Abstract
Objective:
Sickle cell ulcers (SCUs) are a devastating comorbidity affecting patients with sickle cell disease (SCD). SCUs form over the medial or lateral malleoli of the lower extremity, are slow to heal, and prone to recidivism. Some SCUs may never heal, leading to chronic pain and foot deformities. There is no specific and effective therapy for SCUs. Systemic deferoxamine (DFO) has been demonstrated to prevent some of the sequelae of SCD by chelating iron. In this study, we tested the ability of DFO delivered via a transdermal delivery system (DFO-TDDS) to accelerate healing in a murine model of SCU.
Approach:
Excisional wounds were created in a transgenic murine model of SCD expressing >99% human sickle hemoglobin, and healing rates were compared with wounds in wild-type mice. Next, excisional wounds in SCD mice were treated with DFO-TDDS, DFO injection, or left untreated. Wound closure rates, histology, and iron in the healed wounds were analyzed.
Results:
Wounds in SCD mice healed significantly slower than wild-type mice (***p < 0.001). DFO-TDDS-treated wounds demonstrated significantly accelerated time to closure, reduced size, and improved wound remodeling compared with untreated wounds (***p < 0.001) and DFO injection treatment (*p < 0.05). DFO released from the TDDS into wounds resulted in chelation of excessive dermal-free iron.
Innovation:
DFO-TDDS is a novel therapeutic that is effective in healing wounds in sickle cell mice.
Conclusion:
DFO-TDDS significantly accelerates healing of murine SCUs by chelation of excessive free iron and is currently manufactured in an FDA-compliant facility to be translated for treating human SCUs.

Introduction
T
Sickle cell erythrocytes also display an abnormal level of membrane-associated iron and excessive superoxide production. 6 Furthermore, chronic hemolysis leads to an accumulation of heme in plasma and in tissues, 7 resulting in increased reactive oxygen species (ROS) both through the Fenton reaction 8 and from neutrophil- and monocyte-derived oxidative burst. 7 Increased ROS lead to tissue-free radical defense mechanisms, yielding DNA damage, excessive inflammation, and end-organ damage, among them, chronic ulceration. 9,10 Acute vasoocclusive episodes, typical of SCD, result in severe pain, while ulcers are associated with both acute and chronic pain. 11
Patients with SCD manifest the first SCUs in the second decade of life, 12 but the prevalence increases with age. The majority of SCUs are slow to heal and form over the medial or lateral malleoli, but occasionally, the dorsum of the foot and the digits may be affected. 13 With or without specialized care, these wounds can increase in size, become infected and reoccur. 1 In some patients, ulcers can last indefinitely, leading to extreme pain, depression, and long-term deformities. 14 Unfortunately, there is no specific and uniformly effective therapy for treating SCUs. 13 Ideally, a vulnerary agent for SCU would chelate iron in the skin preventing the cascade leading to tissue necrosis.
Systemically delivered deferoxamine (DFO) has been FDA approved since 1968 for treating hemochromatosis in sickle cell patients. 15 SCD patients receive repeated blood transfusions to increase normal hemoglobin and dilute hemoglobin S, which can lead to free iron accumulation and hemochromatosis, most critically in the liver and occasionally in the heart. 10,16 To chelate excessive iron from their blood, SCD patients receive DFO, usually in the form of Desferal (Novartis, Switzerland). 17 DFO maintains a maximal affinity for ferric iron, forming an exceptionally stable hexadentate ligand, ferrioxamine, following chelation. 18,19 The drug is either delivered subcutaneously over 8–24 h with the use of a portable infusion pump (not to exceed 20–40 mg/kg/day), intravenously over 8–12 h (20–40 mg/kg/day for children and 40–50 mg/kg/day for adults,) or intramuscularly, not exceeding a daily dose of 1,000 mg. 17 There has been no evidence of toxicity in adult or pediatric patients when treated within these dose limits, 20 but ototoxicity and retinal abnormalities are reported in higher doses (above 50 mg/kg/day).
We have previously demonstrated that DFO is an effective treatment for healing diabetic murine wounds. 21,22 However, the major challenge has been effective topical delivery of this drug into the ulcer and intact skin for prophylactic use. DFO is a hydrophilic drug with a short half-life and delivery into the hydrophobic stratum corneum by injection or direct application in either liquid or cream form has been inconsistent and not always successful. Hence, we developed a novel transdermal delivery system for deferoxamine (DFO-TDDS) that uses reverse micelles to release DFO in a sustained manner to the healing dermis. 23 We first applied DFO-TDDS on diabetic ulcers created in murine models. DFO-TDDS was effective in both preventing and accelerating wound healing in diabetic mice. 23
In this study, we hypothesize that DFO-TDDS can enhance healing of wounds in sickle cell mice through iron chelation. We first created excisional wounds in a transgenic sickle cell mouse model expressing >99% human sickle hemoglobin (HbSS-BERK) and demonstrated that wound healing is impaired in these mice. Next, we treated excisional wounds in HbSS-BERK with DFO-TDDS or subcutaneously injected DFO. We discovered that DFO-TDDS significantly accelerated healing of wounds in the HbSS-BERK mice by chelation of excessive free iron. Our findings demonstrate an effective therapeutic solution that can be rapidly translated into patients with SCUs.
