Abstract
Significance:
In adult mammals, spontaneous repair of a cutaneous wound occurs slowly and leaves a scar with skin adnexa deficiencies. To accelerate cutaneous wound-healing rates and avoid scar formation, current studies have focused on regenerative therapies.
Recent Advances:
Emerging therapeutics for regenerative wound healing often focus on the use of growth factors and stem cells. However, these therapeutic approaches have limited routine clinical use due to high costs and technical requirements.
Critical Issue:
Understanding the molecular mechanisms involved in the signaling pathways for cutaneous wound healing and neogenic synthesis of the skin components is important for identification of novel targets for the development of regenerative wound-healing agents.
Future Directions:
The Wnt/β-catenin pathway is a well-known key player for enhancement of the overall healing process involving tissue regeneration via crosstalk with other signaling pathways. Strategies that activate the Wnt/β-catenin pathway via modulation of the pathway-controlling regulatory factors could provide effective therapeutic approaches for regenerative wound healing.

Scope and Significance
Our understanding of wound healing has increased over the past several decades along with advancements in the understanding of the molecular mechanisms involved in the process. In recent years, clinicians have been interested in the regenerative medications that increase the rates of wound healing, without scar formation. However, current therapeutic agents (e.g., growth factors) have limited use due to drawbacks such as poor efficacies, high costs, and low delivery rates. In this review, we describe distinctions between repair and regeneration in the wound-healing process; then, we discuss current regenerative therapies and their limitations. Next, we discuss a future direction for the development of novel therapeutics that can induce regenerative wound healing by targeting the Wnt/β-catenin pathway, a key signaling pathway involving skin regeneration. Finally, we suggest an approach targeting CXXC5, a negative feedback regulator of the Wnt/β-catenin pathway, as a safe and effective strategy for the development of regenerative wound-healing agents.
Translational Relevance
Instead of repair, which often results in scar formation, current therapeutic approaches for wound healing aim at stimulating a regenerative response that restores the wounded skin to the preinjured state. Therefore, understanding the molecular mechanisms of the signaling pathways involving regenerative healing is important for the development of regenerative wound-healing agents. The Wnt/β-catenin pathway plays important roles in multiple wound-healing processes, including cell proliferation and tissue remodeling. It also participates in stem cell activation and growth factor expression. Therefore, targeting the Wnt/β-catenin pathway could be an ideal approach for regenerative wound healing.
Clinical Relevance
New therapeutics have been developed subsequent to the biological and technical advances in the field of wound healing. However, current therapeutic agents are limited due to unsatisfactory efficacies, economic burdens, and adverse effects. The Wnt/β-catenin signaling pathway is an attractive target for the treatment of many diseases related to tissue homeostasis, including wound healing. Approaches that search for Wnt/β-catenin pathway activating compounds have been developed. A strategy that activates this pathway via blockade of its negative feedback mechanism could be a potential approach for the development of regenerative wound-healing agents.
Background
Wound healing is a complex process consisting of multiple phases (e.g., inflammation, proliferation, and remodeling) controlled by coordinated interactions among various cells, growth factors, cytokines, and chemokines. 1,2 A failure during any of these phases results in abnormal scar or chronic wound formation. As the elderly population increases, the incidence of chronic and nonhealing wounds is increasing, and the global wound care market is growing. 3 Numerous wound care products and therapies have also been developed.
Conventionally, a skin wound was managed by using antibiotics and wound dressings that prevent infection and ensure sufficient tissue perfusion. 4 However, these traditional therapies cause scars, which result in functional and cosmetic impairments, including increased sensitivity to ultraviolet radiation and deficiencies in skin structures (e.g., sweat glands and hair follicles). With the growing interest in higher-quality wound healing, therapeutic approaches using growth factors and stem cells have been introduced as regenerative medicines for complete recovery of the damaged tissue without a remaining scar. 5 –7 However, these approaches have limitations for routine clinical use due to drawbacks, including high costs, technical difficulties, and delivery. 8 –10 Although a drug delivery system has been advanced, these issues remain to be resolved. 11
Over the past several decades, tremendous improvements have been made in understanding the relationships between signaling mechanisms and the wound-healing process. The transforming growth factor beta (TGF-β) pathway, the Notch pathway, the Hedgehog pathway, and the Wnt/β-catenin pathway are signaling pathways that are important for skin regeneration. The absolute requirements for these pathways during skin development have been characterized through many mammalian studies. 12 Therefore, these pathways have been suggested as targets for the development of regenerative wound-healing agents that contribute to the complete restoration of wounded skin.
In this review, we suggest activation of the Wnt/β-catenin pathway, especially via release of the negative feedback regulation by CXXC5 as a potential therapy for acute wounds. Inhibition of CXXC5 function via blockade of its interaction with Dishevelled (DVL) enhances regenerative wound healing. 13 This approach, which activates the Wnt/β-catenin pathway via blockade of the negative feedback mechanism, is safe and results in minimal scar formation.
Discussion of Findings and Relevant Literature
Repair and regeneration in wound healing
The healing of wounded skin occurs by either simple repair or regeneration. Repair implies reestablishment of the structural continuity of injured or damaged tissue, and it results in scar formation with tissue dysfunction. Regeneration, on the other hand, implies replacement of the injured tissue with newly generated tissue, resulting in full restoration of tissue morphology and functionality.
Wound repair
Wound healing is a dynamic process that restores the structural and functional characteristics of damaged tissues. It includes complex cellular and biochemical interactions involving multiple types of cells, extracellular matrix (ECM), growth factors, and cytokines. 1,2 This interactive process consists of four phases: hemostasis, inflammation, proliferation, and remodeling (Fig. 1). 14 In response to injury, the requirements of each phase coordinately function to achieve tissue repair. Often, however, the repaired skin is not identical to the uninjured skin and induces the formation of a nonfunctioning mass of fibrotic tissue, or a scar. 6
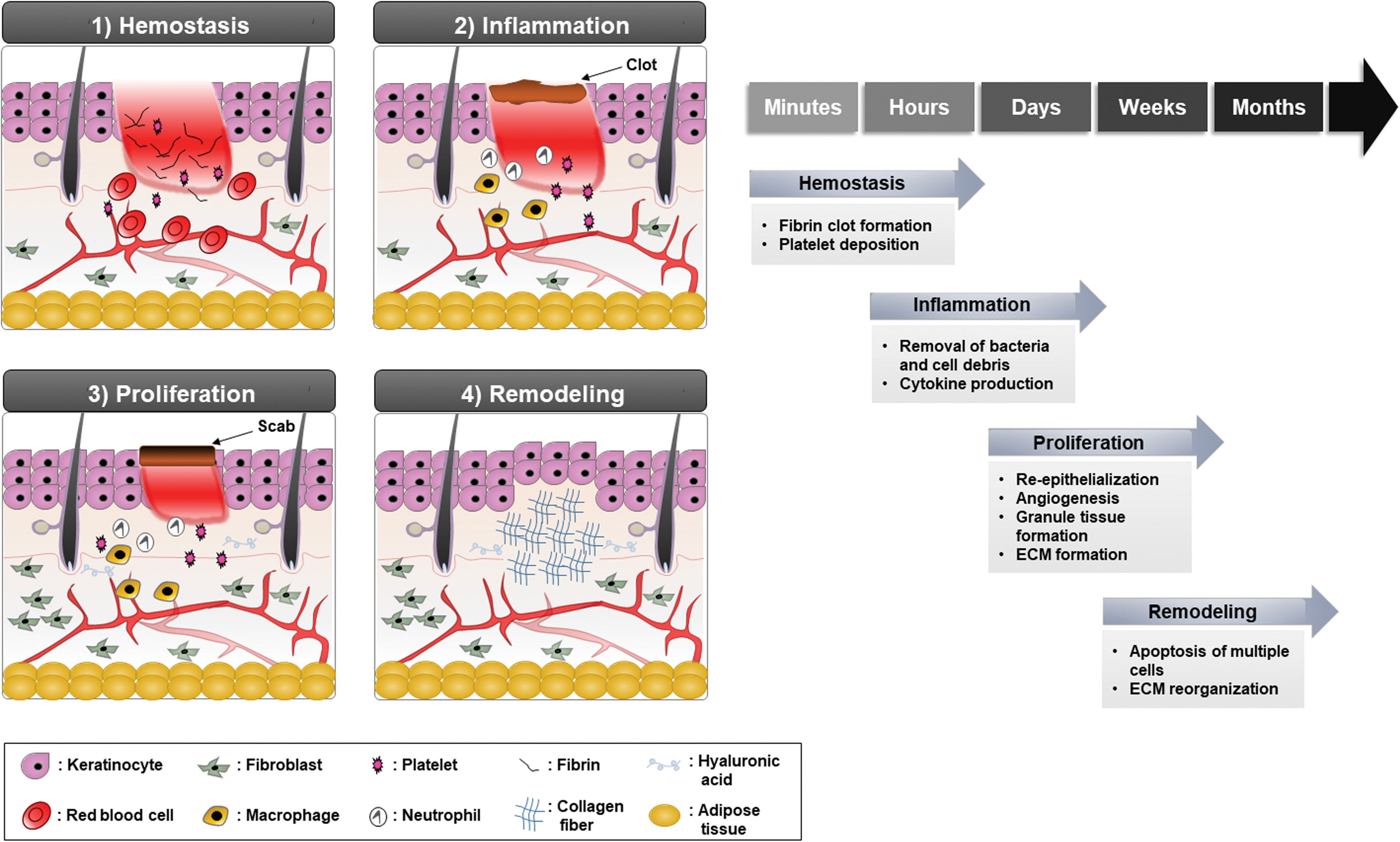
Wound repair phases. The four different stages of the wound repair process.
