Abstract
Objective:
High bacterial load contributes to chronicity of wounds and is diagnosed based on assessment of clinical signs and symptoms (CSS) of infection, but these characteristics are poor predictors of bacterial burden. Point-of-care fluorescence imaging (FL) MolecuLight i:X can improve identification of wounds with high bacterial burden (>104 colony-forming unit [CFU]/g). FL detects bacteria, whether planktonic or in biofilm, but does not distinguish between the two. In this study, diagnostic accuracy of FL was compared to CSS during routine wound assessment. Postassessment, clinicians were surveyed to assess impact of FL on treatment plan.
Approach:
A prospective multicenter controlled study was conducted by 20 study clinicians from 14 outpatient advanced wound care centers across the United States. Wounds underwent assessment for CSS followed by FL. Biopsies were collected to confirm total bacterial load. Three hundred fifty patients completed the study (138 diabetic foot ulcers, 106 venous leg ulcers, 60 surgical sites, 22 pressure ulcers, and 24 others).
Results:
Around 287/350 wounds (82%) had bacterial loads >104 CFU/g, and CSS missed detection of 85% of these wounds. FL significantly increased detection of bacteria (>104 CFU/g) by fourfold, and this was consistent across wound types (p < 0.001). Specificity of CSS+FL remained comparably high to CSS (p = 1.0). FL information modified treatment plans (69% of wounds), influenced wound bed preparation (85%), and improved overall patient care (90%) as reported by study clinicians.
Innovation:
This novel noncontact, handheld FL device provides immediate, objective information on presence, location, and load of bacteria at point of care.
Conclusion:
Use of FL facilitates adherence to clinical guidelines recommending prompt detection and removal of bacterial burden to reduce wound infection and facilitate healing.
Thomas E. Serena, MD

Introduction
An estimated 1
Chronic wounds fail to progress through a timely sequence of repair. It is known that increased microbial load is a key predictor of nonhealing wounds. 7,8 Proliferation of bacteria resulting in moderate-to-heavy loads (>104 colony-forming units [CFU]/g) delays healing 9 –11 and increases the risk of wound complications, including infection, sepsis, and amputation. 12 –14 Guidelines advise that early diagnosis of high bacterial burden is essential to prevent the wound from progression to local or systemic infection. 15 To reduce bacterial burden, clinicians choose from an armamentarium of antiseptic wound cleansers, debridement techniques, and antimicrobial options. This is done without objective information on bacteria at point-of-care and without information on treatment efficacy.
Clinical Problem Addressed
Treatment selection at point-of-care is largely based on evaluation of clinical signs and symptoms (CSS) of infection or high bacterial loads. However, numerous studies have reported that patients with high bacterial burden are frequently asymptomatic. 11,16,17 Furthermore, comorbidities in wound patients (e.g., diabetes and autoimmune disease) can blunt immune responses and exacerbate patient-to-patient variability of CSS. 18 Together, this results in poor sensitivity of CSS for detection of infection, 16,17,19 hindering immediate identification of wounds with high bacterial burden. Quantitative tissue cultures of wound biopsies are the reference standard to quantify bacterial load, but prolonged turnaround time between biopsy and microbiological results limits the rapid decision making needed to effectively manage bacterial burden in wounds. The relative inconsistency of CSS and delays in results from microbiological culture and PCR analysis may explain why 12-week wound healing rates are below 60% 7 and have remained stagnant over the past 40 years, 20 despite tremendous advances in wound treatments.
To address the pervasive problem of bacteria-related delayed healing and facilitate a more proactive approach to treatment planning, objective diagnostic information on bacterial burden in wounds is needed. Point-of-care diagnosis of bacterial burden in wounds is achieved using a handheld fluorescence imaging (FL) device (MolecuLight i:X; MolecuLight, Inc., Toronto, Canada) that detects endogenous fluorescence from bacteria (at loads >104 CFU/g). 21 Macroscopic imaging of bacteria is not possible as bacteria themselves are microscopic. However, when bacteria accumulate at high loads (>104 CFU/g), the fluorophores they collectively emit are detectable through FL. Under safe violet light illumination, common wound pathogens, including bacteria from the Staphylococcus, Proteus, Klebsiella, and Pseudomonas generas, 22,23 endogenously emit red or cyan fluorescent signatures. 23 –26 By detecting these fluorescent signals, FL provides immediate information on bacterial location, without use of contrast agents (Fig. 1). Multiple clinical studies have consistently reported positive predictive values (PPV) of these fluorescent signals averaging 95.6% (range 87.5–100%) to detect moderate-to-heavy loads of bacteria, confirmed by microbiological analysis. 21,27 –29 Recent evidence indicates that the FL procedure facilitates more appropriate treatment selection and timing of advanced therapies (e.g., grafts and skin substitutes) 30 in chronic wounds and burns 27,28,31 –35 ; however these studies lacked rigor and statistical power. The Fluorescence imaging Assessment and Guidance (FLAAG) study, a large, multicenter prospective controlled clinical trial targeting wounds of various type and duration, was established to evaluate the following: (1) whether FL improves detection of wounds with high (>104 CFU/g) bacterial loads and (2) how point-of-care information on bacterial presence and location impacts treatment planning.