Clinical Problem Addressed
There is currently no effective therapy for healing SCUs. Wound care for SCUs is labor intensive and requires multiple clinic visits on a weekly basis. Current treatment protocols include surgical debridement, which is performed to remove dead and infected tissue, and to allow granulation to occur. Compression and dressings that absorb excess exudate while maintaining a moist wound surface are also used. Despite these efforts, SCUs are slow to heal, if at all, and are prone to recurrence. 24 Antibiotics are also prescribed for wounds with obvious purulence, cellulitis, or osteomyelitis, yet there are insufficient data to support either systemic or local antibiotic therapy for SCUs. In this study, we show that clinical-grade DFO-TDDS significantly accelerates healing of excisional wounds in sickle cell mice. Since DFO is approved for systemic use in patients with SCD, we anticipate that local DFO-TDDS can be translated effectively to treat patients with SCUs.
Materials and Methods
Mice
HbSS-BERK mice do not express mouse α- and β-globins but carry the transgenes for human α- and β-sickle genes on a mixed genetic background. 25 These mice simulate human SCD, including hemolysis, reticulocytosis, anemia, extensive organ damage, shortened life span, and pain. 25 –27 Homozygous HbSS-BERK mice were bred and phenotyped for human sickle hemoglobin in Dr. Kalpna Gupta's laboratory at the University of Minnesota as previously described 27 and transported to Stanford under a material transfer agreement. Genotyping for the knockout and hemoglobin transgenes was done by Transnetyx (Cordova, TN). Control mice (wild type) on a C57Bl/6 background [#000664] were obtained from Jackson Laboratories (Bar Harbor, ME). All mice used in the experiments were housed in the Stanford University Veterinary Service Center. NIH and Stanford University animal care guidelines were followed. All procedures were approved by the university's Administrative Panel on Laboratory Animal Care.
DFO-TDDS production
Clinical-grade DFO-TDDS was manufactured, sterilized, and packaged in an FDA- and ISO-13485-compliant facility (TauTona Group, Redwood City, CA) using a formulation previously described. 23
Excisional wound healing
Five-month-old male HbSS-BERK and wild-type mice were subjected to a quantitative and reproducible model of excisional wounding using an established protocol. 28 Briefly, after induction of anesthesia and removal of hair using a shaver and depilatory cream, two 6 mm full-thickness cutaneous wounds were excised on either side of the midline of the murine dorsum using a biopsy punch (Integra, NJ). Each wound was stented with silicone rings (Grace Biolabs, OR) with outer and inner diameters of 16 and 10 mm, and sutured in place to prevent wound contraction. All mice were given buprenorphine slow-release at the time of wounding as analgesic.
Wound treatment
HbSS-BERK mice were randomized into three treatment groups: DFO-TDDS, DFO injection, or untreated. Following wounding, a 6 mm punch of DFO-TDDS was placed on the wound daily or 20 μL of 100 mM DFO solution was subcutaneously injected into the wound daily. The untreated group received no injection or patch. All wounds were covered with an occlusive dressing (Tegaderm, 3M; St Paul, MN). Digital photographs were taken every other day. Wound area was measured using ImageJ software (NIH).
Histology
On closure, wounds were collected, fixed in 4% paraformaldehyde overnight, and embedded in paraffin. For analysis of dermal thickness, paraffin sections were stained with trichrome (Sigma-Aldrich) and average thickness was calculated from three measurements per high-power field per wound.
Perl's Prussian blue stain
Abcam iron stain kit (ab150674; Cambridge, UK) was used to display iron present in tissue sections. Histological sections were deparaffinized and rehydrated. Equal volumes of potassium ferrocyanide and hydrochloric acid solution (2%) were combined to make the iron stain solution. Slides were incubated in the solution for 3 min and then rinsed with distilled water. Slides were then stained with Abcam nuclear fast red solution for 5 min and washed four separate times with distilled water. Slides were finally dehydrated in 95% ethanol, followed by absolute ethanol. Blue stain directly correlates with nonchelated iron in the skin. DFO chelates iron, forming ferrioxamine, which does not react in the Perl's Prussian blue reaction.
Statistical analysis
Results are presented as mean ± SEM. Standard data analysis was performed using a Student's t-test. ANOVA was used to measure significance of wound closure. Results were considered significant for *p ≤ 0.05, **p < 0.01, and ***p < 0.001.
Results
HbSS-BERK mice undergo wound healing impairments compared with wild-type mice
HbSS-BERK and wild-type mice were splinted following wounding to minimize contracture and to replicate human-like wound healing kinetics. 28 Images of the excisional wounds were taken every other day and the wound healing outcomes were assessed by comparing wound sizes at each time point to day 0 within each mouse (Fig. 1A). HbSS-BERK mice demonstrated markedly delayed wound healing compared with wild-type control mice. Differences in the wound area were statistically significant at all time points from day 6 onward until closure (*p < 0.05, **p < 0.01, ***p < 0.001) (Fig. 1B). Time to complete wound closure in the HbSS-BERK mice and wild-type mice was 17.1429 ± 0.4041 and 13.4 ± 0.3055, respectively (Fig. 1C). These results indicate that the HbSS-BERK mice exhibit delayed wound healing.
