Abstract
Background:
Polycystic ovary syndrome (PCOS) is an endocrine disorder frequently characterized by hyperandrogenism that presents acute and chronic health risks. International guidelines for the assessment of PCOS endorse Rotterdam criteria for diagnosis, necessitating thorough clinical and laboratory evaluation. Heterogenous PCOS phenotypes, multiple providers, and subjective visual assessment of PCOS-related symptoms further challenge diagnostic practices. This study describes the patient population with hyperandrogenism and evaluates if Rotterdam criteria are useful to clinicians.
Materials and Methods:
A retrospective population-based study of electronic medical records retrieved from January 1, 2019, through December 31, 2023, of female patients aged 18–99 years with a diagnosis of PCOS or evidence of clinical hyperandrogenism seen at a single academic medical center was evaluated. Demographic characteristics, clinical presentation, and selected laboratory values retrieved within 90 days of diagnosis were compared between groups.
Results:
A population of 2,278 patients were evaluated, out of which 41.4% received a diagnosis of PCOS or presented with hyperandrogenism (58.6%). PCOS patients were mostly found to be White, obese, and aged 18–39 years. Diagnostic evidence of hyperandrogenism was limited; elevated free testosterone and clinical hyperandrogenism were reported in 8.1% and 7.8% of PCOS patients, respectively. Comorbidities including hypertension (24.23%), hyperlipidemia (11.72%), and type 2 diabetes (9.79%) were most prevalent among the sample population.
Conclusions:
Consistent with prior research, our results demonstrate gaps in diagnostic practices and support the need for future studies that evaluate barriers to diagnosis in clinical practice and increase education to support patient’s involvement in the diagnostic practices.
Introduction
Polycystic ovary syndrome (PCOS) is an endocrine disorder that affects up to 20% of reproductive-aged women. 1 Patients with PCOS have increased risk for chronic diseases including diabetes, 2 cardiovascular disease, 3 and mental health disorders. 4 The pathogenesis of PCOS is unclear but likely includes genetic and environmental factors.5,6 Recent estimates reveal annual U.S. health care costs related to PCOS exceed 15 billion. 7 Diagnostic criteria have evolved since guidelines were first introduced in 1990 and, currently, identification of PCOS requires thorough clinical evaluation, laboratory testing, and elimination of other alternative conditions. The 2018 International evidence-based guidelines for PCOS assessment and management 8 endorse the Rotterdam criteria, which relies on the presence of two of the three following conditions: (1) clinical or biochemical hyperandrogenism, (2) oligo-anovulation, and (3) polycystic ovarian morphology (PCOM) on ultrasound imaging.9,10 The 2023 updated guidelines expanded diagnostic criteria to include antimullerian hormone or PCOM in adults but continue to endorse the Rotterdam criteria. 11 However, it is unclear whether these criteria are useful to clinicians, which may explain, in part, visits to multiple providers and delays in diagnosis.9,10,12,13 It is also possible that diagnosis of PCOS is underreported due, in part, to multiple iterations, limitations and ambiguity of diagnostic criteria, 14 heterogeneity of the syndrome, and exacerbation of PCOS presentation from excess weight. 15
The Rotterdam criteria, the current and most widely accepted PCOS diagnostic criteria, broadened the presentation and emphasized the overlap between the syndrome and hyperandrogenism. At present, four phenotypes of PCOS exist, three requiring evidence of hyperandrogenism.9,10 Biochemical evidence of hyperandrogenism is defined by increased total testosterone (TT) or free testosterone (FT) or calculated free androgen index, whereas dehydroepiandrosterone sulfate (DHEAS) or androstenedione (A4) is also a consideration. 16 However, due to differences in assay methodologies and unclear cutoffs, hyperandrogenism should be evaluated largely based on clinical findings. 17 Hirsutism is the most frequently observed demonstration of hyperandrogenism in PCOS, affecting approximately 65%–75% of the population 18 followed by acne and alopecia. Despite the subjective evaluation for hirsutism, diagnosis is critical, as researchers recently proposed that idiopathic hirsutism, a mild androgen excess disorder, is an early stage of PCOS. 19 Unfortunately, a recent survey among North American providers unveiled limitations in diagnostic knowledge and variation in practices, 20 potentially hindering efficient diagnostic practices and delaying management strategies.
