Abstract
Significance:
Cutaneous wound regeneration is vital to keep skin functions and for large wounds, to maintain human survival. In a deep burn, the ability of the skin to heal is compromised due to the damage of vasculature and resident cells, hindering a coordinated response in the regeneration process. Temporal skin substitutes used as first cover can play a major role in skin regeneration as they allow a rapid wound covering that, in turn, can significantly reduce infection risk, rate of secondary corrective surgeries, and indirectly hospitalization time and costs.
Recent Advances:
Skin was one of the first tissues to be bioengineered providing thus a skin equivalent; however, what is the current status subsequent to 40 years of tissue engineering? We review the classic paradigms of biological skin substitutes used as first cover and evaluate recent discoveries and clinical approaches adapted for burn injuries cover, with an emphasis on innovative cell-based approaches.
Critical Issues:
Cell-based first covers offer promising perspectives as they can have an active function in wound healing, such as faster healing minimizing scar formation and prepared wound bed for subsequent grafting. However, cell-based therapies encounter some limitations due to regulatory hurdles, as they are considered as “Advanced Therapy Medicinal Products,” which imposes the same industry-destined good manufacturing practices as for pharmaceutical products and biological drug development.
Future Directions:
Further improvements in clinical outcome can be expected principally with the use of cell-based therapies; however, hospital exemptions are necessary to assure accessibility to the patient and safety without hindering advances in therapies.


Scope and Significance
Temporal skin substitutes used as first cover can play a major role in skin regeneration, especially for deep burns, as they allow a rapid wound covering that, in turn, can significantly improve subsequent clinical outcomes. In this review, we discuss the evolution of biological skin substitutes to include innovative cell-based therapies, with associated advantages and limitations.
Translational Relevance
In a deep burn, the ability of the skin to heal is compromised due to the damage of vasculature and resident cells, hindering a coordinated response in the healing process. The introduction of next generation of biological skin substitutes could stimulate regeneration by the release of a well-proportioned combination of growth factors.
Clinical Relevance
The care of large burn wounds is associated with prolonged hospitalizations and significant costs without mentioning the severe pain, emotional, and physical strain that the burn trauma inflicts to patients. Unmet needs still remain regarding sufficiently advanced wound care therapies to improve healing. Cell-based first covers offer innovative approaches to treat burn wounds and cope with the various burden.
Background
The skin is the largest organ of the human body, having the functions of controlling the body temperature and peripheral fluid balance, allowing sensation (e.g., heat, cold, touch, and pain) and providing a semipermeable protective layer against pathogens. 1,2 Although the skin has a spontaneous self-regeneration ability, in some cases physiopathological conditions hinder this ability: in chronic wounds, such as in diabetic foot ulcer or pressure ulcer, tissue homeostasis is disrupted because of an altered paracrine signaling between keratinocytes and fibroblasts 3 ; in acute wounds (e.g., deep burns), vasculature is damaged or partially damaged, thus limiting the number of cells and factors brought for regeneration. 4 The time to wound closure is correlated with higher risk of complications such as infection, pain, and scarring occurrence. 5