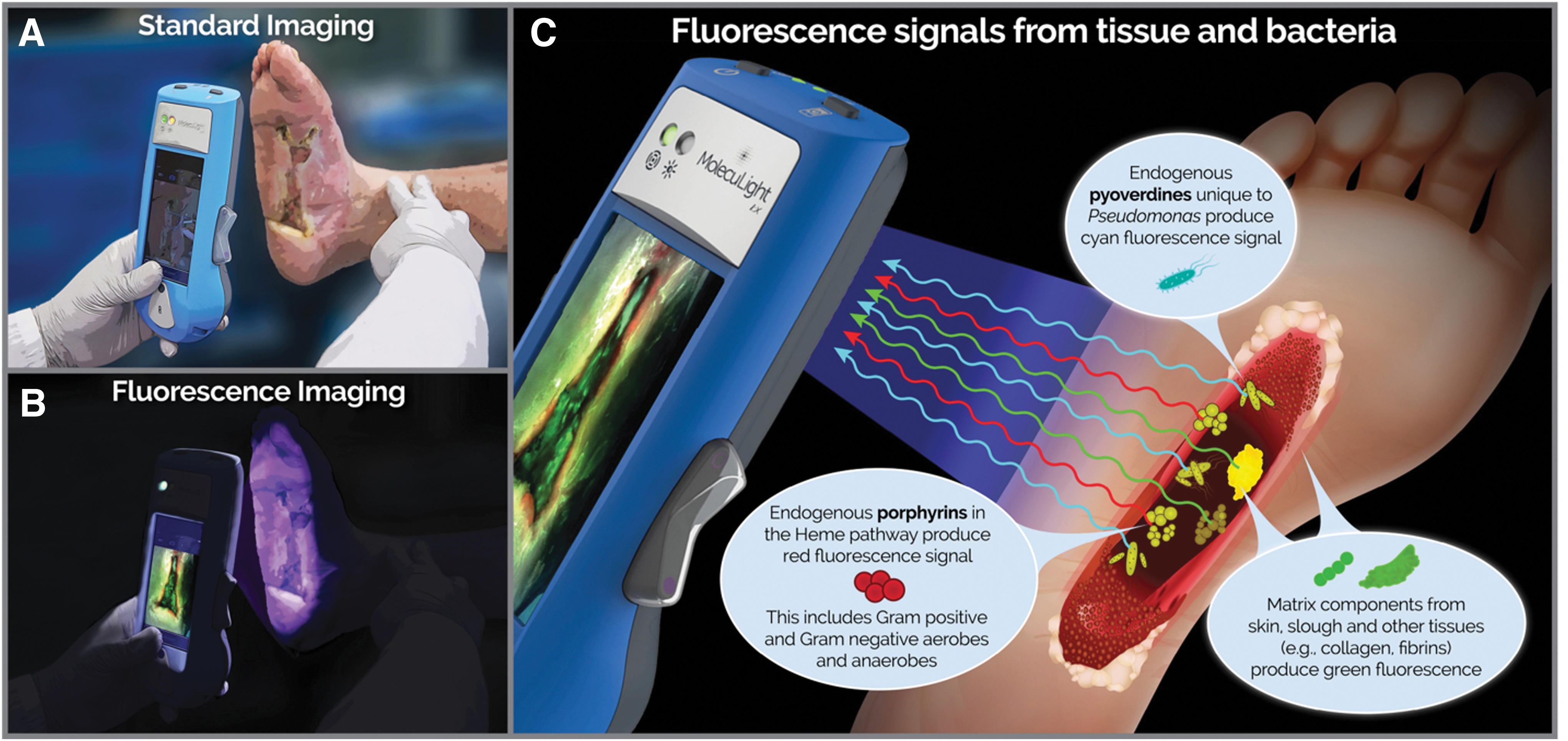
Materials and Methods
Study population and design
This prospective, single-blind, multicenter cross-sectional study (
Assessment of CSS of infection and FL
Clinicians reviewed patient history and visually inspected wounds for CSS using the International Wound Infection Institute (IWII) Wound Infection checklist. 15 Assessment of infection was based on clinician judgment; wounds with ≥3 criteria present were considered positive for moderate-to-heavy (>104 CFU/g) bacterial loads, per guidelines, 15 but if one overwhelming sign or symptom was present, clinicians had the discretion to deem the wound positive for CSS. A 4-week treatment plan was created based on assessment of CSS. Immediately following CSS assessment, standard and fluorescence images were captured with the FL device. To ensure uniform FL, the device is held at a 90° angle to the wound. The device's LEDs emit safe 405 nm violet light to excite fluorophores in the wound up to a penetration depth of 1.5 mm. 36 This excitation wavelength causes most bacterial species in wounds to emit a red fluorescent signal due to endogenous porphyrins in the heme pathway. 23,25 While Pseudomonas aeruginosa also produces porphyrins, 37 it uniquely produces a predominant cyan fluorescent signal due to endogenous pyoverdine, a virulence factor. 26 These fluorescent signals from bacteria that accumulate in a region of the wound at loads >104 CFU/g are detectable by the device. 21,29 Specialized optical filters on the device allow transmission of only relevant fluorescence from tissue and bacteria. 36 Connective tissues (e.g., collagen) produce green fluorescent signals 23,25,26,38 and flaky skin appears a brighter green with white edges. Images where red or cyan fluorescence was observed by clinicians were considered positive for moderate-to-heavy bacterial loads (>104 CFU/g) 21 (Fig. 2). A new treatment plan was documented incorporating information about bacterial fluorescence. Clinicians then completed a survey indicating how FL influenced diagnosis of bacterial burden in the wound, guided procedure, and treatment selection (i.e., frequency of treatment, including cleaning, debridement, and use of topical antimicrobials and antibiotics), or influenced patient care.
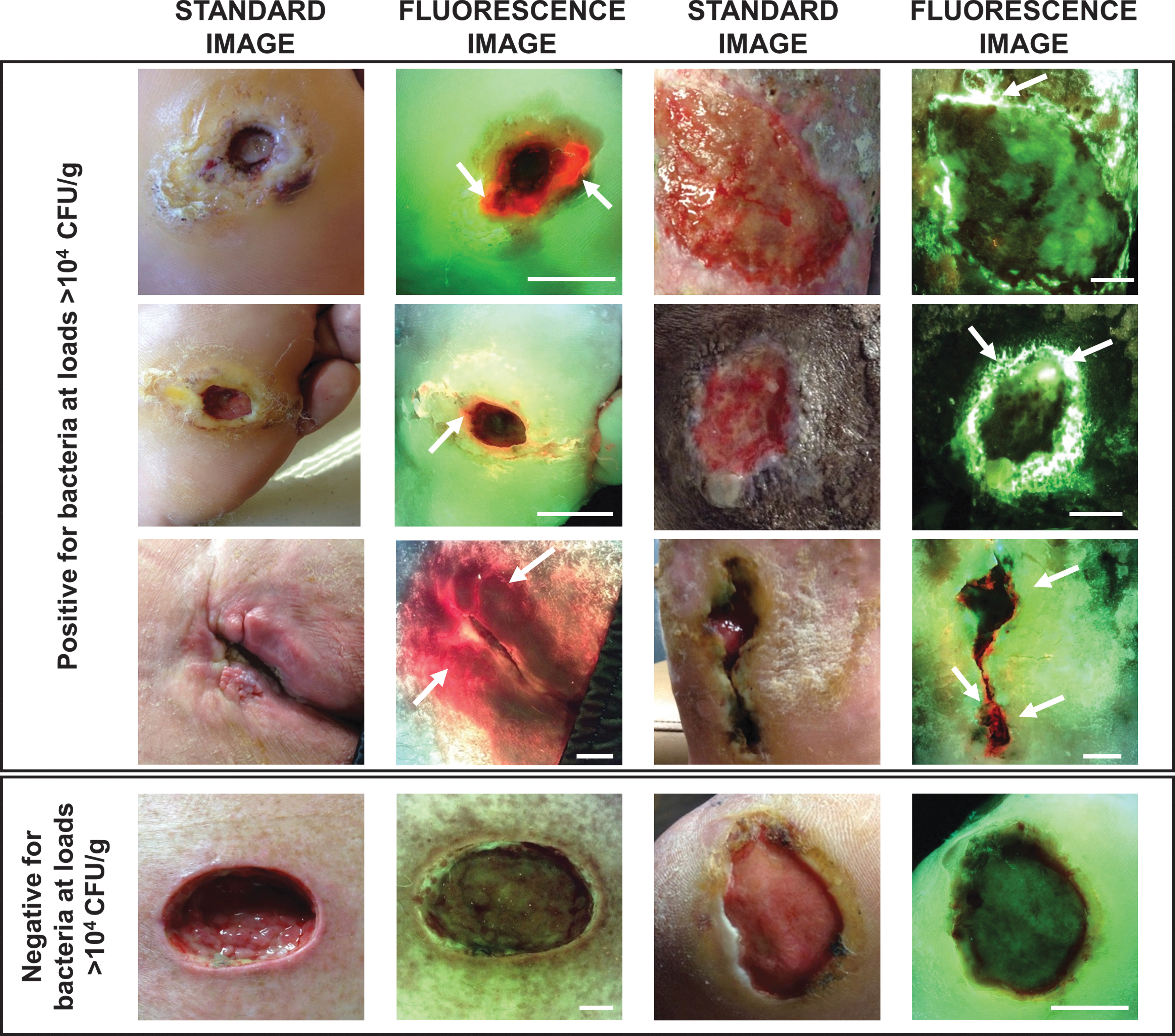
Representative fluorescence images of wounds that were positive or negative for moderate-to-heavy loads of bacteria (>104 CFU/g) in and around the wound bed. White arrows indicate regions of red or cyan fluorescence from bacteria; scale bars represent 1 cm. CFU, colony-forming unit.
Microbiological analysis of total bacterial load
Punch biopsies from wounds were collected to quantify total bacterial load. Up to three biopsies (6 mm diameter) were obtained under local anesthetic: a biopsy from the wound center, or if applicable, a biopsy outside of the wound center from a region of the wound positive for bacterial fluorescence, or region positive for CSS. In wounds where bacterial fluorescence was observed, clinicians were directed to collect a biopsy from the region of the wound that was brightest for bacterial fluorescence. Biopsy samples were cut to a depth of 2 mm (to restrict bacterial contents to the penetration depth of imaging device) and transported in Remel ACT-II transport media to a central