Fibrosis
Adult mammalian skin typically responds to injury by fibrotic repair. Fibrosis or scarring is attributed to the deposition of excess amounts of ECM components, such as collagen. The interposition of fibrotic tissue hinders skin adnexa formation and subsequent tissue regeneration. 6 The inflammatory process could be involved in fibrotic healing. 15 Although inflammation is crucial for protection of the body from infection by foreign organisms at the wound site, the deregulated pro-inflammatory cytokines, including interleukin (IL)-1β and tumor necrosis factor (TNF)-α, contribute to the fibrotic process and can cause a chronic wound state or abnormal wound repair such as a hypertrophic scar and a keloid. 16
Regenerative wound healing
The regenerative healing of wounds is characterized by complete restoration of tissue morphology and function (Fig. 2). In contrast to adult mammalian tissue, injured embryonic tissue can be completely regenerated without scarring. 17 –19
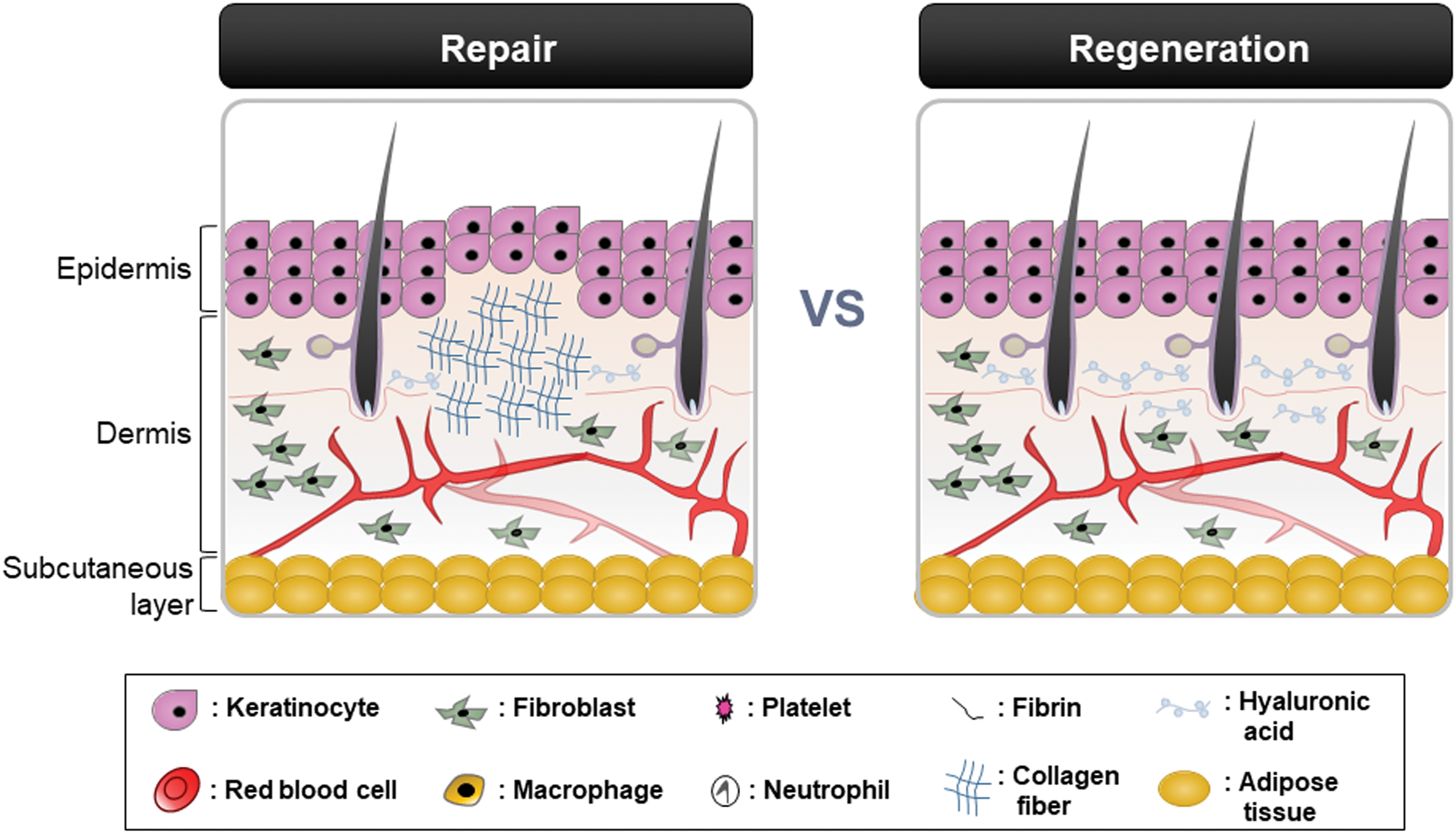
Comparison between repair and regeneration in skin wound healing. Left, Repaired skin. Healing by the repair process fails to restore skin to uninjured status and remains scar due to the alignment of excessive collagen fibers in the dermis. Right, Regenerated skin. The regenerative healing induces de novo synthesis of hyaluronic acid, and it results in complete restoration of skin tissue with the formation of adnexa including hair. Color images are available online.
To determine the key factors mediating regenerative healing, it is important to understand the differences between embryonic and adult wound healing. In addition to the immature inflammatory responses mentioned earlier, embryonic tissue retains a relatively abundant stem cell population compared with adult tissue. Therefore, it is plausible that the restricted regenerative capacity of adult mammals is attributable to lowered population of stem cells or deficiency of proper environmental signals. Recent studies demonstrate that new tissue is generated by de-differentiation and transdifferentiation of adult cells at the edges of the wound. 20,21 In addition, it was demonstrated that the epidermis of wounded adult mice regenerates skin adnexa, including hair and sweat glands, depending on characteristics such as wound size and status of specific signaling, for example, the Wnt/β-catenin pathway. 22 These observations indicate that there are factors leading to an embryonic skin-like environment in the wounded skin area during the healing process of adult mammals. This could occur by de-differentiation of adult cells to a stem cell-like state or recruitment of stem/progenitor cells into the wounded region. The Wnt/β-catenin pathway is closely related with these critical events involving activation of adult stem cells. 23,24 Wnt/β-catenin signaling plays important roles in determination of the fate and proliferation status of progenitor cells during embryonic development, as well as in maintenance of tissue homeostasis during the postnatal period. 25,26 Therefore, it is worth considering the Wnt/β-catenin pathway as a target for the development of regenerative wound-healing agents.
Earlier, we discussed the role of the Wnt/β-catenin pathway in regenerative wound healing and its underlying mechanism in detail; we will briefly review current regenerative therapies and their limitation.
