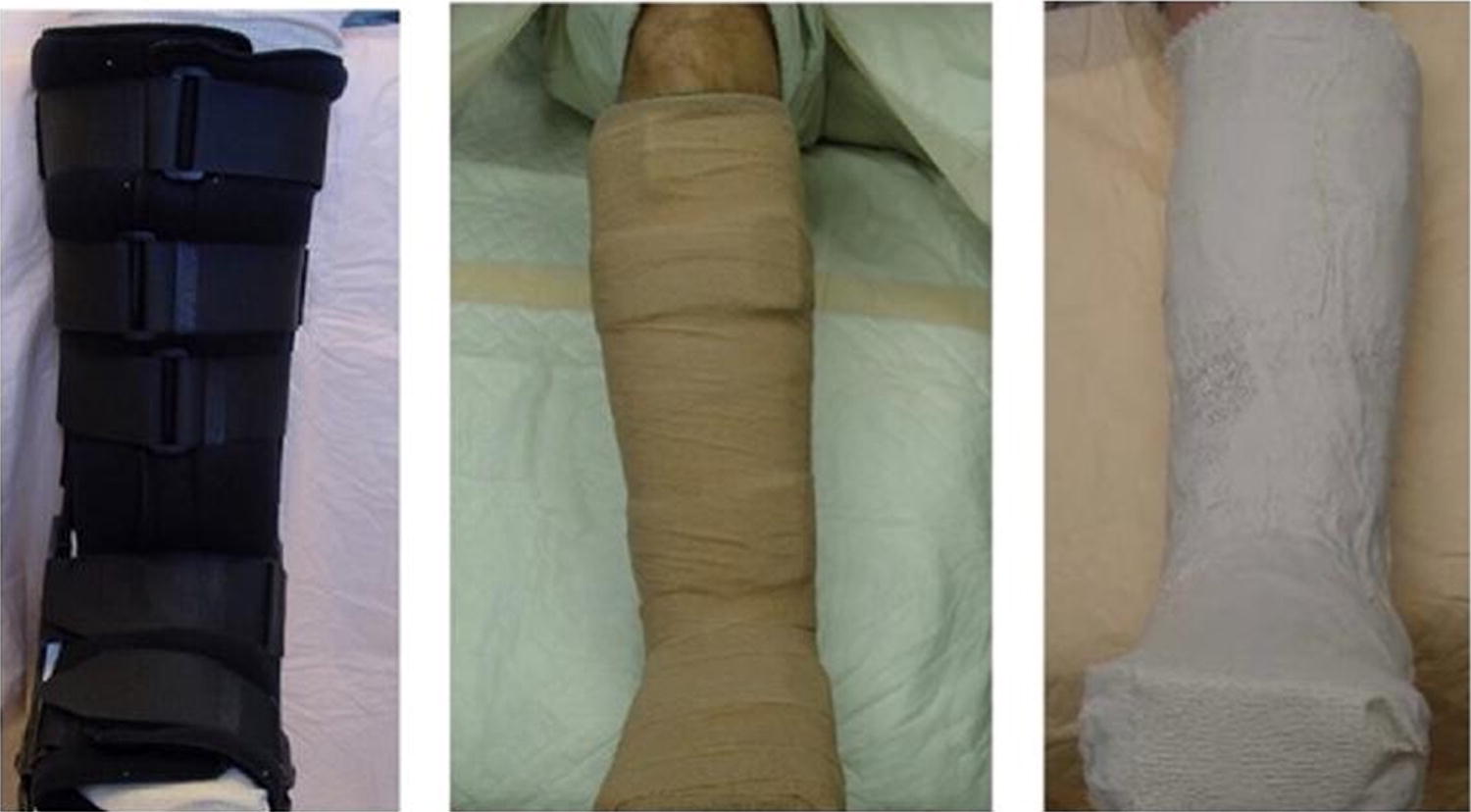
Removable cast (Courtesy of David Armstrong, DPM). Different forms of removable casts.