laboratory (Eurofins Central Laboratory, Lancaster, PA) for microbiological culture analysis of load and species. Fluorescence can only be detected from bacteria that are alive, thus necessitating the use of quantitative culture analysis to confirm the total bacterial loads detected by FL. This method may not fully capture the microbiological diversity in the wound, including some fastidious bacterial species; therefore, every effort was made to provide optimal conditions for bacteria that are challenging to culture. To prepare for analysis, a small portion of the tissue was prepared for Gram staining on a sterile slide. The remaining biopsy sample was homogenized and serially diluted 39 for quantitative microbiological analysis (range of detection from 0 to 109 CFU/g). Diluted biopsy homogenates were cultured on BAP/Chocolate agar (nonselective growth), Columbia CAN agar (select gram positive), MacConkey agar (selective gram negative), or Brucella agar (anaerobes) and incubated at 35°C in the appropriate atmosphere. Aerobe cultures were assessed for growth after 24 h of incubation and incubated up to 48 h; anaerobes were assessed after 48 h of incubation, and then reviewed every 24 h up to 7 days. A wound was considered microbiologically positive if the total bacterial load (the sum of all bacteria from any biopsy) was >104 CFU/g. Matrix assisted laser desorption ionization-time of flight mass spectrometry (Bruker Daltonics) was used to identify bacterial species, as previously described. 40 Microbiologists were blinded to the results of the CSS assessment and FL.
Statistical analysis
One-sided exact McNemar tests were used for comparisons of sensitivity, specificity, and accuracy of detecting bacterial loads >104 CFU/g. Comparisons of predictive values (PPV and negative predictive value [NPV]) were performed using an asymptotic method as described by Moskowitz and Pepe. 41 Sample proportions and 95% confidence intervals (CIs) were used to estimate the diagnostic accuracy characteristics. Fisher's exact test was performed to assess association between fluorescence diagnosis (FL+ or FL−) and reported survey outcomes; statistical significance was set at p = 0.05. All analyses were performed using R version 3.6.2.
Results
Between May 2018 and April 2019, 371 patients with various wound types (DFUs, VLUs, PUs, SS, and others) were screened. Of the 371 patients screened, only 4 (1.1%) were excluded from the study and microbiology data were completed for 350. Basic demographic information along with antibiotic use, wound type, wound duration, and total bacterial load are reported in Table 1. Mean (standard deviation [SD]) age of participants was 60.2 (12.4) and 35.7% were female. Wound duration exceeded 3 months in 69.7% of wounds and delayed healing was observed in 52.9%. No serious adverse event resulting from use of the device was reported. 42
Baseline characteristics of study participants
Wounds that were “microbiology positive” had bacterial loads >104 CFU/g. Fischer's exact test was used to compare microbiology-positive and microbiology-negative subsets of each characteristic described. Statistical significance was set at p = 0.05; bold values indicate significance.
CFU, colony-forming unit; DFU, diabetic foot ulcer; PU, pressure ulcer; SD, standard deviation; VLU, venous leg ulcer.