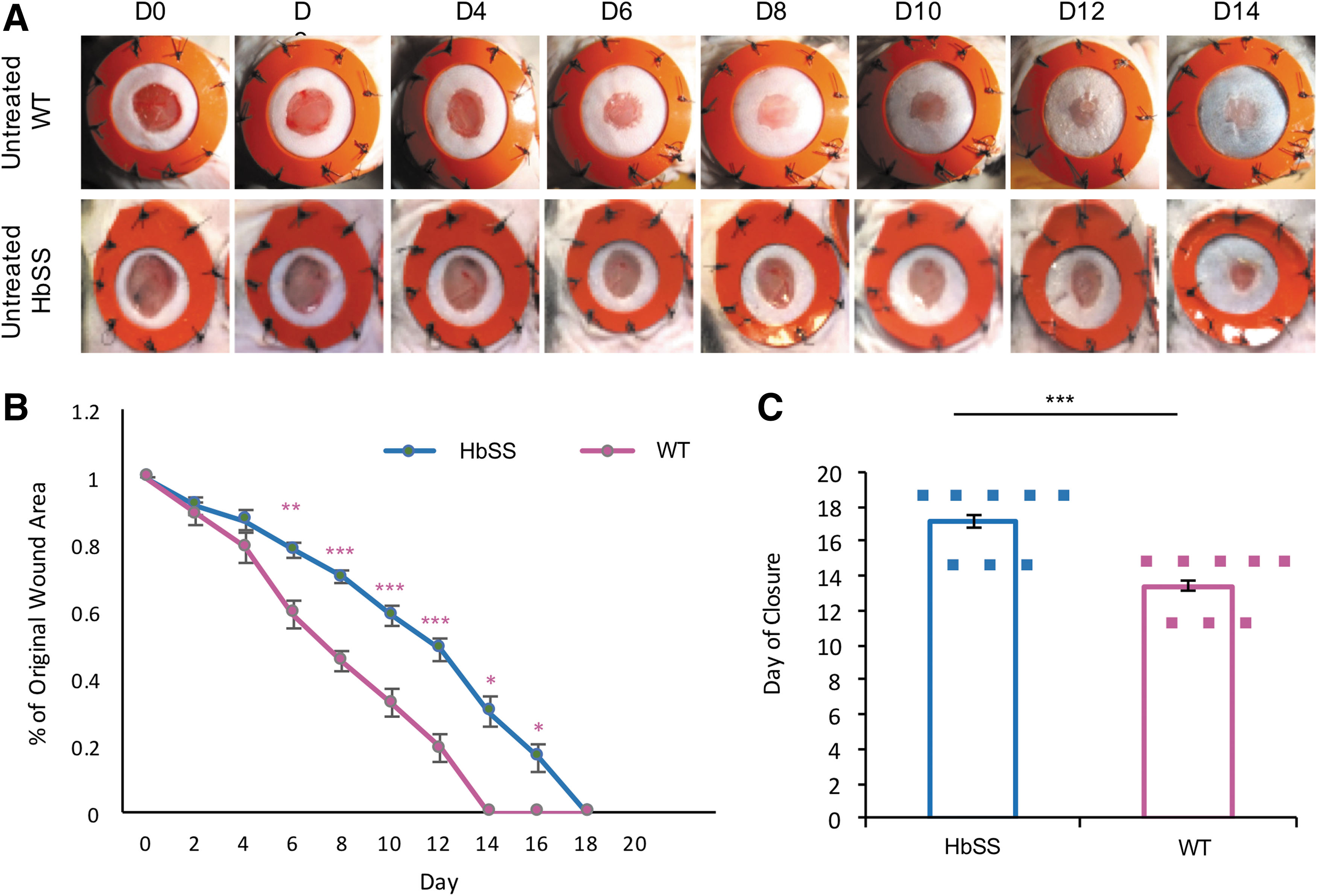
HbSS-BERK mice demonstrate wound healing impairments compared with wild-type mice.
HbSS-BERK wounds treated with DFO-TDDS demonstrate accelerated wound healing
Once we established that HbSS-BERK mice have impaired wound healing, we treated these mice with DFO-TDDS or injected the wounds subcutaneously with DFO solution daily. Wound healing in the treated mice was compared with wounds in HbSS-BERK mice that were left untreated. DFO-TDDS-treated mice displayed significantly accelerated wound closure compared with both the DFO injection (*p < 0.05) and the untreated group (***p < 0.001) (Fig. 2A, B). Injection with DFO solution significantly reduced wound area compared with untreated controls (**p < 0.01), but not as effectively as the DFO-TDDS group. Time to complete wound closure in the DFO-TDDS group, DFO injection group, and untreated groups was 13 ± 0.3660, 14.5 ± 0.6268, and 17.1429 ± 0.4041, respectively. These results demonstrate that DFO-TDDS significantly accelerates wound healing and is more effective than DFO injection at treating wounds in a murine model of sickle cell ulceration.