There are some clinical scenarios in which osseous deformities are too prominent, impeding healing potential. In such cases, surgical intervention at the of apex of the deformity may be required to optimize healing potential. Such examples include bunion/hammertoe corrections, surgical ostectomy (i.e., resection of prominent bone structures) of osseous prominences, Achilles tendon or gastrocnemius lengthening, and Charcot foot reconstructions, among others. These are frequently chronic conditions but may have an acute presentation when a wound develops.
High-velocity wounds and orthopedic considerations in acute wounds
Forward progress in the understanding of wound care and surgical management of wounds has always paralleled military conflict. 93,94 A broad coalition of nations has been at war for nearly 20 years. The predominant mechanism of injury has been the low-tech improvised explosive device. This weapon creates massive amounts of soft tissue trauma, hemorrhage, and devastating wounds. 93,95 Despite this, the advancement of body armor has increased survivability on the modern field of combat. Many soldiers sustaining severe extremity wounds survive what previously would have been fatal injuries. Many of these high-velocity wounds typically seen in combat are increasingly being experienced in the civilian arena. 96 The lessons from the management of these wounds in combat discussed in this section are also applicable off the battlefield.
Initial management and stabilization
The acute management of combat trauma wounds begins at the very point of injury. 93,97,98 Traditionally, the use of tourniquets was not advised; however, a landmark study by Holcomb et al. 94 demonstrated that hemorrhage was the single greatest cause of survivable death on the battlefield. After 20 years of experience, tourniquets are now ubiquitous on the battlefield, and in the civilian sector. Whether on the battlefield or in a Level I trauma center, patients with significant extremity wounds will present with tourniquets “high and tight,” which leads to necessary ischemia of the limb (Fig. 10). The first principle of combat wound management is to limit tissue ischemia, which entails restoration of blood flow to extremities as soon as possible, damage control resuscitation, and aggressive use of fasciotomies. 97 –99 The algorithm of wound management depends on the location of the initial management and should follow standard principles of bone stabilization, restoration of vascular in-flow and out-flow, liberal use of fasciotomies, and damage control resuscitation.
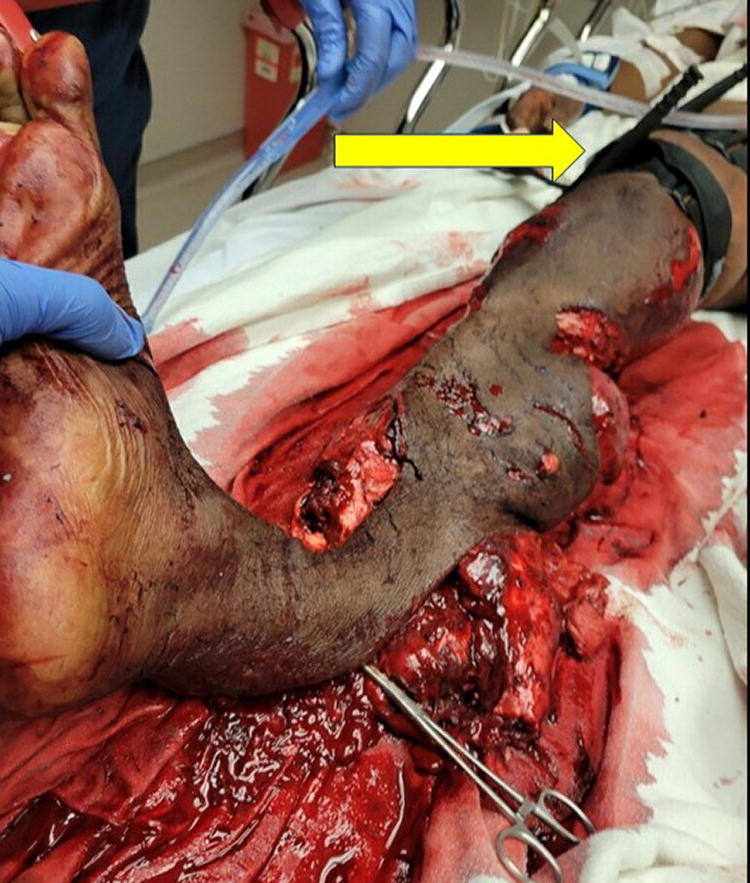
High Velocity wound with tourniquet. (Courtesy of Sam Sheppard, RN, EMT—Memphis, TN). High velocity injury of the lower extremity with major blood loss controlled with application of a tourniquet (yellow arrow) proximal to the injury.