Current regenerative therapies and their limitation
Growth factor-based therapies
Platelet-derived growth factor
Platelet-derived growth factor (PDGF) participates in cell growth and angiogenesis during the wound-healing process. 27,28 PDGF comprises three isoforms: PDGF-AA, -BB, and -AB. Although PDGF-BB is a growth factor first approved by the U.S. Food and Drug Administration (FDA) for application to treat chronic wounds, including diabetic foot ulcers, 29 controversial reports for its efficacy have been emerged. 30,31
Epidermal growth factor
Epidermal growth factor (EGF) induces re-epithelialization by promoting epithelial cell proliferation and migration. 32,33 It also induces angiogenesis and tensile strength of new skin. Clinical studies found that topical application of human recombinant EGF promotes epidermal regeneration of partial-thickness wounds and second-degree burns. 34 –36
Transforming growth factor beta
The main function of TGF-β is regulation of wound contraction and scarring. 37,38 The TGF-β family comprises three functional isoforms: TGF-β1, TGF-β2, and TGF-β3. Although TGF-β1 and -β2 facilitate fibroblast–myofibroblast differentiation and ECM deposition, they often result in fibrosis and scar formation. TGF-β3 promotes ECM reorganization and scar reduction. Administration of Avotermin (human recombinant TGF-β3) showed significant improvement in prevention of scar formation, but it failed to meet its endpoints in a phase III clinical trials. 39,40
Vascular endothelial growth factor
Vascular endothelial growth factor (VEGF) induces the initiation of angiogenesis by promoting endothelial cell proliferation and migration. The VEGF family consists of VEGF-A (VEGF165), VEGF-B, VEGF-D, VEGF-E, and placental growth factor. A clinical study found that VEGF-A enhances vessel formation and improves re-epithelialization of diabetic foot wounds. 41,42 A phase I clinical trial of the topical application of Telberim (recombinant human VEGF) found that it accelerates ulcer healing in patients with chronic diabetic foot ulcer. 43
Fibroblast growth factor
Fibroblast growth factor (FGF) family, such as FGF-1, -2, -7, -10, and -22, have key roles during the wound-healing process by promoting angiogenesis and encouraging connective tissue cell division. 44,45 Especially, FGF-2 (i.e., bFGF) is able to regulate collagen distribution and reduce scar formation. Clinical trials of pressure ulcer and chronic burn wound treatments found that use of FGF-2 results in acceleration of healing rates. 46,47
Limitations
A variety of growth factor-based therapies have been developed, but they have had limited success. The development of recombinant growth factors is limited in their routine usage in the clinic due to high cost and high risk. There are also many obstacles in the clinical applications, because the growth factors have poor skin absorption rates and short half-lives due to their susceptibility to denaturation and proteolytic degradation. 48 Thus, they are prone to be removed by exudation before reaching the wound. To achieve healing, repeated administration of high growth factor concentrations is needed; however, the excess dose requirement could lead to local toxicity and adverse effects such as cancer development. 49 Further, growth factors are required to interact with specific surface receptors of the target cells for exhibition of their biological activities. This ligand–receptor interaction activates a series of intracellular signaling cascades followed by an expression of target genes involved in the wound-healing process. However, some cells within wounds lack growth factor stimuli due to defects in molecular components (e.g., downregulation of receptor). 8,50,51
Stem cell-based therapies
Mesenchymal stem cells
Mesenchymal stem cells (MSCs) have self-renewal and multipotent differentiation characteristics. 52,53 They can release a variety of paracrine factors that enhance wound healing (e.g., PDGF-BB, VEGF, and bFGF) and, subsequently, promote angiogenesis. 54,55 The MSCs have antibacterial and anti-inflammatory properties via the secretion of antimicrobial factors and anti-inflammatory cytokines, respectively. 56 One clinical study found that chronic skin wound healing was improved by local application of bone marrow- and adipose-derived MSCs. 57
Hematopoietic stem cells
Hematopoietic stem cells (HSCs) are self-renewing cells present in the bone marrow. When transplanted into mice, HSCs can differentiate into follicular epithelial cells, sebaceous gland cells, and epidermal keratinocytes. 58 They also promote angiogenesis. Topical application of HSCs led to an improvement in wound closure rates of full-thickness excisional wounds in diabetic mice. 59
Epithelial stem cells
Epithelial stem cells (EpSCs) are quiescent cells, but they self-renew and differentiate into at least one cell type. EpSCs have an important role in stratified epidermal regeneration. 60 They also participate in hair follicle neogenesis in the wound dermis. Transplantation of a bioengineered dermis containing EpSCs into acute wounds induces skin reconstruction and hair formation in goats. 61 Injection of EpSCs enhances vascularization, elastin content, and follicle-like structures when compared with the control group. 62
Limitations
Although stem cell therapy is a fast-growing field in regenerative medicine, many issues remain to be resolved (e.g., low safety, high cost, difficulty in administration, and quality control) before the routine clinical usage. There is currently no FDA-approved stem cell therapy for wound treatment despite numerous attempts. The age of the transplanted cells and the local microenvironment of the injured skin also need to be considered for therapeutic application of stem cells. 63 For example, MSCs derived from old mice rather inhibited wound healing in diabetic mice. 64 A favorable environment, including a sufficient blood supply, receptor, and presence of biological molecules, is necessary for effective use of stem cells as therapeutics for wound healing. These matters, together with the safety and quality control issues, make the clinical use of stem cell-based therapeutics challenging.
Future Direction
Emerging strategies for the development of regenerative therapeutics
Signaling pathway and wound healing
To overcome current limitations of regenerative therapeutics and to discover new therapies, it is necessary to understand the pathways involved in wound healing. Growth factor therapies eventually exert their biological activities through the downstream signaling pathway after interaction with specific surface receptors. The function of adult stem cells residing within tissues are modulated and reprogrammed by their microenvironment, especially molecular pathways. 65,66 Moreover, many mammalian studies have elucidated that flawless regeneration of embryonic skin wound absolutely depends on the activity of signaling pathways that are important for complete restoration of adult wound skin. 12 TGF-β, Notch, Hedgehog, and Wnt/β-catenin pathways are major players for regenerative wound healing.
The TGF-β pathway is differentially involved in the regulation of healing rate depending on the isoforms. 12 TGF-β1 functions as a fibrosis-stimulating factor but TGF-β3 regulates anti-scarring activity. 67,68 Members of the TGF-β superfamily participate in the development of skin or its adnexa, such as hair follicles. 69,70 The Notch pathway regulates epidermal cell differentiation during stages of adult and embryonic development. 71 This pathway also has important roles in the maintenance of skin homeostasis and promotion of angiogenesis. 72 –74 The Hedgehog pathway is involved in skin morphogenesis and angiogenesis. The Hedgehog pathway modulates dermal repair and wound vascularization during the wound-healing process. 75,76 The Wnt/β-catenin pathway plays a role in adult tissue regeneration, 77 –79 and it participates in multiple steps of the wound-healing process together with activation of stem cells residing within skin. 24 Proper regulation of the Wnt/β-catenin pathway is crucial for flawless and complete regeneration of wounded skin. Therefore, we are going to focus on the Wnt/β-catenin pathway as a target for the development of regenerative wound healing agents in the following sections of this review. Initially, small molecules or natural products that activate the Wnt/β-catenin signaling will be introduced, because those are easier to manufacture than growth factors or stem cells. In the later part, we will focus on a new strategy for the regenerative wound healing targeting CXXC-type zinc finger protein 5 (CXXC5), a negative feedback regulator of the Wnt/β-catenin pathway functioning via interaction with DVL. 13,80 This approach blocking the CXXC5–DVL interaction for activation of the Wnt/β-catenin signaling could be an effective and safe way for regenerative wound healing.
Regulation of the Wnt/β-catenin pathway
The Wnt/β-catenin pathway has essential roles in numerous biological processes, including cell proliferation, differentiation, and migration. It is well known for its role in stem cell self-renewal and differentiation during normal tissue homeostasis and tissue regeneration after injury. 78,79,81,82 The Wnt/β-catenin pathway has recently been characterized as a key modulator of cutaneous wound healing; Wnt/β-catenin signaling is upregulated by wounding and is involved in the overall stages of the healing process. 24 Therefore, activation of the Wnt/β-catenin pathway is an attractive strategy for cutaneous wound healing. The rate of development of wound-healing agents that activate the Wnt/β-catenin pathway has increased in recent years.
Wnt/β-catenin signaling pathway
The hallmark of Wnt/β-catenin signaling activation is stabilization and nuclear translocation of β-catenin. The stability of β-catenin is regulated by the formation of a destruction complex consisting of Axin, adenomatous polyposis coli, glycogen synthase kinase-3 (GSK-3), and casein kinase 1 (CK1) in the cytoplasm (Fig. 3). 83 –85 In the absence of Wnt stimuli, β-catenin is subjected to priming phosphorylation by CK1 and subsequent phosphorylation by GSK-3 in the destruction complex (Fig. 3). The recruitment of β-TrCP, an E3 ubiquitin ligase, to the phosphorylated β-catenin results in proteasomal degradation via polyubiquitination. When extracellular Wnt ligands bind to the Frizzled receptor and LRP5/6 co-receptor complex, recruitment of the downstream signal mediators, such as DVL and Axin, is triggered and results in the dissociation of the destruction complex. Ultimately, the β-catenin is freed from the complex, accumulates in the cytoplasm, and is finally translocated into the nucleus. In the nucleus, β-catenin binds with the T cell factors/lymphoid enhancing factors (TCFs/LEFs) for target gene expression. More than 100 Wnt/β-catenin target genes have been identified. 79 Many of these genes (e.g., Axin2, Collagen I, Collagen III, EGFR, Endothelin-1, Fibronectin, Keratin-14, Lgr5, VEGF, and WISP1) have roles in cutaneous wound healing (Fig. 4 and Table 1).