In 82% (287/350) of wounds, bacterial loads >104 CFU/g were observed, confirmed by microbiological analysis (Fig. 3). Median (range) total bacterial load of all wounds was 1.8 × 106 CFU/g (0.0–7.7 × 109 CFU/g). A higher proportion of males (69.7%) than females (30.3%) had microbiology-positive wounds (>104 CFU/g). Of the microbiology positive wounds, 19.5% were on systemic antibiotics, and bacterial load of these wounds averaged (SD) 1.4 × 107 CFU/g (3.1 × 107 CFU/g); over 50% of microbiology-negative wounds (<104 CFU/g) were on systemic antibiotics. Bacterial loads >104 CFU/g were most prevalent in DFUs and wounds of ≥12 month duration. Of the 350 wounds in the study, 183 (52.3%) had bacterial loads >106 CFU/g, which some consider to be indicative of infection 17 ; in 16.9% (59/350) of wounds, bacterial loads >108 CFU/g were observed, while 18% (63/350) of wounds had bacterial loads <104 CFU/g. One hundred and six different bacterial species (51 genera) were detected from 1,053 isolates; species detected included the following: 68 gram positive, 38 gram negative, 78 aerobes, and 28 anaerobes. In 85.7% (246/287) of microbiology-positive wounds (loads >104 CFU/g), mixed bacterial colonization was present. Staphylococcus aureus was the most prevalent species observed, present in 71.1% of microbiology-positive wounds. P. aeruginosa was prevalent in 13.9% (40/287) of microbiology-positive wounds and was associated with presence of cyan fluorescence, as expected. Supplementary Table S1 lists bacterial species detected from all study wounds. An average of 2.8 bacterial species was detected per biopsy collected from the center of the wound. In most wounds, the center of the wound was also the brightest region of fluorescence. However, in 78 wounds, an additional FL-guided biopsy was collected outside the wound center. From these FL-guided biopsies taken outside of the wound center, an average of 3.1 bacterial species was detected. This was significantly higher than the average number of bacterial species detected in biopsies collected from the center of the same wound (2.2; p < 0.001). The inclusion of 98.9% (367/371) of the population screened suggests that these findings are representative of bacterial loads in typical wound populations.
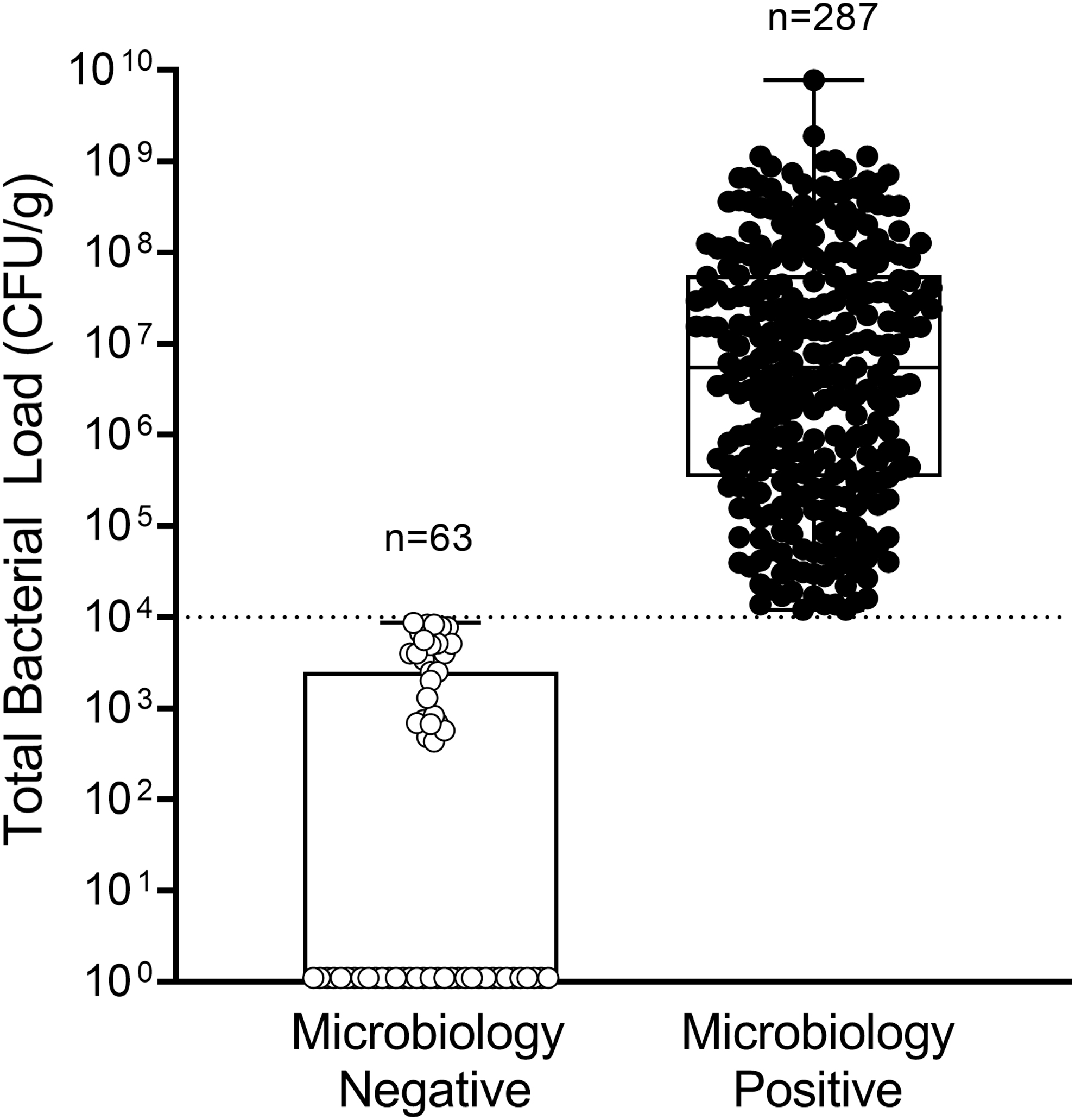
Box plot shows the distribution of total bacterial load (CFU/g) of each wound biopsied (n = 350 wounds total) based on whether wounds were microbiologically negative (bacterial load <104 CFU/g; n = 63) or positive (>104 CFU/g; n = 287). Boxes contain the 25th to 75th percentiles of data set, while center line indicates median bacterial load of all wounds (106 CFU/g). Black whiskers represent minimum and maximum values. Dashed line indicates lowest threshold (104 CFU/g) at which bacteria can be detected using FL. Of the microbiology-negative wound biopsies, 36 had total bacterial load of 0.