Several approaches have been developed over the years to treat cutaneous wounds that have lost their ability of spontaneous healing; split-thickness skin autografting remains the gold standard treatment for covering of large acute and chronic wounds (Fig. 1). 6 However, immediate wound covering by surgery is not always possible such as in large wounds of severely burned patients, as available surface for donor skin is not sufficient for autografting. In such cases, alternative cell-based strategies have been proven to be life saving, namely by the cultured epithelial autografting (CEA), where the patient's own keratinocytes are expanded to form stratified cell sheets that can be applied topically to the patient for wound closure. 7
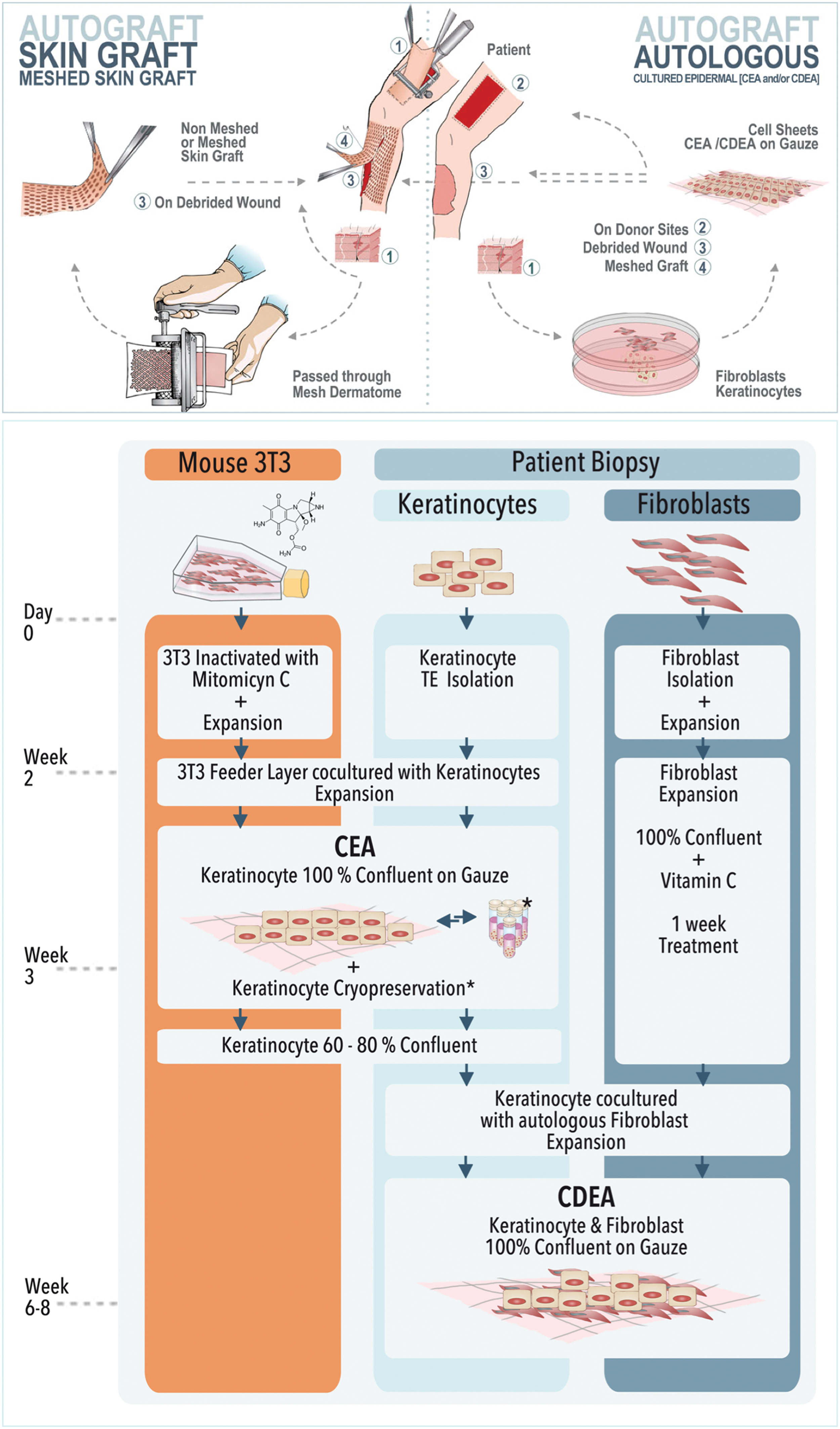
Upper panel: Schematic illustration of split-thickness skin autografting and cell-based autografting; lower panel: Fabrication process of CEA and CDEA. CDEA, cultured dermal–epidermal autograft; CEA, cultured epithelial autografting. Color images are available online.
In the mid-90s, we have also implemented at our hospital a variant cell therapy, namely cultured dermal–epidermal autograft (CDEA), which is based on the same principle of stratified cell sheets as for CEA, but allows to recreate a thicker skin graft, because of a double layer composed of autologous fibroblasts and keratinocytes. 8 CDEA provides better clinical outcome with a skin graft having more functional mechanical properties than a CEA. Nevertheless, CEA and CDEA techniques require a minimum of 3 and 6 weeks, respectively, to expand cells and build bioengineered thin skin grafts, leaving the patient without cutaneous covering for considerable time periods.
Temporary skin substitutes have a crucial function in this specific case, as they are applied on patients during the cell culture phase to prevent fluid loss and decrease pain by nerve covering, but most importantly provide a physical barrier that can reduce infection risks. 9 Indeed, wound infection is thus the major cause of mortality, morbidity, and wound healing delay. 10,11 With >11 million people affected annually by burn injuries worldwide, 12 the prevalence of infection in burn units is ∼66%. 13 Temporary skin substitutes may considerably reduce the costs and burden related to prolonged hospitalization by reducing the risk of infection and thus have a major impact on the complications requiring longer hospitalization periods.
In this study, we aim to present the different types of temporary skin substitutes used as first cover for skin burns, with a major emphasis on our own experience with the history of the different first covers used at the burn unit of our hospital, including a comparison of the performance, costs, and regulatory pathway of each temporary cover.
Evolution of Temporary Skin Substitutes in Clinical Use
Skin substitutes can be classified into two categories: permanent substitutes and temporary substitutes, which we name “first covers” (Fig. 2). Over time, permanent skin substitutes have encompassed all the cell-based grafting approaches, including autografting of cultured and stratified cells with or without the support of regenerative matrices. Temporary skin substitutes have included xenogeneic decellularized skin, allogenic cadaveric human skin, acting mainly as a barrier to external contamination and protection of underlying tissue, but have evolved to more sophisticated bioengineered formulations, allowing rapid would healing. In this section, we will present a brief overview of the different types of temporary skin substitutes.