Once the primary and secondary surveys are completed and life-threatening injuries temporized, the tourniquet(s) can be released, and the extremity evaluated. Often in combat trauma, rapid recognition of injury patterns allows the team to identify those patients who will require massive resuscitation. During the evaluation of the trauma patient, aggressive resuscitation with blood products (packed red blood cells, fresh frozen plasma, platelets, and cryoprecipitate) or even fresh whole blood can be employed to help restore perfusion. Previously, it was thought that the initial time to surgical debridement was the most important factor in mitigating infection risk. 100 –102 It has now been recognized that prevention of infection after trauma requires prompt administration of initial antibiotics and timely admission to a definitive trauma treatment center.
Management of vascular high-velocity injury
When a wounded extremity is identified as having a vascular injury, restoration of blood flow is essential to limit tissue ischemia. All combat surgeons should be comfortable with the placement of vascular shunts. 97 This technique is taught as part of the Advanced Surgical Skills for Exposure in Trauma course (ASSET™) provided by the American College of Surgeons. Due to the potential for prolonged evacuation times and uncontrollable variables, the surgeon should liberally employ the use of fasciotomies. 98,99 In addition to the overall burden of trauma, the ischemia-reperfusion injury and massive resuscitation will create significant swelling within the extremity. The mantra should be: “Big Problems Get Big Incisions!” Care should be taken to ensure that complete fasciotomy of the anterior and deep compartments of the leg, and compartments of the hands and feet be performed as applicable. When in an austere environment, bony stabilization using a simple splint cannot be underappreciated. When a vascular shunt is in place, external fixation of the extremity should be performed to not dislodge or occlude the shunt during medical evacuation. In a larger medical center or location where definitive care of the extremity can be performed, external fixation provides a quick option to stabilize the extremity while resuscitation is ongoing. Definitive vascular repair of arterial inflow, and if possible venous outflow, should be performed as soon as possible.
Once the patient has been stabilized, vascular inflow and outflow established, wide fasciotomies performed, and bony stabilization established, the wounds will begin to declare themselves. This process is combined with extensive and repetitive debridements. In the extensive soft tissue trauma seen in combat injuries, debris can be blasted throughout the soft tissues, even far from the obvious injuries. The soft tissues, while initially viable, may continue to necrose over time. Thus, multiple careful debridements should be performed under loupe magnification before any discussion of reconstruction. During these debridement sessions, the use of high-pressure irrigation (such as pulsatile irrigation) should be avoided as it increases tissue edema, pushes debris further into the tissue, damages the soft tissue, and propagates bacteria into the tissue. 103,104 Warm saline running through cystoscopy tubing combined with a scrub brush provides ample pressure and gentle debridement. Necrotic muscle can be readily identified by applying electrocautery. Necrotic muscle will not contract with the application of electricity, whereas viable muscle tissue will. Sharp debridement of necrotic tissues to healthy, vascularized tissue is the standard. This debridement can be done with a simple 15-blade scalpel or with a hydrosurgery system. Placement of NPWT dressings is extremely helpful through this process. NPWT helps to control tissue edema and fluid shifts, aids in nursing care, develops a stable wound, and helps the surgeon to further distinguish between vascularized tissue and tissue that needs further debridement. This process allows the surgeon time to develop a stable wound, appreciate the injury pattern and understand the best reconstructive options.
