Abstract
Introduction:
Female sterilization is one of the most common methods of contraception in the United States. 1 Historically, procedures were performed through abdominal laparotomy/laparoscopy. Transcervical sterilization was developed as an innovative method of sterilization performed in the outpatient setting without the need for general anesthesia. In 2002, Essure® was approved by the US FDA as a hysteroscopic sterilization method with comparable efficacy to traditional female sterilization. 2,3 In the United States, >750,000 units have been sold. 4 However, Essure has been associated with a growing number of adverse events. From 2002 to 2018, the FDA received 32,773 reports related to Essure. 5 Although the true adverse event rate for certain complications is unknown, migration/expulsion, perforation, and bowel obstruction have been described. 6,7 Chronic pelvic pain is the most common complication, occurring in 4.2% of patients. 8,9 Ultimately, the manufacturer stopped selling Essure in December 2018. Device removal is not mandatory; however, women with symptoms and/or malpositioned devices may desire removal. Removal through hysteroscopy, laparoscopy, and laparotomy has been described; however, there is insufficient evidence to recommend a preferred approach. Removal can be accomplished with salpingectomy for permanent sterilization, or less commonly, in women who desire fertility, device removal with concomitant tubal reanastomosis can be performed. This video illustrates laparoscopic techniques for Essure removal and salpingectomy.
Materials and Methods:
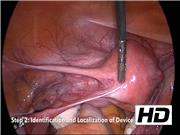
Thirty-two patients underwent Essure removal at Loyola University Medical Center from October 2013 to February 2019. A stepwise approach to laparoscopic Essure removal is shown. 10 The first step is knowledge of device anatomy. The second step is to localize the distal ball tip using laparoscopic instrumentation. For removal through salpingectomy/cornuectomy, mesosalpinx transection is performed in a medial direction toward the cornua. Confirmation of complete device removal is performed before further resection of a small portion of the cornua. Hemostasis can be achieved with bipolar energy obviating the need for suture of the uterine serosa. For removal using salpingostomy/salpingectomy, an incision is made in the isthmus to remove the device before salpingectomy. Lastly, specimen removal through backloading is demonstrated. Surgical “pull/push” and “open/spread” techniques are also highlighted.
Results:
The most common indication for Essure removal was pelvic pain (19 of 32; 59.4%). Estimated blood loss was 53.9 mL and mean length of stay was 0.7 days. Perioperative complications occurred in four patients. Complete and partial resolution of symptoms was reported by 78.1% and 18.8%, respectively.
Conclusion:
The increase in Essure-related complications and market withdrawal of the device has led to a growing need for skilled gynecologic surgeons who offer safe and effective Essure removal. Our video highlights the essential steps in performing effective laparoscopic removal of Essure devices for women seeking surgical management of Essure-related symptoms.
L.C.Y., ownership interest in KLAAS, LLC; L.M. and M.M., no competing financial interests exist.
Runtime of video: 9 mins 10 secs
This video has previously been presented at Society of Gynecologic Surgeons 45th Annual Scientific Meeting, Tuscon, AZ, March 31–April 3, 2019.


Get full access to this article
View all access options for this article.