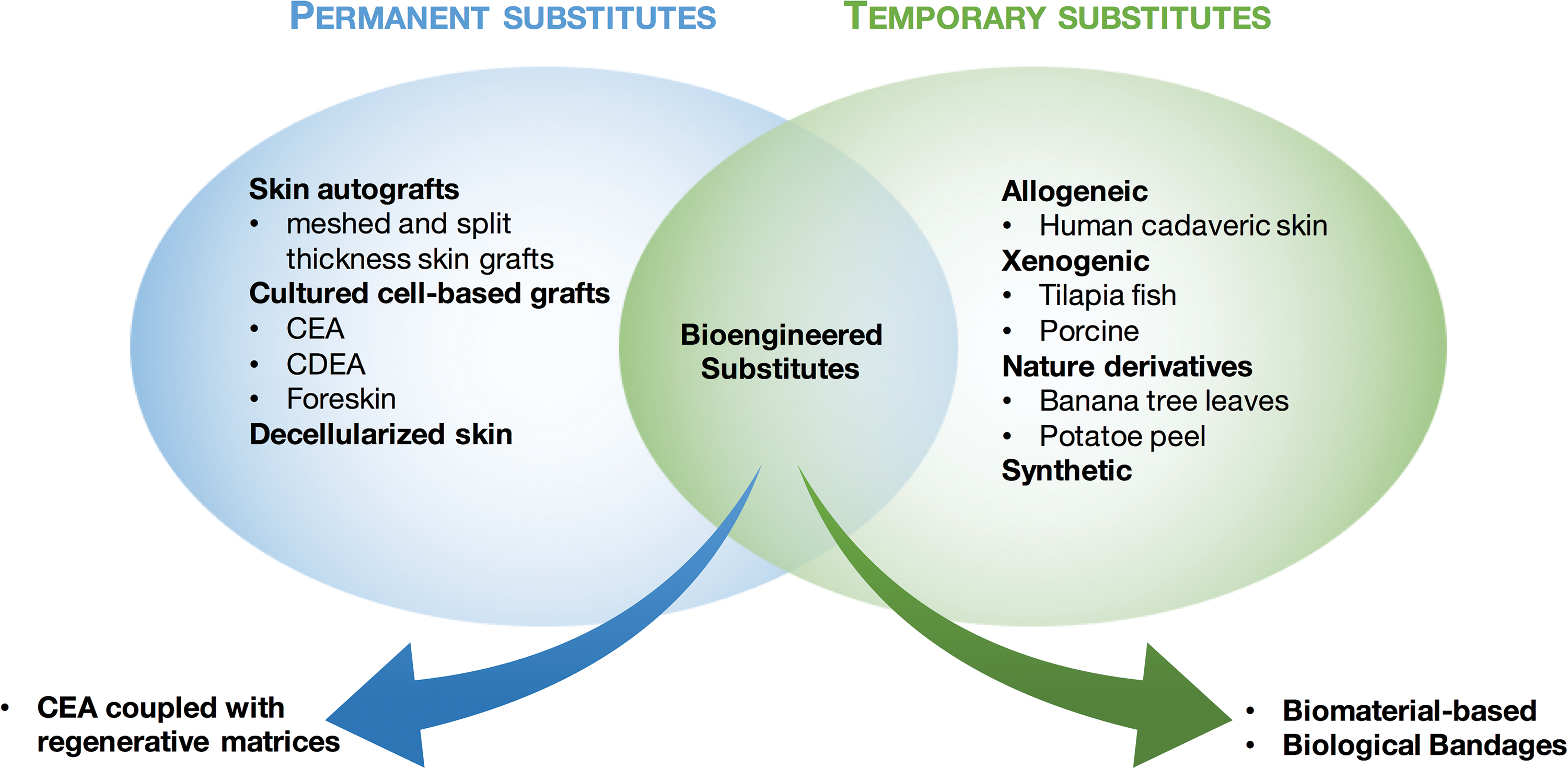
Skin substitutes classified into permanent and temporary substitutes. For both categories, products have evolved from conventional therapies to bioengineered products combining various cells and biomaterials as strategies to improve wound healing. Color images are available online.
Conventional skin substitutes: human cadaveric and xenogeneic
Several temporary skin substitutes were used over time as first cover for partial- and full-thickness burn wounds, for donor sites and meshed autograft protection. They have included human amnion (e.g., Mimedx or Osiris), 14,15 xenografts 16 (e.g., E-Z Derm®, Mölnlycke, Sweden), and human cadaver allografts, such as supplied by the Euro Skin Bank (Beverwijk, Netherland). Porcine xenografts are probably the most frequently used temporary skin replacement worldwide due to easy accessibility and storage, 17 but also because porcine skin is anatomically close to human skin. 18 Skin substitutes are available in several forms: cryopreserved, in glycerol, lyophilized, and decellularized. 16,19 –21 It has been reported that storage in glycerol may be problematic as efficient removal of glycerol residues from the xenograft implies higher degradation rates of the substitute on patients. 22
From our experience, cryopreserved allografts give better protection than glycerol-preserved allografts, probably because of a cryopreservation allowing dermal substitutes to retain the ability to release growth factors and biochemical cues promoting wound healing. 23 It has been shown that an allogenic skin substitute becomes engrafted to the wound after 2–3 weeks, thus the alloepidermis is removed at surgery, leaving the dermal components as a viable bed for a CEA. 24 In some cases, remaining allodermis parts even become re-epithelialized. 25
The benefits of allografts and xenografts include immediate soothing upon application, minimized pain during dressing changes, and minimized protein and water loss, allowing the patient to resume activity without disturbing the healing process.