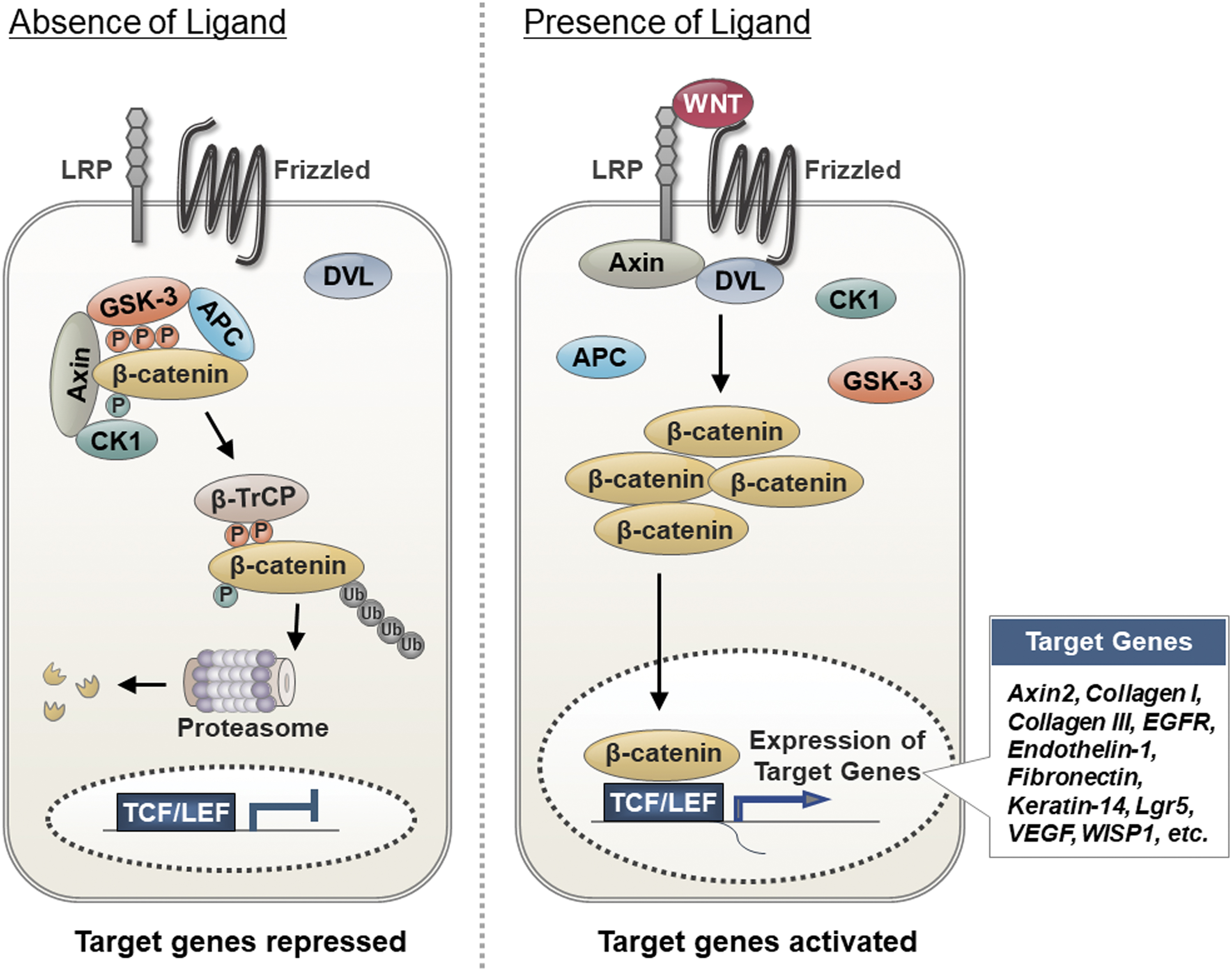
Wnt/β-catenin pathway and its target genes. In absence of the Wnt ligand, the destruction complex composed of Axin, APC, GSK-3, and CK1 is formed in the cytoplasm. By forming this complex, β-catenin is phosphorylated initially by CK1 and subsequently by GSK-3. The β-TrCP E3 linker is recruited to the phosphorylated β-catenin and β-catenin is then degraded by ubiquitin-mediated proteasomal degradation machinery. In the presence of Wnt ligand, it binds to the Frizzled/LRP5/6 receptor complex, leading to the dissociation of the destruction complex. Free β-catenin proteins accumulate in the cytosol and are then translocated into the nucleus for activation of TCFs/LEFs. The activation of TCFs/LEFs transcription factors induces a variety of Wnt/β-catenin signaling target genes, including those involved in skin wound healing (e.g., Axin2, Collagen I, Collagen III, EGFR, Endothelin-1, Fibronectin, Keratin-14, Lgr5, VEGF, WISP-1). APC, adenomatous polyposis coli; CK1, casein kinase 1; GSK-3, glycogen synthase kinase-3; LEFs, lymphoid enhancing factors; TCFs, T cell factors. Color images are available online.
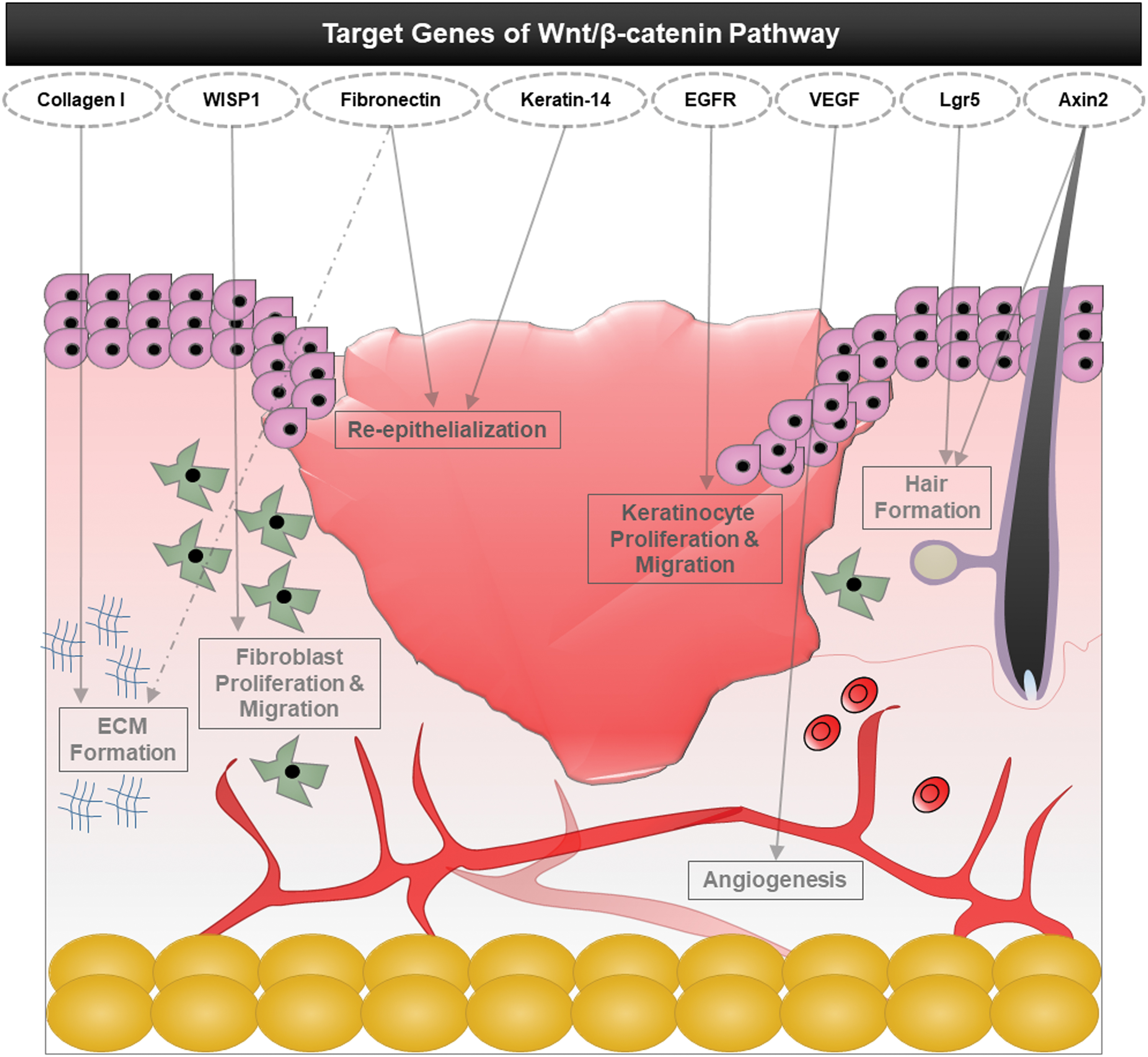
The effects of Wnt/β-catenin pathway target genes on cutaneous wound repair. Various Wnt/β-catenin signaling target genes contribute to the multiple events that occur during wound healing. For example, Collagen-I has a role in ECM formation, WISP1 activates fibroblast proliferation and migration, fibronectin controls re-epithelialization and ECM formation, Keratin-14 promotes re-epithelialization, EGFR regulates keratinocyte proliferation and migration, VEGF enhances angiogenesis, and Lgr5 and Axin2 promote hair formation via activation of hair follicle stem cells. VEGF, vascular endothelial growth factor. Color images are available online.