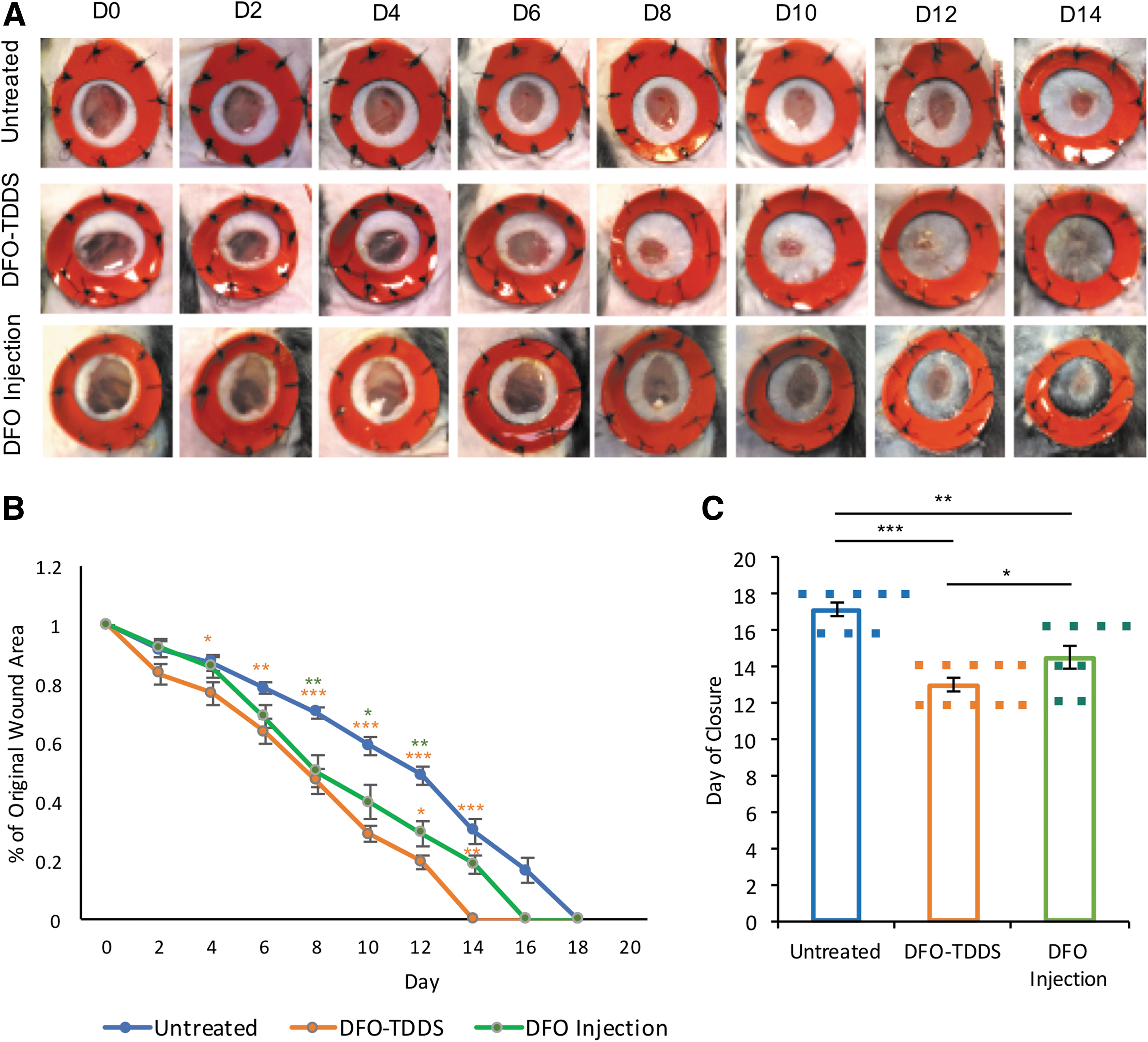
DFO-TDDS accelerates wound healing and is more effective than injected DFO at treating wounds in HbSS-BERK mice.
HbSS-BERK wounds treated with DFO-TDDS demonstrate a thicker dermis
Histological sections of the healed wound were subjected to trichrome analysis to determine collagen deposition in the dermis. DFO-TDDS-treated wounds in the HbSS-BERK mice displayed markedly greater collagen deposition in organized bundles compared with the untreated group and the DFO injection-treated mice (Fig. 3A). The width of collagen across the slides was measured to determine thickness of the dermis. Wounds treated with DFO-TDDS demonstrated significantly higher dermal thickness (*p < 0.001) (Fig. 3B). Greater collagen deposition and a higher dermal thickness are desirable in the healed skin of sickle cell patients, to prevent a wound recurrence at the same site.