Orthopedic considerations in high velocity wounds
The decision of amputation versus limb salvage is driven by many factors. In 2021, the American Academy of Orthopedic Surgeons published a CPG to assist in the decision-making process for early amputation versus limb salvage. 105 This CPG highlights the complex decision-making process that a multidisciplinary team faces. The decision of limb salvage or early amputation is optimally made only when the wounds have stabilized, necrotic tissues have been debrided, and the extent of the injury is fully appreciated by the managing team and patient.
Future directions in high velocity wound care
The future of conflict will likely see large-scale force-on-force battles and require non-surgeons to manage patients for prolonged periods of time. As military engagements continue to increase, weapon systems are becoming increasingly lethal. In this complex environment, the innovation of novel wound management advances in the management of hemostasis, and resuscitation will be necessary to preserve life. Most of the focus on innovation is in the realm of hemostasis and resuscitation; however, along with these advances, management of severe wounds will continue, by necessity, to evolve. War and urban violence will always continue to drive the demand for expert wound management.
Scar management
All wounds heal with distortion of structural architecture and the formation of a scar. The healing response can result in a barely noticeable scar but can also result in hypertrophic scar (HTS) or keloid formation. Reducing scar formation is an important consideration in the treatment of acute wounds.
One of the most effective ways to reduce scarring is to ensure proper wound closure. For incisional wounds, this begins with the choice of the location of the incision. The location and orientation of the incision relative to tension lines will allow for proper wound apposition without tension. 106 Avoiding incisions across joints and areas subjected to significant motion will also reduce scarring. Wound apposition can be achieved through various techniques such as sutures, staples, or adhesives. There are conflicting reports about whether skin staples are superior to subcuticular sutures in closing skin wounds, especially in obese individuals. 107,108 The benefits of adding tissue adhesives to subcuticular skin closure compared to skin staples were also not borne out in a recent meta-analysis. 109 The choice of wound closure technique should be determined based on the wound location, patient factors, tension at the wound edges, time, and cost. In addition to proper wound closure, early and aggressive wound care is also important in reducing scarring. This includes keeping the wound clean and moist and avoiding excessive tension on the wound edges. When wounds or incisions are in areas of high tension, dressing strips that reduce the tension at the wound edges can mitigate scar formation. 110 For wounds where the edges cannot be easily approximated, NPWT can help with wound contraction and reduce the need for complex wound closure (e.g., skin grafting, flaps). 111 Even for wounds healing following primary closure, there is some evidence that incisional NPWT may reduce SSIs. 112
Another strategy to reduce scarring is the use of silicone gel sheets or creams. Silicone has been shown to improve the appearance of scars by reducing their thickness and redness. 113 The mechanism behind this is not fully understood, but it is believed that silicone helps to regulate the production of collagen, which is responsible for scar formation. 114 The use of silicone dressings has also been shown to be effective in reducing scar formation in burn wounds. 115 A more recent meta-analysis of existing data did not provide conclusive support of silicone gel sheeting, calling into question its impact on health-related quality of life as well as cost. 116 Other modalities for managing HTs or keloids, when they develop, include laser treatment, steroid injection, and surgical excision in isolation or in various combinations. 117 Lastly, it is important to note that the management of pain and inflammation can also play a role in reducing scar formation. Pain and inflammation can delay wound healing and contribute to scarring, so the use of analgesics and anti-inflammatory agents may be beneficial. Excessive movement at the wound site can also worsen pain and inflammation and thus impact scar formation.
Abnormal scars can also form following the healing of acute wounds. These can be HTS or keloids. HTS are confined to the site of injury and typically occur in wounds that have taken a long time to heal, especially burns. Keloids are abnormal scars that extend beyond the margin of the original wound and are more commonly found in people of color. Keloids can form even following innocuous or incidental injury. HTS tend to form over several weeks to months and may spontaneously regress after about a year. In contrast, keloids may continue to grow over the years and rarely undergo spontaneous regression. There are many treatment modalities that have been employed for HTS and keloids that are beyond the scope of this article but are referenced here. 117 –122
In conclusion, the reduction of scar formation in acute wounds is an important consideration in wound management. Proper wound closure, early and aggressive wound care, the use of silicone dressings, limiting movement around the wound, and the management of pain and inflammation are all strategies that can be used to reduce scarring. By using a combination of these approaches, healthcare professionals can help to ensure that patients achieve the best possible outcomes following acute wounds.