List of target genes involved in the Wnt/β-catenin pathway that are related to wound healing
ECM, extracellular matrix; VEGF, vascular endothelial growth factor.
The roles of Wnt/β-catenin signaling in the wound-healing process
Many target genes that are transcriptionally induced by activation of the Wnt/β-catenin pathway mediate various functions during the wound repair process (e.g., inducing structural construction of the dermis and epidermis and promoting angiogenesis) (Table 2). Moreover, the Wnt/β-catenin pathway plays key roles in regenerative wound healing (e.g., inducing formation of skin adnexa, such as hair) by promoting the activation of the stem cells. 24
Effects of Wnt/β-catenin pathway activation on wound repair and regeneration
The profile of Wnt/β-catenin signaling activity during the wound-healing process
The Wnt/β-catenin signaling pathway is activated in the dermis of the wound bed soon after a skin injury. This activation is quick and spatially restricted within the wound site. A study displayed that the change of oxygen tension occurring within minutes of skin damage can trigger activation of the Wnt/β-catenin pathway through hypoxia-inducible factor-1α. 86 During the proliferative phase of wound healing, Wnt/β-catenin signaling activity is highly increased in mesenchymal cells. For example, murine dermal fibroblast cultures exhibit an increment in β-catenin protein levels and TCF/LEF-mediated transcriptional activity during proliferation. 87 In human wound samples, the levels of β-catenin and the expression of its target genes (e.g., fibronectin and MMP7) are increased during the dermal proliferative phase. 88
The roles of Wnt/β-catenin pathway in wound repair
Upregulation of Wnt/β-catenin signaling promotes proliferation and migration of dermal fibroblasts, making them differentiate into myofibroblasts. This process helps to reduce the surface area of the developing scar. 87,89 The activated Wnt/β-catenin signaling not only facilitates migration and differentiation of keratinocytes in the epidermis, but it also promotes angiogenesis, follicle regeneration, and epithelial remodeling, which directly enhances cutaneous wound healing. 24,90
The roles of Wnt/β-catenin pathway in stem cell activation
Typically, stem cells are harbored in most adult tissues. However, they easily lose their self-renewal capability in response to stress or aging. The Wnt/β-catenin pathway plays roles in tissue-residing, stem cell activation, and migration to the wound bed in the basal epidermis for regeneration of damaged tissue. 91 In response to the Wnt/β-catenin signaling, TCF/LCF complex modulates the fates of lineages of multipotent stem cells in the skin. 92 For example, Wnt/β-catenin is involved in activation of epidermal stem cells (ESCs), the major source for replenishment of lost cells in the process of wound healing. 93 The elevated Wnt/β-catenin signaling activity not only enhances proliferation of quiescent ESCs, but it also promotes differentiation of ESCs into keratinocytes. 94 Further, elevation of β-catenin activity significantly promotes neogenesis of hair follicles, representing a fully functional inter-follicular epidermis in adult mice. 22,95,96 By contrast, inhibition of Wnt/β-catenin signaling during skin wounding hinders the formation of epithelial adnexa, including hair and sweat glands, resulting in scarring. These findings indicate that the Wnt/β-catenin pathway can trigger regeneration of wounded skin by serving as a niche signal for activation of skin stem cells. 97 The Wnt/β-catenin signaling target gene products, Axin2 and Lgr5, are well-known markers for the self-renewing stem cells in tissues, including the skin and the hair follicle. 77,98
The roles of Wnt/β-catenin pathway in hyaluronic acid synthesis
In wound healing, the main difference between repair and regeneration comes from ECM content. During the healing process, granulation tissue, which is formed at the wound site, is characterized by the proliferation of fibroblasts that produce ECM components, including fibronectin, collagen III, elastin, and hyaluronic acid (HA). 99 In the final stage of wound repair, the granulation tissue is converted to fibrotic scar tissue as collagen III is replaced by collagen I forming collagen fibers. The alignment of excessive collagen fibers in the dermis results in an inelastic collagen scar. In the process of regenerative healing, on the other hand, a much higher amount of HA is present in ECM compared with the repair process, and thus it reduces collagen deposition and subsequent fibrotic scar formation. 100 Wnt3a treatment upregulates the genes involved in HA synthesis in fibroblasts. 101 Further, HA interacts with CD44, a well-known Wnt/β-catenin signaling target, to exert its function in the wound-healing process, including enhancement of cell migration toward wound sites and promotion of angiogenesis as well as direct enhancement of tissue regeneration. 102 –104 Therefore, the Wnt/β-catenin pathway not only induces HA synthesis, but it also regulates biological function of HA for regenerative wound healing.
Crosstalk of Wnt/β-catenin pathway with other signaling pathways
The Wnt/β-catenin pathway cooperates with other signaling pathways during the wound-healing process. It interacts with the TGF-β/Smad pathway, which is a major signaling pathway involved in cutaneous wound healing and dermal fibrosis. TGF-β signaling is transiently activated after a skin injury; β-catenin level is then increased via the inhibition of GSK-3β activity or DKK-1 expression. 105 –107 The proliferation of fibroblasts and their differentiation into myofibroblasts via activation of TGF-β signaling occurs in a β-catenin-dependent manner. These results indicate that the Wnt/β-catenin pathway is a mediator of TGF-β/Smad signaling-induced wound healing. Synergistic activation through mutual interaction of the Wnt/β-catenin and Notch pathways improves wound healing and inhibits scar formation by promoting embryonic stem cell proliferation, keratinocyte differentiation and migration, and follicle regeneration. 94 To reconstitute skin adnexa and obtain complete healing, Wnt/β-catenin signaling also facilitates hair follicle regeneration in wounded skin through the formation of a positive feedback loop with FGF-9 signaling. 108
Therapeutic potential of Wnt activators for regenerative wound healing
Considering the roles of the Wnt/β-catenin pathway during wound healing, it has recently been used as a target for the development of wound-healing agents. Studies have examined small molecules and natural products that activate the Wnt/β-catenin pathway as a potential therapeutic for diverse diseases. 109,110 Some of these are under development for the treatment of skin wounds (Fig. 5 and Table 3).
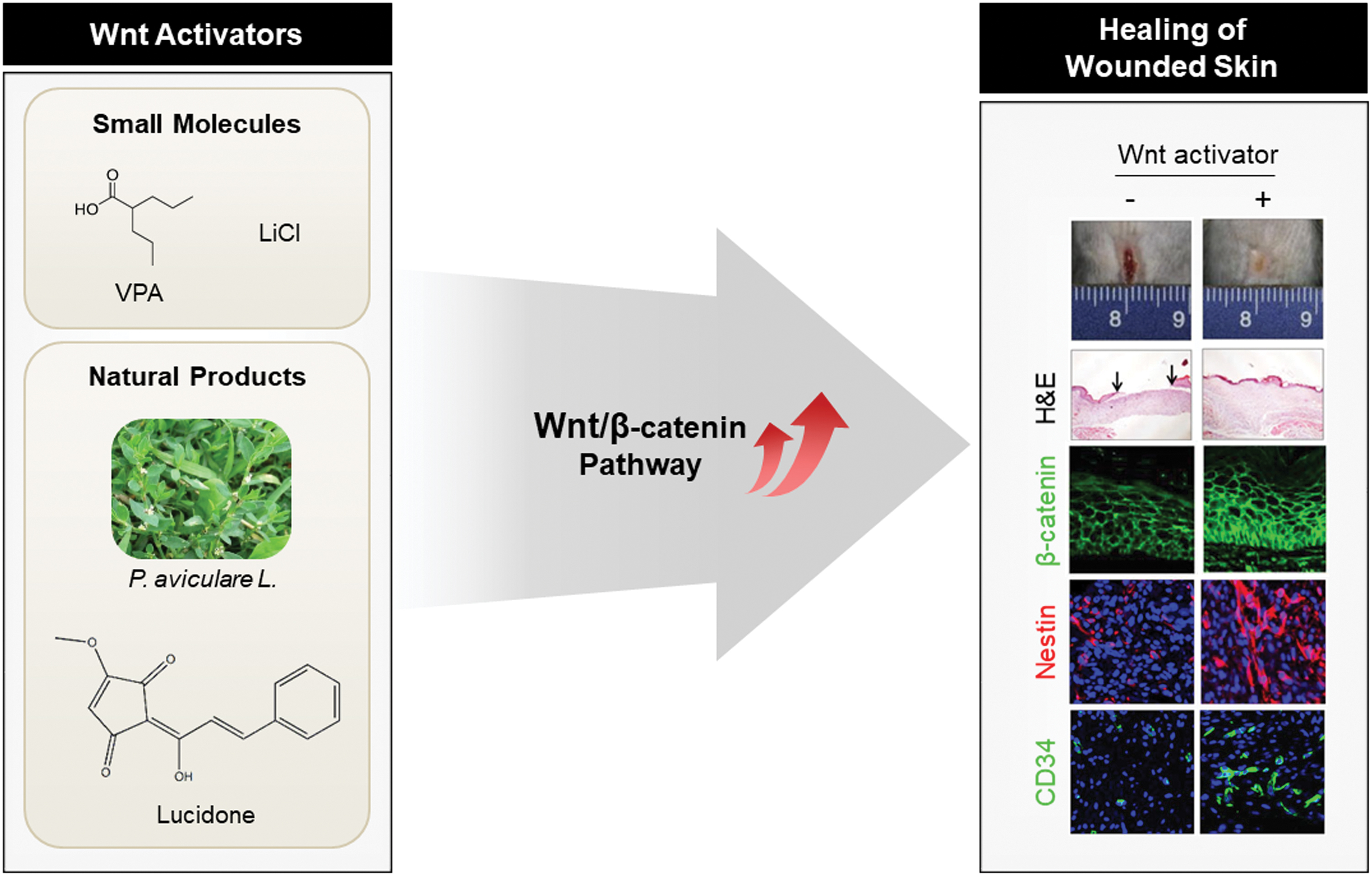
Effects of Wnt activators on healing of wounded skin. The agents that activate the Wnt/β-catenin pathway include small molecules, such as LiCl and VPA, and natural products, such as lucidone and P. aviculare (Polygonum aviculare L.) extract. 112,115 Upregulation of the Wnt/β-catenin pathway via topical application of these agents promotes wound healing with increased expression of stem cell markers such as nestin and CD34. Reproduced with permission from Lee et al. and Seo et al. 112,115 LiCl, lithium chloride; VPA, valproic acid. Color images are available online.