However, their main drawbacks are the rejection-related risks. Indeed, although the incorporated dermis is usually not rejected in a first period, allograft could trigger a chronic severe rejection reaction when the patient's immune system is restored. This can also appear clinically with the formation of unstable or hypertrophic scars. 26 Furthermore, these temporary substitutes may enhance risk of disease transfer and their integration in the recipient site renders their ablation difficult and sometimes leaving painful eschars. 8,27
Synthetic substitutes
Conventional skin substitutes are normally overlayered by low-adherent sterile paraffin gauze (Jelonet from Smith & Nephew Medical Ltd., England). On donor sites or when burn wounds are smaller, gauze or foams only are applied (synthetic dressings without allogenic/xenogeneic substitute) as standard of care. Currently >3,000 types of dressings are available on the market, offering a plethora of options to address all aspects of wound care. 28 In brief, these dressings encompass materials made of semipermeable silicone foams, hydrocolloids, hydrogels, and hydrofibers as reviewed by Borda et al. 29
Although these dressings mainly protect the burn injury, they do not contribute to the recovery of dermal and epidermal tissues. Synthetic substitutes can also be dermal constructs made of nonbiological molecules and polymers not present in normal skin. Although these substitutes offer a stable and biodegradable environment for the regeneration of tissue with a more precisely controlled composition and properties, the use of nonbiological components can present biocompatible issues. 30 Hence, there is still no superior synthetic substitute that can achieve complete healing, especially in case of chronic wounds such as venous leg ulcers, diabetic wounds, and pressure ulcers. 31
Nevertheless, synthetic dressings or substitutes can be available in different formulations, thus several dressings are often functionalized with silver, iodine, or other antimicrobial agents to further prevent infection. 32 –34 However, current antimicrobials in use, most commonly silver, have the limitation to induce skin irritation and staining after prolonged treatment. 35
Bioengineered skin substitutes
Contemporary technological advances in biomaterials have allowed to develop cell-based skin substitutes, such as Dermagraft® or Transcyte® from organogenesis (Canton), which are made, respectively, of polyglycolic acid/polylactic acid scaffolds and collagen-coated nylon mesh, both seeded with allogeneic foreskin fibroblasts secreting growth factors to aid wound healing. 36
Other cell-free yet bioactive dressings have been developed such as Biobrane® (Smith & Nephew, London, England), which consist of a semipermeable silicone film partially imbedded nylon, functionalized with porcine collagen. Biobrane has the benefit of minimizing bacteria proliferation on wound surface by minimizing dead space, controlling water vapor loss (comparable with native skin), and allowing conformability to surface irregularities. The latter property subsequently helps joint movement and early ambulation due to its flexible mechanical properties. A prospective randomized clinical trial comparing both bioengineered products, Transcyte and Biobrane, indicated that the cell-based Transcyte product promotes a faster re-epithelialization, whereas requiring fewer dressing changes and autografting procedures. 37
Nevertheless, biocompatibility still remains the major issue for most of the developed bioengineered products, as they contain either allogeneic or xenogeneic components with risks of inflammation or rejection. To cope with this problem, we have developed at our hospital biological bandages made of clinical-grade progenitor skin fibroblasts of fetal origin, delivered to the burn wound on a collagen matrix. 38 These bandages positively impact wound healing processes, most probably by secreting growth factors, while inducing minimal inflammatory response due to their immune-privileged source. Thus, we were able to show with eight pediatric patients treated with biological bandages alone that they did not require further associated autologous grafting. 38
Likewise, progenitor cells showed remarkable outcomes in the treatment of donor site wounds (Fig. 3) and chronic wounds with positive outcome. 39 The progenitor cells are derived from a Federal Transplantation Program following the Swiss constitution for laws on research on human beings and transplantation medicine (art 119a Cst). Fetal tissue is considered as an organ donation from voluntary pregnancy interruption and can be obtained with specific strict regulation within transplantation law.
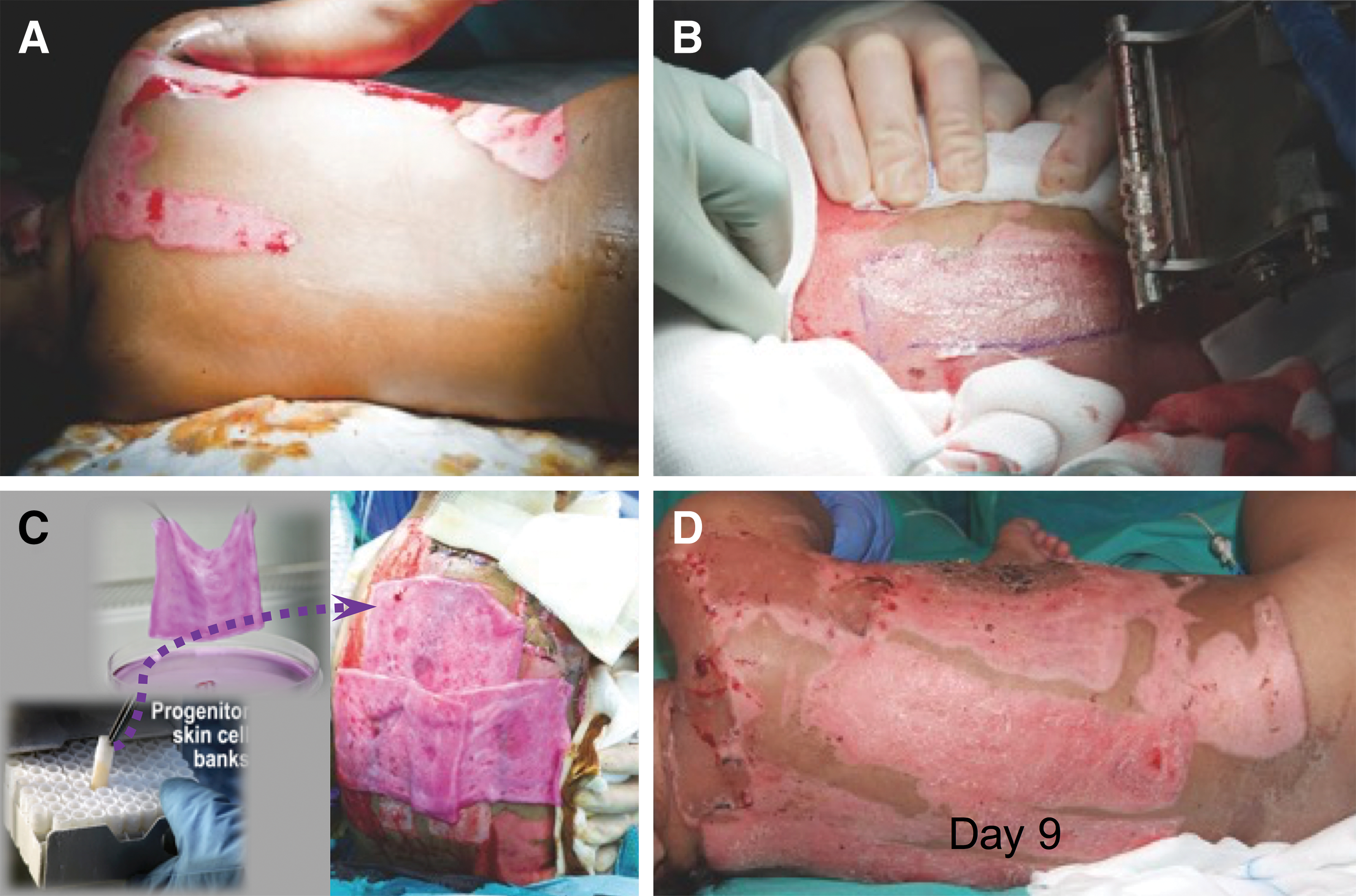
Use of biological bandages as cover for skin donor sites.