The diagnosis of PCOS is accompanied by physical and mental health risks and requires timely consideration. Management practices are symptom-based, but lifestyle behavior modification can support overall health and is recommended for all women with PCOS. Thus, gaps in diagnostic practices can delay timely diagnoses and impede early management of PCOS and related comorbidities. As the clinical differences between PCOS and hyperandrogenism can be subtle, 21 it is the primary goal of this study to describe the population affected by androgen disorders and to better understand the diagnostic practices that clinicians follow in determining these conditions. To achieve this goal, we evaluated the patient population diagnosed with PCOS and assessed if the metrics in the Rotterdam criteria were followed. In addition, we categorized the clinical characteristics, including comorbidities, between patients with a diagnosis of clinical hyperandrogenism and PCOS. The results from this study help elucidate whether diagnoses of clinical hyperandrogenism are associated with findings necessitating medical follow-up after initial diagnosis.
Methods
A retrospective study was completed using electronic medical records (EMR) retrieved from January 1, 2019, to December 31, 2023, of female patients aged 18–99 years with PCOS International classification of diseases [(ICD)-10-CM E28.2)] or hyperandrogenization syndrome. According to the U.S. Census Bureau, the county demographic distribution served by the single academic medical center includes 79.5% White, 13.5% Black or African American, 3.7% Asian, 0.8% American Indian and Alaska Native, and 0.1% Native Hawaiian and other Pacific Islander. 22 Biochemical evidence of hyperandrogenism was defined by increased total T (>60 ng/dL) or FT (>1.0 ng/dL).23,24 Classification of clinical hyperandrogenism was defined as the presence of hirsutism, acne, or alopecia in the patients’ medical record. For each case, the following criteria were collected from the EMR: demographic characteristics, evaluation of self-reported menstrual cycle, ultrasound, comorbidities, signs and symptoms of clinical hyperandrogenism, and screening, metabolic, lipid, and biochemical hyperandrogenism laboratory tests. All laboratory values reported were retrieved within 90 days of diagnosis with PCOS or hyperandrogenism. If multiple labs were available within this timeframe, values closest to the diagnosis date were selected. Medications that can stimulate androgen excess and were prescribed up to 1 year before diagnosis were reported. Conditions of exclusion, via respective laboratory tests, including thyroid disease (thyroid stimulating hormone), hyperprolactinemia (prolactin), Cushing disease (cortisol), and androgen secreting tumors (DHEAS), were assessed up to 1 year before diagnosis. All associated diseases were recorded using ICD-10 codes, and all laboratory values were retrieved using current procedural terminology codes. Provider characteristics including type of ordering provider and department were also collected. University Institutional Review Board approval was obtained for this retrospective chart review.
Recommendations established by the Rotterdam criteria and currently promoted by the 2018 International evidence-based PCOS guidelines, as well as current literature, were used to determine the diagnostic criteria.9,10
Data Analysis
Descriptive statistics were used to assess the patient population with PCOS or hyperandrogenism, and demographic variables were compared between groups using pairwise Student’s t test for continuous variables and using Fisher’s exact test for binary and categorical variables. A comparison of the PCOS and the hyperandrogenism groups was completed to assess if comorbidities and laboratory values differ across groups. Menopause, a natural and transitional phase that occurs 1 year after the final menstrual cycle, is accompanied by low levels of estrogen that increases a woman’s cardiovascular health risks. 25 Given the average age of menopause is 51 years, 26 data for PCOS and hyperandrogenism groups were further analyzed and compared among participants aged ≤50 years. For these, continuous data were compared using the Mann–Whitney test, and frequency comparison was made using Fisher’s exact test. Findings were considered statistically significant at p value <0.001. To analyze the probability of PCOS patients developing chronic inflammatory disease, congestive heart failure, peripheral vascular disease, type 2 diabetes, hyperlipidemia, endometrial cancer, and hypertension were analyzed by assessing the relative risk (RR) in which the RR allowed for the association of PCOS with disease. A RR of 1.0 demonstrated no effect, a RR of <1 indicated reduced risk, and a RR >1 indicated increased risk.