Wnt/β-catenin pathway activators enhancing wound healing
CXXC5, CXXC-type zinc finger protein 5; LiCl, Lithium chloride; VPA, valproic acid; PTD, protein transduction domain; DBM, DVL binding motif; DVL, dishevelled.
Lithium chloride
Lithium chloride (LiCl) is well-known GSK-3 inhibitor and it has a capability for activation of the Wnt/β-catenin pathway. 111 Topical application of LiCl to the wounded skin of rats induced enhancement of the wound closure rate with an elevated β-catenin level. 94 Moreover, thickness of the neoformative epidermis layer and formation of hair follicle structures and sebaceous gland were increased in skin tissues of rats by topically applied LiCl.
Valproic acid
The small-molecule valproic acid (VPA) is known to activate the Wnt/β-catenin pathway by inhibiting GSK-3β. 112 Further, VPA enhances wound healing through promotion of neo-epidermis formation, fibroblast–myofibroblast transition, and cellular proliferation. One study found that when mice were treated with VPA, full-thickness wound sizes were markedly reduced, and healing rates increased. The VPA also induces the expression of stem cell markers (e.g., CD34) involved in neo-vascularization.
Lucidone
Lucidone, a naturally occurring cyclopentenedione isolated from the dried fruits of Lindera erythroccarpa, was reported to increase β-catenin level through the GSK-3β-dependent pathway and to enhance wound healing in both in vitro and in vivo models. 113 Lucidone not only promoted proliferation and migration in both keratinocyte and fibroblast cells, but it also triggered expression of angiogenesis markers in endothelial cells. The healing rate of punched wounds on mice was accelerated by the topical application of lucidone.
Polygonum aviculare L.
Polygonum aviculare L. extract was screened out as a natural product that activates the Wnt/β-catenin pathway. 114 At the cell level, P. aviculare L extract promoted migration of both keratinocytes and fibroblasts. 115 Treatment with P. aviculare L. extract accelerated the healing rates of full-thickness dorsal wounds in mice, compared with a control group. Active ingredients of P. aviculare L. extract (e.g., quercitrin hydrate, caffeic acid, and rutin) have been characterized as components that activate the Wnt/β-catenin pathway and enhance keratinocyte migration.
A novel target, CXXC5 for regenerative wound healing
CXXC5, a negative feedback regulator of the Wnt/β-catenin pathway, suppresses the wound-healing process
Activation of the Wnt/β-catenin pathway is an ideal strategy for regenerative wound healing. However, the development of therapeutics that activate this pathway is limited due to the presence of its own negative regulation systems. Inhibitory factors of the Wnt/β-catenin pathway disrupt skin wound healing. For example, Dickkopf-1 (DKK-1), a secreted Wnt antagonist functioning via interaction with LRP5/6 receptor, hinders proliferation of dermal fibroblasts as revealed by both in vitro and in vivo systems. 116 Consistent with these results, intradermal injection of small interfering RNA (siRNA) for DKK-1 enhances dermal fibroblast functions. Another secreted Wnt antagonist, Frizzled-related protein-1 (sFRP-1), suppresses cell proliferation and ECM production in keloid fibroblasts by inhibiting Wnt/β-catenin signaling through interacting with either Wnt or Frizzled. 117 A mouse model revealed that injection of a neutralizing antibody against sFRP-1 into the palatal wound edge promotes the healing of wounded skin. 118
The function of a negative feedback regulator of the Wnt/β-catenin pathway, CXXC5, is determined by its subcellular location that depends on the tissue type and the cell's physiological status. Cytosolic CXXC5 plays a role as a Wnt/β-catenin signaling inhibitor, whereas nuclear CXXC5 functions as a transcription factor. 119 –123
By binding DVL, cytosolic CXXC5 has a variety of pathophysiological roles by inhibiting the Wnt/β-catenin signaling pathway (Fig. 6). 13,80,124,125 CXXC5 can be transcriptionally induced by the Wnt/β-catenin signaling itself or under a variety of pathophysiological states (e.g., alopecia, osteoporosis, wound formation, and termination of height growth at puberty), and these pathological aberrancies can be restored in CXXC5 knockout mice of the disease model systems. 13,80,124,125
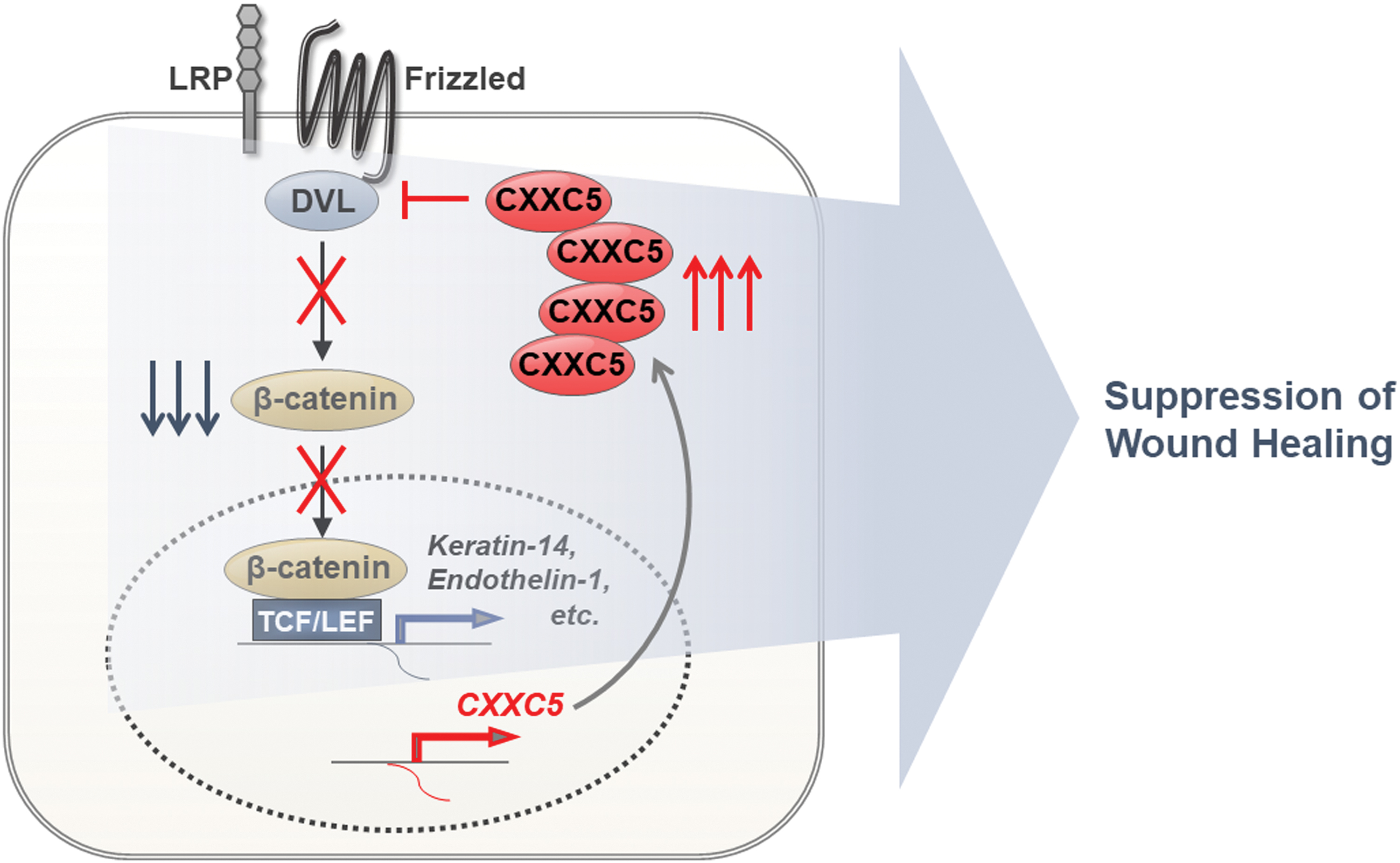
CXXC5 is a negative feedback regulator of the Wnt/β-catenin pathway and it suppresses the expression of target genes involved in wound healing. CXXC5 transcription is induced by strong activation of the Wnt/β-catenin during the wound-healing process. CXXC5 binds to DVL in the cytosol and subsequently suppresses Wnt/β-catenin signaling by blocking the dissociation of the destruction complex by DVL. Inhibition of Wnt/β-catenin signaling in the skin results in repression of wound healing-related genes after inhibition of the wound-healing process. CXXC5, CXXC-type zinc finger protein 5; DVL, dishevelled. Color images are available online.