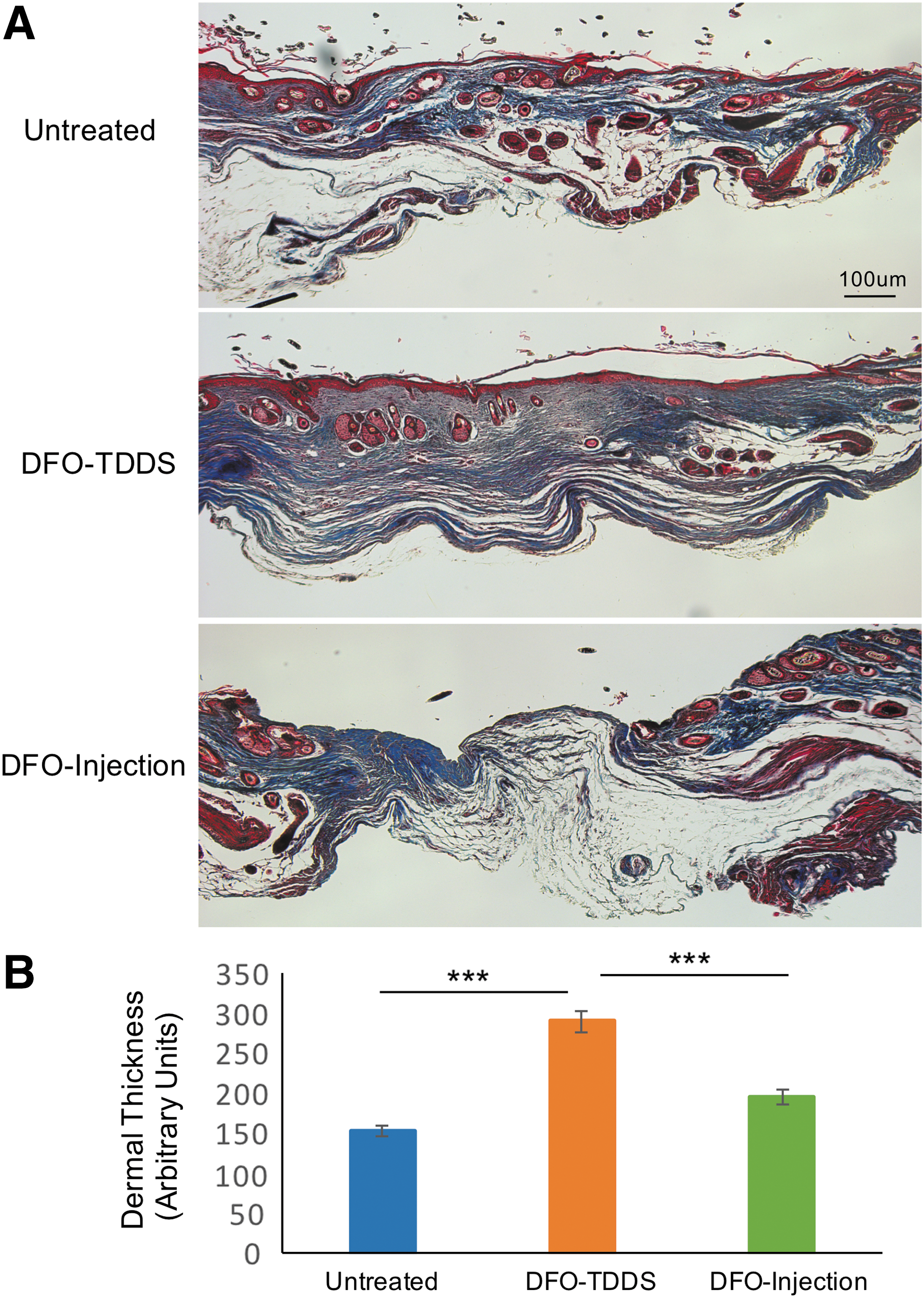
Wounds in HbSS-BERK mice treated with DFO-TDDS display increased collagen deposition and dermal thickness.
DFO-TDDS accelerates wound healing in HbSS-BERK mice by chelation of free iron
Since SCD is characterized by excessive free iron leading to tissue dysfunction, histological sections of the healed wound were subjected to Perl's Prussian blue stain to determine the presence of iron in untreated wounds and wounds treated with DFO. Excessive deposition of iron was observed in the untreated group, and these wound regions highly correlated with lesser dermal thickness and reduced dermal integrity (Fig. 4A). Both DFO-TDDS and DFO injection decreased iron in the skin as evidenced by negligible levels of Perl's Prussian blue stain (Fig. 4A, B). However, the DFO-TDDS treatment group demonstrated a well remodeled wound without excessive cell proliferation, uniformly bundled extracellular matrix, and the return of skin appendages. DFO-TDDS-treated wounds also displayed a thick dermis (more than 500 μm thick), which was not observed in the untreated mice. Interestingly, the DFO injection group showed regions of active cell proliferation and the presence of disorganized extracellular matrix, indicating the wound had not resolved healing in this treatment group. Thus, sustained release of DFO through the TDDS is more effective in healing wounds in sickle cell mice.