Results
A total of 2,278 women aged 18–98 years were diagnosed with PCOS (n = 943; 41.4%) or presented with clinical hyperandrogenism, without a diagnosis of PCOS (n = 1,335; 58.6%), at a single academic medical center between 2019 and 2023. Demographic characteristics of individuals diagnosed with PCOS or hyperandrogenism are summarized in Figure 1. The majority of the patients self-reported as Caucasian/White of not Hispanic or Latino descent (Fig. 1A and 1B). Among the PCOS group, the majority (86.2%) of diagnoses were made in patients aged 18–39 years; however, among the hyperandrogenism group, diagnosis was evenly distributed across age groups (Fig. 1C). Most PCOS (91%) and hyperandrogenism (67.5%) patients presented with a body mass index (BMI) >25.0, which classifies them as overweight or obese (Fig. 1D). This sample population was evaluated primarily by providers in family medicine (29.9%), dermatology (24.8%), and obstetrics and gynecology (22.8%). Analyses of diagnostic practices by hospital specialty (Fig. 2) revealed that the majority of PCOS encounters were conducted by Obstetrics and Gynecology (N = 347) followed by Family Medicine Practice (N = 309). However, the clinical features of hyperandrogenism were primarily reported by different specialties; hirsutism was chiefly reported by Obstetrics and Gynecology (N = 123), acne was most frequently recounted by Family Medicine (N = 292), and alopecia was mainly described by Dermatology (N = 434).
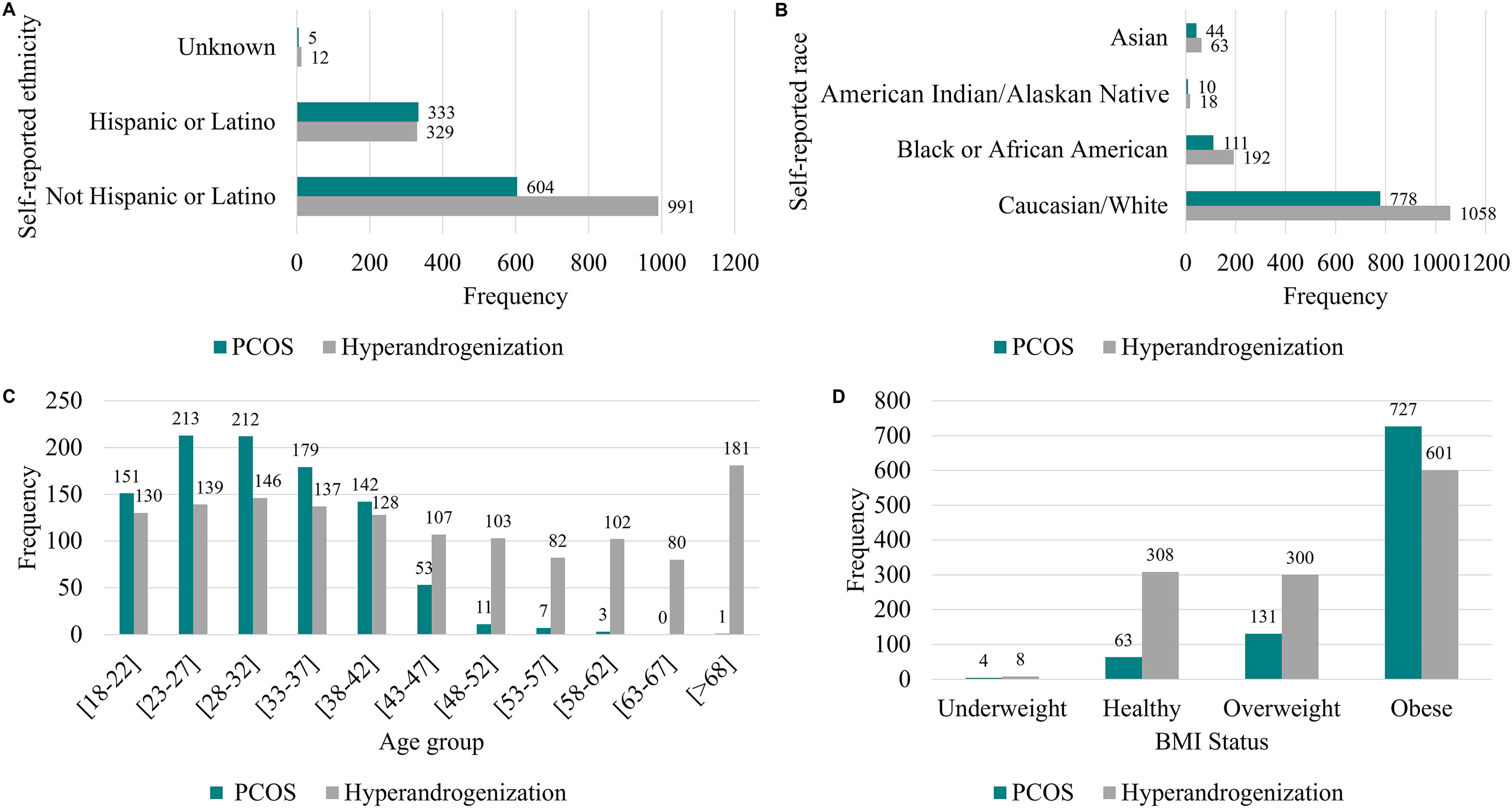
Demographic characteristics of patients diagnosed with PCOS and/or hyperandrogenism.