The benefits of bioactive substitutes may be multiple depending on the severity of the burn wounds. For superficial wound zones, they allow a more rapid healing thus minimizing scar formation, whereas for intermediate zones, it can help to reduce the edema and risks of thrombosis of the capillaries; for deep wounds the bioactive components prepare the wound bed, thus improving subsequent grafting.
Alternative natural skin covers
Interestingly, skin substitutes derived directly from nature have also been used as temporary cover for burn patients, such as banana leaves, 40 tilapia fish skin (e.g., Kerecis Omega3 fish skin) or potato peel. 22 Such approaches are often in use in developing countries, where more sophisticated and more expensive wound cover are not easily available.
Benchmarking of First Covers
In this section, we aim to give comparisons from our own experience between the different types of temporary substitutes used as first covers for burn wounds, thus particular focus will be given to cadaveric and porcine substitutes and the biological bandages, the three types of temporary covers employed at the burn unit of our institution (Fig. 4).
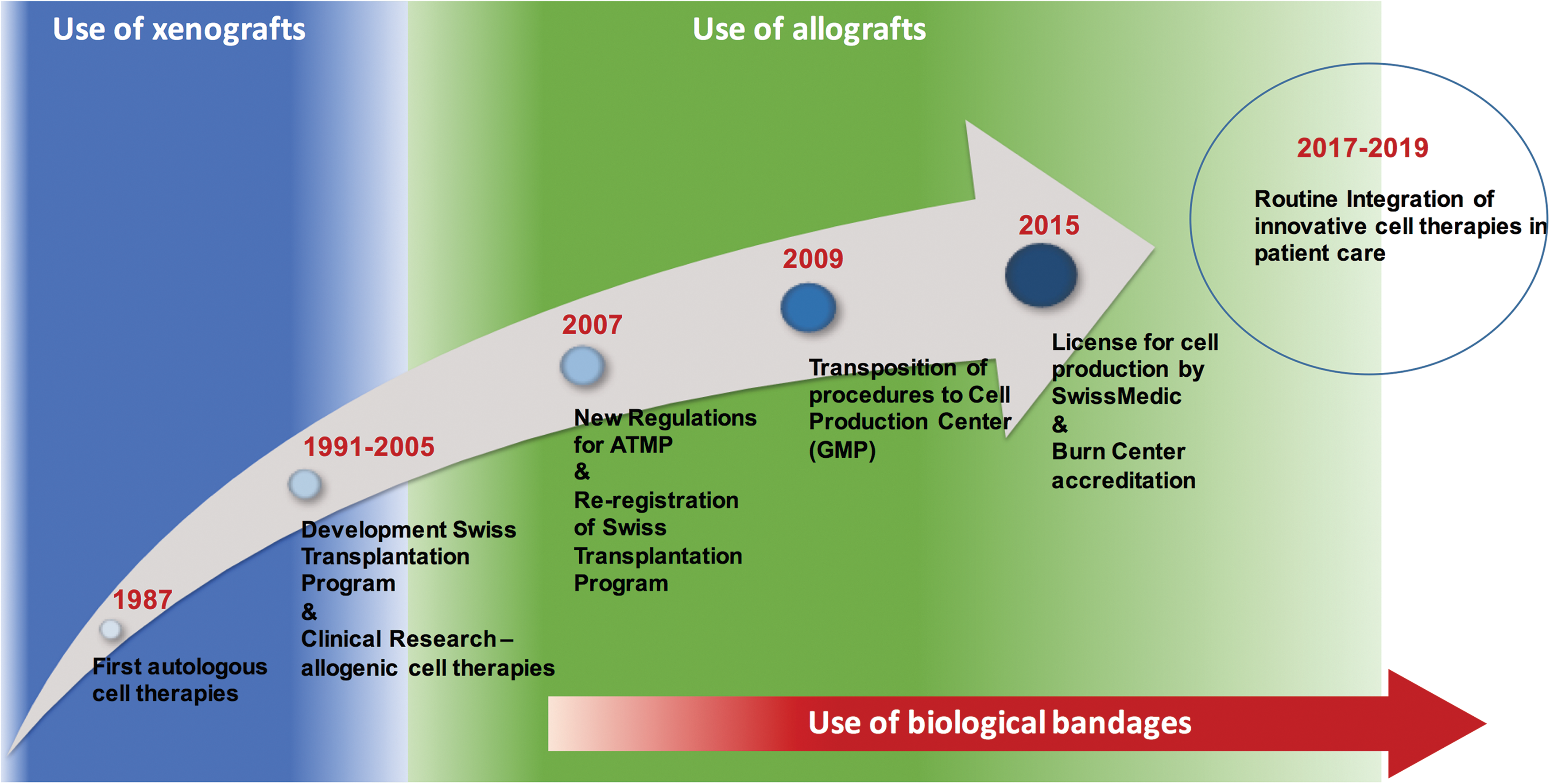
Timeline of evolution of the first covers used at CHUV burn unit and the implementation of cell therapies. Color images are available online.