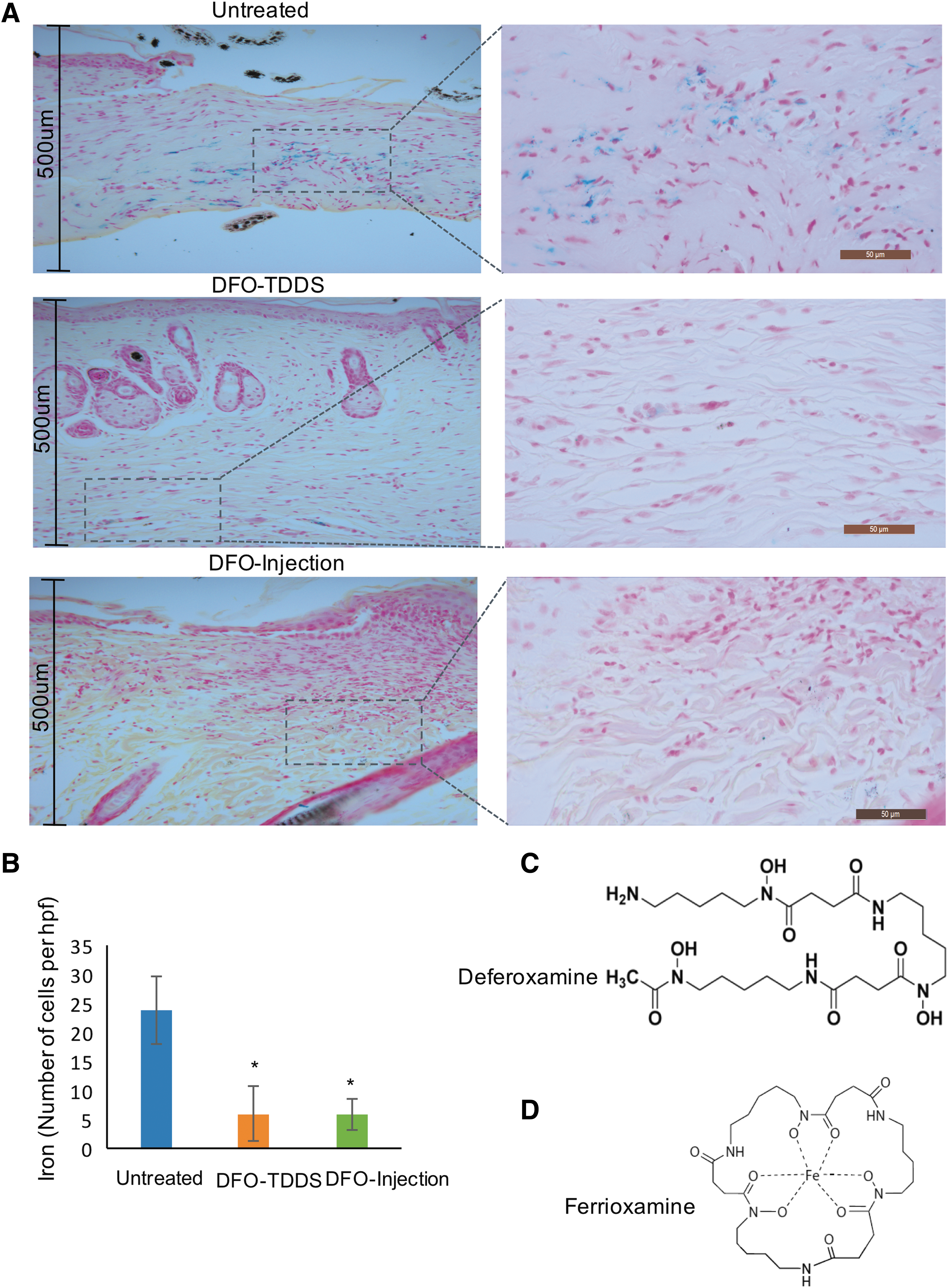
Wounds in HbSS-BERK mice treated with DFO-TDDS display reduced iron by Perl's Prussian blue stain.
Next, to understand why there was reduced Prussian blue stain in the DFO treatment and DFO injection groups, we aimed to understand the chemical reaction by which DFO binds iron. Iron is stable when bound to six oxygen atoms. Several chelators can provide these oxygen atoms, but DFO (Fig. 4C) is known to form the strongest bonds with iron, since it is a hexadentate ligand, sharing six oxygen atoms with Fe3+ to form ferrioxamine (Fig. 4D). Ferrioxamine is difficult to dissociate, even in dilute solutions (10−5M) and thus withholds ferric iron from being available for the Perl's reaction. Thus, our results indicate that in the presence of DFO-TDDS, free iron in sickle cell wounds is chelated and no longer available to generate ROS and cellular damage.