Distribution of diagnoses according to medical specialty in which patients were treated.
Evidence of clinical hyperandrogenism was present in up to 21.6% of the studied population; yet, the use of drug-induced hyperandrogenism was discarded as prescriptions of anadrol, valproic acid, cyclosporine, erythropoietin, psoralens, minoxidil, and streptomycin were rarely reported. The patient population was classified following the Rotterdam criteria, in which PCOS is a diagnosis of exclusion and requires physical and laboratory evaluation (Table 1). Evidence of biochemical hyperandrogenism was defined based on the elevated TT and FT values, 23 and the majority of biochemical features were documented in patients ≤50 years. TT testing was only completed in 92 patients out of the total 2,278 studied population, whereas FT testing was completed in 171 patients. Only 13 patients out of the total 943 PCOS-diagnosed group presented with elevated T, whereas only 108 patients presented with elevated FT. The presence of clinical features was primarily documented in the premenopausal-aged group including hyperandrogenism (hirsutism, acne, or alopecia), which was only reported in 99 PCOS patients aged ≤50 years. Oligo-ovulation, or evidence of menstrual irregularity, was self-reported in 37.43% of the PCOS group (15.5% of the total population). While pelvic and transvaginal imaging were completed for 17.92% and 21.10% of the PCOS population, respectively (7.4% and 8.7% of the total population, respectively), findings are not available to confirm PCOS.
Categorization of Clinical and Biochemical Features of Studied Patient Population and by Diagnosis of Clinical Hyperandrogenism or PCOS, According to the Rotterdam Criteria (Total Population, N = 2,278)
Elevated free and total testosterone cutoff value established based on reference range upper limit (23). Irregular menses established as self-reported by patient. Patients with hyperandrogenism included women who presented with hirsutism, acne, or alopecia.
Percentage calculated out of patients diagnosed with PCOS (N = 943).
Percentage calculated out of patients with PCOS who are ≤50 years old (N = 926).
Percentage calculated out of patients with clinical hyperandrogenism without PCOS (N = 1,335).
Percentage calculated out of patients with clinical hyperandrogenism without PCOS that are ≤50 years old (N = 855).
Percentage calculated out of total population (N = 2,278).
U/S = ultrasound.
PCOS, polycystic ovary syndrome.
Among the entire study population, the PCOS group was significantly younger and had a higher BMI than the hyperandrogenism group (p < 0.001 for both; Table 2). Diagnostic criteria for biochemical hyperandrogenism were limited; specifically, FT (p < 0.001) was only performed in the PCOS group ≤50 years and was significantly higher than the hyperandrogenism group ≤50 years. Laboratory values obtained within 90 days of PCOS diagnosis or presence of clinical hyperandrogenization demonstrated the PCOS group ≤50 years had a significantly higher alanine aminotransferase (ALT) than the comparison hyperandrogenism group. In addition, within the premenopausal-aged PCOS group, hyperlipidemia was present. For example, triglycerides and very low-density lipoprotein (VLDL) among the PCOS group ≤50 years were above normal and significantly higher, whereas high-density lipoprotein (HDL) was undesirable and significantly lower than the comparison hyperandrogenism group. Average cholesterol for both groups was similar and considered in the normal range; however, low-density lipoprotein (LDL) was comparable but not optimal in either group. Among the entire study population, HbA1c was considered normal for the PCOS group (<5.7%) and significantly lower than the 5.72% ± 1.07% observed in the hyperandrogenism group. However, further analyses revealed a comparable mean HbA1c <5.7% for both PCOS and hyperandrogenism groups ≤50 years. Comorbidities including hypertension (24.23%), hyperlipidemia (11.72%), and type 2 diabetes (9.79%) were most prevalent among this sample population. Clinical features of acne, alopecia, and hirsutism did not have a correlation with the presence of PCOS as defined by their risk ratios. RR for the development of chronic diseases was compared between groups and identified only one significant risk difference. Compared with the hyperandrogenism group, the PCOS group had an increased RR (1.46, 95% confidence interval 0.09–22.67) for the development of chronic inflammatory disease.