Dressings for acute wounds
The proper management of acute wounds is crucial to ensure their timely healing and to prevent the development of complications such as infection and scarring, and a moist healing environment is desirable. One important aspect of wound management is the selection of appropriate dressings. Different types of dressings are available for the management of acute wounds, each with its own indications and benefits (Table 4).
Dressing types, indications and benefits
Different types of dressings are available for the management of acute wounds, each with their own indications and benefits as denoted in this table.
The choice of wound dressing is based on the nature of the wound. Options vary depending on whether the wound is closed or left open. In addition, deep or shallow wounds, the presence of excessive drainage or exudate, and whether the wound is moist or dry.
The majority of acute wounds are surgical incisions that are closed primarily. These can be covered with a gauze or foam dressing. The intent for these dressings is to provide a physical barrier as the underlying wound epithelializes. In addition, the dressing absorbs any minimal blood or wound fluid that may drain from the wound and provides some protection against direct trauma. A waterproof covering allows the patient to shower with the dressing in place. These dressings can typically be removed in 2–3 days by which time the incision should have epithelialized considerably. There is some evidence that incisional NPWT reduces SSIs. 123,124 This is presumed to be due to its ability to decrease hematoma or seroma formation, decrease tension at the wound surface, and enhance regional lymphatic flow. For open wounds that are dry, hydrogels impregnated gauze or hydrocolloids are useful to hydrate the wounds. 125 For large bloody, high-velocity wounds, hemostatic gauze, impregnated with kaolin or other substances that induce the intrinsic clotting response in the patient is useful to ensure prompt hemostasis and control of hemorrhage. In addition to the application of direct pressure to the bleeding site, packing with these hemostatic gauze packings and then applying pressure dressings can be life-saving. 126 For wounds that are moist with a large amount of drainage, control of the drainage is important to prevent maceration of the surrounding skin. Application of foam dressings or calcium alginate dressings will help absorb the excess fluid. 