Discussion
Hemoglobin S is formed by a substitution of valine for glutamic acid (GAG→GTG) at position 6 in the β-globin chain of hemoglobin A. 4 The inheritance of two copies of this mutation (HbSS), one from each parent leads to homozygous SCD. HbSS polymerizes on deoxygenation, forming rigid sickle-shaped erythrocytes. These erythrocytes impair blood flow and readily lyse leading to an accumulation of excessive free iron in the plasma and in tissues. 4 The complications of SCD are myriad, but the most common acute events during childhood are pain, stroke, acute chest syndrome, and splenic sequestration. 29,30 As the patient advances into adulthood, progressive vasculopathy and hemochromatosis lead to end-organ damage, including chronic renal failure, stroke, avascular necrosis of bone, and pulmonary hypertension. 31 Patients with SCD often develop SCUs, affecting physical function and the quality of life of the patient. 24,32
DFO is a highly effective and nontoxic iron chelator that has been routinely used to remove excessive iron from patients with hemochromatosis. 33,34 Due to its short half-life, it has been administered by subcutaneous or intravenous infusion, usually over 8–12 h and for 5–7 days/week. 17,35 In this study, we hypothesized that local delivery of DFO to the wound site would improve wound healing in mice with SCD by chelating excessive free iron in the wound. However, the DFO molecule is hydrophilic and relatively large, making cellular diffusion difficult. 19
To enable local delivery to both wounds and intact skin, we used a novel DFO-TDDS that contains DFO encapsulated in reverse micelles. 23 When applied topically, it allows for DFO to easily diffuse through the impermeable stratum corneum and hydrophobic membranes, with targeted delivery into the dermis. 23 We have previously demonstrated that DFO-TDDS improves wound healing in mice by enhancing new blood vessel formation and reducing production of ROS. 23
In this study, we were specifically interested in determining if DFO-TDDS could chelate iron in the skin of sickle cell mice and enhance wound healing. HbSS-BERK sickle cell mice were used for experiments. These mice contain >99% human sickle hemoglobin 25 and display similar tissue impairments as evidenced in patients with SCD, including heme-induced vasoocclusion, 36 decreased epidermal and dermal thickness, 27 hyperalgesia, 27 and mechanical allodynia. 37 In our experiments, HbSS-BERK mice demonstrated slower wound healing compared with wild-type mice. Healed wounds in untreated HbSS-BERK mice displayed increased iron in the dermis by Perl's Prussian blue stain. Regions of high iron accumulation directly correlated with reduced dermal thickness (Fig. 5).
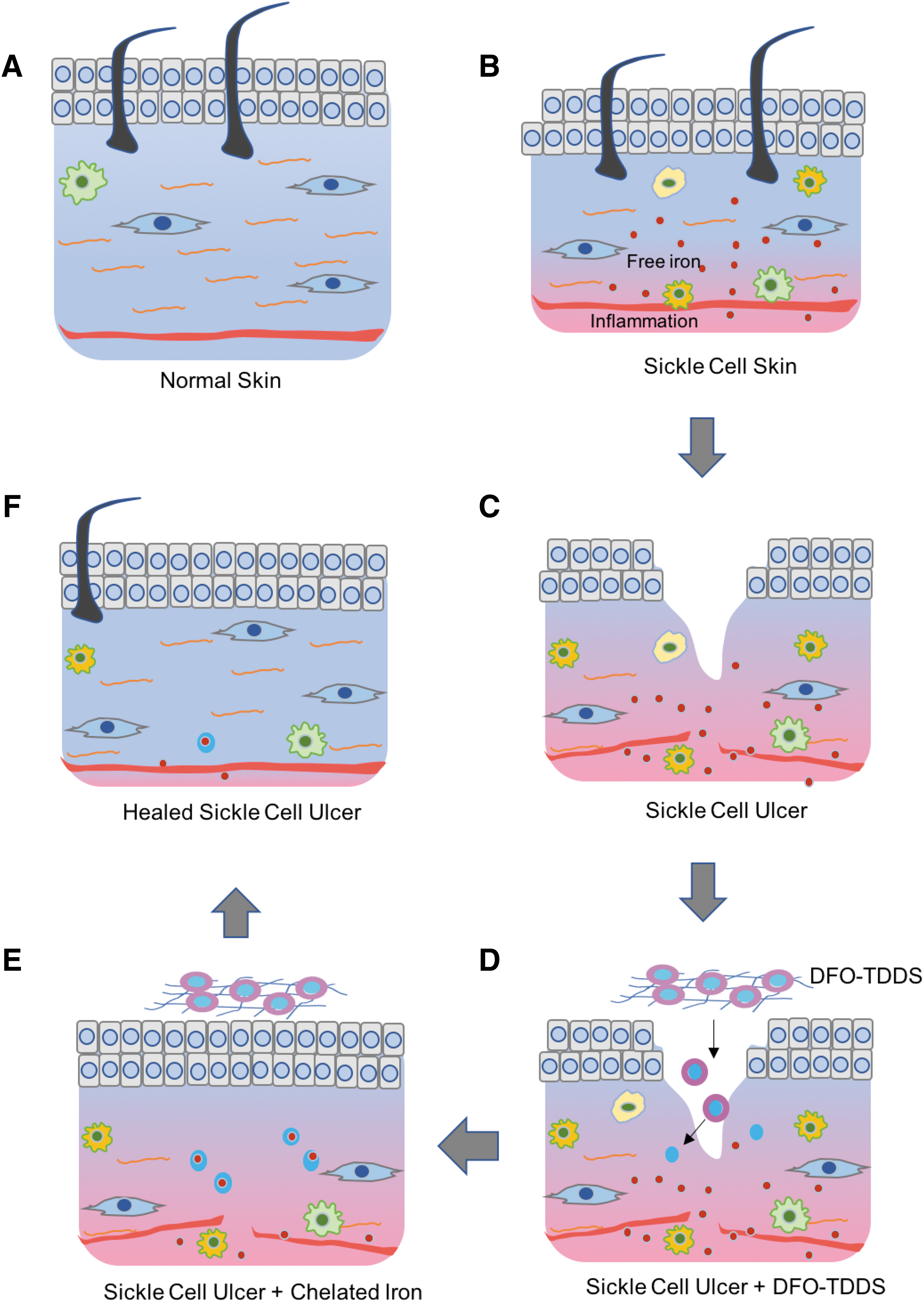
Schematic demonstration of the effectiveness of DFO-TTDS in healing sickle cell ulcers.