Clinical and Biochemical Characteristics of Female Patient Population with PCOS and Clinical Hyperandrogenism (N = 2,278)
Laboratory values obtained within 90 days of diagnosis with PCOS or hyperandrogenization syndrome, which included the presence of clinical hyperandrogenism features hirsutism, acne, or alopecia.
p value comparing PCOS group to clinical hyperandrogenism without PCOS group.
p value comparing PCOS group ≤50 years old to clinical hyperandrogenism without PCOS group ≤50 years old.
BMI, body mass index; DHEAS, dehydroepiandrosterone sulfate; PCOS, polycystic ovary syndrome; SD, standard deviation; ALP, Alkaline phosphatase; BUN, Blood urea nitrogen; ESR, Erythrocyte sedimentation rate; SGOT, Glutamic-oxaloacetic transaminase; TSH, Thyroid stimulating hormone.
Discussion
PCOS is observed in up to 20% of reproductive-aged women and is associated with a myriad of conditions including increased chronic disease risk2–4 ; therefore, timely diagnosis is essential. International evidence-based guidelines support the Rotterdam criteria for diagnosis, which entails thorough clinical evaluation, laboratory testing, and elimination of other alternative conditions.9,10 To further compound the challenge, four phenotypes of PCOS exist based on the presentation of all three Rotterdam criteria (clinical or biochemical hyperandrogenism, oligo-anovulation, and PCOM on ultrasound imaging) or the combination of any two criteria.9,10 Metabolic risks differ between phenotypes as evidenced by the reduced prevalence of hyperandrogenism, insulin resistance, and abnormal lipids in the one nonhyperandrogenic PCOS phenotype. Conversely, the most severe phenotype, containing all three diagnostic criteria, presents the highest metabolic risk. 27 Moreover, excess body weight exacerbates the presentation, 15 and laboratory diagnostic criteria are contentious. 14 It is not surprising that physicians report discrepancies in knowledge and diagnostic practices for PCOS 20 In this retrospective chart review of patients with a diagnosis of PCOS or presentation of clinical hyperandrogenization at a single academic medical center between 2018 and 2023, we observed that most patients are diagnosed with PCOS between ages 18 and 39 years, with the majority of patients presenting a BMI >30. This is consistent with other reports describing PCOS impacting women of reproductive age with high BMI. 28
Despite biochemical hyperandrogenism being one of the criteria for PCOS diagnosis, TT and FT testing were only evaluated in a small percentage of the population studied. This would suggest that greater emphasis is given to the presence of clinical hyperandrogenism; however, only 11% (104 out of 943 patients) of PCOS individuals presented with hirsutism, although it is understood that visual assessment of hirsutism is subjective and cutoff values for diagnosis vary by ethnicity.29,30 Self-assessment of hirsutism may present an opportunity for increased PCOS evaluation, 31 although self-ratings are higher than clinician ratings, 32 as women appear bothered by excess hair growth. 33 Clinical presentation of acne may be overlooked, as a global standard visual assessment tool is not available and specific acne criteria are not described in the international guidelines.8,34 Documentation of clinical features of hyperandrogenism by different hospital specialties in this study reveals added diagnostic challenges. The presence of oligo-anovulation is a limited feature, as it is influenced by reproductive age and oral contraceptives; however, more than one-third of PCOS patients self-reported menstrual irregularity. Ultrasound imaging may have been ordered more frequently, but it is unknown whether imaging for PCOM led to the evidence of PCOS. In addition, we must acknowledge the heterogeneity of PCOS-related symptoms may prompt visits to clinicians inexperienced with PCOS, further challenging diagnostic opportunities. Gaps in diagnostic practices and the subsequent need for increased physician education related to the Rotterdam criteria have been previously reported. 20 Hence, the Rotterdam criteria can have limitations in clinical practice, as only a small percentage of the population was evaluated according to these guidelines.