Taken together, these findings indicate that CXXC5 can be a therapeutic target for diseases caused by suppression of Wnt/β-catenin signaling. The importance of targeting cytosolic CXXC5 function in wound healing was supported by enhanced cutaneous wound healing in mice treated with the protein transduction domain-fused DVL-binding motif (PTD-DBM) peptide, which blocks CXXC5–DVL protein–protein interactions (PPI). 13
A role of CXXC5 during wound healing
In patients with melanoma, β-catenin level was gradually increased, especially during the late inflammatory and early proliferative stages, and it was then decreased during the remodeling stage (Fig. 7). 13 In contrast, CXXC5 level was declined during the early proliferative stage after surgery but it then rose again, which shows the opposite patterns of those of β-catenin in the same wounded area. The inhibitory role of CXXC5 in wound healing was revealed by the increment of the wound closure rate in Cxxc5 −/− mice. 13 Myofibroblast differentiation and collagen production is inhibited by CXXC5 overexpression in human dermal fibroblasts. Both in vitro and in vivo studies found that the inhibitory roles of CXXC5 during the wound-healing process are exerted by the suppression of Wnt/β-catenin signaling via its interaction with DVL in the cytosol. 13 Taken together, these findings indicate that the inhibition of CXXC5 function, especially its cytosolic role related to Wnt/β-catenin signaling inhibition, may be a new strategy for the development of wound-healing agents.
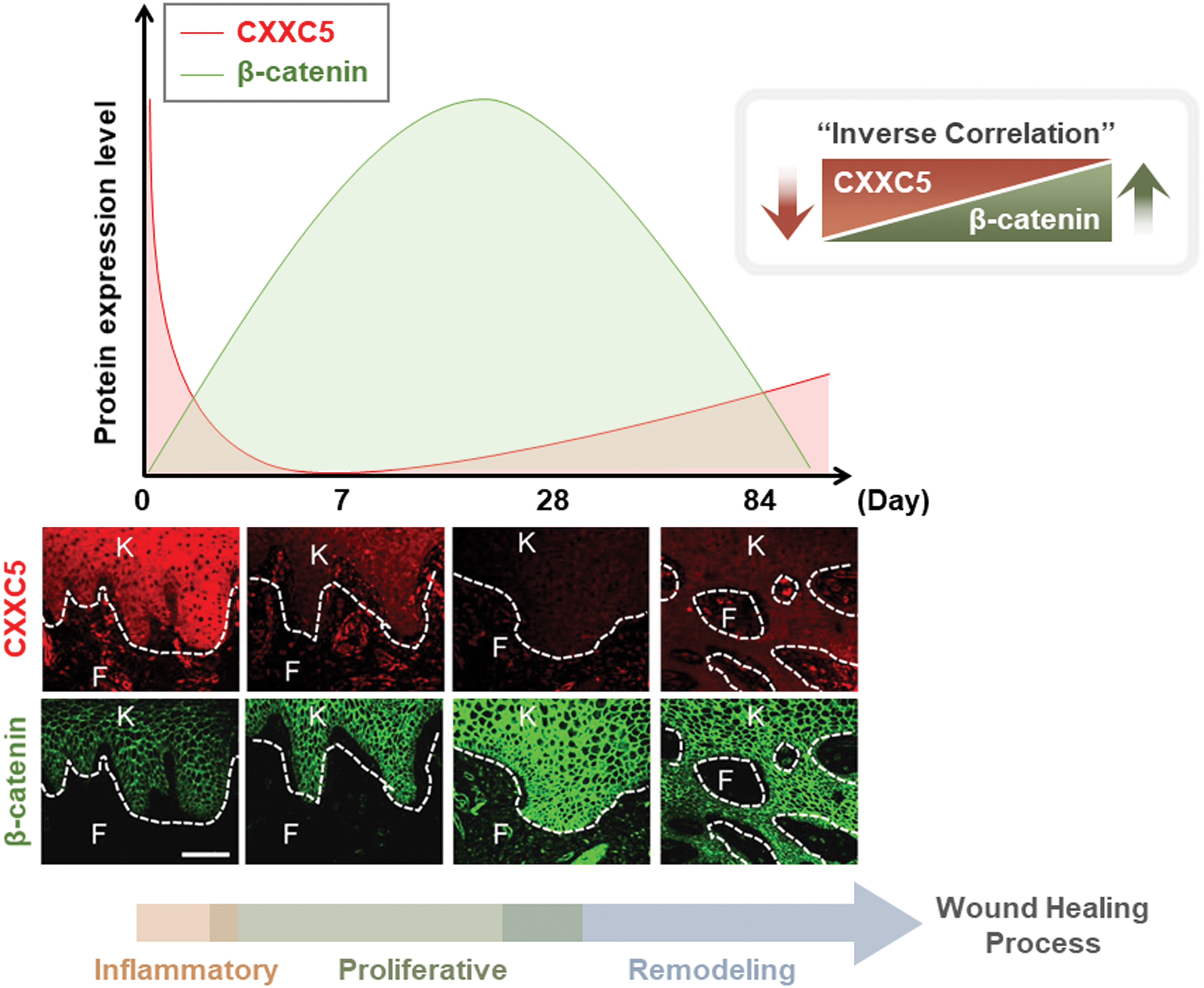
Kinetics of the expression levels of β-catenin and CXXC5 during the wound-healing process. Profiles for CXXC5 and β-catenin expression during the wound-healing process, adapted from a previous study. 13 The tissue samples were from patients with melanoma who underwent surgery. The immunohistochemical images represent expression levels of CXXC5 and β-catenin in wounded skin at 0, 7, 28, and 84 days after surgery. Day 0, intact skin. White dashed lines, the epidermal–dermal junction. F, fibroblasts; K, keratinocytes. Scale bars, 100 μm. Reproduced with permission from Lee et al. 13 Color images are available online.
Effects of PTD-DBM, an interfering peptide against CXXC5–DVL interaction, on wound healing
A PTD-DBM peptide, which contains the sequence of CXXC5 binding to DVL and activates Wnt/β-catenin signaling via interference of the CXXC5–DVL interaction, was developed for practical application (Fig. 8A). 80 The PTD-DBM peptide effectively promotes cell migration in vitro via activation of Wnt/β-catenin signaling. 13 Topical application of the PTD-DBM peptide significantly accelerates the wound closure rate. Increment in wound healing-related markers and critical deposition of collagen occur during the healing of full-thickness wounds on the backs of mice (Fig. 8B). In addition to enhancement of wound healing, induction of alkaline phosphatase (ALP) in PTD-DBM peptide-treated mouse wounds indicates that PTD-DBM peptide promotes formation of neogenic hair follicles (Fig. 8B), proved by the presence of white hair in the healed tissues. 124 Recent clinical studies by the Clinical Peptide Society found that PTD-DBM peptide enhances human hair growth. These observations indicate that activation of the Wnt/β-catenin signaling by the blockade of the CXXC5–DVL interaction with PTD-DBM peptide could be a therapeutic strategy for regenerative wound healing.