From a purely practical point of view, the advantage of the biological bandages over the cadaveric and porcine substitute resides is the fact that it can be easily removed from patients without being painful and leaving necrotic tissues (Fig. 5). Biological bandages, on the contrary to the allogenic substitute, do not require staples to attach to the patient, as they can be simply overlaid with Jelonet followed by a Multivac compress, as a standard of care.
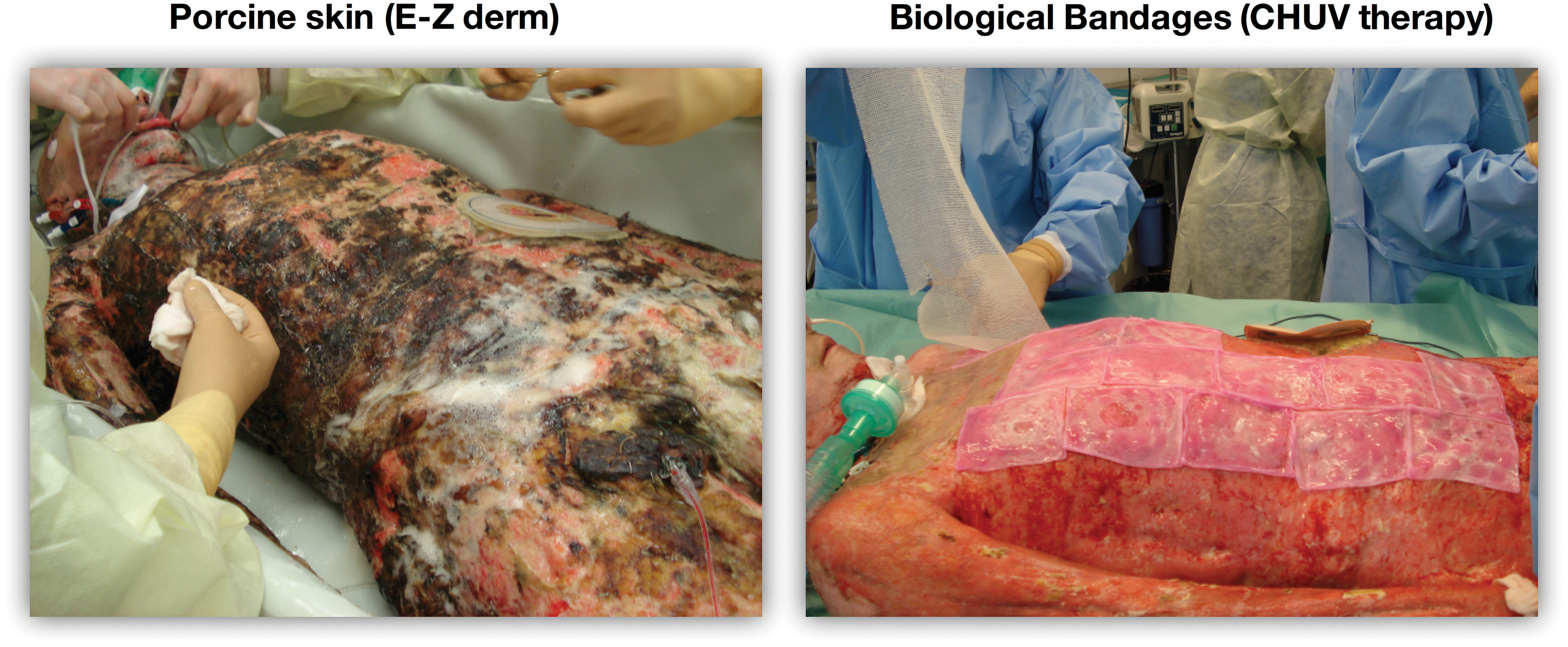
Photographs of a burn patient treated in a first time with E-Z Derm® as first cover (left panel), and with biological bandages (right panel). Biological bandages are applied the same day after E-Z Derm removal and debridement; they demonstrate superior outcomes in comparison with porcine substitute, as shown by the lack of necrotic tissue, the absence of the staples and ease of handling. Color images are available online.
Regarding the costs, the price of porcine cover (E-Z Derm of 8 × 120 cm surface) is 321 Swiss Francs (CHF), whereas a biological bandage (9 × 12 cm) costs 26 CHF. Consequently, to cover 1 m2 of burn surface would cost about 3,340 CHF for porcine cover and 2,550 CHF for a biological bandage, which is 25% less expensive. The most expensive first cover remains human cadaveric skin substitute as it costs ∼12,000 CHF for the same surface of 1 m2 (from Skin Euro Bank, Netherlands).