Laboratory values that were found to be statistically significantly higher in PCOS patients, including the PCOS group ≤50 years, included ALT, FT, triglycerides, and VLDL. Elevated FT in PCOS individuals was expected, as it is a hallmark reported in literature. 16 While ALT was elevated, it is still within the normal reference range, so its clinical utility to assess liver disease is limited at the time of PCOS diagnosis but may be an important marker to continue monitoring as patients age. Metabolic dysfunction-associated steatotic liver disease, formerly named nonalcoholic fatty liver, a condition frequently identified through elevated liver function tests, is associated with hyperandrogenism and more prevalent among women with PCOS. 35 Interestingly, the mean VLDL and HDL values were significantly higher and lower, respectively, in patients with PCOS, including the PCOS group ≤50 years, supporting evidence of dyslipidemia, a modifiable risk factor for cardiovascular disease. A large, retrospective observational case–control study in Canada reported an increased prevalence for adverse health outcomes among women with PCOS across the lifespan including the development of obesity, dyslipidemia, type 2 diabetes, and cardiovascular disease compared with age-matched controls. 36 Although our study findings of decreased frequency for comorbidities hypertension, hyperlipidemia, and type 2 diabetes among the PCOS group are perplexing, it may be a reflection of the age (young), medication use, or short follow-up period postdiagnosis. However, we cannot ignore the fact that PCOS is an underrecognized condition, and it is possible that individuals in the hyperandrogenism group had PCOS but had not received a diagnosis. There is also evidence to support an increased prevalence of cardiovascular risk factors among premenopausal women with hyperandrogenism and independent of BMI 37 suggesting diagnosis of androgen disorders in women necessitates medical follow-up after initial diagnosis.
Limitations
The analyses of our retrospective chart review of patients with a diagnosis of PCOS and clinical presentation of hyperandrogenization are limited to the information obtained from one medical institution and curtail the evaluation of missed or delayed diagnoses in the broader population. Although the race and ethnicity of participants seen at this academic medical center reflect the county demographics, 22 it is plausible that additional diagnostic labs were completed elsewhere, impeding analysis. Lab tests may have also been performed outside the 90-day window of diagnosis, potentially underestimating the frequency of testing. In addition, medication history, including contraceptive use, was not available, limiting definitive conclusions. Because analyses were limited to 5 years, follow-up time may be inadequate for disease progression and development of comorbidities. Although PCOS diagnosis relies on laboratory analyses to identify hyperandrogenism and exclude alternative conditions, we did not evaluate A4 or 17-hydroxyprogesterone to support an alternative condition, nonclassical adrenal hyperplasia. However, given the few DHEAS analyses provided and overall limited biochemical androgens completed, it likely would not have affected outcomes. Furthermore, it is not possible to know whether the low incidence of hirsutism documented in this PCOS group is due to diagnostic criteria or inadequate screening.
Conclusions
The first international evidence-based guidelines for PCOS assessment and management were published in 2018 to illuminate the complexity of the condition and improve clinical practice. Yet, in this retrospective chart review of a single academic medical center over a 5-year period following the publication of the guidelines, we observed diagnostic practices for PCOS and clinical hyperandrogenism among women with a history of established care were limited. Age of diagnosis differed between the groups; diagnosis of PCOS was largely limited to reproductive age, whereas diagnosis of hyperandrogenism was confirmed throughout the adult lifespan. Diagnosis of PCOS is one of exclusion and relies on thorough clinical evaluation and laboratory testing for confirmation, yet evidence of clinical and biochemical hyperandrogenism was infrequently reported. It is plausible that clinical symptoms related to hyperandrogenism including hirsutism or acne that were infrequently reported were observed by clinicians but not pertinent to the appointment visit and not recorded. Diagnostic challenges exist and necessitate coordinated care across the health care spectrum to manage PCOS-related comorbidities. Our findings support the need for future studies to evaluate barriers to diagnosis in clinical practice and increase education to support patient’s involvement in the diagnostic practices.
Footnotes
Author Disclosure Statement
No conflict of interest has been declared by the authors. All authors listed meet the authorship criteria according to the latest guidelines of the International Committee of Medical Journal Editors and agree with the article.
Funding Information
No funding was received for this article.