Diagnostic accuracy of FL was assessed on its own and in combination with information provided by CSS assessment (CSS+FL). Clinicians diagnosed 302/350 wounds as negative for CSS. Addition of FL to CSS improved sensitivity (61.0% [95% CI, 55.3–66.6%]) to detect wounds with bacterial loads >104 CFU/g by fourfold compared to CSS alone (15.33% [95% CI, 11.16–19.50]; p < 0.001, Fig. 4A), consistent across wound types (Fig. 4D). Sensitivity of FL was comparable to CSS+FL. Detection of false positives using CSS and FL was rare, resulting in specificity of 84.1% (95% CI, 75.1–93.2%; Fig. 4B) of CSS+FL, which was comparable to CSS. Specificity of FL remained similarly high relative to CSS across all wound types (Fig. 4E). Diagnostic odds ratio of CSS+FL was 8.3 (95% CI, 4.1–17.0), and was 3.1-fold higher than CSS (2.7 [95% CI 0.9–7.7]; Fig. 4C). PPV of FL (either alone or in combination with CSS) was comparably high (96.0, 95% CI [93.1–98.9] and 94.6, 95% CI [91.3–97.9], respectively) to CSS alone (91.7, 95% CI [83.9–99.5]), but NPV and accuracy of CSS+FL were significantly increased by 64.4% and 2.2-fold, respectively, compared to CSS (Table 2; p < 0.001). CSS alone had poor discriminative power to predict wounds with high bacterial loads (Fig. 5); FL drove improvements in discriminative power to identify wounds with bacterial burden >104 CFU/g at point of care. With FL, high bacterial burden was identified in 131 wounds otherwise missed by CSS. FL provided additional benefits at the time of diagnosis by locating bacterial burden outside of the wound bed in 128/302 (42.4%) wounds negative for CSS. The enhanced sensitivity, accuracy, and discriminative power of FL compared to CSS resulted in identification of a larger proportion of wounds with bacterial loads >104 CFU/g.
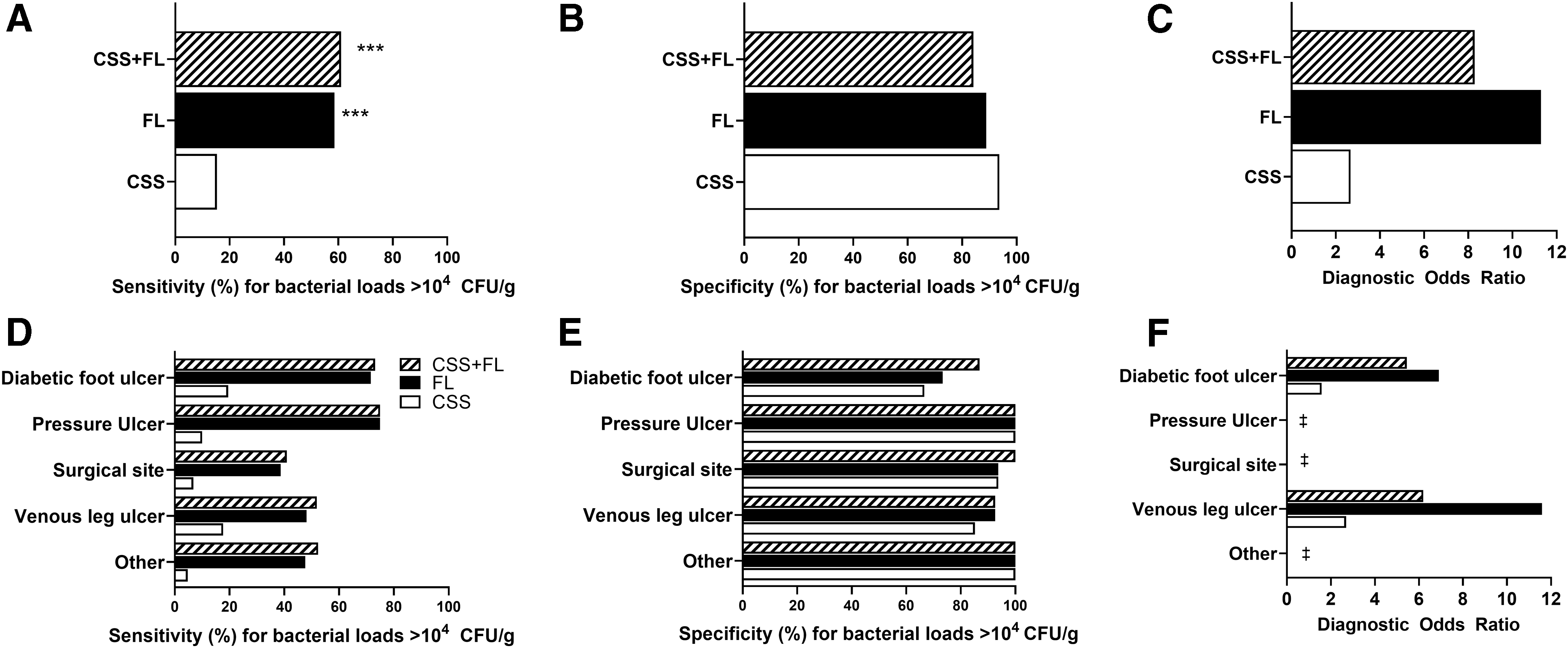
CSS of infection combined with FL were compared with CSS and FL alone at the participant level for sensitivity
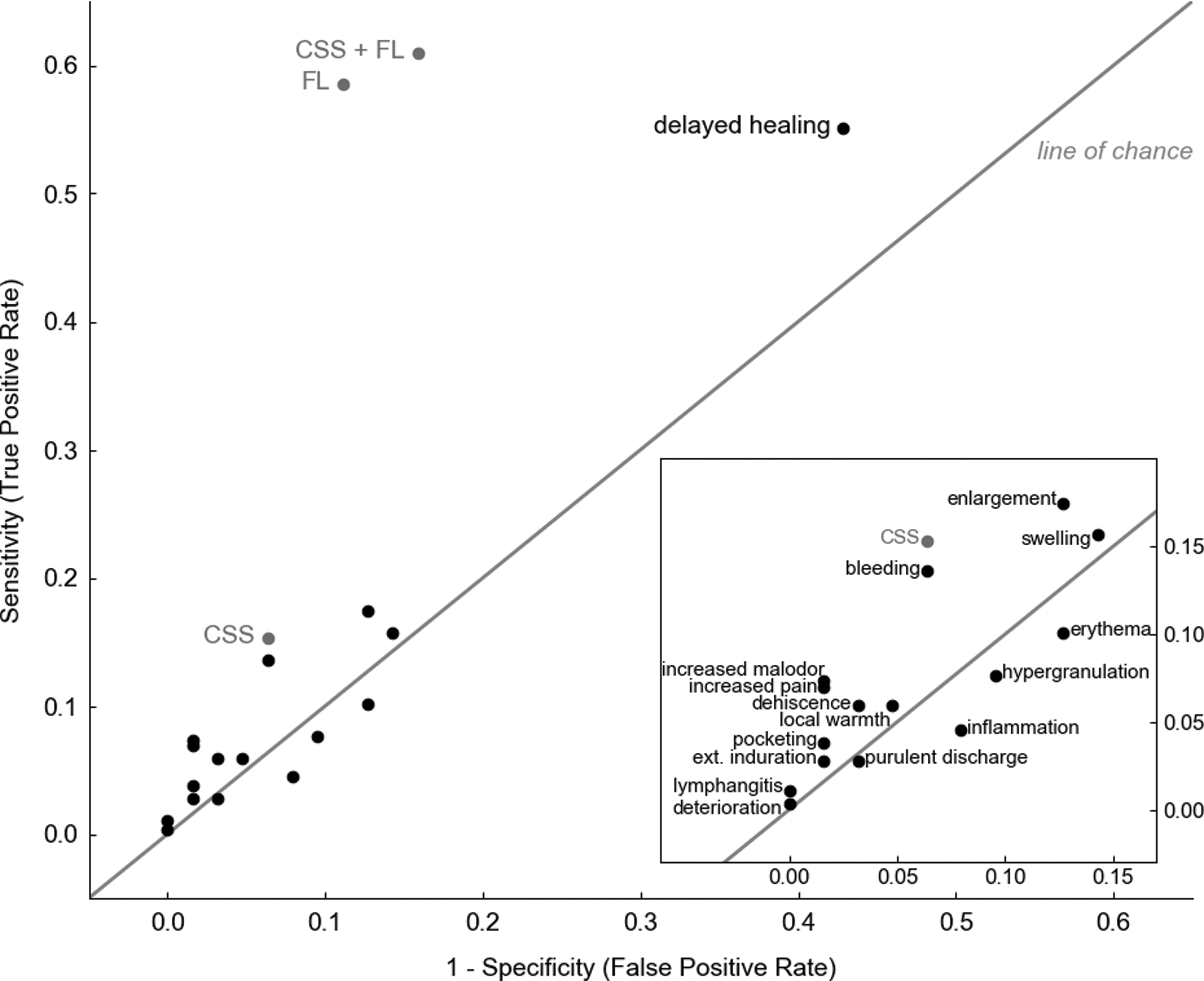
Scatter plot (pairs of sensitivity, 1-specificity) comparing discriminative power of CSS of infection (based on IWII criteria 14 ), individual signs of infection, FL, and CSS+FL. Values in the top left corner indicate high discriminative power. Erythema, hypergranulation, inflammation, and purulent discharge all fell below the line of chance indicating they were no better than “flipping a coin” at predicting bacterial loads >104 CFU/g in wounds. IWII, International Wound Infection Institute.
Estimates of positive predictive value, negative predictive value, and accuracy for detection of bacterial loads >104 CFU/g
Values in bold indicate significance.
PPV, NPV, and accuracy were estimated for CSS, CSS+FL, and FL using microbiological analysis of total bacteria load to serve as ground truth. CSS of infection combined with FL was compared with CSS and FL alone at the participant level. All p-values were derived from one-sided tests.
CI, confidence interval; CSS, clinical signs and symptoms; FL, fluorescence imaging; NPV, negative predictive value; PPV, positive predictive value.