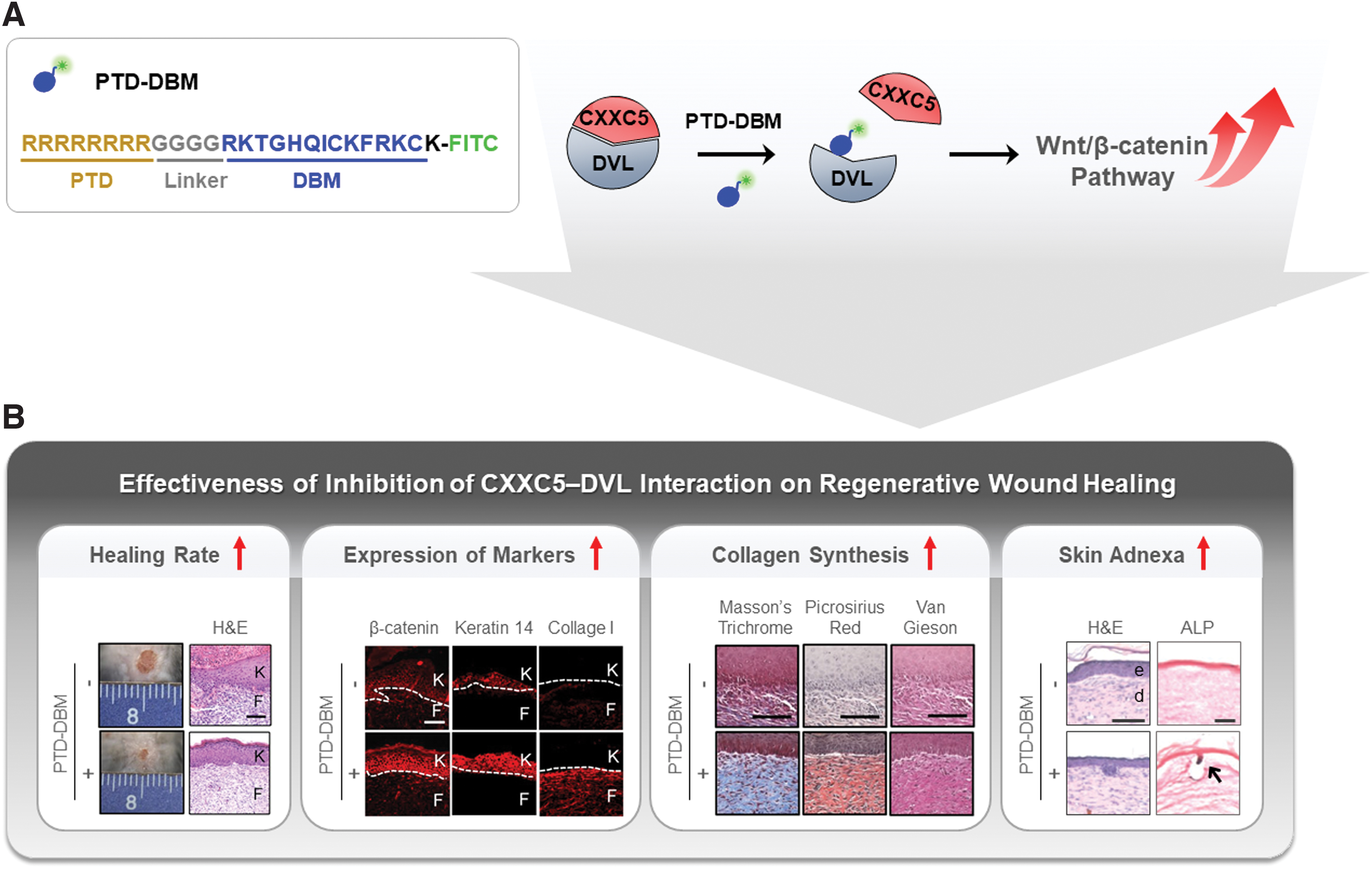
PTD-DBM, a peptide that interferes with the CXXC5-DVL interaction, and its effectiveness on regenerative wound healing.
Perspectives
The Wnt/β-catenin pathway is an attractive target for regenerative wound healing. Although agents activating Wnt/β-catenin signaling stimulate the wound healing process, an appropriate dosing and treatment duration is important because aberrant activation of the Wnt/β-catenin pathway causes fibrotic diseases, including hypertrophic scarring, keloid formation, and skin cancer. However, activating Wnt/β-catenin signaling via releasing the CXXC5-mediated negative feedback loop instead of via direct activation enhances wound healing without leading to unwanted outcomes such as melanoma. The safety of this therapeutic approach was confirmed by the absence of any pathological skin phenotypes, including melanoma-accompanying transformations, in 1-year-old Cxxc5 −/− mice or in mice that received topical application of PTD-DBM peptide for >6 months. Further, PTD-DBM peptide treatment does not induce transcription of cyclin D1 and c-Myc, which are the Wnt/β-catenin signaling target genes frequently overexpressed during cancer development, but it does induce transcription of endothelin-1, which contributes to enhanced wound healing. 13 Finally, the specific blockade of the cytosolic function suppressing Wnt/β-catenin signaling, not the nuclear function acting as a transcription factor, 119,120 further provides conceptual safety of an approach interfering with the CXXC5–DVL interaction for target specificity. Therefore, the CXXC5–DVL interaction is potentially a safe target for regenerative wound healing. A strategy to discover small molecules mimicking the PTD-DBM peptide could be a valuable approach for the development of first-in-class wound healing agents that would be cost-effective and suitable for routine use.
Summary
An acute skin wound is spontaneously repaired within 1–2 weeks. However, due to scar formation, the repaired skin is not identical to intact uninjured skin. As the elderly population with delayed wound healing increases, the need for effective wound-healing agents based on regenerative healing also increases. Current wound care research has focused on regenerative therapies to diminish scar formation, improve the quality of restored skin, and accelerate healing rates.
The Wnt/β-catenin pathway could be viewed as a major target for development of drugs in the field of regenerative wound healing because it can promote the overall wound-healing process by activation of stem cells through interaction with other signaling pathways, including the TGF-β/Smad pathway.
Activators of Wnt/β-catenin signaling, such as small molecules (e.g., LiCl and VPA) and natural products (e.g., lucidone and P. aviculare L. extract), have been characterized as agents enhancing wound healing. However, their effectiveness during the healing process could be restricted due to induction of CXXC5, a negative feedback regulator of the Wnt/β-catenin pathway, during the early stages of wound healing. Therefore, inhibition of CXXC5 function, especially the cytosolic form that suppresses Wnt/β-catenin signaling via interaction with DVL (CXXC5–DVL interaction, PPI), is a target for the development of novel regenerative wound-healing agents. Topically applied PTD-DBM peptide, which interferes with the CXXC5–DVL interaction, effectively enhances the wound-healing process and has the potential to act as a therapeutic agent. The maximal effects of PTD-DBM peptide occur in combination with direct Wnt/β-catenin signaling activators, such as VPA. This combination treatment promotes regenerative wound healing via strong activation of the Wnt/β-catenin pathway; initial activation occurs through VPA-induced GSK-3β inactivation. Subsequent further activation is enhanced via blockade of the CXXC5–DVL interaction by the PTD-DBM peptide. This approach for wound healing activating the Wnt/β-catenin signaling could minimize undesirable side effects (e.g., skin cancer) that can be induced via aberrant activation of the Wnt/β-catenin pathway.
Take-Home Messages
Use of regenerative therapies, including stem cells and growth factors, is a current approach for complete healing of skin wounds.
Multiple signaling pathways are involved in the skin wound healing, and the Wnt/β-catenin pathway is a key player in the wound-healing process.
The Wnt/β-catenin pathway participates in the activation of skin stem cells as well as the overall process of wound healing to enhance regenerative wound healing.
CXXC5, a negative feedback regulator of the Wnt/β-catenin pathway, suppresses wound healing by exerting its function via suppression of this pathway.
Inhibition of CXXC5 function via its binding to DVL enhances the speed and quality of healing in mouse skin wounds, without any adverse effect.
Restoration of the Wnt/β-catenin signaling via blockade of the CXXC5-mediated negative feedback mechanism, not by direct activation, is a potential future strategy for regenerative wound healing.
Footnotes
Authors' Contributions
K.Y.C. conceptualized and supervised the project. S.C. and M.Y. wrote the article and drew the figures. S.C. and K.Y.C. revised the article.
Acknowledgments and Funding Sources
This work was supported by the National Research Foundation (NRF) of Korea grant funded by the Korean Government (MSIP) (2019R1A2C3002751, 2020M3E5E2040018). M.Y. was supported by a BK21 PLUS program.
Author Disclosure and Ghostwriting
No competing financial interests exist. The content of this article was entirely written by the authors listed. No ghostwriters were used to write this article.
About the Authors