127 NPWT is also quite effective in these settings. When there is considerable tissue loss, and a wound cavity, the wounds can be “packed” with moist gauze dressings, calcium alginate/gelling fiber or managed with NPWT (Fig. 11). NPWT promotes granulation tissue formation and enhances wound contraction.
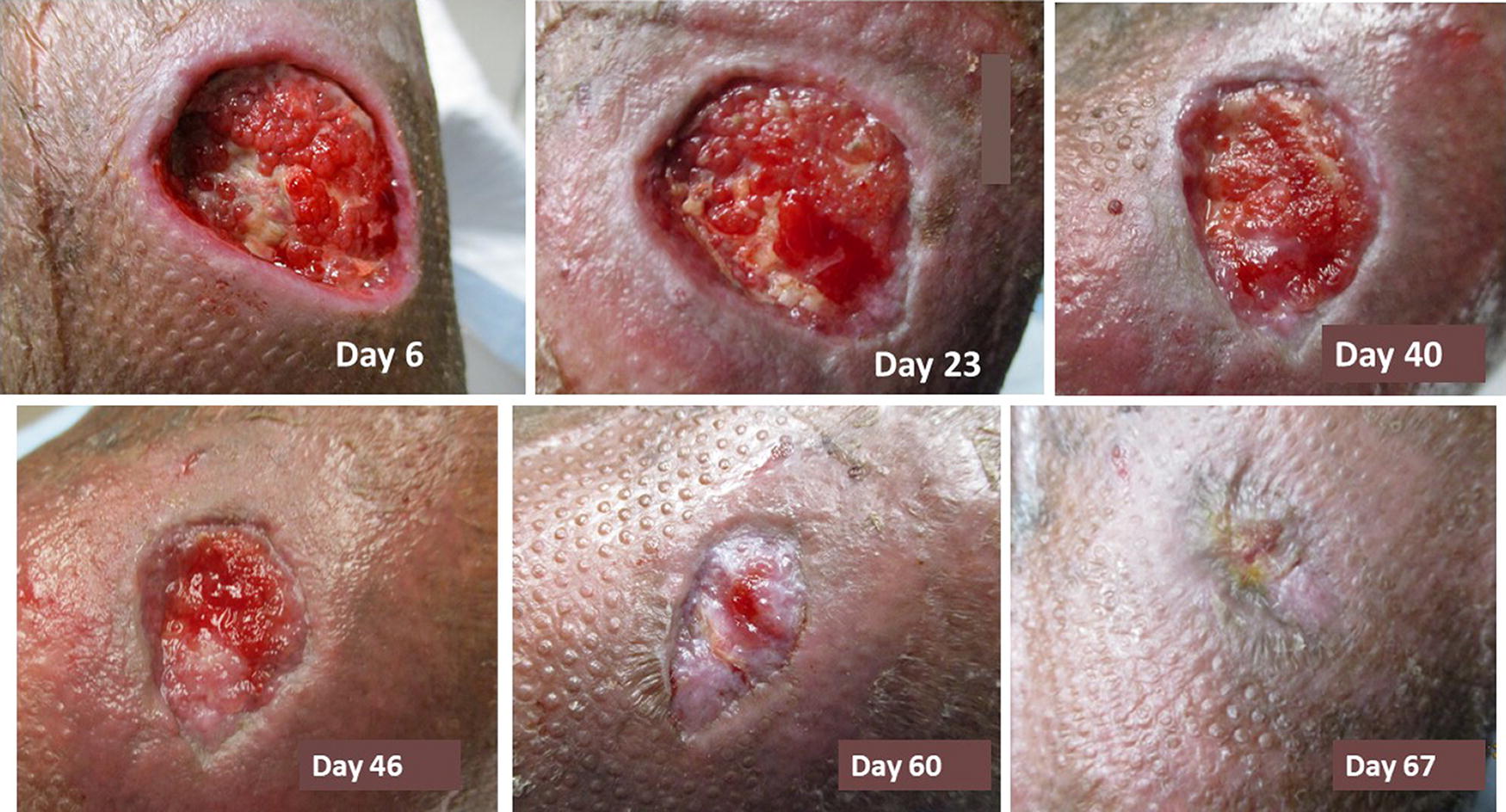
Wound with cavity treated with negative pressure wound therapy with the formation of granulation tissue and skin level healing over time. (Courtesy of Dot Weir, RN).
A systematic review of the literature denotes only weak levels of evidence on the clinical efficacy of modern dressings compared with saline or paraffin gauze in terms of healing, with the exception of hydrocolloids. 128 More wound care research providing stronger levels of evidence is needed.
In summary, the selection of the appropriate dressing is an important aspect of wound management, especially in the care of acute wounds. Hemostatic gauze packings can help with the control of bleeding in the acute setting. Non-adherent dressings, foam dressings, alginate dressings, and hydrocolloid dressings, that provide a moist healing environment, are all effective therapeutic options that can be used depending on the type and severity of the wound. The proper selection of dressing can help to promote healing, prevent complications, and improve patient outcomes.
SUMMARY
Acute wounds are one of the common presentations to EDs. In addition to planned surgical incisions, these wounds can occur in all parts of the body. The general approach to acute wounds includes wound preparation (infection control), pain management, prompt apposition of the wound edges when possible, and the application of appropriate dressings to reduce the possibility of abnormal scar formation. Wounds in specific parts of the body (face, hands, and feet) or sustained as a result of unique mechanisms (burns or high velocity) require special consideration and may need specialized expertise. The approach to wound management in children should take into consideration the developmental stage and psychosocial needs of the child while still ensuring timely and proper wound care. Clinicians should be cognizant of the cost and efficacy of various treatment approaches while selecting the appropriate modality for each individual patient.