The impact of FL information on care planning was evaluated using a clinician survey. The survey asked clinicians to report which aspects of wound care were most impacted by FL. Clinicians reported that FL resulted in improvements to patient care (which includes wound bed preparation, treatment planning, patient engagement, and monitoring treatment efficacy) in 90.0% of study wounds. FL information also resulted in changes to diagnosis of bacterial burden in 52.3% of wounds (Fig. 6). The objective, diagnostic information provided by FL changed clinical treatment plans in 68.9% of wounds (Fig. 6A). FL information guided wound bed preparation in 84.6% of wounds; and had the greatest impact on primarily tissue management (67.4%) and infection control (76.3%; Fig. 6B). Wound care decision making stems from assessment; thus, not surprisingly, assessment was heavily influenced by FL-information (78.6%). Downstream aspects of care, including sampling location (44.6% of wounds), cleaning (42.9%), debridement (48.0%), treatment selection (55.4%), and wound documentation (45.1%), were also influenced (Fig. 6C). Table 3 summarizes the aspects of care that were impacted by fluorescence information and compares impact of that information in wounds deemed fluorescence (bacteria) positive versus fluorescence negative. As expected, changes to care plan, (with the exception of wound assessment, moisture imbalance, and edge advance), were more prevalent among wounds positive for bacterial fluorescence compared to those negative for bacterial fluorescence (p < 0.001), indicating that it was primarily the enhanced detection of bacteria provided by fluorescence information that significantly influenced clinicians' care planning.
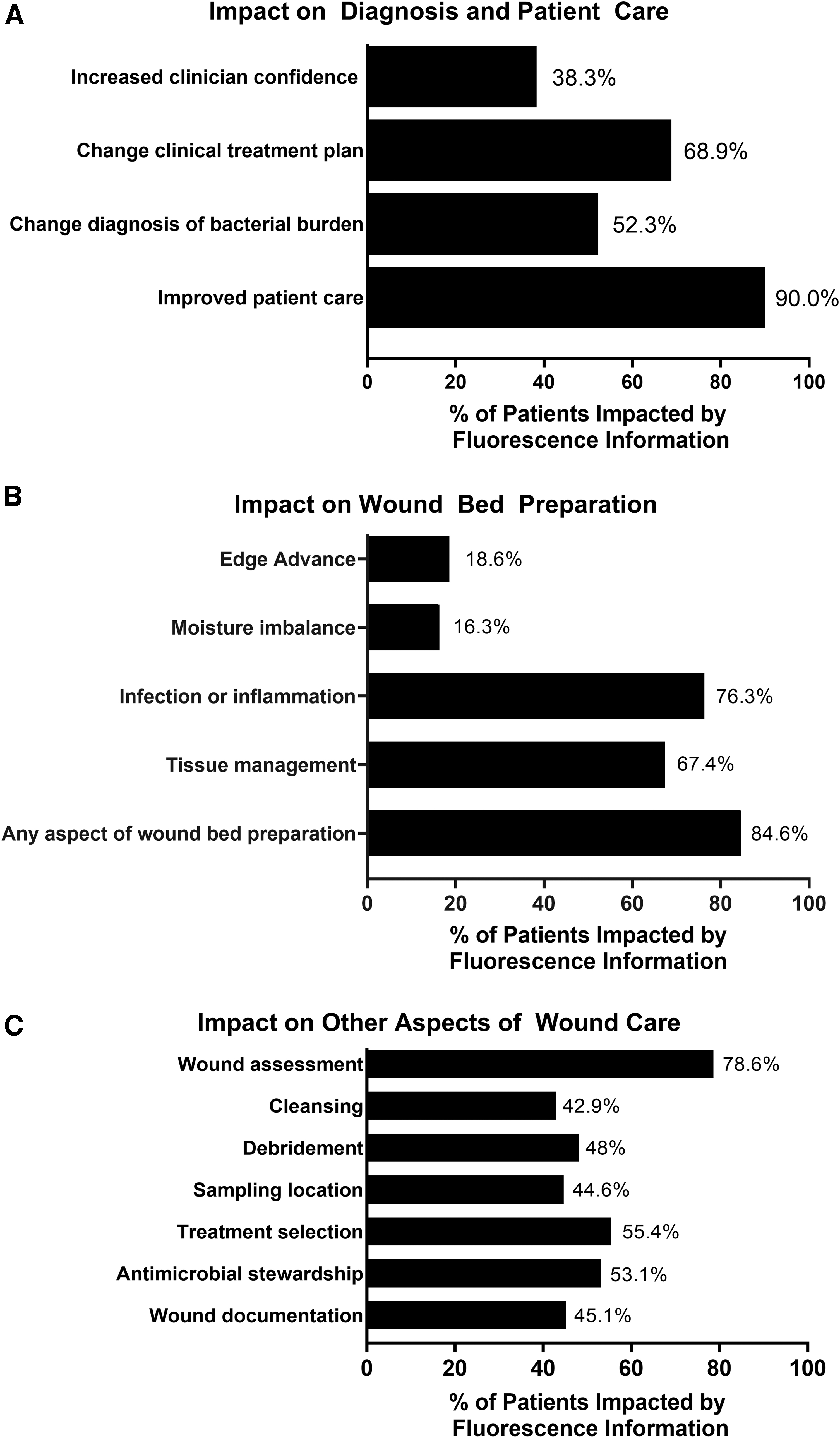
Impact of FL on care plan. Clinicians completed a survey on utility of fluorescence information after capturing images. Clinicians reported on how FL information impacted diagnosis and patient care
Impact of fluorescence imaging on care plan
Clinicians completed a survey on utility of fluorescence information after capturing images. The total number of participants where fluorescence information influenced care plan is listed in column 2. For each survey item, a Fischer's exact test was performed to assess differences between wounds deemed positive (FL+) or negative (FL−) for bacterial fluorescence. Statistical significance was set at p = 0.05; values in bold indicate significance.
Discussion
Bacterial load in wounds is underestimated and the incidence of infection in the wound care population is underreported, 17,18 and therefore undertreated. The presence and severity of bacterial loads in wounds are typically inferred from CSS. 43,44 However, CSS is inherently subjective and frequently fails to detect wounds with moderate-to-heavy bacterial loads. 16,17 More accurate methods to identify wounds with clinically significant loads of bacteria can facilitate better management of wounds according to standard of care practices. 15 In this study, FL of bacteria to detect bacterial loads >104 CFU/g was used in combination with standard of care assessment of CSS to determine if detection of wounds with high bacterial loads (>104 CFU/g) could be improved. Microbiological analysis of wound biopsies revealed median bacterial load of 1.8 × 106 CFU/g, with 36.6% of study wounds having bacterial loads >107 CFU/g. At bacterial loads of 104 CFU/g, clinical signs of infection may not manifest, but delayed wound healing is observed. 9,10 CSS assessment failed to detect 84.7% (155/183) of wounds with bacterial loads >106 CFU/g, a threshold that some consider indicative of infection. 18 CSS (individual and combined criteria) had poor discriminatory power in identifying wounds with bacterial loads >104 CFU/g. Delayed healing, which had high sensitivity, was the clear exception, but had poor specificity, likely due to presence of physical characteristics that may delay healing (e.g., presence of biofilm, vascular insufficiency, and poor offloading). 