Daily application of DFO-TDDS on wounds significantly accelerated wound closure, increased dermal thickness, and resulted in deposition of organized collagen bundles compared with both untreated wounds and wounds injected with DFO solution daily. All mice were given buprenorphine slow-release as an analgesic during wounding to counter pain, which is a characteristic feature of the HbSS-BERK mice. 26,27,38 DFO-TDDS-treated wounds displayed negligible iron in the dermis (Fig. 5), indicating iron chelation by the formation of ferrioxamine. While the DFO injection-treated wounds also displayed negligible iron in the dermis, the wounds in this treatment group were still in the proliferative state with disorganized extracellular matrix, indicating that a single dose of DFO given once a day is not as effective as sustained release of DFO throughout the wound healing process.
Ferric iron (Fe3+) is much more stable under aerobic conditions in relation to ferrous iron (Fe2+) and is of greater significance when assessing the value of different chelators. 18 The positive charge of ferric iron creates a particularly high charge density within the atom, thereby predisposing it to forming bonds with other atoms that possess high charge densities. 18 Chelators such as DFO have regions of polarization that serve as strong binding points for highly charged ferric cations. As DFO has six binding sites with ferric iron, it is labeled a hexadentate ligand. 18 Hexadentate ligands exhibit greater binding strength at lower concentrations than others, such as bidentate ligands, and thus are less likely to dissociate and form hydroxyl radicals. 18 These chemical properties make DFO a highly efficient drug to chelate excessive ferric iron in the skin. Once chelated, ferrioxamine most likely is excreted via exfoliation of epidermal cells, through sweat, or might enter the blood stream and is excreted via the kidney. 39,40
Our results indicate for the first time that removal of iron from the dermis of HbSS sickle cell mice directly correlates with improvement of wound healing. Reducing iron locally in the skin using an FDA-approved chelator, DFO significantly enhances time to wound closure, dermal thickness, and wound remodeling. Sustained delivery of DFO through the DFO-TDDS is more effective in closing wounds compared with a single bolus of DFO injected locally. DFO-TDDS is currently being manufactured, sterilized, and packaged under good manufacturing practice in an FDA-compliant facility. This will allow for rapid translation for treatment of patients with SCUs.
Innovation
SCUs are a significant health care burden. There is no effective therapy for their prevention or treatment. Beyond standard debridement and ensuring that the wound remains clean, few solutions exist. To address SCUs, we have developed DFO-TDDS, which enables DFO to be released into both the intact skin and wounds. In this study, we establish the efficacy of DFO-TDDS for treating SCUs where no alternative strategy currently exists. The TDDS also introduces a technology through which other small-molecule drugs can be delivered into the skin for treating wounds in other impaired states. DFO-TDDS has been scaled for human application in an FDA-compliant facility.
We demonstrate for the first time that the HbSS-BERK murine model of SCD exhibits impaired wound healing
DFO-TDDS significantly accelerates healing of wounds in HbSS-BERK mice
DFO-TDDS is more effective than subcutaneous injection of DFO at treating wounds in HbSS-BERK mice
DFO-TDDS accelerates wound healing in sickle cell mice by chelation of excessive free iron
Footnotes
Acknowledgments and Funding Sources
We thank Joe Rimsa and his staff at TauTona Group for manufacture and delivery of the DFO-TDDS, Yujin Park for her assistance in tissue processing, Ritu Jha for breeding and phenotyping mice, and Theresa Carlomagno and Barb Benson for administrative support.
About the Authors
Author Disclosure and Ghostwriting
G.C.G. and M.T.L. have filed patents on this work. The article was written by the authors and ghostwriting services were not used.