TAKE-HOME MESSAGES
Acute wounds are wounds that heal in a timely fashion, typically by four weeks after healing conditions have been optimized. Early, proper treatment of acute wounds helps to avoid the development of chronic wounds. The goal of acute wound treatment is not to eliminate all microorganisms from an acute wound bed but to prevent the progression of the microbial “bioburden” to levels that impede complete healing in a reasonable time. Tetanus prophylaxis and antibiotics are important adjunctive therapies for all injuries and should be tailored to the individual situation/level of contamination. Pain associated with acute wounds should begin to diminish over time as healing progresses. Acetaminophen and/or NSAIDs are typically sufficient for pain control. Opioids can be considered for more severe pain. When possible, local anesthesia should be provided during cleansing, repair or manipulation of the wound, with consideration given to the possible complications of local anesthetics such as cardiotoxicity and central nervous system symptoms (especially in children). Management of burn injuries is a complex, multi-staged process that starts with first aid and continues through scar prevention and treatment with ongoing psychological support to assure maximal recovery. Minor burns can be treated outside of a burn center so long as the treating team is familiar with burn care and American Burn Association recommendations. Formation of HTS is directly proportional to the time of wound healing, with less than 14–21 days considered to yield lower rates of scarring. Facial lacerations can be closed after 18–24 hours if the edges are even (or later if the edges are excised and freshened) and well irrigated with saline under low pressure. Irrigation is particularly important in contaminated wounds, including dog and human bites, as these carry a higher risk of infection. When a laceration can be optimally closed with surgical tape or glue, this should be done; however, when sutures are needed, in pediatric patients it is often preferable to use absorbable (chromic) sutures to avoid the often painful and sometimes psychologically traumatic procedure to remove the sutures. In hand wounds accurate history-taking and physical examination are the foundation for maximizing outcomes. Plain X-rays are also necessary. Dressings and splinting of hand injuries, either to minimize contractures or to provide comfort during healing, should be appropriate for the specific injury. Superficial infections usually can be treated adequately with oral antibiotics. Management of deep infection of hand or foot wounds or those with suspected bone and joint involvement should include intravenous antibiotic therapy and appropriate surgical debridement. Patients with diabetes may not present with the classic signs of foot infection and, thus, are at risk for more complex treatment courses following an acute puncture wound. Tourniquets, which result in necessary ischemia of the limb, mitigate hemorrhage. The algorithm of wound management depends on the location of the initial injury and should follow standard principles of bone stabilization, restoration of vascular in-flow and out-flow, liberal use of fasciotomies, and damage control resuscitation. During debridement sessions, the use of high-pressure irrigation should be avoided as it increases tissue edema, pushes debris further into the tissue, damages the soft tissue, and propagates bacteria into the tissue. One of the most effective ways to reduce scarring is to ensure proper wound closure. For incisional wounds, the location and orientation of the incision relative to tension lines will allow for proper wound apposition without tension. Effective management of movement and inflammation can also play a role in reducing scar formation.
Footnotes
ACKNOWLEDGMENTS
Nana E. Coleman, M.D., Ed.M. for her editorial contributions.
FUNDING INFORMATION
The expenses (personnel, IT, recording, and transcription) were covered by unrestricted donations (David Zamierowski, M.D., Mimedx, and 3M). No editor or panel member received any payment for their participation.
AUTHOR CONFIRMATION
O.O.O., E.E., A.D.M., R.S.K., R.T., G.S., T.L.W., D.W., R.B.F., B.N.-M., P.Y.L., K.E., D.G.A., N.M., and J.B.S. participated in data gathering, panel discussion, consensus statement voting, article drafting, and critical editing of the article. O.O.O., E.E., and A.D.M. participated in the conceptual design of the project and recruitment of panelists.
AUTHOR DISCLOSURE AND GHOSTWRITING
The authors have no relevant disclosures. The views expressed herein are those of the authors, not the Department of Defense or its elements, nor the institutions of the authors or the wound healing foundation.
ABOUT THE AUTHORS