15,45 Four signs of infection (purulent discharge, inflammation, hypergranulation, and erythema) fell below the line of chance and were ineffective at predicting bacterial loads >104 CFU/g, consistent with previous reports. 16,17 The poor discriminatory power of CSS would have resulted in 84.7% (243/287) of patients with bacterial loads >104 CFU/g receiving inappropriate treatment to address bacteria at the time of assessment. Indeed, a recent meta-analysis of CSS effectiveness concludes “the apparent lack of utility of a combination of findings identified by infectious disease experts (Infectious Diseases Society of America criteria) as useful for diabetic foot infection is both surprising and disappointing, but highlights the difficulty in making the diagnosis.” 17 To overcome stagnant wound healing trends, improved methods of identifying and treating bacterial load need to be prioritized.
Detection of bacteria in wounds using FL has been previously validated through in vitro and in vivo studies that elegantly demonstrated the correlation between intensity of fluorescent signal (from bacterial porphyrins) and bacterial load and showed that FL can detect both planktonic and biofilm-encased bacteria, 23,46 although it cannot distinguish between these two states of bacteria. Biofilm detection and eradication are of tremendous importance in wound care, with biofilm prevalence estimated in up to 90% of chronic wounds. 47 Even without distinguishing between planktonic and biofilm-encased bacteria, the ability of FL to detect bacteria in biofilm and target treatment to regions that potentially contain biofilm is a significant advancement for the field.
In vitro results lack the tissue in which wound bacteria are dispersed and other factors present in the wound that may influence capacity to detect high bacterial loads in wounds. This makes clinical studies critical to assess the true performance of this device to detect bacteria above 104 CFU/g. Consistent with prior clinical studies, 33,35,48 use of the FL diagnostic procedure to detect bacterial loads >104 CFU/g resulted in higher sensitivity (4-fold) and accuracy (2.2-fold), enhanced detection of high bacterial burden in wounds otherwise missed by CSS, and immediately impacted treatment plans. Inaccurate or late diagnosis of bacteria and infection plagues chronic wounds at great costs to the patient and health care systems, 3,4,49 and contributes to some of the 196 daily DFU-related amputations in the United States. 50 Undertreatment and overtreatment can lead to suboptimal wound care, inflated costs, and antibiotic misuse. 51 The robust performance characteristics of FL reported in this study demonstrate the applicability of this diagnostic procedure to facilitate earlier detection of detrimental wound bacterial burden. 15
According to guidelines,
15
intervention is mandated in wounds when bacterial colonization turns into local infection (≥106 CFU/g). Intervention at this critical point prevents further escalation up the infection continuum and damage to host tissue. In this study, FL provided real-time evidence of high (>104 CFU/g) bacterial loads in 131 wounds negative for CSS, prompting intervention in the form of bacterial-targeted therapies (e.g., cleansing, debridement, or use of antimicrobials). The inclusion of FL as part of routine wound assessment provided information on bacterial burden that led to additional improvements in care: Guided wound bed preparation in ≥90% of wounds in this and other studies.