If we put into perspective these numbers with the costs of common dressings used in the standard of care of burn wounds, such as Aquacel (ConvaTec, United Kingdom) or a Jelonet paraffin Tulle Gras dressing, for the same surface a cadaveric cover is one order of magnitude more expensive than Aquacel, and two orders of magnitude more than Jelonet. Nevertheless, these numbers have to be normalized over time as well, since a biological bandage or conventional dressings can be changed several times per week, whereas conventional covers can remain several weeks without being changed.
The benefit of using such high-cost covers should then be weighted by the impact they might have on reducing hospitalization times, knowing that patient care at the Lausanne University Hospital (CHUV) Burn Unit costs 4,000 CHF per day. The main advantage of biological bandages over allogenic and xenogeneic covers, respectively, remains in the fact that it could actively stimulate wound healing. Indeed, as already mentioned, a biological bandage may induce faster healing minimizing thus scar formation, whereas for intermediate zones it can help to reduce the edema and risks of thrombosis in capillaries, and for deep zones it can prepare the wound bed thus improving subsequent grafting.
These advantages can positively impact wound healing that, in turn, may reduce hospitalization times and global costs, besides the fact that biological bandages can readily be prepared within 24 h from an off-the-shelf good manufacturing practice (GMP) cell bank, circumventing storage concerns.
Figure 6A shows the constant increase in the number of biological bandages used at the Lausanne University Hospital per year between 2013 and 2017, whereas the number of patients admitted to the ICU burn unit per year remains constant, which illustrated the implementation of biological bandages in clinical routine in burn care, especially for adult patients. This observation can also be illustrated by the increase of mean number of bandages used per patient over the years for a mean total body surface area (TBSA) per patient that also remains constant over time (Fig. 6B).
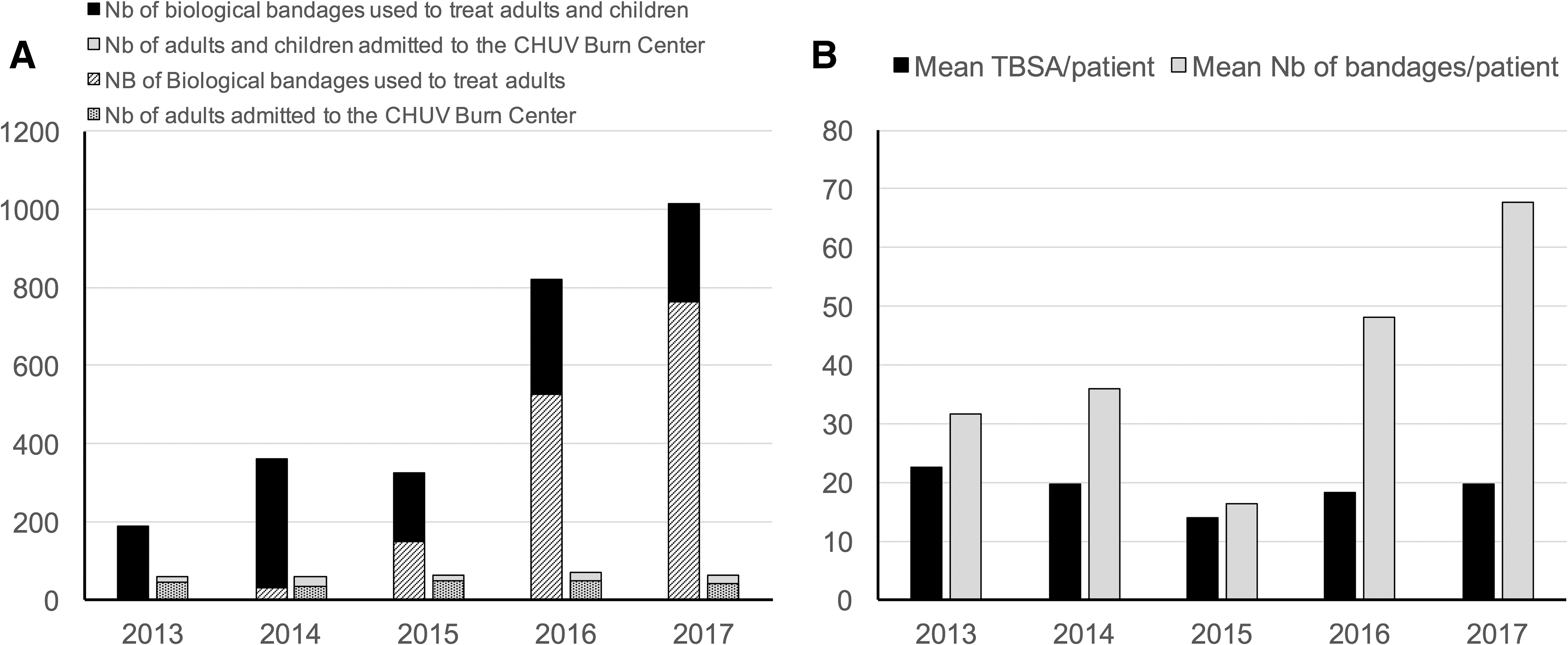
Interestingly, for the same year range, the indicator of care duration normalized by burned surface decreased from 0.6 days/TBSA in 2014 to 0.2 days/TBSA in 2017 (for adults), thus suggesting an improvement in the quality of burn patient care, to which the use of biological bandages may have contributed, as all other aspects of burn care had been standardized previously, except for small improvements in nutrition intake, which was adjusted with supplement intake to cope with the exudative loss of trace elements within the first week. 41 Table 1 summaries the assets that an “ideal” skin cover could have. 22,42
Benchmarking of the biological bandages and conventional allografts used as first covers
Finally, a consideration that might affect the choice of a therapy or another is the use of certain biological material that might be related to religious and/or ethical issues (for instance porcine products for Muslim patients), which requires the informed consent of the patient.