35,52
Information on location of bacterial burden at point of care has been shown to be highly impactful for debridement,
52,53
selection of appropriate cleanser,
30
and general wound bed preparation before application of advanced therapies.
30
Advanced therapies such as cellular and tissue-based products and skin grafts often fail when high bacterial loads are present.
54
–56
Alerted clinicians to unexpected location of bacterial loads.
27,52
In this study, more than 80% of wounds (150/185) positive for fluorescence from bacteria had bacterial burden outside of the wound bed. Treatments to minimize bacterial load (e.g., debridement) are not typically targeted to this region
57
and sampling is rarely performed outside of the wound bed.
58
–60
The FL information in this study provided objective evidence on location of bacteria to facilitate targeted eradication. Provided information on efficacy of antibiotics and guided stewardship decisions without delay.
35
In this study, 56 microbiology-positive wounds were on systemic antibiotics at the time of enrollment. FL revealed the presence of red or cyan fluorescence, indicative of bacterial loads >104 CFU/g in 39.3% (22/56) of these wounds. Biopsy analysis later confirmed the presence of bacteria at loads >104 CFU/g in these wounds. Together, these findings suggest inadequacy of the antibiotic treatment that had been prescribed to those 22 patients.
A recent international position article on antimicrobial stewardship 51 highlighted diagnostic uncertainty in wounds as a key factor contributing to antimicrobial misuse, and recommends the use of rapid, diagnostic testing to ensure judicious use of antimicrobials. In this study, we show evidence that supports this recommendation; FL resulted in more appropriate diagnosis of 46% of wounds with bacterial loads >104 CFU/g compared to CSS and impacted antimicrobial stewardship decisions in 53.1% of wounds. Diagnostic imaging provides actionable information to better implement gold standard wound care.
Strengths and limitations
This study of 350 patients included a heterogenous sample of wounds, across multiple clinical sites. The minimal participant exclusion criteria and diverse wound types included in the study increase the generalizability of results to the overall chronic wound population. Furthermore, the use of wound biopsy and culture analysis to confirm bacteria loads enhanced confidence in the diagnostic accuracy measures reported. However, there were limitations to these methodologies. First, due to the imprecision of soft tissue biopsy trimming, the biopsies were cut to a greater depth than the 1.5 mm excitation limit of the imaging device; thus, it is possible that the biopsy may have detected slightly more anaerobic bacteria than the device was able to. Second, the conditions of culture analysis are not favorable for fastidious bacteria and may have resulted in underreporting the diversity of bacteria species present in the wound. This study focused primarily on high bacterial loads as a contributor to delayed wound healing, but additional systemic factors that were not reported in this study, including vascular insufficiency 61 and protease activity, 62 must also be considered. Clinicians had limited experience using FL in a clinical context before the study, which may have contributed to lower sensitivity to detect bacteria at loads >104 CFU/g than previously observed. In prior FL studies, sensitivity estimates ranging from 72% to 100% were reported, likely due to more clinician experience using the device. 21,28,29,63 As with other diagnostic imaging modalities, 64 –66 we anticipate that the performance measures reported should be improved with increased experience. 67,68 This single time point study meant that effectiveness of changes in treatment plan based on FL could not be measured. Longitudinal randomized controlled trials assessing wound healing may further elucidate the impact of point-of-care diagnostic imaging of bacteria. Evidence from small longitudinal observational studies demonstrate accelerated wound area reduction with use of FL. 32,53 Due to the limited (1.5 mm) depth of excitation 36 and inability to detect non-porphyrin-producing bacteria, including species from the Streptococcus, Enterococcus, and Finegoldia generas (which account for an estimated 12% of the most prevalent wound pathogens 23 and rarely occur monomicrobially 69 ), it is recommended that FL be used in combination with CSS.
Conclusion
The severity of bacterial burden in wounds is grossly underappreciated. Our results from 350 wounds reveal failure of current standard-of-care assessment to detect 84.7% of wounds with bacterial loads >106 CFU/g, which some suggest are indicative of infection. 18 Incorporation of the noninvasive FL diagnostic procedure to wound assessment greatly improved detection of high bacterial burden across a variety of wound types and provided information on bacterial location at point of care. This represents a paradigm shift in wound assessment, in which clinicians now have immediate information on bacterial burden to guide treatment selection and inform the frequency of reassessment to determine the efficacy of selected treatments at point of care. 34,53 The point-of-care information provided by FL facilitates a rapid switch to a more effective bacterial-targeting agent (e.g., cleanser and bandage). 34,70 Study results, collected across 14 study sites from 20 clinicians of varying skill levels, indicate the widespread utility of FL to inform wound assessment, wound bed preparation, and overall treatment planning.
Innovation
Despite advances in wound therapies, wound healing rates in the last 40 years have remained stagnant as clinicians continue to work blindly to address bacterial burden in wounds. In this study, FL increased detection of high loads (>104 CFU/g) of bacteria by fourfold and informed the location and extent of bacteria in wounds. This actionable information enabled early detection of bacteria, especially in highly prevalent asymptomatic wounds, and allowed clinicians to treat bacterial burden without delays. Information provided by this noncontact point-of-care imaging device can be used to inform treatment planning and evaluate the efficacy of selected treatments.
Key Findings
Eighty-two percent of study wounds (287/350) had clinically significant bacterial loads (>104 CFU/g), which were missed by standard-of-care assessment of CSS of infection.
Incorporation of MolecuLight i:X fluorescence imaging device with standard-of-care assessment of CSS increased point-of-care detection of wounds with high bacterial loads (>104 CFU/g) by fourfold compared to CSS alone.
Use of this noncontact point-of-care bacterial imaging device significantly impacted downstream aspects of patient care, including sampling location (44.6% of wounds), cleaning (42.9%) and debridement (48%), selection of antimicrobials (53.1%) and other treatments (55.4%).
Footnotes
Acknowledgments and Funding Sources
Funding for the study was provided by MolecuLight, Inc.; Ironstone Product Development contributed to the study design and conducted data auditing. All authors had access to relevant data, had approved the final version, and were responsible for the decision to submit the article for publication. The authors thank MolecuLight, Inc., for assistance with preparation of the schematic in ![]() .
.
Author Disclosure and Ghostwriting
SerenaGroup research foundation received funding from MolecuLight, Inc., to cover conduct of the study. No competing financial interest exists for other authors. The authors had MolecuLight Inc review the manuscript and provide editorial assistance from an accuracy and regulatory standpoint. No ghostwriters were used to prepare this article.
About the Authors
Supplementary Material
Supplementary Figure S1
Supplementary Table S1
Abbreviations and Acronyms
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