Perspectives on Future Therapies
Despite early excision, immediate wound covering, and topical treatments, the major problematic remaining in burn patient care is the frequency increase of persistent wound infections due to hard-to-treat multidrug resistant bacteria, often Pseudomonas aeruginosa. 43 This is especially true for large burn areas >40–50%. In an effort to cope with burn wound infections, we are currently developing a next generation of biological bandages, coupling antimicrobial dendrimers with progenitor cells, thus allowing a dual effect of controlled infection and enhanced wound healing. 44
In the same perspective to improve wound healing, research endeavors are made to genetically edit keratinocytes to engineer skin equivalents, expressing factors that promote healing. 16 Likewise, studies have shown that mesenchymal stem cells 45 and adipose-derived stem cells 46 can promote wound healing both by direct transfer onto wounds by delivery embedded in scaffolds, as these cells can secrete factors that attenuate inflammation and stimulate angiogenesis, thereby leading to faster wound closure. 47
Although cell-based approaches show significant performance in wound healing, offering promising perspectives, they encounter some limitations due to regulatory hurdles. Indeed, since 2007 new EU regulations consider the application and practice of cell therapies such as “Advanced Therapy Medicinal Products” (ATMPs), imposing to cell therapies the same industry-destined GMPs as for pharmaceutical products. The aim of these regulations is to ensure the safety of these products to a maximum extent. However, these regulations imply considerable delays in research, development, and the production of cell therapies, which would not be of benefit to the patients, without mentioning the increase in costs.
We advocate for “Hospital Exemption,” which would provide a more suitable pathway to customize safe product/therapy for access to industrialized patient care for burn victims. 48 In this respect, revisions on the Therapeutic Products Act (Stage 2) have come into force, from January 2019, in Switzerland. These revisions would simplify and facilitate access to medicinal products for patients and improve conditions for biomedical research. In particular, it will entail a 2-year authorization for the “compassionate use” of a cell-based therapy provided that a clinical study has been approved in Switzerland before the use request.
Other European countries, for instance Belgium, have also applied the same kind of directives for the compassionate use, especially for unproven interventions such as the use of bacteriophages in the treatment of persistent recalcitrant infections, based on article 37 of the Declaration of Helsinki, which stipulates.
“In the treatment of an individual patient, where proven interventions do not exist or other known interventions have been ineffective, the physician, after seeking expert advice, with informed consent from the patient or a legally authorized representative, may use an unproven intervention if in the physician's judgement it offers hope of saving life, re-establishing health or alleviating suffering. This intervention should subsequently be made the object of research, designed to evaluate its safety and efficacy. In all cases, new information must be recorded and, where appropriate, made publicly available.”
Therefore, we are now seeing that our policy-makers are working hand-in-hand with those confronted to help patients in the best manner possible.
Conclusions
In conclusion, temporary dermal substitutes used as first cover for burn wounds can play a major role in skin regeneration as they allow a rapid covering of the wound and thus can significantly reduce infection risk by providing a physical barrier to pathogens, but they can also have an effect on the surface to be grafted and the rate of secondary corrective surgeries; therefore, first covers can indirectly reduce hospitalization time and global costs.
Patient informed consent is also an important matter to take into consideration as the use of various biological products might incite religious and/or ethical issues, which have not been notably documented in the medical literature for skin substitutes.
Advances have been made in the development of dermal substitutes in the past years, and further improvements in clinical outcome can be expected principally with the approach of cell-based therapies; however, hospital exemptions are necessary to assure safety for patients without hindering advances in therapies.
Summary
First covers can improve clinical outcomes in skin regeneration, by allowing a rapid wound covering which subsequently can reduce infection risk, rate of secondary corrective surgeries, and thus hospitalization time and global costs. Recent advances have shown significant improvement of cell-based first cover for burn patients, thus offering promising perspectives in wound healing; nevertheless, cell-based approaches encounter some limitations due to regulatory hurdles.
Further improvements in clinical outcome can be expected; however, hospital exemptions are necessary to assure safety for patients without hindering advances in therapies.
Take-Home Messages
A first cover is a temporary skin substitute that is employed to cover large wounds until autografting.
Over time, first covers have evolved from porcine and human allografts to encompass cell-based therapy approaches.
Biological bandages used as cell-based first covers have shown promising results, as the release of growth factors and cytokines by human progenitor cells helps to prepare the wound bed before grafting and allows faster wound healing, improving thus outcomes.
Regulatory hurdles may impede the use of cell-based approaches, advocating for “Hospital Exemption,” which would provide a more suitable pathway to customize safe therapy for burn victims.
Footnotes
Acknowledgments and Funding Sources
Prof. Applegate acknowledges previous funding from Sandoz and S.A.N.T.E foundations. Current funding from CHUV Priority Project is also acknowledged. Dr. Philippe Abdel-Sayed acknowledges funding from the Marie Sklodowska-Curie Action. The authors thank Ms. Murielle Michetti and Ms. Corinne Scaletta for technical support with artwork.
Author Disclosure and Ghostwriting
Prof. Lee Ann Applegate is the cofounder and interim CEO of Elanix Biotechnologies. Prof. Wassim Raffoul, Dr. Anthony de Buys Roessingh, and Dr. Nathalie Hirt-Burri are cofounders of Elanix Biotechnologies. The content of this article was expressly written by the authors listed and no ghostwriters were used.
About the Authors
